Abstract
The objective of this study was to assess the effect of ficin enzyme as a new antifelting agent acting on wool yarn and fabric. The treatment effect on the dyeing efficiency of luteoline colorant that was extracted from weld (Reseda luteola) was also evaluated. The structural properties of antifelted and non-antifelted fibers were evaluated by scanning electron microscopy (SEM) and Fourier transform infrared (FTIR) spectroscopy. An optimal enzymatic process for antifelting and shrink proofing was achieved at 40 °C for 3 h in alkaline medium at pH 10. The treatment increased the dye absorbance and fixation of luteoline colorant on the wool yarn and fabric. It also improved the fastness proprieties, particularly when a mixed mordant of natural alum and tannin from Acacia meransii were used.
Introduction
Ficin belongs to the cysteine protease class of enzymes, such as papain and bromeline. The amino acid compositions of ficin and papain are similar, although ficin, like chymopapain, contains an additional cysteine residue. The amino-acid sequences around the active site cysteine and histidine residues of ficin are very similar to those around the equivalent residues in papain. 1
Ficin is used in cheese and sausage casing manufacture, and to chillproof beer. Ficin is sometimes included in meat tenderizers, usually in combination with papain and/or bromelain.
Ficin has rarely been used in the antifelting treatment of wool. However, there is extensive research on antifelting treatment with papain, savinase, cutinase, woolase, porcine trypsin, and others.2-5 Because papain has a similar chemical structure to ficin, their antifelting activity may be similar. Results from investigations with papain and other proteases were compared with those in this study using ficin. In some research, enzymatic treatment with papain and savinase as bactericidal enzymes, which belong to the serine protease class, revealed that papain appeared to degrade the scales more efficiently in hydrolysis mode than savinase. Therefore, papain has a higher hydrolytic effect and leads to a lower felting shrinkage than savinase.2,3 During antifelting, wool fabric can lose important proprieties, such as strength and weight, as was observed when comparing the felting shrinkage and strength retention between different proteases. Previous research2,3 has shown that papain causes a reduced mass loss and achieves better strength retention than savinase, but it did not exhibit a higher efficiency simultaneously. Therefore, a higher antifelting effect could be achieved only with a high mass and strength loss. Consequently, it was shown that individual protease treatment did not improve the antifelting property of wool fabrics visibly (i.e., combined cutinase and protease appeared more efficient than the keratinase-protease method, because of the cooperative action between amino acids of both proteases). The area shrinkage of this combined process decreased to 5.2% with a strength loss of 14%.4,6
Other combined processes were investigated using oxidizing agents—hydrogen peroxide, sodium hypochlorite, additive agents based on polyurethane, polysiloxane, and silicone, and chitosan—with commercial protease preparations, such as Perizym AFW, Alcalase 2.5L, Savinase 16L, and papain. 7
The hydrolytic effect of protease can increase the wool fabric wettability.4-6,8,9 The increased wettability can improve the coloration yield on the fiber significantly because the hydrophilicity increases after scales degradation. In the same context, this work investigated the effect of antifelting by ficin on dyeing, to improve the exhaustion and fixation dye rate on wool fiber.
Other techniques were used to improve the wettability and shrink resistance, such as the use of low-temperature atmospheric-pressure plasma on wool fabric under different conditions. The experimental results show that the shrinkage of 30.1% for untreated samples was reduced to ∼1.5% or less depending on the sample position and gases used. 8 A substantial improvement resulted in the wettability of the chlorinated and plasma-treated wool fabrics. The improved wool fiber surface hydrophilicity using a nitrogen plasma could enhance and achieve a better polymer deposition on the wool fiber surface to improve the antifelting properties of wool compared with the chlorination process. 9
We sought to achieve a new zero-adsorbable organic halogen compound (AOX) 10 antifelting processing alternative to conventional processes, such as chlorine/Hercosett processing, and use environmentally-friendly enzymes that were extracted from plants instead.11-13 We chose the fig tree (Ficus carica) plant as the source of a protease enzymes termed ficin. 14 Ficin belongs to the enzyme class that uses the nucleophilic group in cysteine as a catalytic part of the enzyme. Ficin enzymes can break and rebuild the peptide bonds between different amino acids in proteins, and they contribute to anabolic and catabolic processes. The average molecular weight of these enzymes is 25-30 kDa, and they are effective in the range of pH 4-9.5. 15 The enzymes are stable at 40-70 °C for 1 h.16-20
The aim of this work was to study the enzymatic effect of enzymatic antifelting treatment on wool. We tested the antifelting effect on wool yarn and fabric by using ficin that was extracted from fig tree leaves.17,20-27 Various treatment conditions were studied to establish the optimal combination.27-33 The focus was on wool yarn because of its higher accessibility and ease of assessing the treatment effect. The fabric will be studied in detail in the future to confirm the obtained results.
