Abstract
Superhydrophobic surfaces have attracted attention due to their hydrophobic, self-cleaning, anti-icing, and oil/water separation properties. The present study used a nonwoven fabric as a flexible substrate and constructed a high-adhesive superhydrophobic surface by coating the fabric with polydimethylsiloxane (PDMS)-co-polymethyl hydrogen siloxane (PMHS) polymers and embedding mesoporous SiO2 nanoparticles. The water contact angle (WCA) of the superhydrophobic surface was up to 165.2° at a PDMS to PMHS ratio of 4:1. The adhesion to deionized water was 99.7 μN. High hydrophobicity was maintained, even after sandpaper abrasions and flowing water impact. The surface was resistant to acid, alkali, brine, strong oxidation, and heavy metal solutions. The coating exhibited anti-icing and oil/water separation properties. This study provides a facile and effective method for constructing multifunctional superhydrophobic coatings on flexible substrates.
Keywords
Introduction
Superhydrophobicity is a naturally-occurring phenomenon that is reflected in many things, including lotus leaves and insect wings, among others. Non-wetting surfaces that have a high water contact angles (WCA > 150°), and on which droplets can easily slide, are referred to as superhydrophobic surfaces.1,2 The properties of superhydrophobic surfaces give them excellent features such as anti-icing, 3 self-cleaning, 4 and oil/water separation abilities. 5 The formation of superhydrophobic surfaces requires micron or nanometer size roughness and low surface energy.6,7 Based on these two primary conditions, there are many methods to prepare superhydrophobic surfaces currently, such as phase separation, 8 the template method, 9 sol-gel treatment, 10 and etching. 11 However, these methods have some limitations, including expensive reaction equipment, long process routes, high cost, and solvent contamination or deficient membrane strength and membrane area. Therefore, a facile, rapid and effective preparation method for textiles should be explored and developed.
In the creation of a rough superhydrophobic surface, silica particles have been widely-used due to their stability. The structure of the SiO2 molecule is generally regular and ordered due to the high bond energy of the Si-O bond, which contributes to excellent mechanical and chemical stability. Various superhydrophobic surfaces have been prepared using SiO2 nanoparticles. For instance, a bio-based benzoxazine monomer was synthesized using cardanol to provide low surface energy, and SiO2 nanoparticles were used to increase surface roughness. 12 Superhydrophobic surfaces were produced on wood substrates using vinyltriethoxysilane (VTES) modified SiO2 nanoparticles, which were durable and highly resistant to abrasion. 13 The surface of a polyester fabric was sprayed with the adhesive styrene-butadiene rubber (SBR), and the hydro-phobic SiO2 nanoparticles dispersed in the alcohol were sprayed onto the adhesive layer. The resultant superhydrophobic surface exhibited high robustness, mechanical durability, stain resistance, and self-cleaning properties. 14 Hydrophobic SiO2 nanoparticles could be directly applied to common fabrics by simple spray coating without further processes like annealing or heat treatment. The superhydrophobic fabrics showed a water contact angle of 172° and a sliding angle of 3°. 15
Another factor in constructing superhydrophobic surfaces is the requirement of low surface energy. At present, fluoropolymers have been substituted with environmentally-friendly silicones, which also have low surface energy. 16 Silicone refers to an organic/inorganic bi-form polymer composed of a (-Si-O-) backbone chain and two organic groups attached to each silicon atom. 17 Polydimethylsiloxane (PDMS) is an organosilicon compound with two methyl groups attached to a silicon atom and a (-O-Si-O-) backbone chain. PDMS is transparent and is often mixed with other substances to make superhydrophobic surfaces. 18 For instance, a combination of 3-mercaptopropyltri-methoxysilane (MPTS) modified graphene oxide (mGO), and vinyl-terminated polydimethylsiloxane (V-PDMS) was used to produce a superhydrophobic hybrid coating for oil/water separations on polyester fabrics. 19 However, PDMS has mechanical defects, and the coating should, therefore, be optimized to make PDMS an ideal surface. 20 Hydrophobic composite membranes are prepared by cross-linking polymethyl hydrogen siloxane (PMHS) with PDMS. 21 PMHS is a silicone polymer. 22 PMHS has many Si-H bonds that can react with the unsaturated hydrocarbon groups in PDMS. 23 The mechanical strength, antifouling property, flexibility, and texture of the resulting superhydrophobic fabrics were improved because of the PDMS-co-PMHS crosslinked coating. 24
Based on the above research, the thermal curing method was used to prepare the superhydrophobic surface. The process used in this study for the preparation of a superhydrophobic surface is efficient and simple. Nonwoven viscose-polyester blended fabrics were used as the coated fabrics, and PDMS and PMHS were used as the hydrophobic silicone agents. The PDMS and PMHS were crosslinked and polymerized to form a copolymer. The PDMS-co-PMHS with SiO2 superhydrophobic coating was made by doping mesoporous SiO2 nanoparticles in the copolymer solution and constructing a rough hierarchical structure by coating. The surface morphology and chemical structure of the superhydrophobic surface were determined. An in-depth analysis of the mechanical stability (e.g., abrasion and water impact) and chemical stability (e.g., resistance to acid, alkali, and brine) of the surface was also done. In addition to stability testing, high adhesion, anti-icing, and oil/water separation property attributes of the super-hydrophobic coatings were also evaluated.
