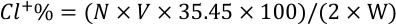Abstract
A novel, water-soluble N-halamine precursor based on s-triazine (2-(aminoethanesulfonic sodium salt)-4-(4-imino-2,2,6,6-tetramethylpiperidine)-6-monochloro-1,3,5-triazine, ATDT) was synthesized and characterized by FT-IR and 1 H-NMR. The cotton fabrics were functionalized with ATDT through the traditional pad-dry-cure method, and optimal conditions were successfully obtained by measuring the active chlorine content. The antibacterial activity of ATDT-coated cotton fabrics was investigated against Staphylococcus aureus and Escherichia coli O157:H7. As a result, 100% of bacteria could be inactivated within a brief contact time. Cell viability was also determined and the results showed that ATDT-modified cotton fabrics had good cytocompatibility to mammalian cells. In addition, the preparation process had little effect on the tensile strength and whiteness of cotton fabrics, although the air permeability showed a slight decrease. The chlorinated ATDT-coated fabrics showed excellent stability against storage and washing.
Keywords
Introduction
Bacterial-induced diseases occur all over the world, seriously affecting people's health and lives.1–4 Bacteria can also cause discoloration, odor, and other issues on textiles. To reduce the bacterial threat to people and textiles, the development of antimicrobial textiles has attracted a great deal of attention.5,6
Many kinds of antimicrobial agents are used on textiles, such as metal ions,7,8 chitosan,9–11 quaternary ammonium salts,12–15 N-halamines,16–19 and other antimicrobial agents. Among these various antimicrobial agents, N-halamines have attracted much attention because of their stability, broad-spectrum activity, regenerability, and efficacy for rapidly inactivating bacteria. N-halamines contain one or more nitrogen-chlorine (N-Cl) bonds, which can be obtained from amine, amide, and imide compounds by exposing them to dilute sodium hypochlorite. N-H bonds are then converted to N-Cl bonds, providing antimicrobial and antiviral properties due to the oxidative chlorine. 20 After eliminating the microbiological threat, the N-Cl bonds are converted back to N-H bonds, which can then be recharged by a simple chlorination process. Therefore, N-halamines have enormous potential for use in water treatment, medical instrument, biomaterial, and textile applications.21,22
Although N-halamines have been known since the 1930s, they were only used to prepare antibacterial textiles in recent years. In the late 1990s, Sun and his coworkers began to use N-halamines as an antibacterial agent for cotton fabrics. In their early works, 1,3-dihydroxymethyl-5,5-di-methylhydantoin (DMDMH) was used as an N-halamine precursor and bound onto cotton fabrics. The chlorinated samples presented good biocidal efcacy against a wide range of microorganisms and washing stability. 23 However, its application was greatly limited due to formaldehyde release. Therefore, many research groups focused on the development of non-formaldehyde and durable N-halamine compounds. Various N-halamines were synthesized containing various reactive groups such as unsaturated double bonds,24,25 siloxane, 26 and epoxy groups.27,28 Sun et synthesized a cyclic amine monomer, 3-allyl-5,5-di-methylhydantoin (ADMH) that can be grafted onto various textile materials to prepare durable and regenerable antibacterial textiles in a continuous finishing process with the assistance of an initiator. 29 Worley et al. prepared a vinyl N-halamine monomer (hydantoin acrylamide, HA) that can be copolymerized with silane-, epoxide-, and hydroxyl group-containing monomers to prepare reactive N-halamine copolymers. The resulting coated cotton fabrics exhibited good antibacterial properties as well as washing and UV light stability.30,31 However, some N-halamine precursors have poor water solubility, and some of them need high temperature and initiator to modify textiles, which might restrict their application and affect the physical-mechanical properties of cotton fabrics. Therefore, developing water-soluble N-halamine precursors with high reactivity to cotton fabrics is an efficient way to solve this problem. 32
In this study, a novel water-soluble N-halamine precursor based on s-triazine (2-(aminoethanesulfonic sodium salt)-4-(4-imino-2,2,6,6-tetramethylpiperidine)-6-monochloro-1,3,5-triazine, ATDT) was synthesized and characterized by Fourier transform infrared (FT-IR) and proton nuclear magnetic resonance (lH-NMR) spectroscopy. Optimal conditions were successfully obtained by measuring the active chlorine content. Chlorine loading can reach 0.29%, which can provide efficient antibacterial properties. The ATDT-coated fabrics were characterized using FT-IR, scanning electron microscopy (SEM), differential scanning calorimetry (DSC), and thermogravimetric analysis (TGA). The antibacterial activity of the ATDT-coated cotton fabrics was measured against Staphylococcus aureus and Escherichia coli O157:H7. To investigate cytotoxicity, the biocompatibility of ATDT-coated cotton swatches was evaluated by cell viability testing. The storage stability, washing stability, tensile strength, whiteness, and air permeability of ATDT-coated cotton fabrics were also investigated.
