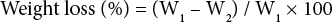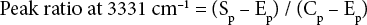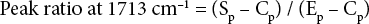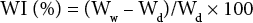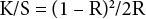Abstract
A new, rapid, and eco-friendly technique for identification of polyethylene terephthalate (PET) composition in blend fabric was developed. This technique could replace a conventional composition identification of PET using toxic chemicals such as halogenated organic solvent or strong inorganic acid. Choline-based deep eutectic solvents (DES), such as ethylene glycol-choline chloride, were used as a treatment medium under microwave irradiation. The PET portion of the blend fabrics, such as 65/35 and 50/50 PET/cotton was completely removed by DES containing 5% NaOH (w/v) after 100–140 s of microwave irradiation. Various instrumental analyses confirmed the removal of PET. Finally, a commercial sample was also tested as a practical application of the new test method.
Keywords
Introduction
Polyethylene terephthalate (PET) is one of the most common textile materials for many years and has been used in various applications. More than 50 million tons of PET is consumed annually and its application has been expanded to many non-clothing areas. 1 PET/cotton blends are also widely used in clothing applications, especially as men's shirt fabric. Composition identification of homogeneous or blend fabric is thus needed to confirm fraudulent labeling in clothing. Microscopic identification could classify natural fibers, but is generally impossible for synthetic fibers. Since the blend fabrics contain a single yarn made of two or more different fibers, its fiber composition is especially difficult to determine. Mechanical separation is very tedious to perform and common chemical identification methods mainly require very strong acids or toxic organic solvents such as 70% sulfuric acid and halogenated organic solvents such as hexafluoropropanol for PET.2-4 70% Sulfuric acid requires personal protection during the identification or separation process due to its strong acidity. It also not only dissolves cotton and other cellulosic fibers, such as linen and rayon, but also could easily dissolve several synthetic fibers, such as acetate, nylon, and vinal in addition to silk. This would limit application of this solvent to separation of other cotton blends with nylon or acetate. 4
PET is also very widely used in non-textile applications such as water bottles. Since a large use of PET generates tremendous amounts of waste, producing negative impacts on atmosphere and biosphere, its recycling methods have been much studied.1-6 The common method for recycling PET water bottles has been cleaning, followed by production of a new granulate that can be used for the extrusion process. In addition, various chemical reactions such as alcoholysis, aminolysis, glycolysis, or hydrolysis have also been used.7,8 Moreover, halogenated organic solvents that are used to dissolve PET produce significant environmental problems, such as volatile organic chemicals.9,10 Use of enzymes has also been also examined for PET recycling as an eco-friendly method. 11 In this regard, these processes showed certain limitations for commercialization, such as high costs for chemicals and energy, and long process times.1,10 Additional recycling methods, using microwave irradiation along with a metal catalyst, substantially reduced the time required for PET decomposition.8,9,12
Ionic liquids have been recently studied to replace volatile organic solvent systems. 2 For example, an ionic liquid, such as 1-allyl-3-methylimidazolium chloride, was used to dissolve the cotton portion from cotton-polyester blend fabrics. 2 These conventional ionic liquids show very low volatility and flammability, but they carry some limitations, such as higher cost (around 10 times higher than organic solvents), similar or higher toxicity than organic solvents, and low biodegradability in the long run, adversely affecting the environment.13-18
Deep eutectic solvents (DES) are similar to ionic liquids in many ways. They are composed of various combinations of anionic and cationic compounds, such as hydrogen bond donors and quaternary ammonium compounds.14,19 DES is a low transition mixture that has a freezing point lower than 150 °C when the two reagents are mixed. 14 Choline chloride (ChCl) melts at 302 °C and is widely used as a cationic compound due to its safety, economy, and for environmental reasons. 15 It often produces freezing point depression up to 27 °C when mixed with a hydrogen bond donor, such as an alcohol, amide, or carboxylic acid in DES.16,19 This is a significantly greater freezing point depression compared with typical imidazolinium-type ionic liquids, consequently resulting in a liquid state at much lower temperatures. Moreover, DESs are generally much less toxic than typical ionic liquids and are much easier to produce by a simple mixing of two components.14-19
Unlike a conventional heating method, microwave irradiation is a simple, convenient way to heat the materials. 8 Microwave radiation is located between the infrared and radio frequencies of electromagnetic radiation.20,21 There are two mechanisms involved in heating the materials by microwave irradiation: the first is electric permittivity caused by molecular oscillation of the dipole moment, and the second is electric conductivity caused by ionic oscillation. 22 The common factor between these mechanisms is heat generation caused by vibration of the molecules corresponding with the specific frequency of microwave. Then, microwave energy is transitioned to heat energy that can increase temperature of the material.21,22 It is known that electric conductivity has a greater influence in the heating process than electric permittivity. 22 Ionic DES, therefore, could produce greater heat energy than a nonionic solvent, such as water, during microwave irradiation. 22
Our previous study indicated that PET was dissolved by DES made of ethylene glycol and choline chloride in the presence of small amounts of alkali. 19 Other DESs made of urea and choline chloride did not dissolve PET. A recent acute toxicity study made on Cyprinus carpio fish also revealed that DES made of ethylene glycol and choline chloride was practically harmless, while another DES made of zinc chloride was highly toxic. 17 Therefore, the present study examined the feasibility of using a specific DES for identification of PET in PET/cotton blend fabric. Moreover, if the dissolution of PET is possible by DES, it can also be used as a solvent system for PET recycling. This study was carried out using microwave irradiation to facilitate the rate of the process. To confirm removal of PET from the blend fabric and/or to investigate the mechanism of PET dissolution, various instrumental methods such as Fourier transform infrared spectroscopy (FTIR), X-ray diffraction (XRD) analysis, scanning electron microscopy (SEM), differential scanning calorimeter (DSC), thermogravimetric analysis (TGA), and high performance liquid chromatograph (HPLC) were used to investigate the identification method and the potential mechanism of PET dissolution.
