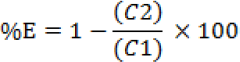Abstract
A key element in reducing human UV exposure is making proper UV protective goods with good ultraviolet protection factor (UPF) protection. The current research is focused on the synthesis of UV-protective functional dyes containing benzophenone-based UV absorbers and its application on wool and silk fabrics. Novel mono azo acid dyes were synthesized by the coupling of diazonium salt solutions of different aromatic amines with 4-hydroxybenzophenone, and these were applied on wool and silk. The fabrics dyed with the synthesized dyes showed less than 5% UVA and UVB transmission, indicating good protection against UV radiation. Dyed fabric also gave good to excellent washing, rubbing, and light fastness, and UV protection properties, even after ten washing cycles.
Introduction
Sunlight is necessary for all living organism on earth, but exposure to UV radiation from sunlight is the primary reason for many skin diseases, including skin cancer. 1 Many studies revealed that ozone layer depletion is a foremost cause of increased skin cancer cases, especially in Australia and New Zealand. The thinning of the ozone layer means higher levels of human UV exposure, which increases skin cancer risk. UV radiation is capable of causing changes in the DNA sequence of an organism and acts as a tumor initiator as well as a tumor promoter. 2 These risks may be remarkably decreased by reducing the exposure of skin to UV radiation. 3 Generally, protection against harmful UV radiation is improved by applying sunscreen containing a UV absorber on exposed body parts. 4
In the last decade, awareness about apparel grade protective textiles that can protect the human body from the effect of harmful UV radiation has increased tremendously. 5 The most effective protection from UV radiation is obtained with next to skin apparel, which depends on fabric composition such as type of fiber, fabric thickness, grams per square meter (GSM), yarn compactness, weave structure, and the dyes and finishes used. 6 A key goal in reducing exposure to UV radiation is producing appropriate UV protective textiles with an ultraviolet protection factor (UPF) greater than 40. These kinds of textiles are essential in high altitude areas for protection against skin disease.7–10
Conventional textiles have low UPF values. To make them UV protective, they need further treatment with certain natural or synthetic chemicals. Commercially-available synthetic UV protective finishes include benzophenone derivatives,11,12 benzotriazole derivatives, other benzophenone-based compounds, 13 titanium dioxide formulations, and others. 14 Solumbra fabric, which is rated at 100+ sun protection factor (SPF), was awarded the prestigious Gold Triangle Award by the American Academy of Dermatology. It blocks more than 98% of UVA and UVB rays and was developed for some of the most medically sun sensitive people.15,16 Application of commercial UV protective finishes is an additional process in textile wet processing, increasing the overall cost of the product.
In the past few years, natural colorants were found to have excellent UV protection properties owing to the presence of UV-blocking components. UPF-enhancing natural dyes from plants, namely Diospyros kaki, Dioscorea cirrhosa, Millettia sp. (Jixueteng), Ecliptae herba, and microcap nucuma, extracted using environmentally-friendly solvents, show UV-protection on cotton and silk fabrics. 17 Wool, cotton, and silk fabrics dyed with pomegranate, curcumin, cutch, and red onion peel extract also show excellent UV-protection properties. 18 UV-protective properties of cotton fabric dyed with green, black, and red teas,19,20 madder, weld, cochineal, 21 lavender, daphene, roth, curry plant, 22 and neem nanoparticle 23 are also reported in the literature. Wool fabric dyed with marigold flower, honeysuckle extract, and Indian rhubarb also show excellent UPF values.24–26 However, reproducibility, cost efficiency, inadequate degree of fixation, and low color fastness properties are some of the limitations of natural colorants.27–29
The concept of natural colorants with UV-blocking components encouraged us to synthesize novel acid dyes with built-in UV-blocking functionality. UV absorbers such as benzophenone, benzotriazoles, acrylonitriles, and triazines are very commonly used as additives in the plastic, paint, polymer, and coating industries, where they can reduce light-induced degradation. UV absorbers proficiently absorb high-energy UV radiation and convert it into harmless heat radiation. 30 Benzophenone is an excellent UV absorber31,32 that prevents the photo-degradation of many vinyl polymers. 33 Compounds such as 4-hydroxybenzophenone, 2,4-dihydroxybenzophenone, and 2-hydroxy-4-me-thoxybenzophenone bearing hydroxyl and methoxy (as auxochrome) groups and a keto (chromophore) group have widely used applications as a polymer additive. 34 In the literature, a few benzophenone-based acids, reactive, and disperse azo dyes are also reported.5,32,35–39
The present communication reports the synthesis of acid azo dyes by the diazo coupling process using 4-hydroxybenzo-phenone as a coupler, and its application on wool and silk fabrics. The wool and silk fabrics dyed with synthesized acid dyes were evaluated for UPF, UVA and UVB transmission, photo-stability, and fastness properties initially and after 1, 5, and 10 washing cycles.