Experimental
Materials
Bleached cotton fabric was obtained from the Qingdao Fenghuang Dyeing & Printing Co. Ltd., China. Cyanuric chloride and sodium thiosulfate (Na2S2O3) were provided by J & K Scientific Co. Ltd., China. 4-amino-2,2,6,6-tetramentylniperidine (TEMP) was purchased from Shanghai Macklin Biochemical Co. Ltd., China. Taurine was purchased from Shanghai Titan Scientific Co. Ltd., China. Potassium iodide (KI), sodium hydroxide (NaOH), and sodium carbonate (Na2CO3) were purchased from Sinopharm Chemical Reagent Co. Ltd., China. Starch was provided by Ruijinte Chemical Co. Ltd., China. Acetone was provided by Sanhe Chemical Reagent Co. Ltd., China. Phosphate buffer solution (PBS, 0.05 M, pH = 7.4) was provided by Wuhan Boster Bioengineering Co. Ltd., China. Trypsin-EDTA was obtained from Gibco, USA. All reagents and medicines were used without further purification. NIH 3T3 cells were supplied by Shanghai Zhong Qiao Xin Zhou Biotechnology Co. Ltd., China.
Synthesis of ATDT N-Halamine Precursor
The synthetic route of ATDT preparation is shown in Fig. 1. Cyanuric chloride (0.22 mol) and acetone (240 mL) were added into a 500-mL four-necked flask equipped with a mechanical stirrer, a thermometer, and a constant pressure dropping funnel. The solution was cooled to 0–5 °C and stirred for 20 min to ensure complete dissolution of cyanuric chloride. A solution of 2-aminoethanesulfonic sodium salts prepared from 0.2 mol taurine and 0.2 mol NaOH in 60 mL water was added dropwise over 1 h at 0–5 °C with stirring. After addition was complete, the mixture was maintained at the same temperature for another 5 h and 10 wt% sodium carbonate was used to control pH at 5–6. The mixture was then filtered and further washed with acetone several times to remove the residual reactants. After drying at 45 °C to a constant weight, the intermediate product ASDT was obtained in 77.2% yield.
ATDT synthesis.
1 H-NMR (400 MHz, DMSO-d6) δ (ppm): 3.18-3.24 (t, 2H), 3.80–3.86 (t, 2H). FT-IR (cm–1): 3337, 2926, 1582, 1556, 1503, 1401, 1303, 1216, 1185, 1172, 1142, 1049, 977, 854, 793.
ASDT (0.1 mol) and water (240 mL) were added to a 500-mL four-necked glass flask equipped with a mechanical stirrer, a thermometer, and a constant pressure dropping funnel. After stirring at 40–50 °C for 20 min for complete dissolution of ASDT, the mixture of TEMP in 50 mL water was added dropwise over 1 h. The mixture was then stirred at 40–50 °C for another 5 h. Afer filtering and drying at 45 °C to a constant weight, a white product ATDT was obtained in 72.7% yield. 1 H-NMR (400 MHz, D2O-d2) δ (ppm): 0.90-1.31 (m, 14H), 1.58–1.76(m, 2H), 2.60–2.70 (m, 2H), 3.43–3.55 (m, 2H), 4.06–4.27 (m, 1H). FT-IR (cm–1): 3246, 3034, 2846, 1607, 1582, 1532, 1354, 1315, 1188, 1156, 1038, 983, 889, 802, 766.