The effect of antifelting treatment on the dyed wool yarn and fabric was studied to improve the absorbance of luteoline dye extracted from the Reseda luteola plant.34-36 Mordanting during the dyeing was used by mixing the mineral mordant (natural alum) and organic mordant (tannin from Acacia mearnsii).37-39
Experimental
Materials
Wool Fiber and Fabric
Wool fiber was from the Chaouia Wrdigha region (Sardi breed) in Morocco. White fleece was compacted and homogenized into a fleece of 1.9-2.5 kg. The fiber fineness was 50-54 using the Bradford scale and the fiber medium length was 7.3 cm. The fabric was manufactured from the described fiber, with a plain weave and a density of ∼125 g/cm2.
Enzymes
Enzymes were extracted from Ficus carica tree leaves. Fig trees grow in several regions of Morocco. The extraction method consisted of maceration in distilled water for 24 h in acidic medium at room temperature (RT), followed by specific preservation in a freezer and purification.16,21,22,28
Natural Dye
Luteoline dye was extracted from weld (Reseda luteola), which grows in several regions in Morocco.37,40-42 The extraction method consisted of water-solubilizing the dried and powdered plant at a high temperature. As described below, dye extraction can also be achieved by maceration in water at low temperature.
Chemicals
Alkali reagent, sodium carbonate (Na2CO3), and acidic reagent, acetic acid (CH3COOH), were analytical grade and obtained from Sigma-Aldrich (Germany). A Marseille soap type, that can be obtained from a supermarket, was prepared from vegetable oil.
Bath
A 250-mL flask was used. Heating was done using a thermostat hotplate (Scilogex MS-H280-Pro, Scilogex, USA).
Enzymatic Treatment of Wool Yarn
Enzymatic Extraction and Proteolytic Activity
Dried fig leaves (10 g) were macerated in 100 mL of distilled water for 24 h at pH 5 and RT (20-25°C). The blend was filtered using filter paper, frozen at -15 °C for 6 h, defrosted at RT with soft stirring for 1 h, and centrifuged at 5000 rpm for 10 min. The supernatant was used in the antifelting treatment. 12
The proteolytic activity of ficin (like protease or keratinase) was determined utilizing casein as substrate according to Silva et al. 43 It was determined by measuring the concentration of the tyrosine resulting from the hydrolysis of casein according to Green and Stackpoole, 44 and Lowry et al. 45 It was found to be nearly 93 μg/mL at pH 5.2 and 35 °C.
For the following treatment baths, to keep the same enzymatic concentration and activation, 750 mL of homogeneous enzymatic extract was prepared from 75 g of dried fig leaves using the conditions described above.
Yarn Treatment: Optimization of pH Medium
Seven yarns (1 g each) were placed separately in seven baths that contained 50 mL of enzymatic extract. Each bath was treated separately at pH 4, 5, 6, 7, 8, 9, and 10 at RT for 24 h. At the end of the treatment process, the yarns were rinsed in cold water.
Yarn Treatment: Temperature Optimization
Three yarns (1 g each) were placed separately in three baths that contained 50 mL of enzymatic extract. The first bath was heated at 40 °C and kept at this temperature for 3 h at pH 10. The second and third baths were kept at 20 °C and 30 °C for 3 h at pH 10, respectively. The yarns were rinsed in cold water at the end of the treatment.
Yarn Treatment: Time Optimization
Five yarns (1 g each) were placed separately into five baths that contained 50 mL of enzymatic extract. The five baths were heated at 40 °C, kept at this temperature for 30 min, 1 h 30 min, 2 h, 2 h 30 min, and 3 h, and rinsed thereafer in cold water.
