Abstract
Para-aramid fiber was fibrillated in a dimethyl sulfoxide (DMSO)/tetrabutylammonium fluoride (TBAF) solution at 80 °C. Physical gelation of the dispersion liquid occurred by cooling to 20 °C at varying rates of 0.5 to 2.8 °C/min. The solvent was then replaced with acetone and the gel was dried by supercritical CO2 to obtain an aerogel. The fibril diameter decreased from ∼40 nm to 20 nm using a TBAF/DMSO ratio of 1:22 to 2:11, respectively. The specific surface area (SSA) of aerogel reached 702 m2/g at a fibril diameter of 20 nm. Prepared aerogel fiber with a diameter of 536 μm had a translucent appearance, an SSA of 473 m2/g, and a low density of 0.067 g/cm3. It bent freely and repeatedly without deformation or breaking.
Introduction
Aerogels are nano-porous materials with attractive properties such as large specific surface area (SSA), low density, and low thermal conductivity. There are two typical types of aerogels. One type is silica-based aerogels and the other is cellulose-based aerogels. Silica-based aerogels have outstanding thermal insulation property and high transparency. 1 However, they have limited applications due to their low mechanical strength and poor flexibility. Cellulose-based aerogels2,3 are flexible and have excellent thermal insulation properties. However, they burn easily because cellulose is a flammable material. Therefore, it is essential to find new materials to overcome the shortage of existing aerogels. Aerogels from para-aramid fiber would be valuable as these fibers are widely used as protective materials, since they have high modulus, strong strength, heat resistance, and fame resistance.
The aerogel preparation process using para-aramid was developed in our previous research. 4 We obtained an aramid aerogel having a large SSA area of 510 m2/g and a low density of 0.083 g/cm3 through supercritical drying of a physical gel prepared from a fibril dispersion liquid. However, the gelation mechanism of fibril dispersion wasn't clarified and the obtained aerogel did not have enough flexibility.
In this study, we investigated how to form the gel structure from the aramid fibril dispersion liquid while influencing the structure of the aramid aerogel and improving its flex-ibility. The aramid aerogel fiber can be used in protective clothing, such as firefighter clothes or space suits.
Experiments
Materials
Para-aramid yarn (Kevlar 29) was provided by the DuPont-Toray Co. Ltd. Trihydrate of tetrabutylammmonium fluoride (TBAF) was purchased from Tokyo Chemical Industry Co. Ltd. Ethanol, dimethyl sulfoxide (DMSO), and acetone were purchased from Wako Pure Chemical Co. Calcium hydride was obtained from Nacalai Tesque Inc. All chemicals were used without further purification.
Preparation of Aramid Dispersion and its Gelation
The aramid fibril dispersion liquid was prepared by fibril-lation of an aramid fiber by a reported procedure. 5 DMSO and TBAF were mixed to obtain the solution with weight ratios of 11:1, 11:2, and 22:1. An excess amount of CaH2 was added to the solution to remove water, since water competes with aramid molecules upon interaction with TBAF. The solution was stirred for 48 h to fully react CaH2 with water in the solution. The dehydrated DMSO/TBAF solution was separated as the supernatant by centrifugation. The aramid yarn was washed with deionized water and ethanol, and then dried in an oven for 3 h at 120 °C.
The dried fiber (1.0 wt%) was put into the dehydrated DMSO/TBAF solution during heating at 80 °C and stirred at 250 rad/min for 1 h to fibrillate the aramid fiber. We defined this mixture of fibrils as the aramid dispersion. The hot dispersion was poured into a mold of the desired form and cooled down to 20 °C at various cooling rates to obtain the aramid physical gel. We used a glass dish for preparing a bulk gel and a glass capillary as a mold for preparing a fiber-shaped aerogel. The gel was kept at 20 °C until the total time, including the cooling time and the holding time, reached 3 h. The aerogel fiber was prepared by extruding the aramid dispersion of DMSO:TBAF = 11:1 from a syringe equipped with a polytetrafluoro-ethylene (PTFE) tube to the atmosphere by a syringe pump. The syringe was kept at 80 °C and the air atmosphere was at 20 °C. The inner diameter of tube was 0.96 mm, and the length was 63 mm. The extrusion rate was 9.2 mm/s.
