Abstract
Black liquor (BL), a byproduct generated during the kraft pulping process, was used as a source of alkaline lignin (AL) to stabilize aqueous dispersions of carbon black (CB). Black liquor-treated carbon black (BL-CB) samples were prepared by varying the BL/CB/water ratio and characterized by scanning electron microscopy (SEM), thermogravimetric analysis (TGA), and Brunauer-Emmett-Teller (BET) surface area. Improvement in dispersion of BL-CB with increased BL content was observed. The dope dyed viscose fibers were spun by addition of BL-CB to the viscose solution. These fibers exhibited deeper color strength than those obtained from conventional CB formulations at a comparable CB loading on the fiber.
Introduction
The kraft process is a commonly-used pulping method to obtain dissolving grade pulp for producing regenerated cellulosic fibers.1,2 Black liquor (BL) is a waste generated during this process. 3 About 7 tons of BL byproduct is produced for 1 ton of pulp production; most of the BL is used as fuel in the recovery boiler. 3 Apart from the cooking chemicals like NaOH and Na2S, BL also consists of organic matter such as lignin, hemicellulose, their degradation products and extractives.2,3 Viscose fibers are regenerated cellulosic fibers obtained via a complex process involving derivatization of cellulose into sodium cellulose xanthate, which is subsequently dissolved in NaOH solution to give a spinnable viscose solution.4,5 This polymer solution is passed through spinnerets in an acidic bath to regenerate fibers. Dope dyed or spun dyed viscose fibers are inherently colored; regenerated cellulosic fibers spun from a viscose solution contain a pigment formulation.6,7
Carbon black (CB) is a commonly-used black pigment to produce black and other dark shades. The inherent hydro-phobicity of CB limits its uniform dispersion in aqueous media. The high alkalinity of viscose solution (pH > 13) makes dispersion of CB more difficult, leading to its agglomeration. Non-uniform dispersion of CB in viscose solution results in batch-to-batch as well as within a batch variation in shades of fibers. 7 To improve CB dispersions, surfactants are usually incorporated in the formulations.8,9 Surface modification treatments (e.g., ozone, 10 H2O2, 11 acid treatment, 12 anodic oxidation, 13 and polymerization14–16) have also been used to impart hydrophilicity to CB for obtaining uniform, stable aqueous dispersions.
Chemicals derived from the petrochemical industry are extensively used to synthesize surfactants. In recent years, due to growing environmental concern, bio-based surfactants have gained more attention. Lignin is one such biomaterial tailored to produce surfactants for various applications. 17 Lignosulfonate from bagasse, 18 Caribbean pine, 19 and oil palm empty fruit bunches 20 was synthesized and used as a surfactant in the oil industry. Sodium lignosulfonates (SLS) of different molecular weights were used as dispersants for coal water slurries. 21 SLS was adsorbed on the surface of coal particles, increasing the absolute zeta potential value to stabilize the slurry. Alkaline lignin (AL)-based surfactant, synthesized by attaching hydrophobic carbon chains and hydrophilic sulfonic groups, gave superior surface activity compared to lignosulfonates. 22 Cationic amphiphilic lignin derivatives were synthesized from kraft lignin and thoroughly characterized. 23 Kraft lignin, acetylated lignin, and lignosulfonates were modified with polyethylene glycol to obtain nonionic polymeric surfactants, and their effect on dispersion of gypsum was investigated. 24
Lignin, acetylated lignin, and propionated lignin were used to disperse multi-walled carbon nanotubes (MWCNTs) at high loadings in aqueous media. 25 Derivatization of lignin hydroxyl groups resulted in a new class of surfactants that imparted anti-static properties to MWCNTs containing polyolefin films. 25 In another study, lignin was superior to sodium dodecylbenzene sulfonate in stabilizing carbon nanotubes (CNT) dispersions. 26 Aqueous dispersions of graphene at high concentrations were obtained by exfoliation of graphite. 27 The mechanism of graphene stabilization was explained using Fourier transfer infrared (FTIR), fluorescence spectroscopy, and zeta potential measurements. It was suggested that AL was adsorbed on the graphene surface due to π- π interaction between the two, and the electrostatic repulsive forces between the negatively-charged functional groups of lignin stabilized the graphene sheets.
