Abstract
The level dyeing index (LDI) is a measure of dye performance influenced by the dye migration process, ensuring the maximum uniformity of dye redistribution onto the fabric surface. The current study evaluates the level dyeing performance on polyester according to the energy level (low and high) and chemical classes (azo and anthraquinone based) of the three disperse dyes studied. The best levelness was obtained using C.I. Disperse Red 73 (an azo-based, low-energy level disperse dye), which exhibited the highest migration index (MI%) value. LDI results were obtained from the ratio of the exhaustion at the critical dyeing temperature (ECDT% and the final exhaustion Ef%), and the migration index (MI%). Each dye's LDI can be used to determine the compatibility of disperse dyes for combination dyeing.
Introduction
Disperse dyes are best suited for dyeing synthetic textiles and show better substantivity for hydrophobic fibers, such as polyester, nylon, and acetate, characterized by finely-ground water insolublity. 1 Low molecular weight (400-600 g/mol) dye particles are essential for efficient diffusion into materials, resulting in level dyeing. Disperse dyes often include azo, anthraquinone, or diphenylamine-based groups that are mainly non-ionic and contain no water solubilizing groups in their structure. They are available as either a dispersible powder or a concentrated aqueous dispersion.2-4
The Society of Dyers and Colourists (SDC) classify the dyeing characteristics of disperse dyes, including build-up, leveling properties, rate of dyeing, and the temperature range properties of the dyes on a range of hydrophobic fibers. Low-energy disperse dyes contains small dye molecules with low polarity, giving better leveling and rapid dyeing properties with poor heat resistance. More polar and higher molecular weight (MW) dyes have low dyeing rates and poor migration, but show good sublimation fastness. These are categorized as high-energy disperse dyes.1,5-7
The liquid-phase adsorption of C.I. Disperse Orange 30 onto activated carbon was previously studied for contact time, initial dye concentration, and pH at 30 ± 1 °C temperature. The Langmuir isotherm model was best suited and the adsorption rates followed the pseudo-second order model.8-11 Some attempts were made to study adsorption isotherms, kinetics, and thermodynamic parameters on polyester fabric at various temperatures. It was observed that the free volume of the polymer-dye system influences the dyeing kinetics. The adsorption time curves were determined by studying the dyeing kinetics of direct dyes on cotton fiber. Several experiments used synthesized monoazo disperse dyes and their thermodynamic parameters were examined.12-17
The effects of ultrasound on the K/S values, fiber, and disperse dye were observed by Wang et al. 18 using polyethylene terephthalate (PET) fabric. The report concluded that the ultrasonic power improved the K/S values as the dye particle size was reduced in solution.
The effect of temperature on the dyeing rate constant, diffusion coefficient, and time of half-dyeing of polyester fibers dyed with azo and anthraquinone-based disperse dyes was studied by Georgiadou et al. 19 Shibusawa et al. 20 examined the sorption isotherms of azo- and anthraquinone-based disperse dyes on cellulose film, acetate, nylon-6 film, and PET fibers. Taher 14 studied the possibility of reducing the temperature, dye use, and energy used in the conventional disperse dyeing of micro-polyester (0.22 dtex) and micro-nylon-6 (0.06 dtex). Disperse dyeing using nano-dye particles was studied to reach high exhaustion values comparable to those obtained with the conventional dyeing method at various temperatures. Different finishing conditions were also studied. 21
The present study focuses on the establishment of the level dyeing index (LDI) that represents even dyeing performance (i.e., the leveling properties of different energy levels and different chemical classes of disperse dyes). The LDI of disperse dyes is an indication of the combined effects of dye migration and exhaustion values. By the LDI, a dye compatibility study can be performed to ensure better level dyeing for combination shades.
Materials and Methods
Fabric
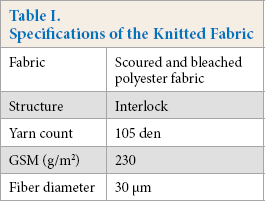
In this study a 100% polyester interlock knit, scoured, and bleached fabric (Micro Fibre Group, Narayangonj, Bangladesh) was used. The specifications of the fabric are listed in Table I.