Finishing and Chlorination
ATDT was dissolved in 100 mL distilled water to prepare a 2∼10 wt% finishing solution, and 1∼5 wt% NaOH was added. Then, the cotton fabrics were immersed in the coating solutions for 15 min and padded with two dips and two nips to achieve a wet pick-up of 100%. After drying at 90 °C for 5 min, the coated fabrics were cured at 110–150 °C for 1–5 min. Finally, the coated cotton fabrics were washed thoroughly with distilled water and dried at 45 °C for 1 h.
To activate the N-halamine functionality, the ATDT-treated cotton fabrics were immersed in a 10 wt%, pH 7 aqueous bleaching solution at room temperature (RT) for 1 h. Then the samples were washed thoroughly with distilled water and dried at 45 °C for 1 h. The oxidative chlorine content on the cotton fabrics was determined by the iodometric/thiosulfate titration method and calculated by Eq. 1. 33
Cl+% is the weight percent of oxidative chlorine on the samples, N and V are the normality (equiv/L) and volume (L) of the titrant sodium thiosulfate, respectively, and W is the weight (g) of the ATDT-coated cotton fabrics.
Measurement and Characterization
1H-NMR spectra of ASDT and ATDT were recorded on a Bruker AVANCE III HD 400MHz spectrometer (Bruker, Germany) using D2O and DMSO-d6 as solvents.
The FT-IR spectra of ASDT, ATDT, and ATDT-coated cotton fabrics before and after chlorination were measured using a Nicolet iS 50 FT-IR spectrometer (Thermo Fisher Scientific, USA) ranging from 400 cm−1 to 4000 cm−1 with a resolution of 4 cm−1 and an average of 32 scans.
The surface morphology of control cotton and ATDT-coated cotton fabrics before and after chlorination were characterized by a Tescan Vega3 scanning electron microscope (SEM) (Tescan, Czechoslovakia).
TGA was carried out on a thermogravimeter (TG209F3, Netzsch Instruments Co. Ltd, Germany) from ambient temperature to 800 °C at a heating rate of 10 °C/min under nitrogen. Differential scanning calorimetry (DSC) was measured by using Tadsc 250 (TA, USA) at a heating rate of 5 °C/min under a nitrogen atmosphere from ambient temperature to 300 °C
The antimicrobial activity of ATDT-coated cotton fabrics was investigated against S. aureus and E. coli O157:H7 according to AATCC TM100-2012. 27 Bacterial suspension (25 μL) of ∼107 colony forming units (CFU)/mL was added to the middle of two pieces of 2.54 × 2.54 cm2 cotton swatches by a sterile weight to allow sufficient contact between the bacteria and the cotton fabrics. After 5, 10, and 30 min of contact, the samples were centrifuged with 5.0 mL of sterile sodium thiosulfate (9.5 mL of PBS and 0.5 mL of 0.05 M of sodium thiosulfate) to remove any oxidative chlorine. Then, 100 μL solution from the centrifugal tube was inoculated on nutritional agar plate. The plates were incubated at 37 °C for 24 h. The antimicrobial activity was evaluated by the colony counting method.
Cytotoxicity of ATDT-coated cotton fabrics was determined using 3-(4,5-dimethyl-2-thiazolyl)-2,5-diphenyl-2-H-tetra-zoliumbromide (MTT) assays. Before this test, all samples were cut into round meshes and UV-sterilized for 30 min on 24-well tissue culture plates. Then NIH 3T3 cells were seeded in 24-well tissue culture plates with a seeding density of 2 × 104 cells/well. After culturing for 24 h and 48 h at 37 °C in a humidified atmosphere of 5% CO2, MTT assays were performed. The cell viability was characterized as the percentage with respect to the control.