Experimental
Materials and Reagents
The materials and reagents used in the work are shown in Table I. The water used in the entire experiment was deionized.
Specifications and Manufacturer Information for Materials and Reagents
Preparation of Nano-Silica Particles
Deionized water (17 mL), ethanol (3 mL), and CTAC (35.5 mg), were added to a 50 mL three-necked flask and stirred for 10 min. This step was followed by a dropwise addition of 1.1 mL of TEA, and the mixture was stirred at room temperature (RT) for 20 min. The mixture was transferred to a water bath maintained at 60 °C, and 1.4 mL of TEOS was added dropwise with vigorous stirring for 3 h. The resulting white precipitate was mesoporous silica nanoparticles. The above white precipitate was dispersed in absolute ethanol, collected by centrifugation at 5000 rpm for 5 min, and the supernatant was discarded. The template removal solution was prepared by mixing 2 mL of 37% HCl with 16 mL of ethanol, and the obtained sample was dispersed in the mixed solution and kept shaken for 15-20 min. Finally, the sample was washed with ethanol 2-3 times, and dried in an oven at 40 °C. A white powder of mesoporous silica nanoparticles was thereby obtained.
Preparation of PDMS-co-PMHS with SiO2 Composite Coating
The cross-linked products of PDMS and PMHS were used as low-surface energy coating materials. The low surface energy coating was prepared as follows. The mixtures of PDMS and PMHS, with mass ratios of PDMS to PMHS set as 1:1, 2:1, 3:1, 4:1, 5:1, and 6:1 were stirred with a smart magnetic stirrer for 10 min. The initial stirring speed should be controlled at 200 rpm and the various PDMS-co-PMHS coating liquids were obtained after uniform mixing.
The SiO2 particles are required for the construction of roughness. Under the optimal cross-linking ratio of PDMS and PMHS prepared above, that is, the coating showed the maximum WCA, SiO2 particles, with mass ratios to polymer of 5%, 6%, 7%, 8%, and 9%, were added into the mixture of PDMS and PMHS (4 mL), respectively. After stirring at a gentle stirring speed of 200 rpm for another 10 min, the PDMS-co-PMHS with SiO2 composite coatings were prepared.
Fabrication of PDMS-co-PMHS with SiO2 Superhydrophobic Coating
The nonwoven fabric (4 × 4 cm) was fixed on the experiment table, and 1 g of PDMS/ PMHS with SiO2 composite coating liquid was transferred to a glass piece and placed on one end of the nonwoven fabric. The coating liquid was quickly scraped from one end of the nonwoven cloth to the other with another piece of glass. The coated nonwoven fabric was dried in a constant temperature oven and cured at 120 °C for 10 min to obtain the PDMS-co-PMHS with SiO2 coated nonwoven fabric.
Characterization
Superhydrophobicity
A contact surface measurement system (JC2000C, Shanghai Zhongchen Digital Technology Equipment Co. Ltd.) was used to test the wettability of the sample surface by measuring the WCA value. To minimize error, each sample was tested at five random points and averaged.
Morphology Characterization
The surface morphology of the PDMS/PMHS with SiO2 coatings was examined using a field emission scanning electron microscope (SEM, S-4800-I, Hitachi, Japan). The surface topography and roughness of the PDMS-co-PMHS nonwoven fabric and PDMS-co-PMHS with SiO2 nonwoven fabric were evaluated by atomic force microscopy (AFM, Dimension Icon, Bruker, USA).
Chemical Structure Characterization
The chemical compositions of the original nonwoven fabric surface, and the nonwoven fabric coated with PDMS-co-PMHS and PDMS-co-PMHS with SiO2 were characterized by infrared-Raman spectroscopy (Nicolet 6700, Thermo Fisher, USA). The types and elemental composition on the surface of the fabrics were analyzed using energy-dispersive X-ray spectroscopy (EDS, S-4800-I, Hitachi, Japan).