Specifications of the Knitted Fabric
Disperse Dyes
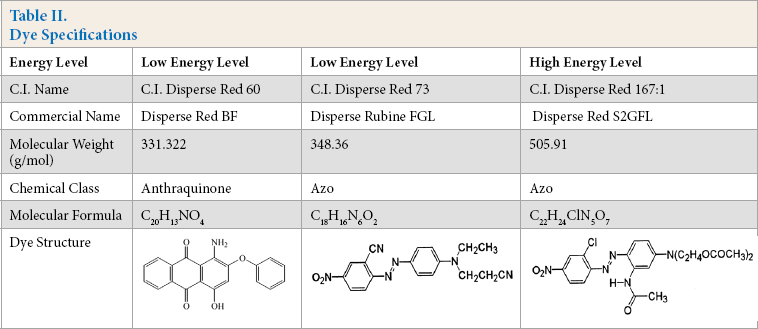
The dyes used in these experiments were Disperse Red BF (C.I. Disperse Red 60, with a maximum absorbance at λmax = 550 nm), Disperse Rubine FGL (C.I. Disperse Red 73, λmax = 620 nm), and Disperse Red S2GFL (C.I. Disperse Red 167:1, λmax = 590 nm). All the dyes and required chemicals used in the studies were obtained from Orient Chem-Tex Ltd.
These dyes were used according to their energy levels. Dyeing was carried out at various dye concentrations (0.01, 0.05, 0.1, 0.5, 1, 1.5, 2, 2.5, and 3 wt.%) for measuring dye buildup. Then, dye migration and rate of dyeing were observed at maximum saturation of each dye, which was obtained from build-up values. The dyes used, their structure and molecular weight, as well as energy levels are listed in Table II. The experiments were carried out five times and the average data were used for calculating all test parameters.
Dye Specifications
Dyeing Procedure
Dyeing of all samples was performed in a lab dyeing machine at 130 °C for 60 min with a liquor ratio (LR) of 1:10, and a pH maintained at 4.5 to 5.5. The dyeing machine used a programmed temperature maintaining system by a heating and a combined air-water cooling unit. Specially designed dye vessels with a septum by which small amounts of auxiliaries can be added during the dyeing process were used. The recipes used for dyeing are presented in Table III.
Dyeing Recipe

The following procedures were adopted for dyeing the samples. First, each individual sample and the required amount of water were added to the dye pot and the lab dyeing machine was started. After the bath temperature was raised to 40 °C, the dye was added, and the temperature was raised to 80 °C at a rate of 1-2 °C/min. During dyeing, fabric samples were collected at 80, 90, 100, 105, 108, 111, 114, 116, 118, 120, 122,124, 126, 128, and 130 °C At 130 °C, dyed fabric samples were also obtained after 5, 10, 20, 30, 40, 50, and 60 min. For each case (temperature and time conditions), color strength (K/S) values were used to measure the percent dye exhaustion after reduction cleaning.
After dyeing, fabrics were subjected to after-treatment. First, reduction cleaning was carried out at 80 °C for 20 min using hydroze (3 g/L) and caustic soda (2 g/L). A hot wash was then carried out at 60 °C in water and a cold wash was done at room temperature (RT). Finally, the dyed and washed samples were dried in a dryer for 30 min at 60 °C
Color Build-Up
Fabric dyed at 130 °C at various dye concentrations and absorbance values were measured at the same wavelength using UV-Vis absorption spectroscopy. Maximum dye saturation was considered as the color build-up for each dye. K/S values were determined using the Kubelka-Munk equation (Eq. 1).
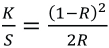
K is the absorption coefficient of the dye, S is the scattering coefficient of the dye, and R is the percent reflectance. By putting the values of percent reflectance in Eq. 1, the K/S values were obtained.