For stability testing, the antibacterial cotton fabrics were stored at RT for 30 days and the oxidative chlorine contents were measured every 5 days. The washing stability test was carried out by monitoring the oxidative chlorine content of antibacterial cotton fabrics after washing and rechlorination was performed according to the literature. 34 In this test, the chlorinated samples were immersed in conical beakers containing 0.5 wt% AATCC standard powdered detergent (without optical brighteners) aqueous solution. The beaker was fixed in a 25 °C shaking bath at the speed of 120 rpm for 45 min, which was defined as one washing cycle. The oxidative chlorine content on the cotton fabrics after washing was determined by the iodometric/thiosulfate titration before and after rechlorination. Each sample was tested three times and the average value was obtained.
The air permeability of cotton control, cotton-ATDT, and cotton-ATDT-Cl was measured using a YG461E-III fully automatic permeability instrument (Ningbo Textile Instrument Factory) at a pressure of 100 Pa, according to GB/T 5453-1997. 35 Each sample was tested 10 times and the average value was obtained.
The whiteness of cotton control, cotton-ATDT, and cotton-ATDT-Cl were measured using a WSD-3U fluorescence whiteness meter (Beijing Kangguang Optical Instruments Co. Ltd, China) according to GB/T 17644-2008. 36
The tensile strength of cotton control, cotton-ATDT, and cotton-ATDT-Cl were measured according to GB/T 3923.1-1997. 37 The size of testing samples was 300 × 50 mm2. Each sample was tested 5 times and the average value was obtained.
Results and Discussion
Structural Characterization of ATDT
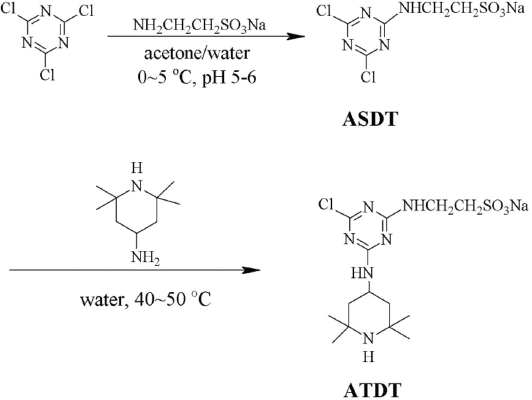
The 1 H-NMR spectra of TEMP, ASDT, and ATDT are shown in Fig. 2a. For the 1 H-NMR spectrum of TEMP, the signals at 1.07–1.23, 0.84–0.94, and 1.71–1.81 ppm were attributed to the protons of methyl (a) and methylene (b and c) groups, respectively. The signal at 3.01–3.13 ppm corresponded to the methine proton (d) in TEMP. For the 1 H-NMR spectrum of ASDT, the signals at 3.18–3.24 and 3.80–3.86 ppm were attributed to two sets of methylene protons (f and e). For the 1 H-NMR spectrum of ATDT, the chemical shift appeared at 0.90–1.31 (-CH3, C-CH2-C), 1.58–1.76 (C-CH2-C), 2.60–2.70 (S-CH2-C), 3.43–3.55 (C-CH2-NH-) and 4.06-4.27 (-NH-CH-), respectively. Compared with TEMP, the higher chemical shifts of protons at (b) and (d) in ATDT were due to the strong electron absorption effect of the triazine ring.
(a) 1H-NMR spectra of TEMP, ASDT, and ATDT. (b) FT-IR spectra of ATDT, ASDT, TEMP, taurine, and cyanuric chloride.