Mechanical Stability
Resistance to abrasion was determined using a self-made device. Briefly, a piece of PDMS-co-PMHS with SiO2 non-woven fabric (2 × 2 cm) was fixed to the bottom of a 50 g weight, and the weight was placed on 600 mesh sandpaper and pulled horizontally at a speed of 2 cm/s. Each 10 cm covered was recorded as one time, and the WCAs of the nonwoven surface after abrasion for 10, 20, 30, 40, 50, and 60 times were measured. The strength and elongation at break of the original and PDMS-co-PMHS with SiO2 non-woven fabric were tested using a desktop electronic fabric strength machine (YG(B)026PC, Wenzhou Darong Textile Instrument Co. Ltd. China).
The fabric with a surface angle of 45° facing outward was perpendicular to a water outlet by 20 cm. The water flow rate was adjusted to about 3 m/s and 500 mL of water was recorded as one cycle. After exposure to a specific volume of water, the fabric was dried, and the WCA was recorded.
Chemical Stability
The superhydrophobic fabrics were immersed in a series of solutions at various pH values, including strong acid solutions, alkali solutions, brine, and heavy metal solutions for specific durations, to evaluate their chemical stability. The samples were removed from solution at 2, 4, 6, 8, and 10 h, and the surface of the nonwoven fabrics was rinsed with distilled water and dried at 150 °C for 10 min.
Anti-Icing Performance
The original nonwoven fabric and PDMS-co-PMHS with SiO2 nonwoven fabric were fixed on a glass piece with double-sided tape. Reactive Red dye solution (0.2 mL) was dropped on the surface of the samples using a pipette and placed in the freezer at a temperature of -18 °C. The samples were taken out every 10 min to observe the icing of the water droplets. The icing condition of the two kinds of nonwoven fabrics was separately recorded using a microscope and a camera until both fabrics were frozen.
High Adhesion Performance
PDMS-co-PMHS with SiO2 nonwoven fabric was cut into long strips (2.5 × 5 cm) and flatly pasted on a glass sheet. Water droplets were dropped on the sample surface, and the volume of water droplet was gradually increased from 2 μL. Then, one end of the glass slide was slowly raised to increase the angle of the glass slide to the horizontal plane. When the water droplet was slipped, the volume of the droplet was recorded and the number of slip angles were measured. The adhesion was calculated according to Eq. 1.
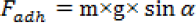
m is the mass of the water drop, g is the acceleration by gravity, and α is the angle between the cloth surface and the horizontal plane.
Oil/Water Separation
To perform the absorptive capacity test, various oils or organic solvents were used as the target adsorbed liquid. A 2 × 2 cm PDMS-co-PMHS with SiO2 nonwoven fabric weighing 0.1 g was used to calculate the oil absorption efficiency X according to Eq. 2.
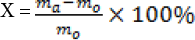
ma is the weight of the wet sample with oil or organic solvent (g), and m0 is the weight of the dry sample (g).
Results and Discussion
Superhydrophobic and Morphology
The construction mechanism of the PDMS-co-PMHS with SiO2 superhydrophobic surface on nonwoven fabric is shown in Fig. 1.
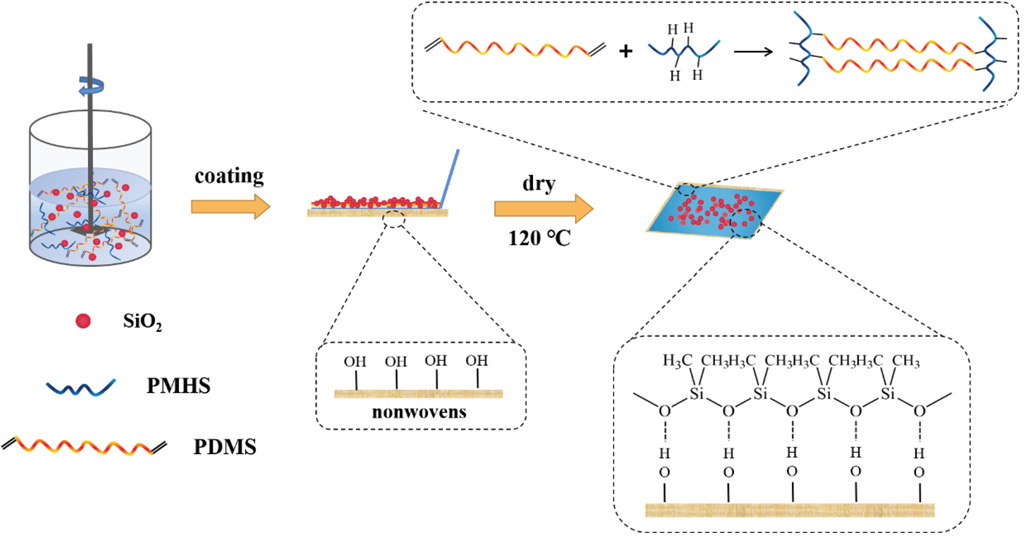
Copolymerization reaction between PDMS and PMHS, and construction mechanism of PDMS-co-PMHS with SiO2 superhydrophobic coatings on nonwovens.