Experimental
Materials
4-Hydroxybenzophenone, o-aminobenzenesulfonic acid, m-aminobenzenesulfonic acid, m-aminobenzoic acid, p-aminobenzoic acid, sodium hydroxide, sodium nitrite, sodium carbonate, dimethylformamide (DMF), acetone, methanol, conc. H2SO4, and conc. HCl were purchased from Sigma Aldrich Ltd. Lyogen WSN leveling agent was procured from Archroma. Ready for dyeing woven wool fabric (weight 173 g/m2, 55 ends/inch, and 48 picks/inch) and woven silk fabric (weight 51 g/m2, 320 ends/inch and 146 picks/inch) were purchased from Kiran Treads.
All compounds were purified by recrystallization and confirmed by thin layer chromatography (TLC) analysis (silica gel 60 coated aluminum plates (Merck). The UV-Vis spectrum of the synthesized dyes was recorded on a UV-Vis spectrometer (Specord 21, AnlytikJena). The L*, a*, b*, C*, h, K/S, DEcmc, and %STR-WSUM of dyed fabrics were obtained using a X-Rite Color i7 (illuminant D65 and 10° observer).
Synthesis of Mono Azo Acid Dyes
This is the general diazotization procedure for the preparation of the monoazo acid dyes
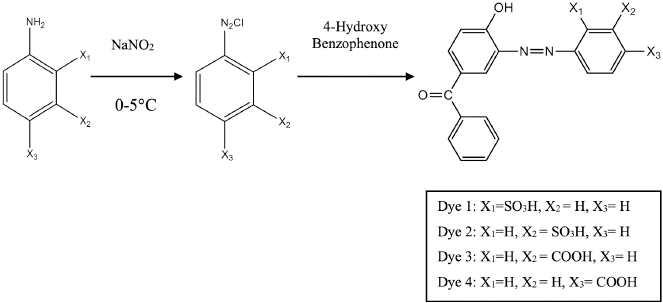
Synthesis of monoazo acid dyes using 4-hydroxybenzophenone.
Na2CO3 (0.01 mol, 1.059 g) and NaOH (0.01 mol, 0.4 g) were dissolved in 50 mL of H2O and gradually stirred until dissolution. 4-Hydroxybenzophenone (0.01 mol, 1.98 g) was added to the solution, followed by continuous stirring until complete dissolution, and then the solution was cooled to 0∼5 °C. The diazonium salt solution was added dropwise to the coupling component over a span of 30 min. During the entire addition, the pH of the reaction mass was adjusted to between 8 and 9. The reaction mass was stirred continuously for 2–3 h. After that, the reaction mixture's pH value was reduced to 2–3 using a 10% w/w aqueous solution of HCl. The reaction mass was filtered using Whatman filter paper and the filtered dye was further purified using DMF-ether to give dyes
Dyeing Wool and Silk
Dyeing of wool and silk fabrics was carried out in a laboratory dyeing machine (R.B.E. Electronics) with a mass to liquor ratio (MLR) of 1:20 and the depth of the shade was 1%. The dyeing bath was adjusted to acidic pH (4.5–5) using 10% acetic acid solution and Lyogen WSN was used as a levelling agent. Wool and silk fabrics were then dyed in the prepared dye bath. Dyeing was initiated at room temperature (RT) and the temperature was gradually raised to 100 °C. Dyeing was carried out at 100 °C for 1 h, followed by cooling to 50 °C. After completion of the dyeing cycle, the dyed fabrics were thoroughly washed with warm and cold water followed by air drying.
To measure the percent exhaustion initially and after the dyebath liquor was collected, the absorbance of the collected solution was measured on a UV-Vis transmission spectrophotometer. The percent exhaustion (%E) was calculated from spectroscopic measurements according to the following formula.