In the current study, we used BL from the pulp industry as a source of AL to stabilize aqueous dispersions of CB. A range of black liquor-treated carbon black (BL-CB) samples were prepared by varying the CB/BL ratio. These dispersions were added to viscose solutions and black fibers were spun via the dope dyeing process. Color properties of fibers obtained from BL-CB samples were compared to those obtained from a commercial CB formulation.
Experimental
Materials
Powdered CB (grade N234) was obtained from SKI Carbon India. BL (17% solid content) was obtained from Harihar Unit of Grasim Industries Ltd. Standard viscose solution was used for spinning of fibers. Viscose fibers were regenerated in a spin bath consisting of H2SO4, Na2SO4, and ZnSO4. Deionized water (DI) water was used for preparation of solutions and slurries.
Characterization of BL
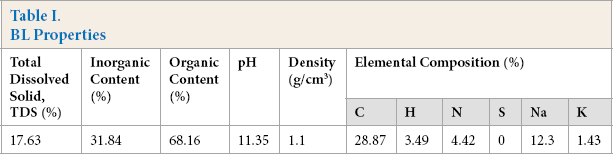
Thorough characterization of BL was carried out for total solids, inorganic and organic content, pH, and density. 28 Additionally, elemental analysis was carried out by CHNS analysis and inductively coupled plasma-optical emission spectrometry (ICP-OES). These results are summarized in Table I.
BL Properties
Preparation of BL-CB samples
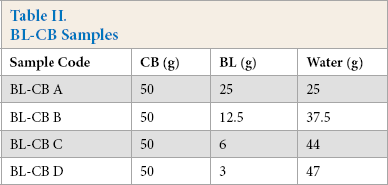
Powdered CB (50 g) was stirred in a stainless steel reactor attached to an overhead stirrer. To this, the desired amount of BL, premixed with an appropriate amount of water, was added slowly under stirring. The mixture was stirred for 1 h using an overhead stirrer. The resulting BL-CB was dried at 120 °C until a constant weight was obtained. Four different ratios of CB/BL/water were used to prepare BL-CB samples (Table II).
BL-CB Samples
Slurry Preparation and Addition to Viscose Solution
BL-CB powder (2.5 g) was added to 100 mL of DI water under stirring. The resulting slurry was ultrasonicated for 1 h. The required amount of BL-CB slurry was added to 150 g of viscose solution and mixed thoroughly (15 min) using an overhead stirrer. BL-CB containing viscose solution was spun into fibers.
Characterization of Powdered BL-CB
BL-CB samples in powdered form were characterized by scanning electron microscopy (SEM), thermogravimetric analysis (TGA), and Brunauer-Emmett-Teller (BET) surface area analysis techniques. TGA of BL-CB powder was carried out on a Mettler Toledo TGA Analyzer by heating from room temperature (RT) to 800 °C under a nitrogen flow. BET surface area analysis was performed using Nova 2000 E Quanta Chrome instrument.
Images of CB samples were captured on a HITACHI S-4800 scanning electron microscope (SEM). Aqueous BL-CB dispersion (0.5 wt%) were ultrasonicated (Equitron ultrasonic bath) for 20 min at 50 kHz at 100% power, and a drop of the dispersion was place on a glass coverslip mounted on an SEM sample stub. It was subsequently dried in a hot air oven and coated with gold on a combined sputtering and carbon coating system (Quorum, Q 150R ES) before being analyzed by SEM.
Characterization of BL-CB Dispersions
Characterization of the BL-CB aqueous dispersions was carried out on a Malvern Zetasizer Nano ZS. BL-CB slurries (2.5%) were diluted to 500 ppm. To obtain diluted samples representative of the behavior and dispersion state of initial samples, BL-CB was uniformly dispersed via stirring and ultrasonication of the initial samples. The required amount of BL-CB slurry was taken out using a micro pipette and added to DI water under stirring to maintain the original dispersion state.