The superhydrophobic property of the as-prepared nonwoven fabrics was examined by measuring the water contact angle (WCA). Fig. 2 shows a typical picture of water droplets on the surface of nonwoven fabrics prepared by copolymerization of various proportions of PDMS and PMHS coatings. On the surface of different nonwoven fabrics, water droplets were hemispherical, elliptical, or approximately spherical in appearance.

Images of water droplets on different types of nonwoven fabrics. (a) original, (b) PDMS:PMHS = 1:1, (c) PDMS:PMHS = 2:1, (d) PDMS:PMHS = 3:1, (e) PDMS:PMHS = 4:1, (f) PDMS:PMHS = 5:1, (g) PDMS:PMHS = 6:1, (h) PDMS:PMHS = 4:1+ 5% SiO2, (i) PDMS:PMHS = 4:1 + 6% SiO2, (j) PDMS:PMHS = 4:1+ 7% SiO2, (k) PDMS:PMHS = 4:1+ 8% SiO2, and (l) PDMS:PMHS = 4:1+ 9% SiO2.
While the water completely drenched the original non-woven fabrics due to the hydroxyl groups in the viscose fiber structure, the PDMS-co-PMHS coated fabric exhibited hydrophobicity, and were, therefore, not drenched by water droplets. The WCA values increased with increasing proportions of PDMS and PMHS. When the ratio of PDMS to PMHS was 4:1, the WCA value of the treated nonwoven fabric reached a high of 148.49°. Further increments in the proportion of PDMS and PMHS resulted in reduced WCA values. The WCA value of the treated nonwoven fabrics without a SiO2 rough surface ranged from 120° to 150°. Moreover, the nonwoven surface constructed by PDMS-co-PMHS with SiO2 was superhydrophobic, and the WCA value hit a high of 165.2° (Fig. 2k). When the amount of SiO2 added increased from 5% to 8%, the WCA value gradually increased from 152.65° to 165.43°, showing superhydrophobicity. However, when the added amount reached 9%, the WCA value decreased. This indicates that SiO2 nanoparticles had constructed a rough hierarchical structure on the surface of the nonwoven fabric, and superhydrophobicity was successfully achieved after modification by PDMS-co-PMHS coatings with low surface energy.
SEM images of the surface of the original and resultant superhydrophobic fabrics taken at different magnifications are illustrated in Fig. 3. The differences between the hydrophobicity of the various fabrics were evident, even at low-magnification. The SEM images of the fabrics after modification, the fibers covered under the PDMS-co-PMHS layer, are represented in Figs. 3b and d, respectively. The images indicated that the PDMS-co-PMHS polymers successfully coated the fabric surface.

SEM images of nonwoven fabrics. (a) untreated nonwoven fabric at ×3500, (b) untreated nonwoven fabric at ×6000, (c) nonwoven fabric coated with PDMS-co-PMHS with SiO2 at ×3500, and (d) nonwoven fabric coated with PDMS-co-PMHS with SiO2 at ×6000.
As shown in Fig. 3a, the original fiber has a clean and smooth surface, devoid of any roughness. However, obvious differences in surface morphology between the original and modified fabrics were observed from the high-magnification SEM images (Fig. 3c). Unlike the original fabric, the modified surfaces had many protrusions, demonstrating that the SiO2 nanoparticles adhered to the fibers strongly. The fibers coated with the layer of PDMS-co-PMHS polymers were mainly coated on the fiber surface. Changes in diameter were negligible, indicating that PDMS-co-PMHS coatings had good film-forming properties and the roughness of the fiber surface was increased. The PDMS-co-PMHS layer protected the coated SiO2 nanoparticles. The resulting contact angle sizes and durability of the fabric hydrophobicity were due to this modification.
AFM was used to study the surface profile of the nonwoven fabric. As shown in Fig. 4, the PDMS-co-PMHS coating with an average roughness (Ra) of 242 nm had a relatively smooth surface, and a root mean square roughness (Rq) of 354 nm. Compared with the PDMS-co-PMHS coating, the microstructure of the PDMS-co-PMHS with SiO2 coating significantly changed. A large bulge was formed on the surface of the coating, resulting in an increase in average roughness (Ra) to 618 nm and the root mean square roughness (Rq) to 777 nm. These results indicate that the SiO2 nanoparticles successfully constructed a rough surface on the PDMS-co-PMHS with SiO2 coating, which can trap a lot of air in the contact surface between water droplets and solids. Also, the contact area was reduced, but the contact angle increased.