Synthesis of ASDT and ATDT was also confirmed by FT-IR (Fig. 2b). The absorption peak at 3337 cm–1 corresponded to the -NH- group, indicating a successful reaction between cyanuric chloride and taurine. 38 Absorption peaks at 1172 cm–1 and 1046 cm–1 were attributed to the asymmetric stretching vibration and symmetric stretching vibration of -SO3Na. For the FT-IR spectrum of ATDT, the peak at 3246 cm–1 was associated with the skeleton vibration absorption peak of the piperidine amine ring. The vibration absorption peaks of -CH3 and -CH2- on the piperidine amine ring appeared between 2846 cm–1 and 3034 cm–1. 39 In addition, the absorption peaks of the triazine and piperidine amine rings were between 1354 cm–1 and 1582 cm–1, and the peaks at 1188 cm–1, 1038 cm–1, and 802 cm–1 belonged to the vibrations of -SO3Na and C-Cl groups. 40
Preparation of Antibacterial Cotton Fabrics
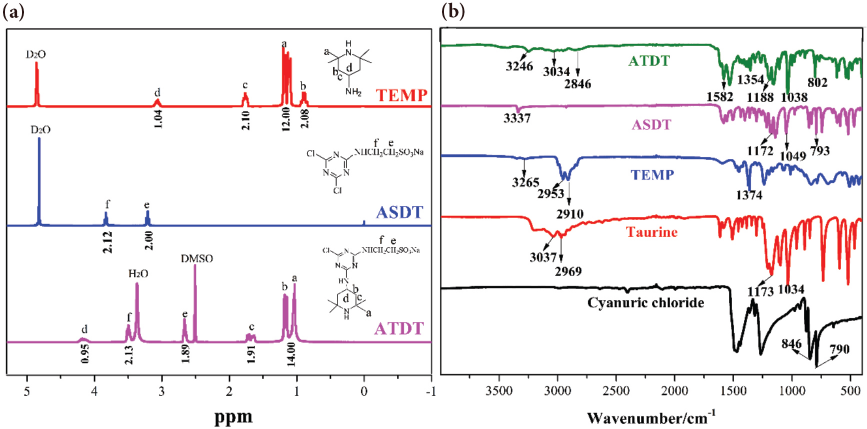
The effect of ATDT concentration, curing time, curing temperature, and NaOH on the oxidative chlorine content of ATDT-coated cotton fabrics were investigated and the results were presented in Fig. 3. The increase in the concentration of the solution led to a sustained increase in the oxidative chlorine content of ATDT-coated fabrics. The oxidative chlorine content reached a maximum when the solution concentration reached 8%. The oxidative chlorine content increased with the extension of the curing time and the chlorine loading reached 0.33% at 5 min curing time. Other factors including curing temperature and the alkali dosage were also studied. The oxidative chlorine content reached the maximum when the curing temperature and alkali dosage were 150 °C and 2%. After considering various factors, the optimal process was obtained (4% ATDT, 150 °C, 5 min, and 2% NaOH), in which the chlorine loading was 0.29%. Previous studies have shown that the chlorine content of treated cotton fabrics was 0.10%, which can provide rapid inactivation of microorganisms and facilitate industrial application.23,27,41
Oxidative chlorine content of cotton-ATDT and cotton-ATDT-Cl fabrics prepared under various conditions.
Characterization of ATDT-Coated Cotton Fabrics
FT-IR spectra of control cotton and ATDT-coated cotton fabrics before and after chlorination are shown in Fig. 4. Compared with control cotton (Fig. 4a), the sample of ATDT-coated cotton fabrics (Fig. 4b) showed new characteristic vibrational bands at 805 cm–1 (triazine ring), 1533 cm–1 (piperidine ring), and 1565 cm–1 (the stretching vibration of C=N), showing that ATDT was successfully coated onto the cotton fabrics. After chlorination with sodium hypochlorite (Fig. 4c), the characteristic vibrational bands were correspondingly shifted to 814 cm–1, 1542 cm–1, and 1588 cm–1 due to the electron withdrawing effect of the oxidative chlorine.
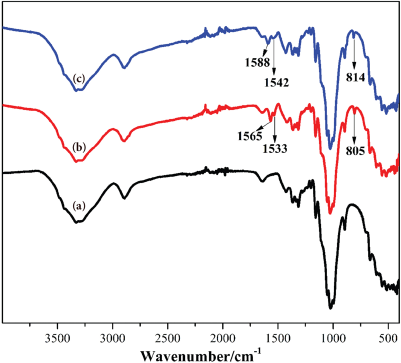
FT-IR spectra of (a) cotton control, (b) cotton-ATDT, and (c) cotton-ATDT-Cl fabrics.