High magnification optical imaging of conventional CB and BL-CB slurries was carried out to understand their dispersion behavior. BL-CB slurry (2.5%) was diluted in DI water, 5% NaOH solution, and viscose solution corresponding to their loading in fibers (1, 2, and 3%). These diluted samples were passed through a quartz flow cell (0.2-mm thick). The flow cell was illuminated from the bottom and a high magnification camera (DFK MKU130-10x22, The Imaging Source) was placed above it. Images at high magnification were captured and compared.
Fiber Spinning
Dope dyed viscose fibers containing conventional CB as well as BL-CB samples were spun on a lab-scale spinning setup. CB-containing viscose solution was filled in a syringe pump (100 mL) and fixed on an infusion pump setup (DBK Instruments). A jacketed glass spin bath was used to regenerate the viscose solution coming out of the spinneret. The spin bath consisted of H2SO4, Na2SO4, and ZnSO4. The temperature of the spin bath was maintained by circulation of hot water (Equibath). The fibers emerging from the spinneret were pulled, stretched, and collected by a two-drum winder (DBK Instruments). The fibers were given the required stretch between the two drums and collected on the 2nd drum. After the required amount of fibers was collected on the 2nd drum, they were cut in the form of staple fibers (length ≈ 45 mm), washed, and dried.
Color Measurement
Measurement of color values of dope dyed viscose fibers was done by using a color spectrophotometer (Konica Minolta CM-2500d). Dope dyed viscose fibers were opened and aligned parallel before winding on a rectangular cardboard strip (8 × 2.5 cm). These sample strips were then measured for their color values in terms of L*, a*, and b* by the color spectrophotometer.
Tensile Property Measurement
To determine preliminary performance properties of dope dyed viscose fibers, their tensile properties (e.g., denier, tenacity, and elongation) were measured on Vibroscop-400 and Vibrodyn-400 instruments (Lenzing Instruments). Fibers were conditioned at 25 ± 2 °C and 65% relative humidity (RH) for 24 h before testing.
Results and Discussion
BL-CB Powder and Dispersions
Four different ratios of CB/BL/water were used for the treatment of powdered CB by BL (Table II). Table III summarizes the results of BET surface area, TGA, particle size analysis, and zeta potential. The untreated CB had the greatest BET surface area. Among various BL-CB samples, the surface area decreased with increased amounts of lignin in BL-CB samples. This can be attributed to reducing the amount of CB in BL-CB samples.
BL-CB Powder and Dispersion Properties
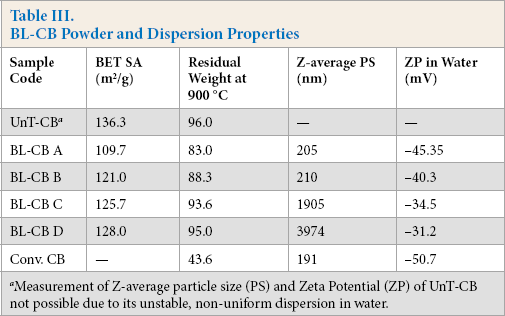
Measurement of Z-average particle size (PS) and Zeta Potential (ZP) of UnT-CB not possible due to its unstable, non-uniform dispersion in water.
Fig. 1 shows the TGA curves for dried BL, untreated CB (UnT-CB), and different BL-CB samples. Dried BL samples underwent thermal decomposition over a wide temperature range similar to lignin. 29 Weight loss at lower temperatures (<200 °C) was ascribed to moisture removal, as well as formation of smaller molecules from lignin decomposition, while weight loss at higher temperatures was due to the decomposition of the polymeric structure of lignin. UnT-CB underwent only 4% weight loss on thermal decomposition. Among various BL-CB samples, weight loss increased with increasing lignin content (Table III), which can be attributed to the loss of lignin coated on CB particles.
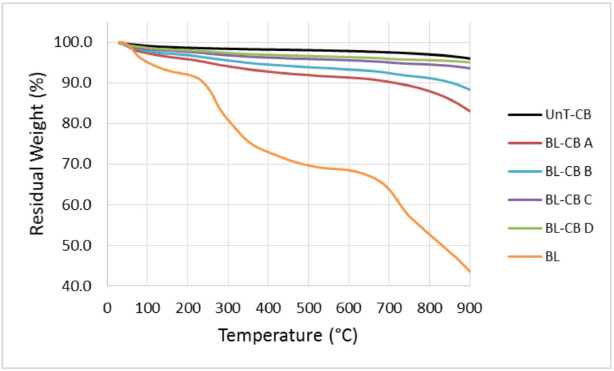
Thermogravimetric analysis profiles of dried BL, UnT-CB, and various BL-CB samples.