Yarn Soaping Test
The treated and untreated yarns (control sample) were soaped with 0.3−0.5 g/L detergent and rinsed at 40 °C with normal agitation. The soaping procedure was carried out similarly to three 5A wash cycles (ISO 6330 5A standard)5,6 for felting shrinkage testing. 5A represents the wash cycle program for the Wascator machine including the following procedure: normal agitation during heating and washing, temperature of 40 °C, soap concentration of 0.3−0.5 g/L, washing time of 12 min, and rinsing. The wool fabric shrinkage was measured using IWS Test Method 31.5,6
The results were expressed as a percentage of area shrinkage after relaxation and given as the arithmetic means of three parallel runs.
Enzymatic Treatment of Wool Fabric
Enzymatic Extraction and Fabric Treatment
Dried fig leaves (20 g) and the same yarn extraction protocol was followed for the fabric case. Wool fabric (1 g) was placed in the bath that contained 50 mL of enzymatic extract. The bath was heated at 40 °C and kept at this temperature for 3 h at pH 10.
Fabric Soaping Test
The treated and untreated fabric (control sample) were tested as described for the treated yarn test.
Dyeing of Yarn with Weld Extract
Mordanting
Wool yarn (2 g) was placed in a bath that contained 100 mL of distilled water and 1 g of alum. The bath was heated at 90 °C and kept at this temperature for 1 h. The yarn was rinsed and placed in a bath that contained 100 mL of tannin extract (prepared from 1 g of Acacia mearnsii bark). The bath was heated at 90 °C and kept at this temperature for 1 h.
Preparation of Extract Dyes
The weld plant (6 g) was stirred in 100 mL of water at 90 °C for 1 h and 10 g sodium carbonate was added to reach pH 10. The solution was filtered using filter paper and was well-preserved for the dyeing phase.
Dyeing
Two mordant yarn samples (1 g), one of which was antifelted as described previously, were placed separately into two baths that contained 50 mL of the weld extract at 70 °C for 1 h. Sodium carbonate (2 g) was added to reach pH 8, with a liquor ratio (LR) of 1:50.
Soaping
The dyed yarns were soaped with 0.5 g/L Marseille soap at 60 °C for 15 min at an LR of 1:50.
Dyeing of Fabric with Weld Extract
Mordanting
Wool fabric (4 g) was placed in a bath that contained 100 mL of distilled water and 2 g of alum. The bath was heated at 90 °C and kept at this temperature for 1 h. The fabric was rinsed and placed in a bath that contained 100 mL of tannin extract (prepared from 2 g of Acacia mearnsii bark). Likewise, the bath was heated at 90 °C and kept at this temperature for 1 h.
Preparation of Extract Dyes
The weld plant (4 g) was stirred in 100 mL of water at 90 °C for 1 h, and 10 g sodium carbonate was added to reach pH 10. The solution was filtered by filter paper and was well-preserved for the dyeing phase.
Dyeing
Two mordant fabric samples of 2 g, one of which was antifelted as described previously, were placed separately into two baths that contained 50 mL of the weld extract at 70 °C for 1 h, and 2 g sodium carbonate was added to reach pH 8, with an LR of 1:50.
Soaping
The dyed fabric was soaped with 0.5 g/L Marseille soap at 60 °C for 15 min with an LR of 1:50.
Analysis
UV-Vis Spectrophotometry
A UV-Vis spectrophotometer (Thermo, Helios Epsilon (USA)) was used at wavelengths from 325 to 1100 nm with a spectral bandwidth of 1 nm. Spectrophotometer calibration was achieved by using distilled water as a standard solution.
Dye exhaustion and fixation measurements were made as follows. A solution (1 mL) was removed from each dye bath for measurement. Each sample of yarn and fabric was diluted to 20 mL and 30 mL, respectively, using distilled water. Absorbance measurements were made at 380 nm.
Colorimetry
Reflectance (R%) values of dyed samples were measured on a Thermo Scientific SPE (USA) instrument (D65/10°) based on CIE L*a*b* colorimetric space.
Infrared Spectroscopy
A Thermo Scientific TM IS 5 Fourier transform infrared (FTIR) instrument (USA) was used at wavelengths from 800 to 2500 cm−1.
Scanning Electron Microscopy
The morphology of the studied samples was examined by scanning electron microscopy (SEM) using a Carl Zeiss EVO 10 SEM (GmbH, Jena, Germany) operated at 15 kV. Before SEM observations, the samples were coated with a thin conductive gold layer using an ion sputtering apparatus.
pH Meter
An Adwa AD1000 pH meter (Szeged, Hungary) was used; this multimeter can measure pH, oxidation-reduction potential, and temperature.