Experimental
Materials
Four different types of fabrics were used in this study: 100% PET, PET/cotton blend fabric (65/35 and 50/50 by weight percent, hereinafter named PC63 and PC55, respectively), and 100% cotton fabric. All the fabric specimens (5 × 5 cm) obtained from FITI, Korea, were plain weave structures. Blended knit cloth with 45% PET and 55% cotton (PC45) was also examined to confirm applicability of the identification method to commercial fabric. Chemicals such as ethylene glycol (99% purity, EG), choline chloride (99%, ChCl), sodium hydroxide (NaOH, 98%), and anhydrous sodium carbonate were purchased from Daejung Chemicals, Korea. Other chemicals for analyses and characterization, such as trifluoroacetic acid and acetonitrile, were purchased from Aldrich, Inc., Korea, were reagent grade, and used without further purification.
Preparation of Specimens and DES
Fabric specimens were washed with 1% aqueous sodium carbonate solution at 80 °C for 10 min, followed by rinsing several times with deionized water to ensure removal of impurities. The fabrics were then dried at 100 °C for 10 min in an oven. In general, 0.45∼0.53 g of the specimens were used throughout the study.
DES was prepared in a very simple way as described in the previous study. 19 A mixture of EG and ChCl in a 2:1 mole ratio was heated in a beaker to 80 °C with continuous stirring. The beaker containing homogeneous and transparent DES was stored in a desiccator with the exclusion of air. The DES prepared was stable and reusable at least for a week. Finally, 5% NaOH (w/v) was added in the DES and the solution was stirred for three days at 45 °C for complete dissolution of alkali, and this DES was hereinafter labeled DES-N. The disparity between DES and DES-N was evaluated with conductivity measurement using an electric conductive meter (CM-42X, CT-58101B) and FTIR spectroscopy.
Identification of PET Composition
The fabric specimen was placed in an Erlenmeyer flask containing DES solution containing 5% NaOH (DES-N) at 50:1 liquor-to-fabric ratio. The flask was sufficiently shaken to ensure a complete wetting of the fabric, followed by microwave irradiation. A screening study indicated that DES-N dissolved PET more rapidly by ∼100 s. Therefore, microwave irradiation times were set as 100, 120, and 140 s for DES-N. It is known that DES shows very high electric conductivity, which is much higher than water, and thus is especially suitable for the microwave heating process. 19 High temperature is easily reached within a very short period of time. Therefore, the flask system was heated for 30 s, cooled for 10 s, and again heated for 10 s to reach a specified time. After irradiation, the flask was completely cooled to room temperature (RT) and the remaining fabric was removed from the flask. Then 20 mL of deionized water was added into the flask to precipitate the dissolved PET portion of the fabric, followed by filtering to collect the dissolved PET. The remaining fabric and the filter paper containing dissolved PET were dried in the oven for 10 min at 100 °C and placed under standard conditions (20 ± 1 °C, 65 ± 2% relative humidity (RH)) for at least 24 h before weighing for identification.
Evaluation of Weight Loss
To identify the composition of PET in the blend fabric, the weight of the fabric residue was measured after the treatment. Since the PET portion in the blend fabric was expected to dissolve in DES, the residual fabric should be cotton and the precipitate from distilled water was PET. Therefore, weight loss of the fabric was calculated using Eq. 1.
W1 was the initial fabric weight and W2 was the weight after treatment. PET composition identification experiments were carried out in duplicate. Weight loss data were then presented with standard deviations.
Characterization
FTIR spectroscopy (Bruker, Vertex 70, Germany) was used to characterize the blend fabric before and after DES treatment. The spectrometer was equipped with an attenuated total reflectance (ATR) attachment at a spectral region of 4000-600 cm−1 with 64 scans and 2 cm−1 resolution. Ratios of absorption peaks at 3331 cm−1 for the cellulose hydroxyl peak and at 1713 cm−1 for the ester carbonyl peak were used to evaluate the composition of PET from the blend fabric. Peak ratios were obtained using Eqs. 2 and 3.
Sp, Ep, and Cp, were peak height values of the specimen (blend fabric), 100% PET, and 100% cotton at the specific peak, respectively. Therefore, peak heights of 100% cotton and 100% PET at 3331 cm−1 were regarded as 1 and 0, whereas peak heights of 100% PET and 100% cotton at 1713 cm−1 were considered as 1 and 0. The ratios of peak height for the blended specimens were calculated based on these reference values.