Solvent Exchange and Supercritical CO2 Drying
The obtained gel was soaked in acetone for six days to remove TBAF and DMSO in the gel. The gel was then dried with supercritical CO2 (scCO2) to remove acetone and an aerogel was formed. The gel was placed in a TSC-WC-0025 autoclave from Taiatsu Techno Corp. which was heated to 80 °C. CO2 was introduced into the autoclave and pressurized to 10 MPa by the SCF-Get high pressure pump from JASCO Corp. This condition was maintained for 4 h. CO2 was released from the autoclave to atmospheric pressure for 3 h with a step of 0.01 MPa maintained by the BP-2080 back pressure valve from JASCO Corp.
Analytical Methods
Dynamic viscoelastic measurements were performed on the aramid dispersion with an MCR302 temperature tunable rotary rheometer from Anton Paar Gmbh. The measurements were at a strain of 10% and an angular frequency of 20 rad/s by the flat plate system with a diameter of 25 mm. Transmission spectra were measured for the aramid dispersion using a USB-2000 spectrometer from Ocean Optics Inc. The measurements were performed on the aramid dispersion cooled at various cooling rates, similar to the preparation of the physical gel, in a glass optical cell with an optical pass length of 2 mm. A halogen lamp was used as a light source. Volume shrinkage of the aramid gels after solvent exchange and scCO2 drying were calculated from the diameter and height or length of the gel, with the cylindrical shape measured by a caliper square. The aerogel SSA was evaluated using the Brunauer-Emmett-Teller (BET) method with nitrogen adsorption on a BELSORP 28 SA-SP system from MicrotracBEL Corp. The aerogel microstructure was observed with a JSM-7600 thermal field emission scanning electron microscope (SEM) from JEOL Ltd. Evaluation of aerogel fiber flexibility was performed by observing the fiber with VB-7000 and VB-7010 optical microscope systems from Keyence Corp. before and after fiber bending.
Results and Discussion
Effect of Cooling Rate on Physical Gelation
Gels were prepared from the aramid dispersion in DMSO:TBAF = 11:1 at cooling rates of 6, 10, and 13 °C/min to investigate the effect of cooling rate on the structure and properties of the gel. The results of dynamic viscoelastic measurements for the aramid dispersion are shown in Fig. 1. Gelation temperature was observed at ∼30 °C regardless of the cooling rate. The elastic modulus after gelation decreased with increased cooling rate.
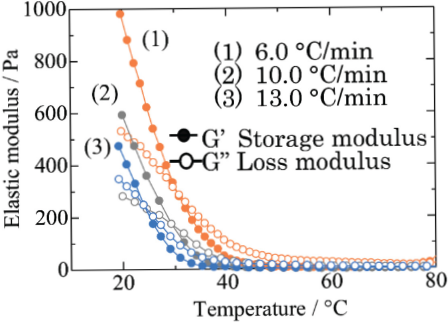
Effect of cooling rate on dynamic viscoelasticity of aramid dispersion in DMSO:TBAF = 11:1.
Transmission spectra of the aramid dispersion cooled at 2.8 and 0.5 °C/min are shown in Fig. 2. Absorption bands below ∼550 nm were blue-shifted with cooling in both spectrums. The shift at 2.8 °C/min was smaller than the one at 0.5 °C/min. An absorption band was observed for the aramid fiber in the region below 420 nm. It has reported that molecular absorption bands are blue shifted by forming H-aggregation of molecules stacking predominantly face-to-face since bonding electrons stabilized by aggregate formation absorb light at higher energy, namely, shorter wavelength, for their excitations. 6 The blue shift of absorption bands here indicates that the aggregation of aramid molecules was progressing with cooling.
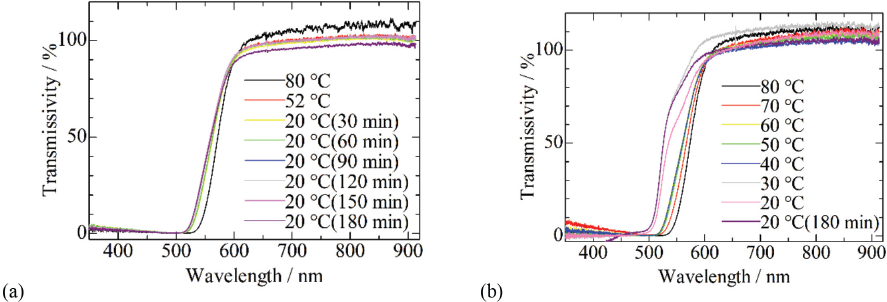
UV and visible light transmission spectrum of aramid dispersion in DMSO:TBAF = 11:1 during cooling from 80 °C to 20 °C at (a) 2.8 °C/min and (b) 0.5 °C/min.