Mass Loss
The mass loss percentage was evaluated and calculated by determining the absolute dry mass of the yarn and fabric before and after any enzymatic treatment.
Tensile Strength
The tensile strengths of wool yarn and fabric were measured by using a yarn and fabric tensile strength tester according to methods ASTM D2256 and ISO13934.1,5,6 respectively. The tensile strength expressed as a maximum load in the warp direction for the fabric and as a maximum stretching for the yarn until bursting. It was given as the arithmetic mean of three different samples.
Statistical Tests
The number of replicates in each optimization experiment (for the yarn and fabric) was at least three. The average values were carried out from the average of three replicate measurements.
Results and Discussion
Enzymatic Treatment of Wool Yarn
Treatment pH
Table I shows the length variation of antifelted and non-antifelted yarns after the soaping test. The antifelted yarns were treated at seven pH values for 24 h at average temperatures of 20 and 25 °C. All antifelting measurements were achieved three times to ensure the reproductivity of the results.
Shrinkage and Mass Measurements of Antifelted Yarns at Various pH Values after 24 hours of Enzymatic Treatment
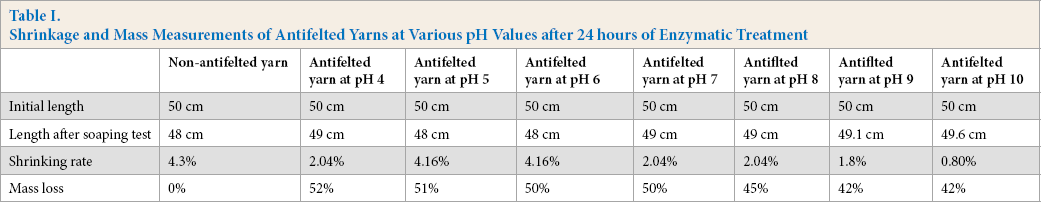
The variation in shrinkage was irregular for the range of tested pHs. Effective treatments resulted at pH 4 and from pH 7 to 10. This behavior could be explained by the higher activity of the ficin enzyme in slightly and mildly alkaline solution, and the lower activity in acidic medium, even though less shrinkage occurred at pH 4.
A large antifelting effect was established at pH 10 for 24 h of treatment at average temperatures of 20 and 25 °C. This optimal condition can ensure a good shrinkage resistance after the soaping test.
The mass losses of the yarn samples before and after protease treatments were measured and are shown in Table I. An obvious decrease of all treated yarn masses resulted from the long treatment time. This mass decrease was considered sufficient to describe the wool proprieties without the requirement for a strength measurement after antifelting treatment.
A slightly higher mass loss resulted for samples treated in acidic and neutral media (pH 4 to 7), and a lower mass loss resulted at pH 9 and 10. This result may be explained by the higher diffusion ability of ficin inside the fiber in acidic and neutral media. In acidic medium, the enzyme, with cysteine being positively charged (-SH2+), had less interaction with the fiber due to the positive charges on wool amino groups at a lower pH. This led to a lower electrostatic retention, which caused an enhanced accessibility of enzyme inside the fiber. In the basic medium, the enzyme acted mostly on the fiber surface with less diffusion into the fiber.
These values confirmed an optimal pH of 10, which corresponded to the higher antifelting effect (0.8%) and relatively lower mass loss (42%). However, such a large mass loss was unacceptable. Therefore, reduced treatment time and increased temperature were required to obtain the same antifelting effect with the least fiber mass and strength loss.
Treatment Time
Table II shows the length variation of antifelted yarns after the soaping test. The antifelted yarns were treated at five treatment times: 30 min, 1 h, 1 h 30 min, 2 h, and 3 h at 40 °C.
Shrinkage and Property Measurements of Antifelted Yarns at Various Treatment Times
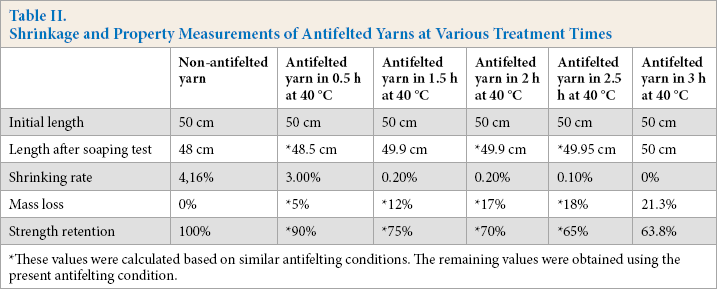
These values were calculated based on similar antifelting conditions. The remaining values were obtained using the present antifelting condition.