Since the fiber morphologies of cotton and PET were quite different, SEM (COXEM, CX-100S, Korea) could be effectively used to study surface characteristics of the blend fabrics after DES treatment. Thermal characteristics of the blend fabrics were evaluated by TGA (Mettler, USA) and DSC (Perkin Elmer, USA). TGA was measured in the 25 °C to 700 °C range at 10 °C/min rate under nitrogen atmosphere. The temperature was increased from 0 °C to 265 °C with a 10 °C/min rate for DSC analyses, cooled very rapidly to 0 °C, and again increased to 265 °C with a 10 °C/min rate. The second heating curve was used to estimate the composition of PET within the blend fabric. XRD could be another way to characterize the PET/cotton blend fabric after dissolution treatment, since each fiber has a specific crystalline structure. The blend fabric before and after treatment was evaluated by XRD (D2 Phaser, Bruker, Germany), using a Co-Kα (λ = 0.1524 nm) source at 30 kV and a 10 mA operating range. The sample was scanned at a speed of 2°/min with 0.02 increment for a 2Θ range of 40° to 10°. To obtain the ratio between cotton and PET, the height of the base line peak at 39° was subtracted from the crystalline peak of each fiber (i.e., cotton at 15.2° for the 101 plane and PET at 25.8° for the 100 plane).
Precipitated Residue Analysis
To investigate mechanism of PET dissolution, an Ultimate 3000 HPLC (Thermo Dionex, USA) was used with a VDSpher C-18 column (VDSoptilab, Germany, 4.6 × 250 mm, 5 μm) at 1 mL/min flow rate and 10 μL injection volume at 30 °C. Two potential decomposition compounds, such as bis(2-hydroxyethyl terephthalate) (BHET) and terephthalic acid (TPA) were used as references in HPLC analysis. BHET was dissolved in 100% methanol, whereas TPA was dissolved by 0.2 N NaOH in methanol. Both references were extracted by one hour of ultrasonic treatment. The ratio between specimen and extracted solvent was 1:100. Trifluoroacetic acid (0.3% in deionized water) and acetonitrile were used as eluents. A UV detector at 241 nm (190-400 nm diode array detector scanning) was used and the result was analyzed by Chromeleon 6.8 software (Thermo Fisher Scientific, USA).
Hydrophilicity and Dye Affinity
Affinity toward water will depend upon composition of cotton and PET in the blend fabric. The specimen could thus show difference in contact angle and water of imbibition (WI) after dissolution treatment. A contact angle analyzer (DSA100, Germany) was used to evaluate the contact angle of the fabric with 3 μL of deionized water. For WI, the specimen was completely dried at 85 °C for 1 h and weighed (Wd). It was then impregnated in deionized water for three hours, followed by centrifuging at 1800 rpm for 30 min, and its weight was again measured (Ww). The WI was calculated by Eq. 4.
PET is commonly dyed by disperse dyes. Therefore, removal of PET from the blend fabric could significantly influence its dyeing characteristics with disperse dyes. The fabric was thus dyed with a disperse dye (Papilion Orange F-GR, KISCO, Korea) in an infrared dyeing machine (DL-6000, Daelim Starlet, Korea) at a 40 rpm/min rotation speed. The dyeing bath was composed of dye (3% owf) at a 50:1 liquor ratio (LR). Dyeing was started at 30 °C, raised to 120 °C at a 2 °C/min rate, maintained at 120 °C for 10 min, and then decreased to 60 °C for 15 min. Surface reflectance of the treated and dyed fabric was measured by a Color Eye CM-512M3 (X-Rite, USA). K/S values were calculated by the Kubelka-Munk equation (Eq. 5).
R is the reflectance of the dyed materials at λmax= 470 nm.
Results and Discussion
Potential Mechanism of PET Degradation
Dissolution of PET from the blend fabric was expected to involve glycolysis because of the presence of EG within DES. DES also contained 5% NaOH, implicating a potential hydrolysis reaction. In addition, the ChCl in DES could facilitate the dissolution process during glycolysis12,19 or alkaline hydrolysis 23 as a catalyst. It was also shown that DES was a highly active catalyst for the fast and mild glycolysis of PET. 1 Therefore, the dissolution of PET from the blend fabric in the present system could occur very rapidly due to the combined effects of glycolysis, alkaline hydrolysis, and catalytic activities of ChCl alone and ChCl with EG as DES. Use of microwave irradiation to heat DES could further elevate the reaction rate because of the high electronic conductivity of DES. 19 As shown in Scheme 1, PET could be decomposed into various products such as BHET, mono-2-hydroxyethyl terephthalic acid (MHET), TPA, terephthalic acid, disodium salt (TPA-Na2), and other oligomers, along with EG during DES treatment.