C1 and C2 are the before and after dyeing concentrations of dye solution respectively.
Fabric Washing
The undyed and dyed wool and silk fabrics were washed 10 times according to ISO 6330-2012 standard test method to study the durability of the UV protective properties of the synthesized dyes. UPF and fastness properties of the dyed wool and silk fabrics were evaluated after 1, 5, and 10 washes.
Analysis
Determination of UPF Factor and UV Transmission
The UPF and UV transmission through the dyed sample was measured on a spectrophotometer (Labsphere UPF TesterV-2000F Fabric Analyzer). Percent transmission of UVA and UVB radiation, and UPF values, were determined using AATCC TM183-2010.
UV Exposure and Color Fading Measurements
UV light exposure and color fading measurements were carried out using a customized method based on AATCC TM186. Samples were exposed for 100 h at 60 °C temperature at an irradiance of 0.77 W/m2. Color difference between unexposed and exposed samples and color strength after 100 h of exposure was measured on a reflectance spectrophotometer (Color i7, X-Rite).
Fastness
Lightfastness of the wool and silk fabrics dyed with synthesized dyes were tested on a Xenon arc lamp apparatus following ISO 105-BO2. Washing fastness was tested on a Washometer (SDL Atlas) following ISO 105-C10-C(3). Change in shade and adjacent fabric staining were reported as per ISO gray scales for color change and staining. Rubbing fastness was tested on a MAG rubbing tester using ISO 105 X12:2016(E), including both dry and wet rubbing. Staining of cotton rubbing cloth was assessed with the gray scale for staining under a suitable illuminant.
Dye Toxicity Testing
The determination of aromatic amine toxicity derived from azo colorants was conducted by subjecting a dyed wool fabric to direct reduction for non-disperse dyes as per EN ISO 14362-1 2017.