The optimal antifelting time was established at 3 h of treatment at 40 °C. This time registered a shrinkage of 0.0%, with a relatively lower mass loss (21.3%) and lower strength retention (63.8%). Compared with the results from a treatment time of 24 h, these results were much improved.
Treatment Temperature
Table III shows the length variation of antifelted yarns after the soaping test. The antifelted yarns were treated at three treatment temperatures: 20, 30, and 40 °C for 3 h.
Shrinkage and Property Measurements of Antifelted Yarns at Various Temperatures
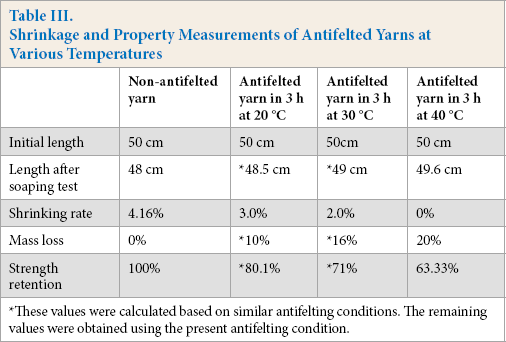
These values were calculated based on similar antifelting conditions. The remaining values were obtained using the present antifelting condition.
The optimal antifelting temperature was established at 40 °C for 3 h of treatment. These conditions gave a shrinkage of 0.0%, with a similar mass loss (20%), and strength retention (63.33%) as achieved previously.
Therefore, we can conclude that 40 °C had a positive effect on boosting the enzymatic hydrolysis of the fiber scales. A further increase in treatment temperature above 40 °C was unnecessary, avoiding further fiber mass and strength degradation.
Optimal Yarn Treatment Summary
The antifelting property of wool yarns was improved significantly over untreated wool yarns by using ficin treatment under optimal conditions for the yarn. These were established at 40 °C for 3 h at pH 10, which achieved an antifelting shrinkage of 0% with an equivalent mass loss of 20% and a strength retention of 63.33% compared to the non-optimal treatment conditions described previously.
The mass loss for the protease-treated wool included the fiber-scale and interior protein segment degradations. Consequently, a reduction in treatment time from 24 h to 3 h and limiting the treatment temperature to 40 °C led to less degradation of fiber mass and strength. Nevertheless, the high strength loss (36.67%) for the yarn under optimal conditions could be reduced in various ways, for instance, by reducing the antifelting efficiency until the shrinkage reached 2% or 3%. Also, it could be reduced by twisting the treated yarns to increase their strength or make fabric with them. Mostly, these yarns are used to make fabrics or nonwoven materials.
Similar results found by other researchers will be discussed in the fabric treatment section.
Enzymatic Treatment of Wool Fabric
Because the fabric was composed from the same wool yarn, the optimized antifelting conditions investigated for the yarn were considered to be similar to those of the fabric. The optimal pH for antifelting yarn treatment was established at pH 10, 40 °C and 3 h (Table IV). However, the shrinkage of the fabric (4.47%) was greater than that of the yarn. This may be because the complex fabric weave could hinder enzyme accessibility to the inter-yarn space. The least mass loss and greater strength retention of the fabric resulted compared with those obtained for the yarn under optimal conditions. Shrinkage resistance of the fabric could be improved by increasing the quantity of ficin enzyme in the antifelted extract, but this could cause more fiber damage.