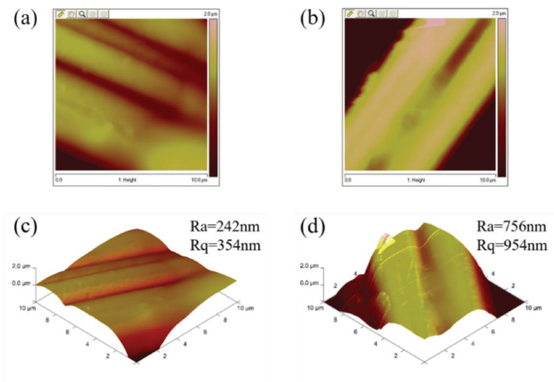
(a) 2D surface structure of PDMS-co-PMHS coating, (b) 2D surface structure of PDMS-co-PMHS with SiO2 coating, (c) 3D image of PDMS-co-PMHS coating, and (d) 3D image of PDMS-co-PMHS with SiO2 coating.
Chemical Structure and Composition
The chemical structure of the fabric surface modified with PDMS-co-PMHS with SiO2 coatings was analyzed by Fourier transform infrared (FTIR) spectroscopy. As shown in Fig. 5a, only a single band at 1016 cm−1 was detected, corresponding to the benzene C-H in-plane bending vibration, 25 and indicating the presence of polyester fiber in the fabric. Five additional absorption bands at 804, 1021, 1086, 1264, and 2963 cm−1 were detected on the surface of the PDMS-co-PMHS with SiO2 nonwoven fabric. The bands around 800 and 1100 cm−1 were assigned to symmetric and asymmetric stretching vibrations of Si-O-Si in a siloxane network, suggesting that the synthesized matrix is a composite of siloxane and organic molecules.26,27 The adsorption bands at 804 cm−1 correspond to the stretching vibrations of Si-C and symmetric stretching of Si-O-Si groups from PDMS. 28 The band at 1021 cm−1 was the characteristic peak of Si-O-Si from PDMS-co-PMHS. 29 Due to the 1021 cm−1 peak occlusion, the 1016 cm−1 peak was no longer visible here. The peak at 1264 cm−1 represented the deformation vibration of -CH3 in Si-Me2 groups. During the cross-linking reaction, the Si-H bonds of PMHS reacted with the Si-OH bonds of PDMS, and no band was observed at around 2200 and 3250 cm−1, which could be attributed to Si-H groups and OH groups, indicating that the cross-linking reaction was complete. 30
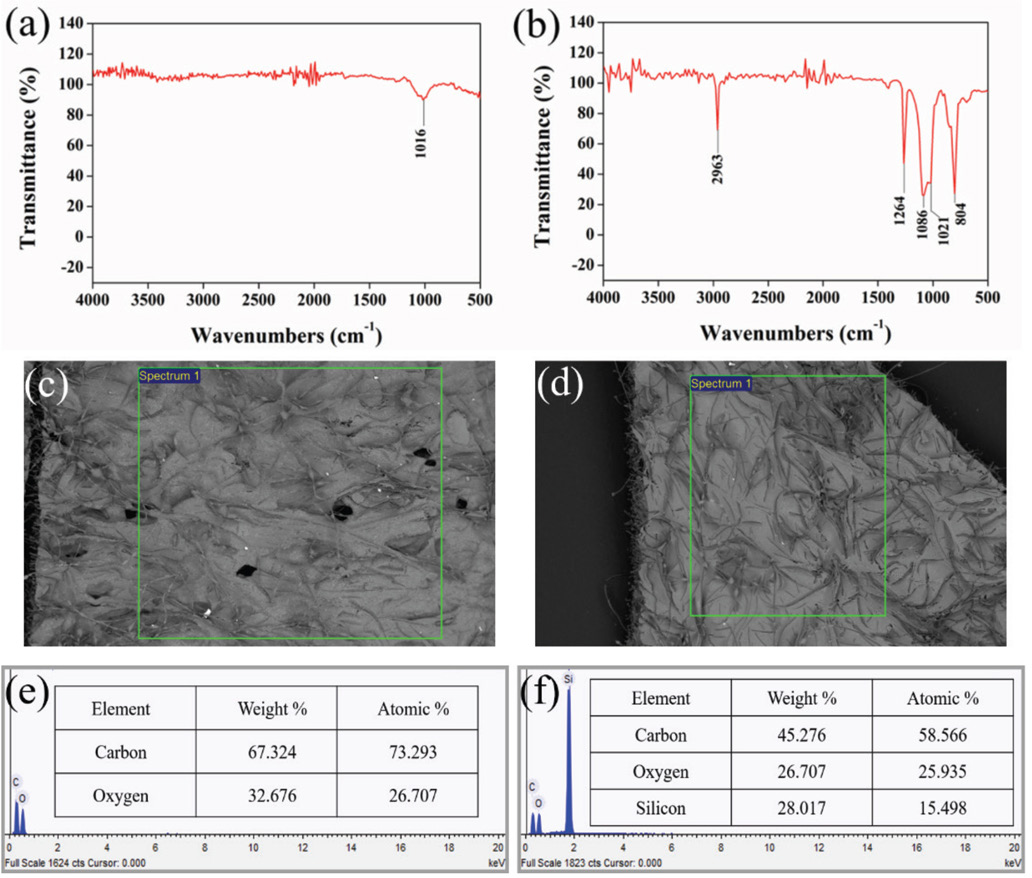
FTIR spectra. (a) Original nonwoven fabric, (b) PDMS-co-PMHS with SiO2 fabric. EDS analysis. (c) and (e) PDMS-co-PMHS coating, and (d) and (f) PDMS-co-PMHS with SiO2 coating.