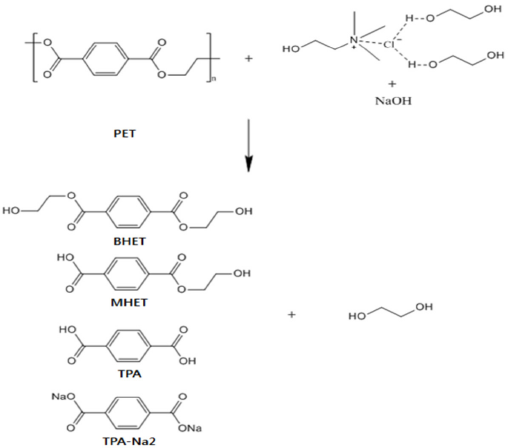
Potential degradation compounds by treatment of PET using DES under microwave irradiation.
Instrumental Characterization
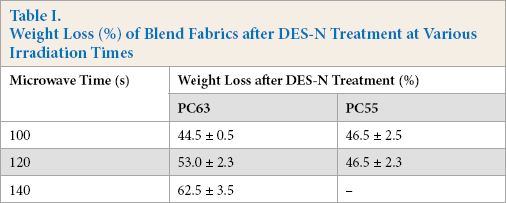
Since the PET portion from the blend fabric was dissolved by DES, the weight of the specimen substantially decreased after the treatment. Weight losses of the blend fabrics are shown in Table I after DES-N treatment. Weight losses expected for PC63 and PC55 were about 65% and 50%, respectively. The results demonstrated that PC63 and PC55 specimens required around 140 s and 100 s, respectively, to reach the targeted weight loss. While considering the complex nature of the blend fabrics, these weight loss data must be verified by other methods, such as instrumental analyses.
Weight Loss (%) of Blend Fabrics after DES-N Treatment at Various Irradiation Times
To characterize the DES-N solvent, its electric conductivity was measured. Electric conductivity of DES-N was 911 mS/m at RT. This indicated that microwave radiation could heat DES-N much more rapidly than deionized water, showing 0 mS/m electric conductivity. Nevertheless, the water was still boiling within 60 s of microwave irradiation at a 50:1 LR. The rate determining factor for PET dissolution thus could be something other than electric conductivity of the dissolution medium. It is known that at molar heat capacities, the Cp of DES and water at 303.15 K were 190.8 ± 0.4 J mol−1K−1 and 72.82 ± 0.2 J mol−1K−1, respectively. 24 The higher Cp value of DES was due to its greater molar mass (87.92 g/mol) than that of water (18.015 g/mol). The Cp value of the binary mixture of DES was expected to decrease with an increasing amount of water. Since Cp is the quantity of heat needed to raise the temperature of one mole of a substance by one degree, the greater Cp of DES implies that it absorbs more energy than water at a given irradiation time. This could consequently result in greater dissolution of PET within the blend fabric. Therefore, DES-N could readily dissolve PET more rapidly and effectively than deionized water, requiring less time to achieve the targeted weight loss.
Since PET and cotton fiber within the blend show characteristic IR peaks, removal of PET by DES treatment could also be identified by differences in FTIR absorption peaks. As explained previously, the OH stretching peak at 3331 cm−1 for cotton and the ester carbonyl stretching peak at 1713 cm−1 for PET were selected as reference peaks. As shown in Figs. 1 and 2, the height of the ester carbonyl peak gradually decreased with increased irradiation time, regardless of the specimen type. The peak ratio at 3331 cm−1 should be close to 1 for complete removal of PET with increased irradiation time, whereas the ratio at 1713 cm−1 should be close to 0 under the same condition. Results in Fig. 2 indicated, however, that the peak ratio at 1713 cm−1 after 140s was almost 0 after the DES-N treatment, whereas the ratio at 3331 cm−1 increased to 0.7−0.8, but it never reached 1. This implied that at 140s, PET was almost removed from the blend, but the OH peaks on the fabric surface were not high enough to correspond to that of pristine cotton.
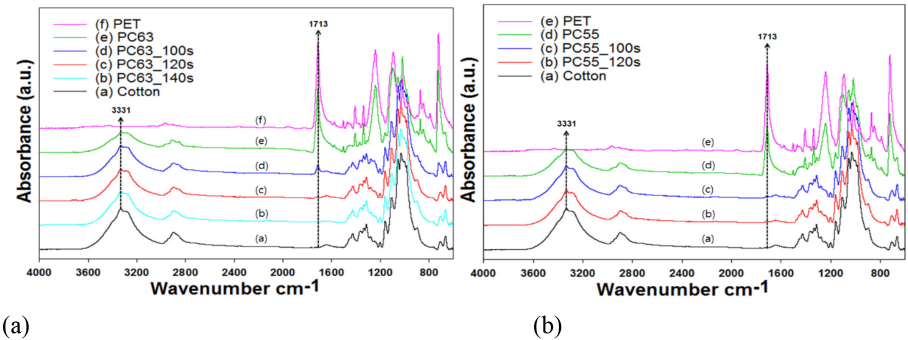
FTIR spectra of DES-N-treated (a) PC63 and (b) PC55 blend fabrics.