Shrinkage and Property Measurements of Antifelted Fabric under Optimal Conditions
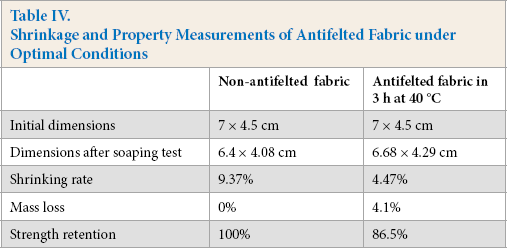
A comparison of these results with those of Wang et al., 4 who used individual and combined enzymatic treatments, showed that savinase treatment reduced the area shrinkage of cutinase-pretreated fabric to 6.6%, and that treatment with cutinase and papain gave a shrinkage of 4.7%. This combined treatment achieved a greater antifelting effect because of the cooperative actions between amino acids of both proteases. It is suggested that cutinase pretreatment destroyed the integrity of the scale epicuticle and facilitated protease accessibility to the wool cuticle, which allowed the succeeding proteolytic reaction to occur easily in the underlying protein layer. 4 According to Wang et al., 5 the area shrinkage of the combined cutinase and protease was 5.2% with a strength loss of 14%.
The ficin treatment in the present study showed a similar antifelting effect compared with the combined process with cutinase-papain or cutinase-savinase, and was more effective than the individual proteases cutinase, keratinase, and papain, which have a similar enzyme structure to ficin. These results may be due to the easy accessibility inside the fiber and the higher activity of ficin cysteine-protease under optimal treatment conditions (pH 10, 40 °C, 3 h).
However, as described by Dong and Xu 3 and Zhang et al., 6 fibers pretreated with an oxidizing agent, such as hydrogen peroxide followed by enzymatic treatment using savinase, woolase, or papain yielded the best antifelting effect with a lower felting shrinkage (0.83%, 0.95%, 2.49%, respectively), but with significant fiber mass and strength degradation, especially with savinase (mass loss 12.27%; strength retention 43%).
The well-known commercial antifelting agent chlorine-Hercosett (chlorination treatment followed by coating with a polyamide-epichlorohydrin resin) is certainly the most effective of all the treatments investigated, and the least expensive shrink-resist treatment. 46 It has become an industry standard for making machine-washable wool. Nevertheless, it affects the handle properties of wool fabrics and its application process produces an effluent containing AOX coming from both the chlorinating agent and the Hercosett resin, making it environmentally hazardous. 47
Characterization of Antifelting Treatment
Microscopy Analysis
A microscopic view of the untreated yarn shows the original form of the fiber scales. Antifelted yarns showed smaller fiber scales (Fig. 1). This result illustrates the efficiency of the ficin enzymes extracted from fig tree leaves. The antifelted yarn that was treated at pH 10 showed more scale destruction than the yarn that was treated at pH 8 because of the double effect of the enzymes and alkaline medium. The wool keratin was less resistant to the highly alkaline medium.

Morphological structure of (a) non-antifelted and antifelted yarns at (b) pH 8 and (c) 10.
FTIR Analysis
FTIR analysis was performed for three yarn structures: a washed yarn, an antifelted mordanting yarn, and a non-antifelted mordanting yarn (Table V).
Infrared Spectroscopy Analysis
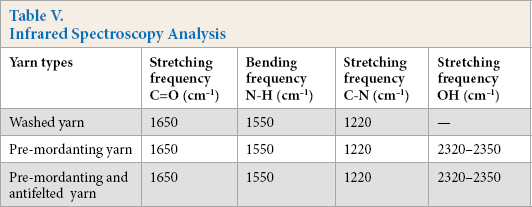
The FTIR spectra of these yarns revealed the existence of similar peaks between 800 and 1700 cm−1, which may be attributed to the keratin structure (Fig. 2a and b). However, the non-antifelted dyed yarn spectra contained peaks from 2320-2350 cm−1, which are attributed to the hydroxyl stretching bond in the carboxylic acid or aromatic group. 48 The spectra in Fig. 2d was similar to that of the tannin extracted from quebracho 49 that contained basically catechin (condensed tannin) like that from Acacia mearnsii. The broad band at 3371 cm−1 revealed the existence of intramolecular hydrogen-bonded hydroxyl groups in chelate form. 48
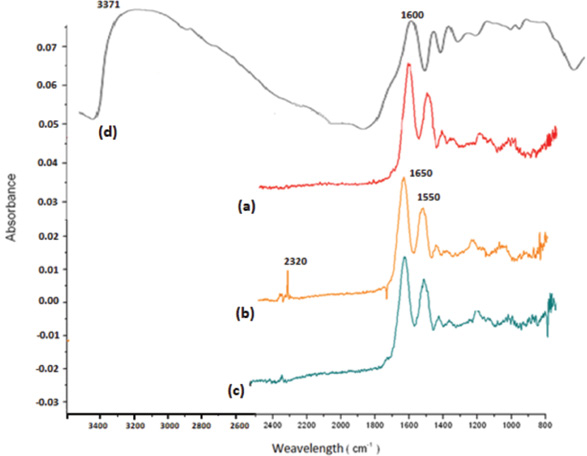
FTIR spectra of (a) washed and non-antifelted yarn, (b) non-antifelted and pre-mordanted yarn with alum-tannin, (c) antifelted and pre-mordanted yarn by alum-tannin, and (d) tannin extract.