EDS analysis was performed to examine the surface chemical composition of the sample. Figs. 5e and f show the EDS diagram of PDMS-co-PMHS nonwoven fabric and PDMS-co-PMHS with SiO2 nonwoven fabric, respectively. The test results show that the PDMS-co-PMHS treated and the PDMS-co-PMHS with SiO2 treated nonwoven fabrics were composed of C, O, and Si elements. Following the introduction of SiO2 into PDMS-co-PMHS coatings, the mass fraction of C decreased by 6.995%, and the atomic proportion decreased by 6.062%. The mass fraction of O increased by 1.337%, and the atomic proportion increased by 2.386%. The mass ratio of Si increased by 5.658%, and the atomic proportion increased by 3.675%. The large increase in the ratio of Si gave the coating low surface energy and roughness.
Mechanical and Chemical Stability
Abrasion tests were performed as previously described. 31 As shown in Fig. 6a, the surface of the PDMS-co-PMHS with SiO2 nonwoven fabric was gradually damaged, and the WCA value decreased with an increase in the number of abrasions. After the 20th abrasion, the WCA value of the sample surface was 152.59° and it still had superhydrophobic properties.
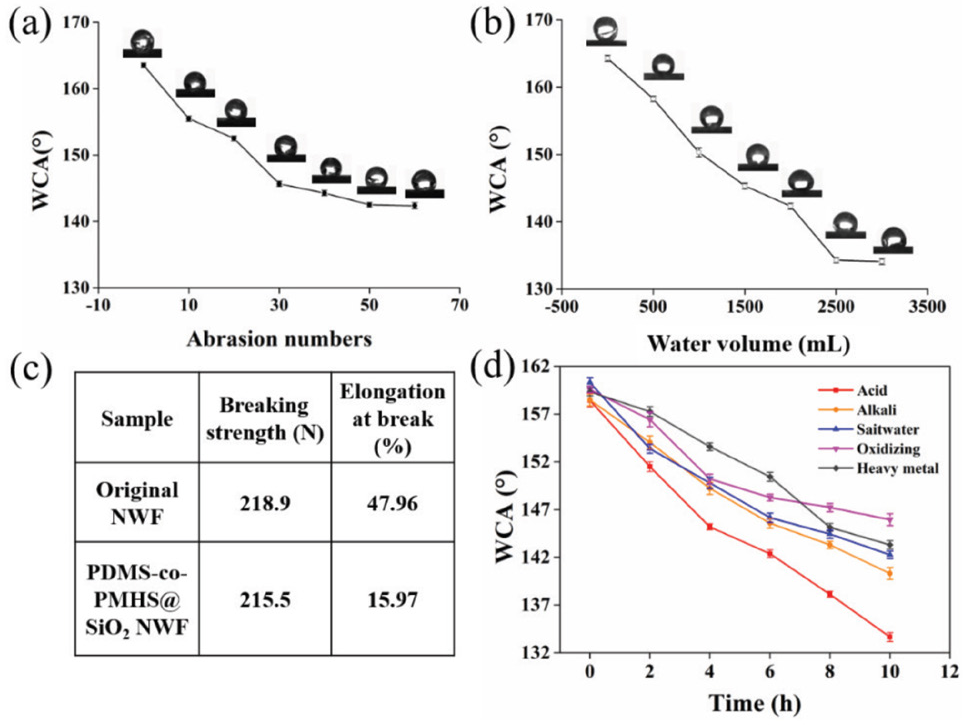
(a) WCAs after various cycles of abrasion, (b) WCAs after various cycles of water impact, (c) breaking strength and elongation at break, and (d) WCAs after exposure to various chemical reagents.
After the 60th time, the WCA value of the sample surface was 142.25°. These findings reveal that the PDMS-co-PMHS with SiO2 nonwoven fabric prepared by this method had excellent resistance to abrasion.
As shown in Fig. 6b, an increase in the volume of water impact resulted in a gradual decrease in the WCA value of PDMS-co-PMHS with SiO2 treated fabric. The WCA value after the impact of 1000-mL water was 150.32°, and was reduced to 134.02°, following an impact of 3000-mL water.