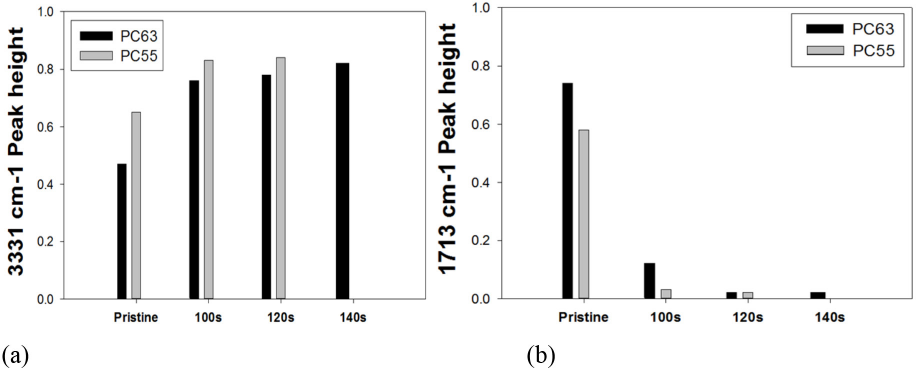
Variation of reference absorption peaks of PC63 and PC55 blend fabrics treated by DES-N at different microwave irradiation times. (a) 3331 cm−1 and (b) 1713 cm−1.
TGA analysis in Figs. 3 and 4 indicated that the maximum decomposition peak (Tp) of PET occurred at 431 °C, whereas that of 100% cotton was 360 °C. TGA and its 1st derivative curves of the PC63 blend fabrics in Fig. 3 gradually resembled to that of 100% cotton with increased irradiation time. In the case of PC63, the Tp of PET at 430 °C was still visible at 100 s of irradiation, whereas in PC55 in Fig. 4, the same Tp totally disappeared at 100s. This was probably because of disparity in the woven structure of two blend fabrics (i.e., PC55 was less dense than PC63).
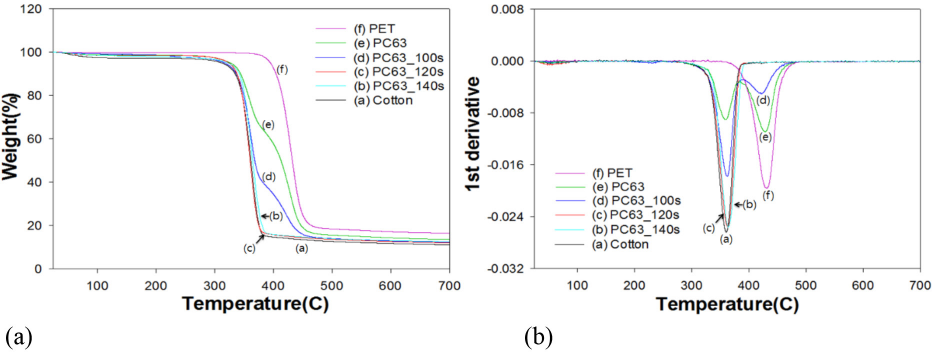
TGA graphs of PC63 blends treated by DES-N. (a) TGA curve and (b) 1st derivative.
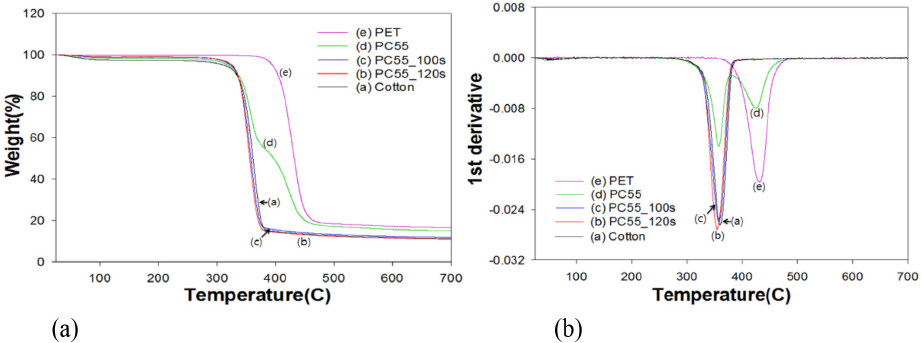
TGA graphs of PC55 blends treated by DES-N. (a) TGA curve and (b) 1st derivative.
100% cotton fabric was also treated by DES-N to observe its effect on pure cotton. 100% cotton fabric lost weight (∼4%) by DES-N treatment after irradiation for 140s, probably due to the presence of alkali. The irradiation time was also increased to 160s with DES-N. Visual observation indicated that the cotton specimen treated by DES-N still maintained its original structure. Nevertheless, too long irradiation time should be avoided during the composition identification process to minimize the effect of DES-N on the cotton portion within the blend fabric.