For non-disperse dyes, the textile test specimen was directly subjected to reduction with sodium dithionite in a citrate-buffered aqueous solution at 70 °C in a closed vessel. Derived amines were then transferred to a t-butyl methyl ether phase by liquid-liquid extraction and cleaned up on a diatomaceous earth column. After concentration of the t-butyl methyl ether extract, amine detection and quantification were conducted by gas chromatography/mass spectrometry (GC/MS). 40
Results and Discussion
Elemental and TLC Analysis of Synthesized Dyes
The dyes obtained were in powder form and in color ranges from yellow to brownish yellow. Composition was confirmed by elemental analysis (Table I). TLC was used for checking purity of dyes using a hexane:ethyl acetate:methanol (3:1:3) solvent system and a single spot was obtained for all dyes.
Properties of Synthesized Acid Dyes
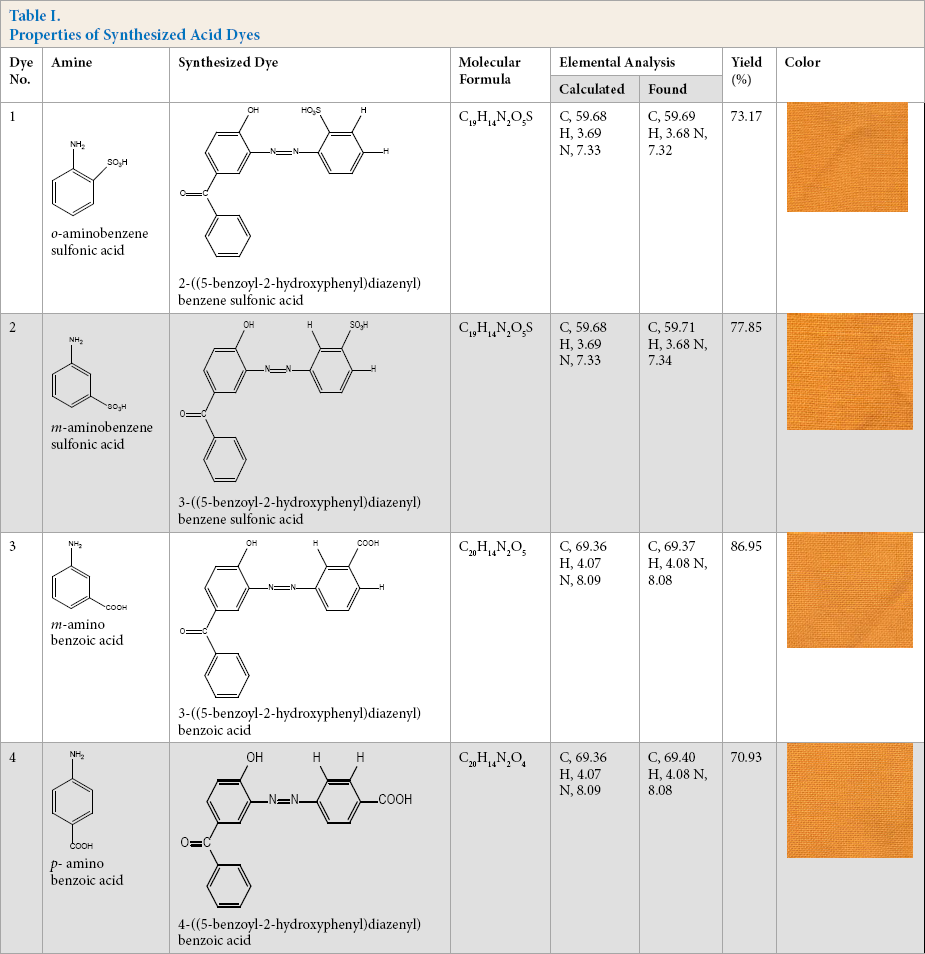
Dye Spectral Data
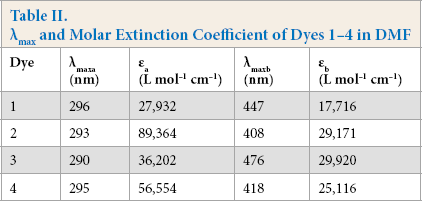
Dyes
λmax and Molar Extinction Coefficient of Dyes 1–4 in DMF
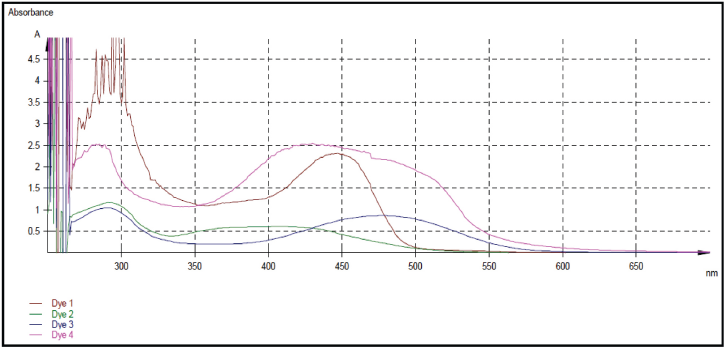
UV-Visible absorption scan of Dyes
Dyeing Properties of Dyes
Wool and silk fabrics were dyed with dyes
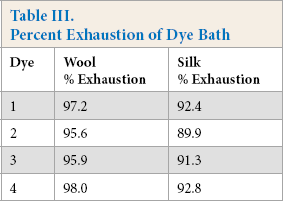
Percent Exhaustion of Dye Bath
Color Parameters of Fabrics before and after UV Exposure
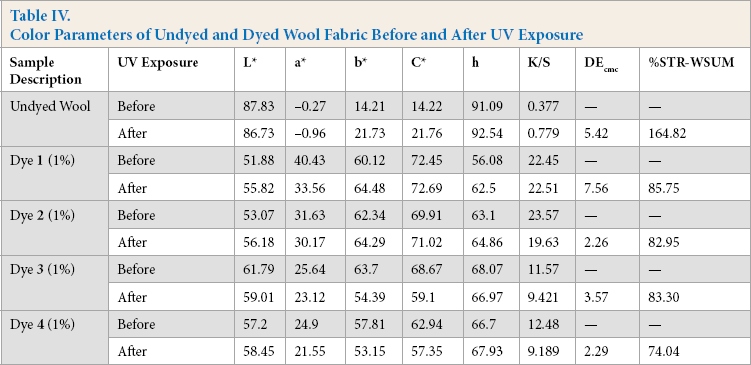
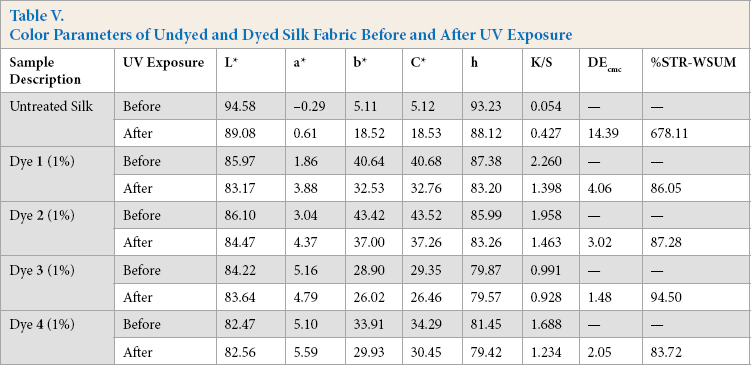
To check the lightfastness of the dyed UV protective azo acid dyed textile material specimens, they were exposed to a UV light source and resistance to degradation was rated based on the color change of the specimens. Samples were exposed for 100 h at an irradiance of 0.77 W/m2 at 60 °C and evaluated with AATCC method [customized]. The results of this experiment are given in Tables IV and V. Color parameters such as L*, a*, b*, C*, h, and K/S of unexposed and exposed fabrics were measured on a reflectance spectrophotometer. Color strength after exposure and the color difference between unexposed and exposed samples were also reported.