Nevertheless, in the pre-mordanted yarn, this band shifted to 2320-2350 cm−1 and became sharp. This may be due to a residual amount of intramolecular hydrogen-bonded hydroxyl groups, with the majority of -OH groups in tannin having been reacted with different groups of keratin. This finding indicates the existence of polyphenolic components that comprised the Acacia meranssi tannin. The mordanting antifelted yarn spectra revealed the similar peak in the same range of 2320-2350 cm−1, but at a minor intensity (Fig. 2c).
Besides the appearance of tannin in the spectra, there was no clear difference between the antifelting and non-antifelting spectra. Therefore, FTIR analysis did not serve as an identification method for wool antifelting treatment.
Effect of Antifelting Treatment on Dyeing
Dye Exhaustion and Fixation
In Tables VI and VII, the absorbances of the initial and final exhausted dye and residual soaping baths are presented, respectively. The exhaustion dye rate on the antifelted yarn and fabric was greater than the non-antifelted yarn. This can be explained by the greater fiber accessibility to the dye, due to the partial destruction of fiber scales. The mordant tannin-alum complex contains several active sites that could react strongly with the fiber and luteoline. This led to high dye absorption and fixation.
Exhaustion of Luteoline in Dye Bath
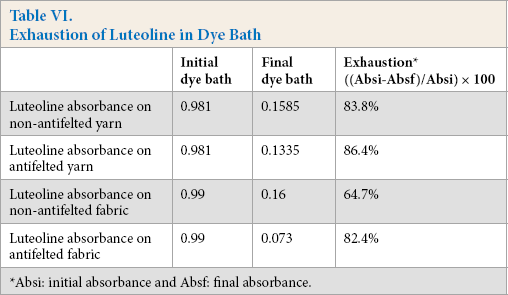
Absi: initial absorbance and Absf: final absorbance.
Fixation of Luteoline in Dye Bath
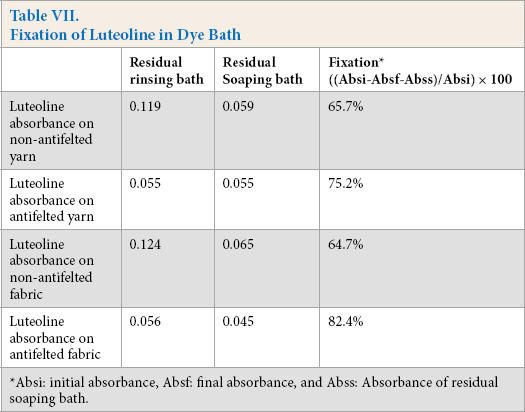
Absi: initial absorbance, Absf: final absorbance, and Abss: Absorbance of residual soaping bath.
The effect of antifelting on the fabric dye absorbance of the yarn was important, possibly because of the greater scale destruction on the fabric. Ficin can cleave the bound fatty acids on the wool surface and the disulfide cross-links, giving the wool improved wettability and shrink resistance. The wettability is an important factor to improve the dye absorbance or exhaustion in the dye bath. After antifelting treatment, the dye absorbance increased when the wettability increased. This improvement in wettability was previously observed when the wool fabric was antifelted. Wang et al. 5 found that the contact angle of the protease-treated wool fabric decreased to 66°. The authors attributed this higher wettability to the effect of cutinase pretreatment that removed the hydrophobic lipids partially from wool epicuticle, increased the keratinase accessibility towards the substrate of the disulfide cross-links, and made the subsequent proteolytic reactions effective in removing the wool scales. 5 This study proposes that the combined actions of cutinase and keratinase-protease may be replaced by ficin action and by using optimal treatment conditions.