This aggregation was caused by hydrogen bonding and pi-pi interaction among para-aramid molecules. The progress of molecular aggregation was prevented with the increased cooling rate. The network structure formed from aramid fibrils with a diameter of ∼30 nm was observed in the SEM image of the aerogel shown in Fig. 3. Similar structures were observed for all para-aramid aerogels independent of cooling rate.

SEM image of cross section of aerogel prepared from aramid dispersion in DMSO:TBAF = 11:1 at a cooling rate of 2.8 °C/min.
From these results, the gelation of the aramid dispersion proceeded with the association of fibrils formed by aggregation of aramid molecules during cooling. When the aramid dispersion is cooled rapidly, the molecular motions are restricted before aggregating. This cooling condition results in obstruction of the association of and poor entanglement among fibrils. The gel obtained from high cooling rates had a low elastic modulus. Volume shrinkage of the gel and the SSA of the aerogel are shown in Table I. The aerogel cooled at 2.8 °C/min from the aramid dispersion using DMSO/ TBAF = 11:1 had a greater SSA of 203.6 m2/g compared to 158.6 m2/g at 0.5 °C/min. The greater SSA of aerogel cooled at 2.8 °C/min is attributed to the large shrinkage caused by the low elasticity of the gel in the solvent exchange step as shown in Table I.
Volume Shrinkage of Gel and SSA of Aerogel
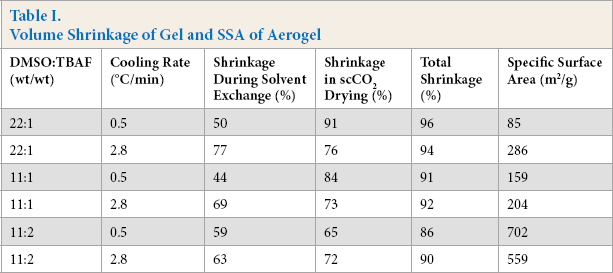
We estimated that the interaction between aramid molecules was enhanced during solvent exchange because TBAF preventing hydrogen bonding among molecules was eliminated from the gel. Gel shrinkage was minimized using scCO2 drying, which resulted in greater SSA of the aerogel for gels prepared with a high cooling rate. These trends for the aerogel from the aramid dispersion at DMSO:TBAF = 11:1 were also observed from the aramid dispersion at DMSO:TBAF = 22:1, but not 11:2. The differences in the behavior of the 11:2 aramid dispersion from the others is under investigation.
Effect of TBAF Concentration on Aerogel Morphology
The fibril diameter distribution measured from SEM images is shown for the aerogels prepared with various TBAF concentrations and cooling rates in Fig. 4. The fibril diameter decreased from ∼40 to 20 nm with increasing TBAF concentration.
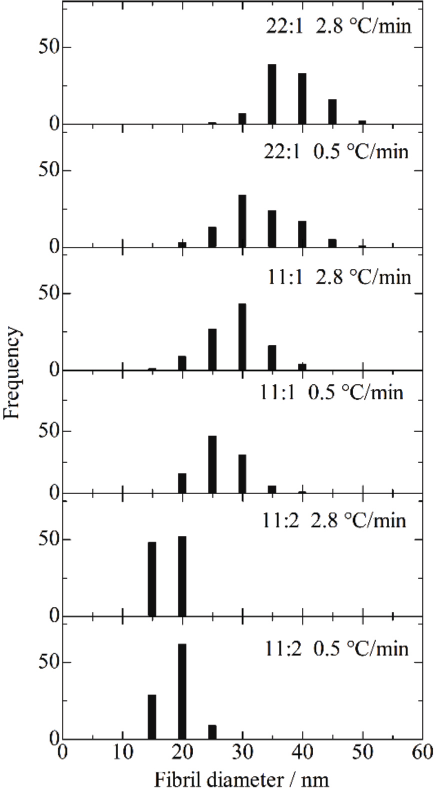
Histograms of fibril diameters measured from SEM images of aerogels.
The TBAF concentration controls the amount of hydrogen bonding among aramid molecules in the fibrillation or cooling processes, thus influencing the fibril diameter. It is reported that diameter of fibril decreased as interaction among aramid molecules was inhibited. 7 The transmission spectrum is shown for the aramid dispersion prepared using the ratio of DMSO:TBAF = 11:2 in Fig. 5. The blue shift of the absorption band as mentioned for Fig. 2 wasn't observed during cooling of the aramid dispersion. The spectrum at 80 °C in Fig. 5 was similar to the spectrum of the dispersion prepared with the 11:1 ratio at the same temperature in Fig. 2.