Application of the PDMS-co-PMHS polymer and SiO2 nanoparticles on nonwoven fabric altered the mechanical performance of the prepared fabric. The mechanical performance characteristics of the treated fabric, including tensile strength and breaking elongation, were assessed and the results presented in Fig. 6c. The breaking strength of PDMS-co-PMHS with SiO2 fabric slightly decreased, and the elongation at break was significantly reduced due to a lot of PDMS-co-PMHS polymer network being deposited on the surface of the fabric, making the fabric more firm and worsening tensile properties.
It can be seen from Fig. 6d that the superhydrophobic coating has good oxidation resistance. After being soaked in a KMnO4 solution for 10 h, its WCA value decreased by only 8.6%, from 159.64° to 145.86°. The superhydrophobic coating after treatment in pH 12 NaOH, 3.5 wt.% NaCl, and CuCl2 solutions also showed good stability, which, after being soaked for 10 h, the WCA values dropped to between 10.2% to 11.3%. HCl, with a pH value of 1, had a greater effect on it. After being soaked for 10 h, the WCA value of the sample decreased by 15.5%, from 158.35° to 133.85°.
High Adhesion
To better observe the shape of the water droplets, a small amount of reactive dye was dissolved in water. It can be seen from Fig. 7 that when the droplet volume was increased from 2 to 4 μL, the droplets could adhere to the surface of the coated fabric. The shape of the droplets was not noticeably deformed when the sample was placed vertically. When the volume was increased to 8 μL, although the droplet still adhered to the surface, the liquid was obviously deformed and sagged due to the effect of gravity. The adhesion test was continued with 10 μL of deionized water at an angle change. It can be seen from Fig. 7 that when the sample was placed vertically, the water droplet still adhered to the surface, but the shape changed significantly. After the sample was rotated 180°, the water droplet hung on the superhydrophobic surface without falling down, proving that the superhydrophobic surface had excellent adhesion.

Morphological images of deionized water droplets at various angles.
Water droplets with a volume of 12 μL would slip of during tilting, and the slip angle obtained after repeated measurements was 58°. The data was brought into Eq. 1 and the calculated Fadh was 99.7 μN. The surface of the PDMS-co-PMHS with SiO2 nonwoven fabric not only had superhydrophobic properties, but also showed high adhesion to water droplets. This was because there was a large amount of fluff on the surface of the nonwoven fabric, and the fluff increased the force of the water droplets and the fabric surface, thereby generating higher adhesion.
The dye droplets (10 μL) were dropped on the surface of the samples and then placed on a heating plate. As shown in Fig. 8, the droplet shape began to change at 50 °C, and it remained at a similar shape until 70 °C. The shape was further elongated and deformed at 80 °C, but still adhered to the sample surface. The increase in temperature had no effect on the adhesion of PDMS-co-PMHS with SiO2 nonwoven fabric to droplets and revealed excellent high adhesion at higher temperatures.

Morphological images of droplets in various states and temperatures.
The adhesion test was performed using a more viscous defatted sheep blood, and the adhesion of the droplet on PDMS-co-PMHS with SiO2 nonwoven fabric was also improved (Fig. 9). The morphology of the droplet did not change significantly at a volume of 10 μL, even though the sample was rotated by 90°. A slight deformation at a volume of 12 μL was observed. The volume was increased to 14 μL, and the droplet morphology changed significantly when rotated to 45°. When the droplet volume was 16 μL, it began to slide during tilting and the sliding angle measured was 68°. Fadh was calculated by Eq. 1 to be 173.6 μN. Compared with the deionized water adhesion results, the slipping angle increased by 17.24%, and Fadh increased by 74.12%. The viscosity of the liquid had a greater impact on the adhesion of PDMS-co-PMHS with SiO2 nonwovens. The adhesion of the sample was enhanced with increased liquid viscosity.
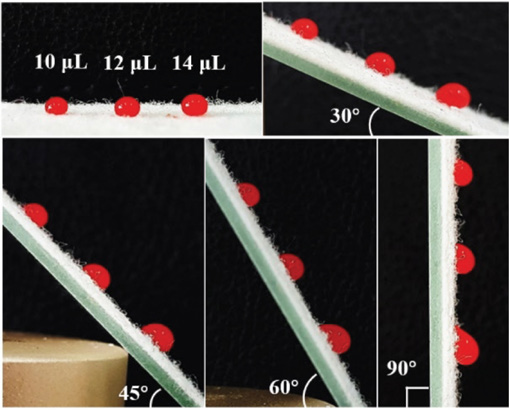
Morphological images of defatted sheep blood at various angles.