Fig. 5 showed DSC curves of PC63 and PC55 specimens treated with DES-N at 100 s irradiation. Results indicated that the exothermic melting peaks at ∼60 °C were detected for both specimens, but the peak of PC63 was greater than that of PC55 due to its greater PET composition. Melting enthalpy values of 100% PET, PC63, and PC55 were 47.19 J/g, 31.67 J/g, and 21.38 J/g, respectively, which were directly proportional to the level of PET within the blends. In addition, after 100 s of DES-N treatment, the enthalpy of the exothermic melting peak decreased considerably (i.e., 25.49 J/g for PC63 and 14.29 J/g for PC55). Decrease in the enthalpy value due to the DES-N treatment was obviously greater with the PC55 specimen than that of PC63. This again revealed that PET removal by DES-N was easier for PC55 probably due to its loose structure. The enthalpy decrease measured by DSC was somewhat less dramatic than those obtained by the ATR method in FTIR analysis, because the removal of PET could occur from the surface to the interior. The ATR method in FTIR mainly evaluated the surface characterization of the materials.
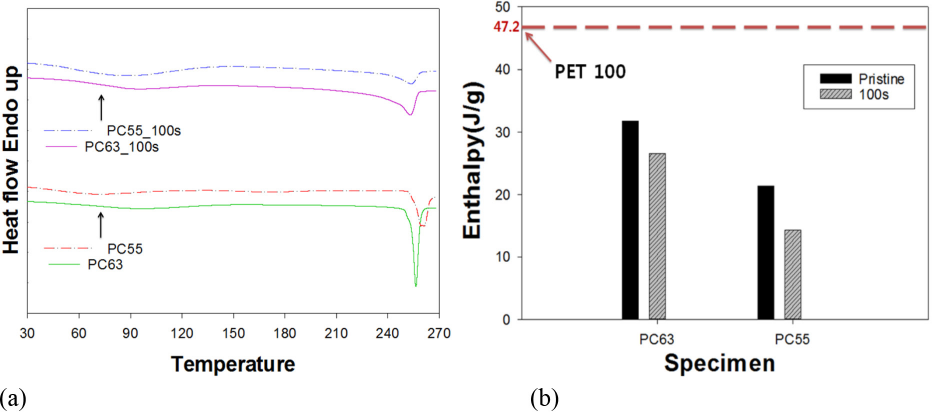
DSC curves (a) of PC63 and PC55 fabrics treated by DES-N for 100 s of microwave irradiation and (b) their enthalpy values.
XRD crystalline peaks of PET and cotton were quite different from each other, This could also be used to characterize the specimens treated by DES. The XRD spectra of the treated specimens would be similar to that of 100% cotton. Crystalline peaks of 100% PET were observed at 2Θ = 17.9°, 22.9°, and 25.8°, which were almost identical to the 2Θ values obtained in the literature. 25 On the other hand, cotton showed the (002) peak at 2Θ = 23.7°, which was very close to the 2Θ = 22.9 peak of PET. Therefore, the reference values used for composition analyses were the 2Θ = 25.8° (100) peak of PET, and the 2Θ = 15.2° for the (101) peak of cotton. As shown in Figs. 6 and 7, the PET reference peak reached almost 0 with increased microwave irradiation time. Results also attested that the crystalline peaks for cotton tended to decrease with increased irradiation time, confirming slight crystalline degradation of cotton by the DES treatment.
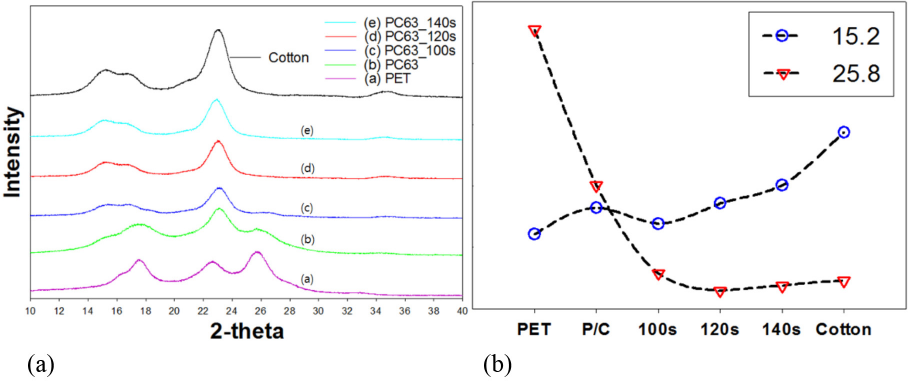
(a) XRD spectra of PC63 fabrics treated by DES-N and (b) ratio of 2Θ = 15.2° and 25.8° peaks.
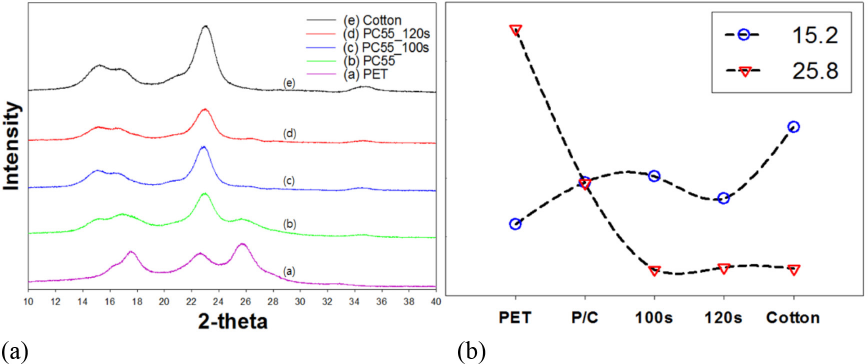
(a) XRD spectra of PC55 fabrics treated by DES-N and (b) ratio of 2Θ = 15.2° and 25.8° peaks.