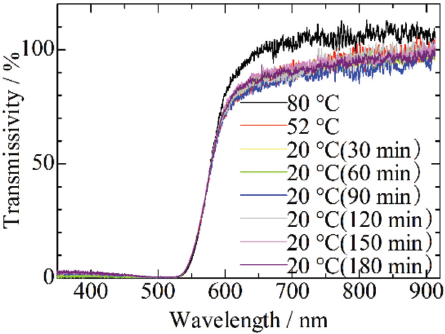
Transmission spectrum of aramid dispersion in DMSO:TBAF = 11:2 during cooling at 2.8 °C/min.
Aramid molecule aggregation did not proceed during cooling in the case of the dispersion prepared with the 11:2 ratio. The diameter of aerogel fibrils prepared from the dispersion with the 11:2 ratio was smaller compared to the one prepared with the 11:1 ratio. The aerogel prepared with the 11:2 ratio and at 0.5 °C/min gave the greatest SSA of 702 m2/g, since the aerogel fibrils were finer than the other aerogel fibrils.
Preparation of Aerogel Fiber
To overcome aerogel fragility, the para-aramid aerogel fiber was prepared. First, we tried to fabricate a fiber-shaped aerogel with the aramid dispersion of DMSO:TBAF = 11:1 under the same conditions for the bulk gel. The photo of the fiber-shaped aerogel prepared with the 1.032-mm diameter glass capillary is shown in Fig. 6. The obtained fiber-shaped aerogel had a translucent yellow appearance. It was constructed from the nanofibrillar network of molecular chain bundles similar to the bulk aerogel as shown in Fig. 3. Photos of the bending test for the fiber-shaped aerogel are shown in Fig. 7. The fiber-shaped aerogel bent freely without deformation or breaking of the gel.

Fiber-shaped aramid aerogel.
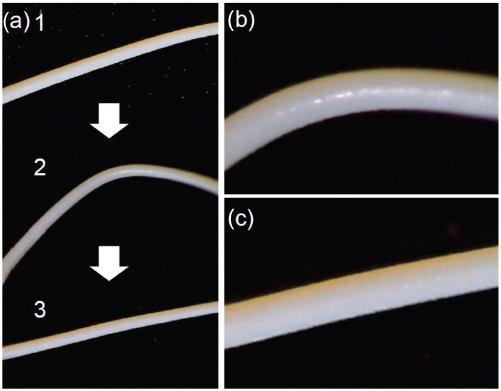
Bending test of fiber-shaped aramid aerogel. (a) 1-initial state, 2-bent state, 3-relaxed state, (b) enlarged photo of bent point in (a) 2, and (c) enlarged photo of relaxed point in (a) 3.
Next, we tried to fabricate aerogel fiber with continuous physical gelation of the aramid dispersion by extruding the aramid dispersion from a syringe. The physical gel prepared by this method provided the aerogel fiber after the solvent exchange and scCO2 drying process. The obtained aerogel fiber with a diameter of 536 µm had a large SSA of 473 m2/g and a low density of 0.067 g/cm3. It had a similar appearance and structure of the fiber-shaped aerogel. It also bent freely and repeatedly.
Conclusion
We investigated how to create a flexible aramid aerogel fiber by forming the gel structure from an aramid fibril dispersion. The gelation of the aramid dispersion proceeded by association of fibrils made through aggregation of aramid molecules during cooling of the aramid dispersion. The physical gel cooled at a high rate had less association in the aramid dispersion using a low concentration of TBAF. This structure prevented gel network destruction during supercritical drying, although the network structure changed and the gel shrank during the solvent exchange in acetone. The fibril diameter decreased with the increased amount of TBAF in the dispersion. The SSA of aerogel reached 702 m2/g by decreasing the fibril diameter to 20 nm. The prepared aerogel fiber with a diameter of 536 µm had a translucent appearance, an SSA of 473 m2/g, and a low density of 0.067 g/cm3. This fiber bent freely and repeatedly without deformation or breaking.
Footnotes
Acknowledgement
The authors wish to thank Professor Koji Nakane of the University of Fukui for the BET measurements of aerogels. This study was supported by a 2016 research grant from Maeda Kosen Foundation and a 2016 industry-academia collaboration grant from Fukui Bank Ltd.