Anti-Icing Performance
The assessment of the dispersion state of the aqueous solution on the cloth surface and the icing condition were done using a test liquid of Reactive Red dye dissolved in water. It can be seen from Fig. 10 that when the dye droplet was dropped on the surface of the original fabric, it spread rapidly and laid flat on the surface, filling the fibers with red liquid. However, when dropped on the surface of the superhydrophobic nonwoven fabric, the drop assumed an elliptical shape. After being placed in the refrigerator for 0.5 h, the surface of the original nonwoven fabric was frozen, and ice particles formed (Fig. 10a). However, the water droplets on the surface of the PDMS-co-PMHS with SiO2 nonwoven fabric hardly changed. After 2.5 h, the water droplets on the PDMS-co-PMHS with SiO2 nonwoven fabric were frozen (Fig. 10b), indicating that the sample surface had excellent anti-icing properties. The surface of the superhydrophobic fabric was composed of low surface energy components and a rough structure. Subsequently, the amount of water hanging was reduced, and the contact area of the droplets with the surface was also reduced, thereby delaying icing.
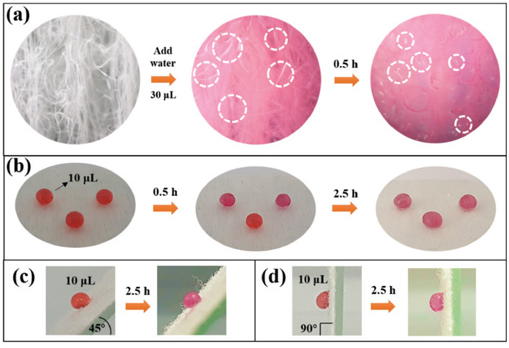
(a) Digital images of droplets on the surface of the original fabric, and (b)-(d) droplets frozen on superhydrophobic fabric surface at different angles.
Moreover, Figs. 10c and d showed that the frozen droplets still adhered to the sample surface when the angle to the horizontal plane was 45° and 90°, respectively. In addition, the freezing time did not changed.
Oil/Water Separation
Adsorption efficiency is the ratio of the weight change of nonwoven fabric before and after adsorption. The results of testing of the adsorption efficiency are shown in Fig. 11. The least absorbed solvent by the PDMS-co-PMHS with SiO2 nonwoven fabric was ethyl acetate, with an absorption amount of 0.1665 g and an absorption efficiency of 166.5%; the most absorbed solution was chloroform, and the absorption amount and the absorption efficiency were 0.3085 g and 308.5%, respectively. The PDMS-co-PMHS with SiO2 nonwoven fabric also displayed excellent absorption capacity for other kinds of oils and organic solvents. For a substrate with low surface energy, the spreading ability of the liquid depends on whether the surface energy of the fluid is greater than that of the solid. In the current experiment, the surface tension of water was greater than that of the superhydrophobic coating, and the surface was hydrophobic. However, the surface tension of the oil was less than that of the super-hydrophobic coating, and the surface was not oleophobic. Consequently, the superhydrophobic coated sample could separate oil and water.
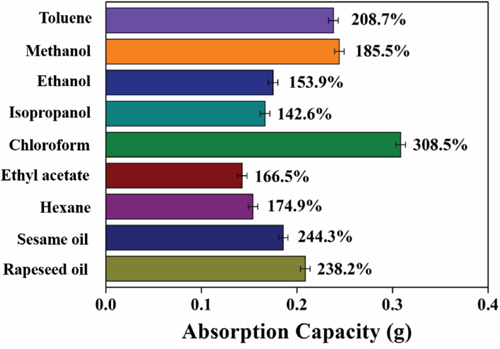
Absorbency of oil and organic solvents.
Conclusion
The present study successfully prepared PDMS-co-PMHS with SiO2 superhydrophobic membrane-embedded mesoporous SiO2 nanoparticles by coating and thermal curing methods. A nonwoven fabric was used as a flexible substrate, and the resultant superhydrophobic fabric has excellent superhydrophobicity with an average WCA value of 159° and a peak WCA value of 165.2°. The mechanical/chemical structure, stabilities, and multiple functional properties of the superhydrophobic fabric were characterized by SEM, EDS, AFM and FTIR. The superhydrophobic fabric had excellent mechanical stability, as shown by its resistance to abrasion, tensile strength, and water impact. Also, the superhydrophobic fabric was chemically stable, as indicated by its ability to maintain a high WCA value under acid, alkaline, brine, strong oxidation, and heavy metal solution conditions. The superhydrophobic surface showed high adhesion, which was 99.7 μN and 173.6 μN to deionized water and defatted sheep blood. The superhydrophobic fabric also had oil/ water separation and anti-icing properties. This study, therefore, presents a simple and effective strategy of constructing a super-hydrophobic surface on a flexible substrate.
Footnotes
Acknowledgments
This work was supported by the Key Project of Hebei Education Dept. (Grant No. ZD2020112) and the Open Project Program of Fujian Key Laboratory of Novel Functional Textile Fibers and Materials (Grant No. FKLTFM1901).