Color Parameters of Undyed and Dyed Wool Fabric Before and After UV Exposure
Color Parameters of Undyed and Dyed Silk Fabric Before and After UV Exposure
After 100 h of UV exposure, most of the wool fabrics dyed with dyes
UV Protective Properties of Fabrics before and after Washing
The UV protection properties of the undyed and dyed wool fabrics were evaluated initially and after 1, 5, and 10 washes as per AATCC 183-2010. The UPF values, and percent transmission of UVA and UVB radiation for undyed and dyed wool and silk fabrics are given in Table VI. The UPF value of undyed wool fabric was 17, whereas the dyed wool fabrics using dyes
UPF and Percent Transmission of UVA and UVB Radiation of Undyed and Dyed Wool and Silk Fabric
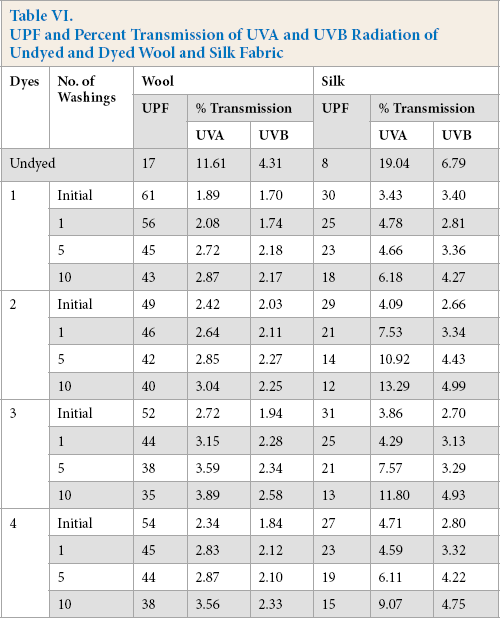
UVA and UVB transmission values of untreated and treated substrates using dyes
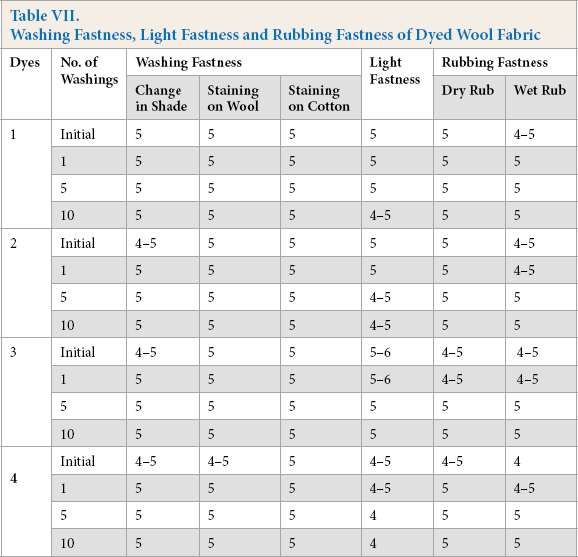
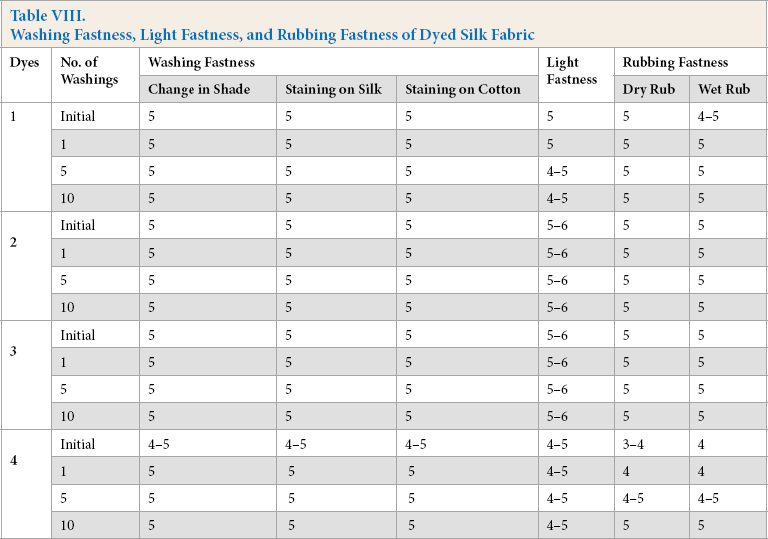