To further prove successful ATDT deposition onto the cotton fabrics, the surface morphology of control cotton and ATDT-coated cotton fabrics before and after chlorination were determined by SEM (Fig. 5). Compared with the smooth surface of control fibers (Fig. 5a), an irregular film appeared on the samples of cotton-ATDT before and after chlorination and the fibers became rougher (Figs. 5b and c), indicating that ATDT was successfully loaded onto the surface of cotton fibers.

SEM of (a) cotton control, (b) cotton-ATDT, and (c) cotton-ATDT-Cl fabrics.
Thermal Analysis
The DSC curve of cotton control, cotton-ATDT, and cotton-ATDT-Cl are shown in Fig. 6a. The melting point of cotton-ATDT (163.83 °C) was higher than that of untreated cotton fabrics (160.59 °C) due to the introduction of sulfur-containing groups and the density of the intermolecular hydrogen bonding. However, after chlorination, cotton-ATDT-Cl gave a higher melting point of 165.54 °C in comparison to cotton-ATDT. 32
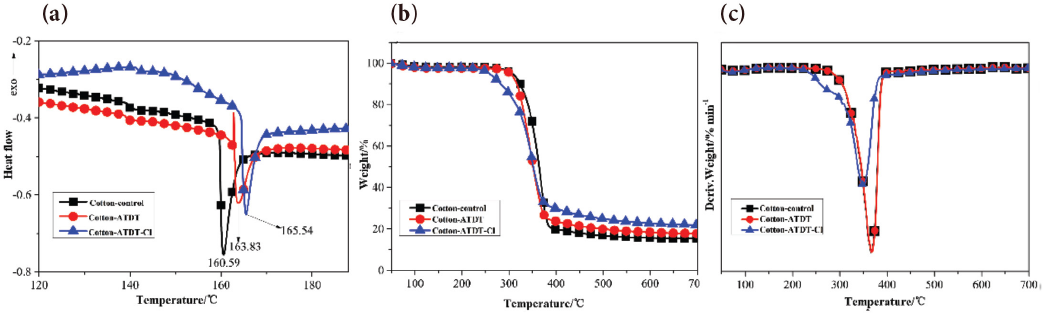
(a) DSC, (b) TGA, and (c) DTG curves of cotton control, cotton-ATDT, and cotton-ATDT-Cl fabrics.
The TGA and difference thermogravimetry (DTG) curves of cotton control, cotton-ATDT, and cotton-ATDT-Cl under nitrogen atmosphere are given in Figs. 6b and c respectively. For uncoated cotton, the temperature at 5 wt% weight loss (T5%) and maximum weight loss temperature (Tmax) were 307 °C and 368 °C, and the residual was 15% at 700 °C.