Fig. 2 shows the SEM images of UnT-CB and BL-CB A samples. It can be seen that UnT-CB is in agglomerated form, whereas the BL-CB A exists as well-separated CB particles embedded in the BL. The improved dispersion of BL-CB is attributed to the presence of AL on the CB surface that acts as a stabilizer and prevents agglomeration of BL-CB particles together. Lignin molecules consist of a large number of aromatic rings, which facilitate interactions between AL and CB particles, resulting in the coating of CB particles with AL. Additionally, the negatively-charged AL causes electrostatic repulsion between BL-CB particles, resulting in stable dispersions.

SEM images of (a) untreated UnT-CB N-234 and (b) BL-CB A.
Among the various BL-CB samples, the particle size decreased and the zeta potential increased with increasing BL content, indicating improvement in the dispersion (Table III). The improvement in dispersion stability can be attributed to the increased total negative charge due to increased AL content. At lower concentrations of BL (3 and 6%), the stabilization offered by AL was insufficient, as indicated by the lower zeta potential values, to impart stabilization; larger particle sizes were also observed due to agglomeration. This validates the hypothesis that the CB particles were stabilized via electrostatic repulsion due to the negatively-charged AL. Thus, the dispersion stability increased with increased BL content.
To further understand the dispersion behavior of various CB samples, high magnification optical images were captured. For these analyses, 2.5% CB slurries were diluted to simulate the actual loadings corresponding to those used to spin fibers containing 1, 2, and 3% CB. Fig. 3 shows the high magnification optical images of various CB dispersions at 1% CB content (920 ppm CB in viscose solution). UnT-CB showed the greatest tendency to agglomerate, while the agglomeration tendency decreased with increasing BL content. These visual observations are in accordance with the particle size and zeta potential measurements carried out on very dilute dispersions (Table III).

High magnification optical microscopy images of (a) BL-CB A, (b) BL-CB B, (c) BL-CB C, (d) BL-CB D, and (e) UnT-CB dispersions.
The agglomeration behavior of BL-CB A in water, 5% NaOH solution, and viscose solution was thoroughly investigated at three different concentrations corresponding to 1, 2, and 3% CB loadings on fibers (Fig. 4). There was no significant impact of increasing the CB loading from 1 to 3% in water, indicating that AL was able to keep CB well dispersed in water. The extent of agglomeration was substantially greater in 5% NaOH and viscose solution, indicating the detrimental impact of alkaline environment on CB dispersion. At the same CB concentration, the agglomeration tendency followed the trend: 5% NaOH > viscose solution > water. The lower agglomeration tendency in viscose solution than in 5% NaOH solution can be ascribed to the higher viscosity of polymeric viscose solution, which offers resistance to agglomeration of CB particles. Based on the reports of CNTs 30 and graphene 27 stabilization in aqueous medium, as well as those of CB reported in the current study, it can be concluded that AL acts as a surfactant facilitating dispersion of CB. The greater alkaline stability of BL-CB samples makes them a potential substitute of the conventional CB formulation for production of viscose fibers.

High magnification optical microscopy images of BL-CB A at different concentrations in (a-c) water, (d-f) 5% alkali, and (g-i) viscose solution.
Fiber Spinning and Color Properties
Various BL-CB samples were added to viscose solution and spinning experiments were carried out under standard spinning conditions. For the samples BL-CB C and D, fiber spinning was not smooth, therefore, fiber samples could not be obtained. Fiber spinning was found to be smooth for samples BL-CB A and B. These observations were in accordance with particle size and zeta potential measurements of various BL-CB samples, which indicated larger sized particles and lower zeta potential for samples C and D, resulting in unstable dispersions.