Reflectance Measurements
Both antifelted and non-antifelted yarn samples showed the seam curve profile, which indicates a homogeneity in the applied colorant (Fig. 3). The high reflectance values were in the 630-nm wavelength range, which is attributed to the yellow-orange color. The higher reflectance of the antifelted yarn was 51.39% at λ = 630 nm, and for the non-antifelted yarn it was 45.65% at λ = 630 nm.
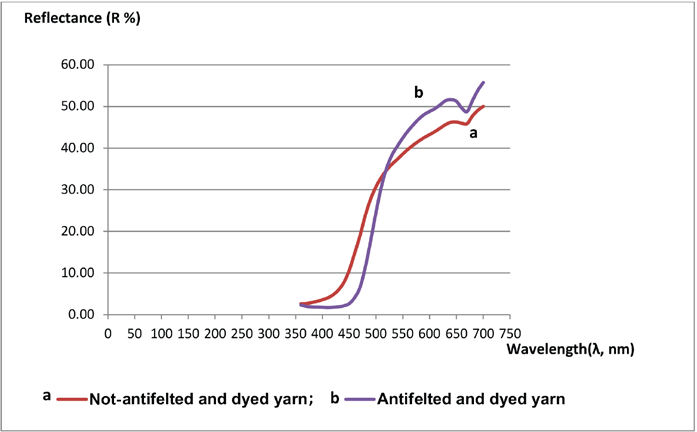
Reflectance curves of non-antifelted dyed yarn and antifelted dyed yarn.
Therefore, the antifelted yarn absorbed and fixed more colorant than the non-antifelted yarn. This result confirmed the calculated values of exhaustion and fixation.
Washing and Rubbing Fastness
In Table VIII, washing and rubbing fastness test50,51 measurement results are presented for the dyed antifelted and non-antifelted yarns. These tests confirmed the solid fixation of dyes by using antifelting. As described previously, the antifelting effect improved the dye diffusion significantly, due to the destruction of a large portion of the scales. Effective fixation of dyes on the fiber occurred because multiple bonds were established between the mordant complex tannin-alum and the complex antifelted dyed fiber. The alum coordinated with the hydroxyl groups of luteoline and keratin, and the tannin hydrogen bonded with luteoline and keratin. The antifelting treatment effect increased the chemical activity of the fiber surface by cleaving the cystine disulfide cross-link. This allowed further bond creation between cysteine and dye. The dye fixation and rubbing fastness increased as a consequence. The excellent rubbing fastness that was achieved by antifelting and mordanting fibers resulted because of resilient dye fixation on the fiber surface.
Washing and Rubbing Fastness of Dyed Yarns
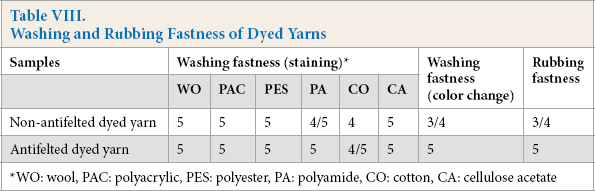
WO: wool, PAC: polyacrylic, PES: polyester, PA: polyamide, CO: cotton, CA: cellulose acetate
Conclusions
Antifelting treatment with ficin enzymes extracted from fig tree leaves ensured a high shrinkage resistance of the resulting wool yarn and fabric, compared with the untreated yarn and fabric, but produced some mass and strength loss. Application of these enzymes were optimized on the yarn, along with the mordanting process that was investigated previously.
The shrinkage property was significantly improved by using ficin treatment under optimal conditions for yarn and fabric. At 40 °C for 3 h at pH 10, the antifelting shrinkage for the yarn and fabric was 0% and 4.37% with a mass loss of 20% and 4.1% and a strength retention of 63.33% and 86.5%, respectively. The measured percentages of the residual strength and mass loss for the wool fabric samples after ficin enzyme treatment under optimal conditions were improved.
The antifelting effect improved the dye absorbance and fixation on wool yarn and fabric. This improvement resulted because of the partial destruction of fiber scales, which increased the wettability and accessibility of the fiber for the luteoline dye.
Despite the tight and dense fabric weave, the dye absorbance and fixation were superior. This result confirmed the effectiveness on the use of these enzyme types, along with the mordanting complex tannin-alum that can interact, particularly through coordination and hydrogen bonding, with dyes and fiber. Washing and rubbing fastness tests confirmed that the deep solid shade remained on the fiber.