Migration Index
Fabrics were dyed with disperse dyes at the build-up concentration at 130 °C for 45 min. Then, half of the dyed sample was separated and another white fabric was dyed under the same conditions. Leveling properties were measured by the percent dye migration at the maximum saturation level for each dye after 15, 30, 45, and 60 min (Fig. 1). The dye migration properties were measured using Eq. 2. The migration index (MI) was calculated for each dye by using the K/S values of dyed and migrated samples. 22
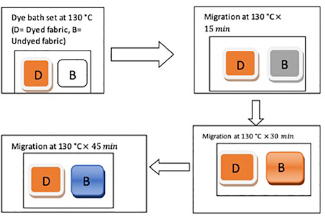
Schematic diagram of disperse dye migration test.
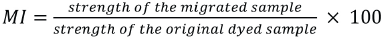
Dyeing Rate
The dyeing rate was obtained from percent dye exhaustion values at various temperature ranges. Exhaustion percentages determined by the amount of dye in the liquors were calculated by Eq. 3.
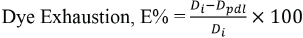
Di is the initial dye weight (mg) and Dpdl is the dye weight (mg) in the post-dyeing liquor.
Critical Dyeing Temperature
For high temperature dyeing with disperse dyes, fiber swelling occurs at 85-90 °C. The solubility of disperse dyes increases with increasing temperature, and less time is required to achieve higher exhaustion. The temperature at which the dyeing rate increases suddenly is considered the as critical dyeing temperature (CDT).
LDI
The LDI was measured from exhaustion values (at CDT and final exhaustion) and migration index (MI) by using the concept of the level dyeing factor for reactive dyeing, where dye substantivity was calculated based on salt addition. 22 For disperse dyes, the temperature effect was considered in the application of the LDI as exhaustion depends on dyeing temperature. Eq. 4 was used for LDI calculation.
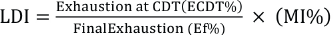
Results and Discussion
Color Build-Up
The color build-up values increased with concentration in all cases. Fig. 2 shows the absorbance values increased more rapidly for the 1% dye concentration; up to this level, the absorbance value rate increase was slow, indicating the saturation level for most of the dyes. Fig. 3 represents the surge value of absorbance for the low MW azo-based dye when compared with the other two dyes.
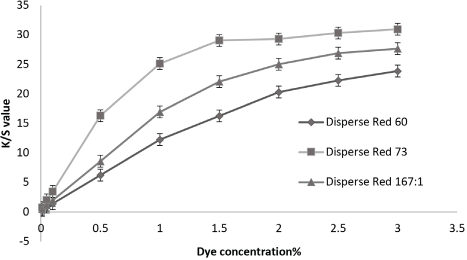
The effect of low MW azo and anthraquinone, and high MW azo, disperse dyes on color build-up.
Dyeing Rate
The highly crystalline polyester fiber swells at high temperatures and low MW disperse dyes quickly enter the fiber. But for higher MW dyes, the diffusion may not be effective. The dyeing rate is affected by the dye M W. Dye mobility increases as low MW dyes are transferred into fabric from the surface with the rise in temperature and dyeing time. 23
Fig. 3 shows that the azo-based low-energy dye had a slightly greater percent exhaustion than the high MW azo disperse dyes. This is consistent with the influence of chemical classes and energy levels on the dyeing rate. 3 The azo-based dyes impart a stronger (generally 2 or 3 times) tinctorial strength then the weak anthraquinone-based dyes. 7
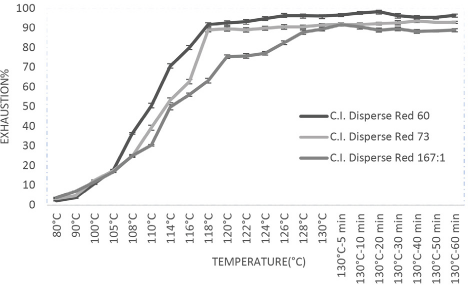
Effect of MW and chromospheres group on dyeing rate.