Table IV summarizes the L*, a*, and b* values of fibers spun from BL-CB and those spun from a conventional CB formulation. In both cases, the fiber lightness (i.e., L* value) decreased with increased loading of BL-CB in the fibers, indicating increased color depth. This is attributed to the increased absorption of light at greater CB content. At the same loading of BL-CB, fibers containing BL-CB A were found to have lower L* values than corresponding fibers containing BL-CB B. This can be attributed to the greater CB content in BL-CB B (96.1%) than in BL-CB A (92.2%).
L*, a*, and b* Values of Various Fibers
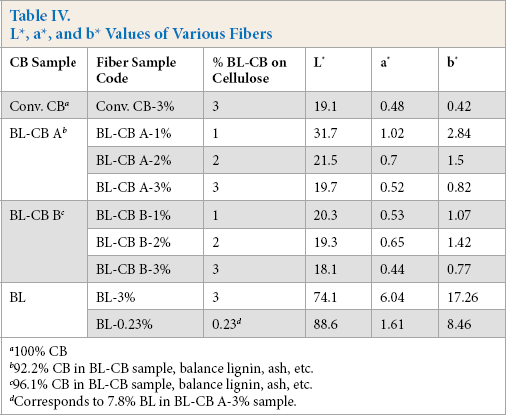
100% CB
92.2% CB in BL-CB sample, balance lignin, ash, etc.
96.1% CB in BL-CB sample, balance lignin, ash, etc.
Corresponds to 7.8% BL in BL-CB A-3% sample.
For comparison, black viscose fibers were also spun from a conventional CB formulation at 3% CB loading on fiber (Conv. CB-3%). Although the BL-CB A-3% fiber sample had a slightly lighter shade, BL-CB B-3% had significantly deeper shade than the Conv. CB-3% sample. Interestingly, the BL-CB B-2% fiber sample, containing less CB than Conv. CB-3%, had comparable L* values. This was attributed to the superior dispersion of BL-CB B-3% in viscose solution than the Conv. CB formulations, resulting in a deeper shade. These results indicate the potential of BL-CB to achieve deeper fiber shades at significantly lower loadings of CB, which in turn, can result in smoother operations, superior fiber properties, and improved fiber processability.
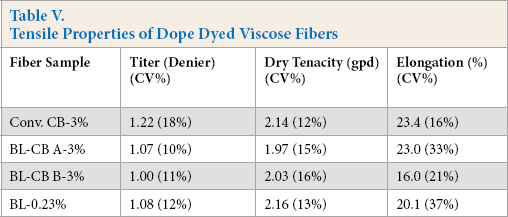
Spinning experiments were also carried out with only BL as pigment to obtain dope dyed viscose fibers. Two different loadings of BL were used: (1) 3% on cellulose (i.e., equivalent to BL-CB A-3%) and, (2) 0.23% on cellulose (i.e. equivalent to the actual BL present in BL-CB A-3%). The BL alone did not impart black color to the resulting fibers as indicated by the significantly greater L* values (Table IV). Thus, in BL-CB samples, BL acted only as a dispersant and the dark color of the fibers was due to CB. The tensile properties of fibers spun from BL-CB were comparable to those obtained from conventional CB formulations, as well as those obtained using only BL (Table V).
Tensile Properties of Dope Dyed Viscose Fibers
Conclusion
Black liquor (BL) generated during kraft pulping was used as a source of alkaline lignin (AL) to stabilize carbon black (CB) dispersions. Treatment of CB with BL resulted in well-separated CB particles. The presence of AL on the CB surface resulted in electrostatic repulsion between black liquor-treated carbon black (BL-CB) particles, which stabilized the dispersions in aqueous media. Dispersion of BL-CB samples in aqueous media improved with the increased content of BL. Alkaline stability of BL-CB was superior to that of conventional CB formulations, making them an attractive option for incorporation into the highly alkaline viscose solution. Dope dyed viscose fibers spun using BL-CB samples had greater color depth (i.e., a lower L* value) than those obtained from commercial CB formulations. However, the inherent inconsistency of AL content in BL on fiber color properties needs to be further studied.
Footnotes
Acknowledgment
The authors would like to thank Birla Carbon India Pvt. Ltd., India and Aditya Birla Science and Technology Co. Pvt. Ltd., Navi Mumbai, India for their support.