Fastness Properties
Wool and silk fabrics dyed with dyes
Washing Fastness, Light Fastness and Rubbing Fastness of Dyed Wool Fabric
Washing Fastness, Light Fastness, and Rubbing Fastness of Dyed Silk Fabric
Dye Toxicity Testing
To test dye toxicity, wool fabric dyed with synthesized acid dyes
Banned Amine Testing of Wool Fabric Dyed with Synthesized Acid Dyes a
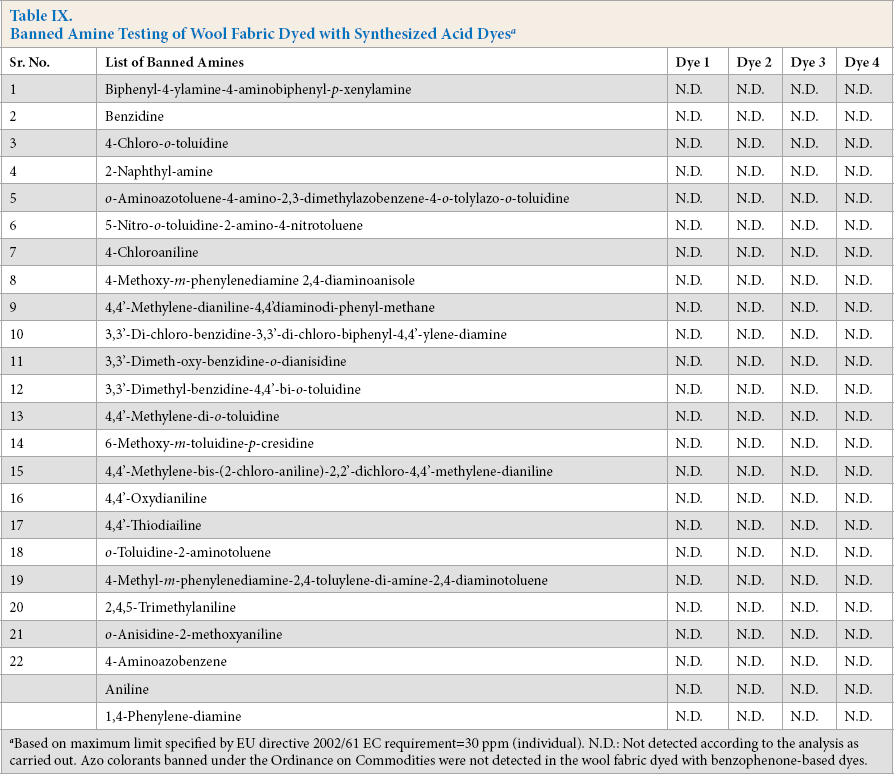
Based on maximum limit specified by EU directive 2002/61 EC requirement = 30 ppm (individual). N.D.: Not detected according to the analysis as carried out. Azo colorants banned under the Ordinance on Commodities were not detected in the wool fabric dyed with benzophenone-based dyes.
Conclusion
UV protective dyes
Footnotes
Acknowledgement
The author acknowledges the financial support received from Ministry of Textiles, Govt. of India, for their support and encouragement in carrying out his college work.