After treating with ATDT, T5% and Tmax values had no significant change, but greater residue (18%) was obtained. After chlorination, T5% and Tmax values of cotton-ATDT-Cl decreased to 258 °C and 350 °C, respectively, and the char residue at 700 °C reached 22%. This might be attributed to the introduction of sulfur and chlorine elements, which can improve the thermal properties of cellulose fibers. 42
Antimicrobial Efficacy
The antibacterial efficacy of cotton-ATDT-Cl against S. aureus and E. coli O157:H7 was measured using cotton control and cotton-ATDT and the results are presented in Fig. 7. The control cotton showed no biocidal properties and S. aureus and E. coli survived after 30 min of contact time. The ATDT-coated cotton fabrics before chlorination presented the same results. After chlorination, the samples had excellent antibacterial properties—no bacterial survival was detected on culture plates and all bacteria was inactivated within 5 min. This might be due to the high water-solubility of ATDT, which can promote contact between bacteria and the oxidative surface of fabrics, enhancing biocidal efficacy. 29
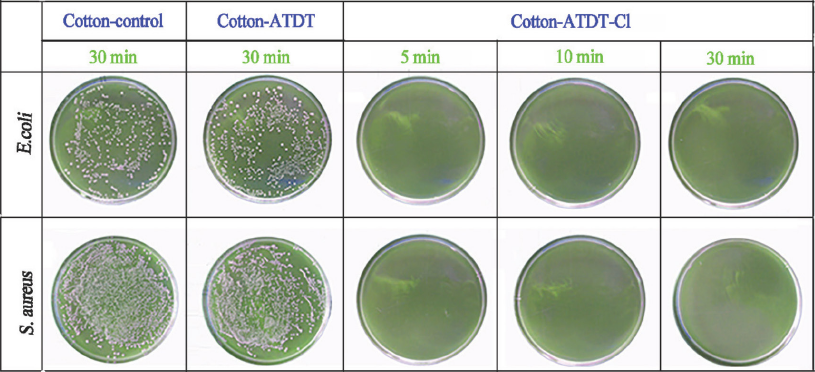
Antimicrobial efficacy of cotton control, cotton-ATDT, and cotton-ATDT-Cl fabrics.
Cytotoxicity Evaluation
To determine the normal cell cytotoxicity of ATDT-coated antibacterial cotton fabrics, NIH 3T3 cells were seeded, and MTT assays were performed. As shown in Fig. 8, after 24 h of cell seeding, all cotton samples showed slightly lower numbers of cell viability compared with the blank. However, the control cotton and Cotton-ATDT-Cl exhibited significant reduction, probably due to the surface hydrophilicity difference from Cotton-ATDT and tissue culture plate. 27 After incubation for 48 h, ATDT-coated cotton samples displayed similar cell viability. However, with respect to the cytocompatibility level of control cotton, ATDT-coated antibacterial cotton fabrics had potential for use in biomedical applications.
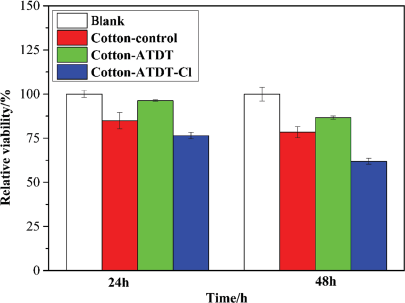
Results of MTT cell cytotoxicity assays.
Storage and Washing Stability
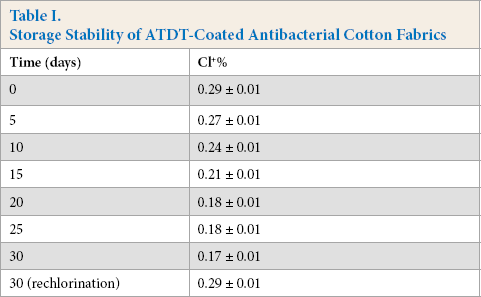
The storage stability of oxidized chlorine on cotton-ATDT-Cl at RT was investigated and the data shown in Table I. Chlorine loading decreased slightly and only 0.02∼0.03% of oxidative chlorine reduction was observed over 5 days. After 30 days of storage, 63% of the oxidative chlorine remained, decreasing from an initial 0.29% to 0.17%, which provided good biocidal efficacy.43,44 In addition, all of the oxidized chlorine can be recovered after rechlorination, which means that the chemical bond between N-halamine compounds and cotton fibers was not broken, and that the loss of oxidative chlorine content was mainly caused by the dissociation of N-Cl.
Storage Stability of ATDT-Coated Antibacterial Cotton Fabrics
The washing stability and rechargeability of oxidized chlorine on ATDT-coated antibacterial cotton fabrics were investigated and the data is shown in Table II. The results show that about 78.1% of oxidative chlorine was retained after five washing cycles, and 96.8% of the chlorine could be recovered after rechlorination. This indicated that the covalent bonds between N-halamine compounds and cotton fibers was very stable to washing, and that the loss of oxidative chlorine content was mainly due to the reduction of N-Cl groups on N-halamine compounds.