Surface Morphology Analyses
As shown in Fig. 8a, PET fibers appeared with a smooth surface in untreated PET63 fabric. After irradiation for 100 s, some PET fibers were fractionized as shown in Fig. 8b. This demonstrated that this irradiation time was not sufficient to completely remove PET fibers from the PC63 specimen. However, PET fibers almost disappeared in the micrographs of Figs. 8c and d after 120 s or longer irradiation time. On the other hand, SEM analyses in Fig. 9 showed that the PC55 blend was not a real blend fabric, but rather a mixture fabric that was woven with cotton in the weft direction and PET in the warp direction. PET in such a mixture fabric would be more readily dissolved by DES because the PET fiber was only positioned in the warp yarn. Some PET lumps were detected as shown in Fig. 9b due to the fiber melting by microwave irradiation, but cotton fiber was only observed in Fig. 9c after 120 s of irradiation.

SEM micrographs of PC63 fabrics treated by DES-N. (a) PC63, (b) PC63 for 100 s, (c) PC63 for 120 s, and (d) PC63 for 140 s.

SEM micrographs of PC55 treated by DES-N. (a) PC55, (b) PC55 for 100 s, and (c) PC55 for 120 s.
Affinity toward Dye and Moisture
Since the PET portion in the blend fabric only had an affinity toward disperse dye, the PC63 and PC55 specimens could be examined by the dye affinity method. 100% PET fabric and untreated blend fabrics showed a strong orange color, whereas the color of the treated fabrics diminished with increased irradiation time (not shown). As shown in Fig. 10, K/S values of the specimens treated by DES-N decreased with increased irradiation time, regardless of fabric types.
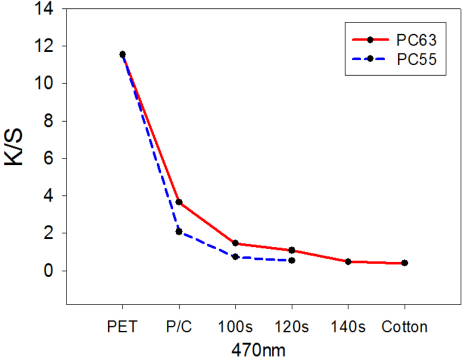
K/S values of dyed PC63 and PC55 fabrics treated by DES-N at λmax= 470 nm.
Similarly, cotton and PET fibers have opposite characteristics in terms of moisture sorption (i.e., highly hydrophilic versus hydrophobic). Since DES treatment removed the PET portion, moisture sorption would increase with irradiation time. Contact angle measurement was carried out only on the PC63 specimen, because of immediate wetting of PC55 due to the nature of the mixture fabric. As shown in Figs. 11a and b, the surface of the untreated PC63 fabric was highly hydrophobic even after 3 min of wetting. On the other hand, PC63 treated for 100 s showed immediate wetting after dropping water on it as shown in Fig. 11c. The WI value was also measured after water imbibition and centrifugation as shown in Fig. 12. As expected, WI values continuously increased in both specimens with increased irradiation time during DES treatment due to the gradual removal of the PET portion.

Contact angle evaluation of PC63 treated by DES-N. (a) PC63, (b) PC63 3 min after water dropping, (c) PC63 treated for 100 s immediately after water dropping.
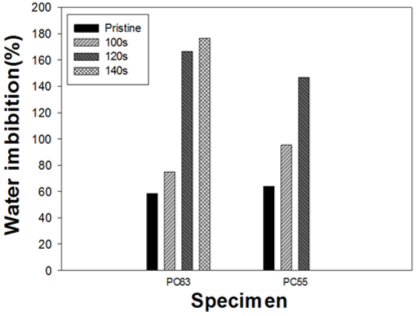
WI of PC63 and PC55 treated by DES-N.
Residue Analyses by HPLC
As shown in Fig. 13a, HPLC evaluation of the filtered PET residue revealed the amount of TPA produced during the DES treatment of blend fabric. The amount of TPA was 52,880 mg/kg (5.28%), while BHET was 2779 mg/kg (0.28%), indicating a much greater level of TPA than BHET in the PET residue. The small production of BHET in the residue suggested that the mechanism of PET degradation was more complex than a conventional glycolysis reaction, which produced mainly BHET. The simultaneous influence of glycolysis and alkaline hydrolysis could attribute to the complex mechanism of PET degradation in DES treatment. Moreover, amounts of TPA or BHET in the residue were much less than we expected, suggesting the production of oligomers during PET degradation, rather than smaller molecules such as monomer (TPA) or trimer (BHET). In addition, it was known that the presence of quaternary ammonium compounds such as ChCl could act as a catalyst in the glycolysis reaction of PET. 23 Therefore, control of the decomposed residues could be an important task for PET recycling and this study is currently undertaken.