Disperse Dye Leveling
Leveling properties of dyes depend on dye migration during and after dyeing. It depends on how much disperse dye dyeing time is allowed since the dyeing mechanism is based on physical attachment of the dye on the fiber. Fig. 4 plots the percent migration of the disperse dyes for 60 min. It shows that migration values were greater for azo-based low MW disperse dye, whereas azo-based higher MW (energy level) dyes gave poor migration. From this result, it is seen that dye migration was influenced greatly by the MW of dyes rather than their chemical classes.
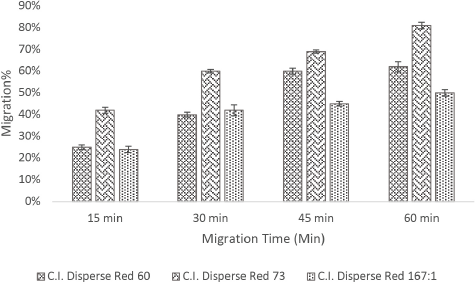
Effect of low MW azo and anthraquinone, and high MW azo, disperse dyes on percent migration.
LDI
Fig. 5 shows that if the dye had a higher value of migration and final exhaustion, due to its lower exhaustion value at CDT, the LDI was also low (i.e., the dye showed poor levelness). The percent LDI directly effects dye migration, and ECDT% and Ef% values.
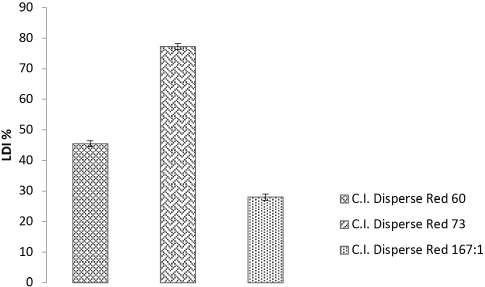
LDI% for low MW azo and anthraquinone, and high MW azo, disperse dyes.
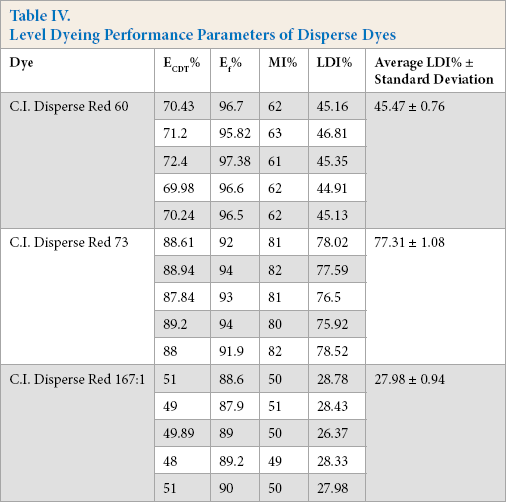
The results Table IV show that azo-based low-energy dyes had better leveling properties during dyeing as these dyes had low molecular weights. High-energy azo dyes gave the lowest LDI values due to lower exhaustion and migration properties. The average migration value, and ratio of ECDT% and Ef% values, for Disperse Red 73 were greater than values for the other two dyes. Since the ECDT%, Ef%, and migration values were greater, the LDI was also greater. Therefore, the low MW azo-based Disperse Red 73 dye showed the greatest leveling property.
Level Dyeing Performance Parameters of Disperse Dyes
Conclusions
Differences in dyeing behavior of disperse dyes indicate that dyeing is a function of the dye structure and molecular weight as well. Color build-up properties of dyes were used to find the saturation level at which the concentration of the dyeing was carried out. Dye build-up, migration, and the dyeing rate is related to their energy level, and for the dyes in this study, the low-energy azo-based disperse dyes had the highest LDI value. Therefore, it can be posited that disperse dyes leveling properties are properly balanced according to chemical classes and molecular weight of dye. It could also be affected by the dye particle size, which was not specified by the dye supplier. The lack of particle size information was a limitation of this study.