Washing Stability of ATDT-Coated Antibacterial Cotton Fabrics a
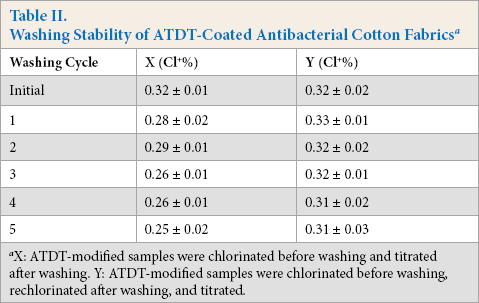
X: ATDT-modified samples were chlorinated before washing and titrated after washing. Y: ATDT-modified samples were chlorinated before washing, rechlorinated after washing, and titrated.
Air Permeability, Whiteness, and Tensile Strength
For functional textiles, air permeability, whiteness, and tensile strength are important for evaluating the quality of functional agents and finishing methods. These were measured and the results are presented in Table III and Fig. 9.
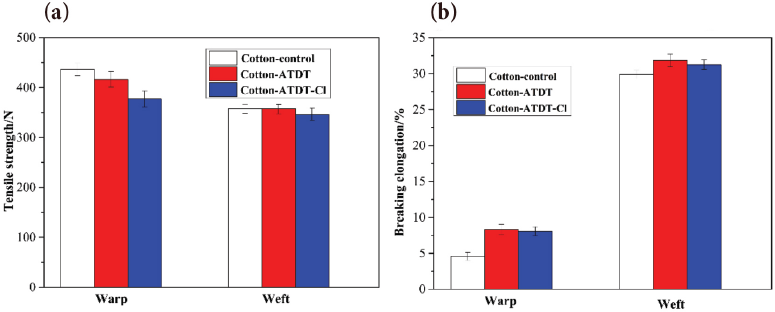
Results from (a) tensile strength and (b) breaking elongation studies.
Air Permeability and Whiteness of Cotton Samples
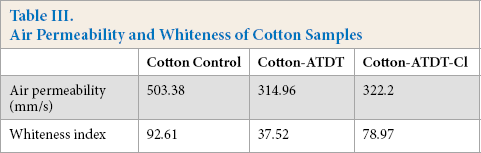
Compared to the cotton control, air permeability decreased by 64% in ATDT-coated cotton fabrics due to the chemical deposition onto surface limiting air space between cotton fibers. Cotton fabrics after coating with ATDT had a higher yellowness index, possibly due to higher curing temperature. However, the whiteness of ATDT-coated cotton fabrics after chlorination was greatly improved due to the strong oxidizing properties of dilute household bleach.
The tensile strength of cotton control, cotton-ATDT and cotton-ATDT-Cl is shown in Fig. 9. Compared to cotton control, a slight loss of tensile strength was observed. Greater than 86% of tensile strength can be maintained in the warp and weft directions for ATDT-coated cotton fabrics before and after chlorination. However, the breaking elongation of cotton-ATDT and cotton-ATDT-Cl significantly increased, which might be due to the stable cross-linking of ATDT with cotton fabrics, giving the cotton fabrics good ductility. 45
Conclusions
A novel water-soluble N-halamine precursor based on s-triazine (ATDT) was synthesized and deposited onto cotton fabrics using the traditional pad-dry-cure method. FT-IR, SEM, DSC, and TGA were used to confirm the successful coating of ATDT to the surfaces of the cotton fabrics. After soaking in sodium hypochlorite, the ATDT-coated cotton fabrics showed excellent antibacterial efficacies against S. aureus and E. coli O157:H7, and all of the bacteria was inactivated within 5 min. ATDT displayed good cytocompatibility and excellent storage stability. ATDT-coated cotton fabrics had good washing stability, and the rechargeability of deactivated chlorine was excellent. The air permeability, whiteness, and tensile strength were also investigated and the results showed that the ATDT coating had little effect on the physical and mechanical properties of cotton fabrics.