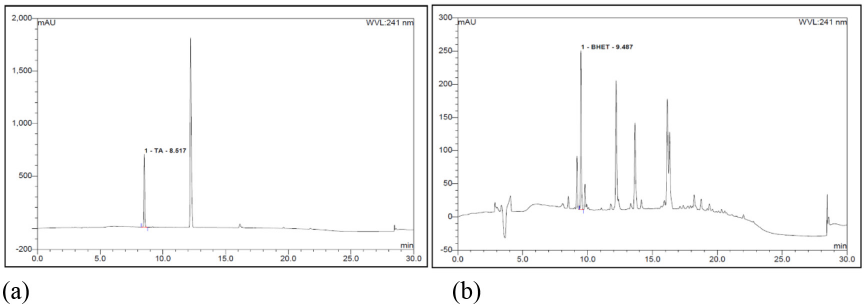
HPLC spectra of the PC63 blend fabric treated by DES-N. (a) TPA and (b) BHET.
Composition Identification for Commercial Specimen
To examine the feasibility of the newly developed identification method on a commercial specimen, a pink-colored, knit fabric, PC45 (PET/cotton of 45/55) was selected. With DES-N, weight losses at 100 s and 120 s were 45% and 48%, respectively. Therefore, the loose knit fabric required less time to dissolve PET completely from the blend fabric. This result indicated that the microwave irradiation time must be optimized through the screening study to obtain a correct result. As shown in Fig. 14, the FTIR spectra of the DES-N treated PC45 fabrics were very similar to those of the PC63 and PC55 fabrics. The Tp peak in the TGA indicating the presence of PET completely disappeared at 120 s in the 1st derivative curve in Fig. 15b, just like other blend fabrics shown in Figs. 3 and 4. The SEM micrographs in Fig. 16 showed that PC45 treated by DES-N degraded from the end of the fiber and the PET fibers mostly disappeared after 120 s of irradiation.
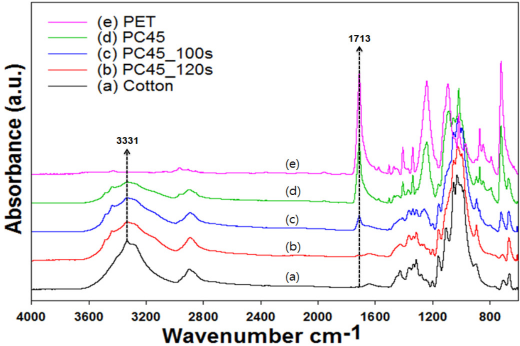
FTIR spectra of PC45 treated by DES-N.
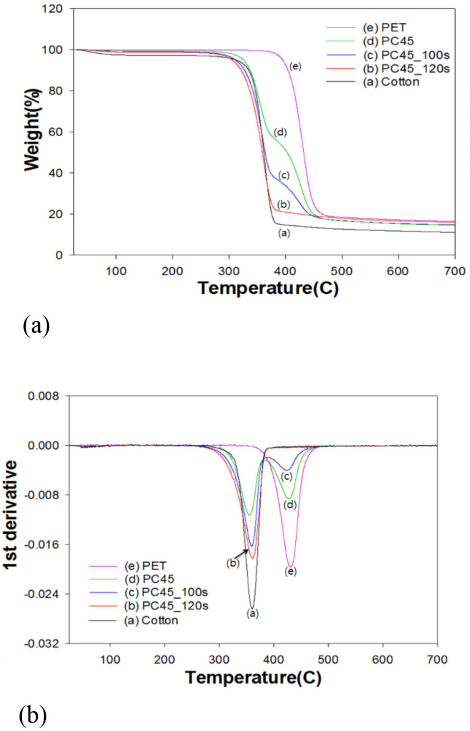
TGA of PC45 fabrics treated by DES-N. (a) TGA curve and (b) 1st derivative.

SEM micrographs of PC45 fabrics treated by DES-N. (a) PC45, (b) DES-N for 100 s., and (c) DES-N for 120 s.
Conclusions
A rapid and sustainable method was developed to identify the composition of PET in PET/cotton blend fabric, which is one of the most common fabrics in the clothing market. This technique used DES-N made of EG and ChCl in the presence of a small amount of NaOH under microwave irradiation. Results indicated that the PET composition in the blend fabrics was identifed within 120∼140 s. This technique was much faster and more eco-friendly compared to conventional identification methods involving strong acid or fluorinated organic solvents. Two PET/cotton blend fabrics, PC63 and PC55, were selected and the composition of PET was confirmed by weight loss. Furthermore, the blend fabrics treated by DES-N were characterized by various instrumental methods such as FTIR, TGA, DSC, XRD, and SEM to further substantiate the removal of PET. Affinity towards a disperse dye and moisture also confirmed the removal of PET. PET residue analyzed by HPLC revealed that the amounts of TPA and BHET were rather small, but more oligomer residues were produced. This was probably due to the presence of glycolysis, alkaline hydrolysis, and the catalytic action of ChCl alone and DES. Feasibility of the method was tested on a commercial fabric and the result indicated that the technique was readily applicable to the identification of PET in the PET/cotton blend fabric as a sustainable method.