Abstract
The ink absorption of polyamide-coated polyamide textile products containing kaolin is poor. Citric acid was used to modify kaolin, and the adsorptive performance of rhodamine B was studied. The modified kaolin with the best adsorptive ability was selected. The results showed that kaolin modified with a 2.0 M citric acid solution was strongly adsorptive. During the interaction between citric acid and kaolin, H+ acts on the surface and interlayer of kaolin, and on Al3+ and Si4+ in the kaolin. This results in an increase in the specific surface area and a smaller particle size. After using modified kaolin in the polyamide wet coating, the ink-absorption of polyamide-coated polyamide fabrics improved and the resulting pH value met Oeko-Tex Standard 100 requirements for textile use.
Introduction
Polyamide wet coatings are used to improve the printing properties of polyamide or polyamide blended fabrics.These coatings are prepared by dissolving waste nylon 6 fiber in methanol and anhydrous calcium chloride, followed by the addition of filler to form the coating slurry. After being immersed in the coagulating bath, a layer of film is formed on the fabric surface, and then dried and fixed to prepare a polyamide-coated trademark fabric with good performance and meeting various printing standards.1,2 These products are low cost, have excellent hand, and so forth.3,4 However, as used in existing trademark fabric, polyamide-coated poly-amide fabrics containing kaolin as the filling agent still have many problems, such as poor adhesion to the fabrics, poor ink-absorption, high pH value, and damage to the cutter.
The printing ink absorption of polyamide-coated poly-amide fabrics is influenced by many factors and is closely related to the structure and properties of fillers used for coating. Owing to its small particle size and large specific surface area, kaolin has good adsorption performance.5,6 The adsorption capacity of the printing ink is improved, as well as the printing ink absorption of the polyamide-coated polyamide fabric.7,8
Citric acid was used to modify kaolin in this study to improve the properties of the polyamide coating slurry. Various concentrations of citric acid in modified kaolin were tested for adsorption of rhodamine B dye. The interactions of citric acid with kaolin were investigated. The application of modified kaolin in the polyamide wet coating process and its effect on the printing ink absorption and pH value of the coated polyamide textile were analyzed.
Experimental
Materials
Anhydrous calcium chloride (analytical grade), polyether NJ-220, printing ink, polyamide 6 fabric (30 g/m2), and waste polyamide 6 fiber (technical grade) were supplied by Huzhou Xinli Label Belt Co. Ltd. Calcined kaolin was purchased from Shijiazhuang Yanfeng Mineral Products Co. Ltd. Absolute methanol and citric acid (analytical grade) were purchased from Hangzhou High Crystal Fine Chemical Co. Ltd. Rhoda-mine B (analytical grade) was purchased from Tianjin Yongda Chemical Reagent Development Center.
Preparation and Characterization of Modified Kaolin
Citric acid solutions (0, 0.5, 1.0, 1.5, and 2.0 M) were prepared. The kaolin was added at a 1:10 solid to liquid ratio. After stirring at 40 °C for 24 h, the filter cake was washed with distilled water five times, and then dried and ground, and the modified kaolin was obtained.
A standard working curve for rhodamine B was prepared. The absorbance curve of the rhodamine B solution was measured in the 400-800 nm wavelength range, using a Lambda 35 UV-Vis spectrophotometer (Perkin Elmer), to determine the maximum absorption wavelength of absorbance (Fig. 1). Rhodamine B solution concentrations (1, 2, 3, 4, 5, 6, 8, and 10 mg/L) were measured at the maximum absorption wavelength of 554 nm and plotted (Fig. 2).
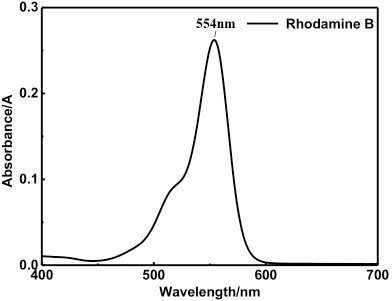
Absorption curve of rhodamine B.
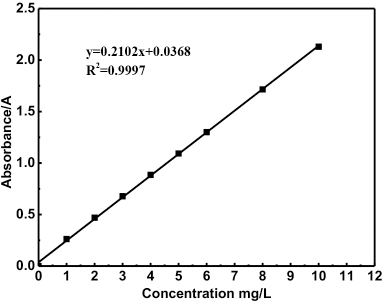
Standard working curve of rhodamine B.
The citric acid modified kaolin was then evaluated for its ability to adsorb rhodamine B. Modified kaolin (0.1 g) was placed in a 100-mL Erlenmeyer flask, 50 mL of a 0.1 mg/L solution of rhodamine B was added, and the mixture kept in a constant temperature water bath at 30 °C. The absorbance at 554 nm was measured by UV spectrophotometric determination of the supernatant obtained by centrifugation after 10, 20, 30, 40, 50, 60, 90, and 120 min. 9 Adsorptive capacity was calculated according to Eq. 1.
The specific surface area was analyzed by the Brunauer-Emmett-Teller (BET) method using a Micromeritics ASAP 2020HD88 instrument. The particle size and particle size distribution of kaolin were measured using a Mastersizer 2000 laser particle size analyzer (British Malvern).Te surface morphologies of modified kaolin before and after the modification were analyzed by a Phenom Pro desktop scanning electron microscope (SEM, Phenom-China Instrument). The chemical structure of modified kaolin was determined using a Vertex 80 Fourier transform infrared (FTIR, Bruker) spectrometer. The crystal structure of kaolin was determined at room temperature (RT) using an ARL X'TRA X-ray diffraction (XRD, Thermo ARL) in the range 2θ-scale from 10° to 85° with a scanning speed of 3°/ min.
Preparation and Characterization of Polyamide-Coated Test Materials
Polyamide-coating slurry was prepared based on a literature procedure. 10 The citric acid modified kaolin (22 g) and 100 g of methanol were added into three 250-mL flasks, stirred for 30 min at RT, then heated to 65 °C, and 35 g of calcium chloride was added to the flask. When the mixture became a stable emulsion, 16 g of waste polyamide 6 was added to the three flasks. After the waste polyamide 6 dissolved completed, the suspension was stirred for 1 h. Finally, 2.3 g of polyether NJ-220 was sequentially added at RT for another 25 min to the three flasks. The polyamide coating slurry preparation was now completed.
Glass Plate Coating
According to the preparation method for coating film on a glass plate in the literature, 11 a 10 × 10 cm glass plate was used. Four strips (0.16 mm of the total thickness) of parallel transparent tapes were attached on the glass plate ∼7 cm between strips. A certain amount of polyamide-coated slurry was uniformly applied to the top of the glass plate and the glass plate was tilted 30° slowly into the water bath. After 5 min, the completely solidified coating film was taken out and set at 100 °C in an electro-thermal constant temperature blower drying box for 90 s. The film was then removed from the plate for testing.
Polyamide Fabric Coating
Polyamide 6 fabrics were coated with the polyamide-coating slurry on both sides using the wet coating method, then the coated fabrics were put slowly into a 25 °C water bath to solidify. After 3 min, the coated fabrics were placed in a continuous heat setting machine for 90 s at 160 °C.
Testing
The ink length on polyamide-coated polyamide fabric 12 was determined as follows. Polyamide-coated polyamide fabric was cut into 15 × 4 cm coated fabric samples, placed in a constant temperature and relative humidity (RH) box (23 ± 2 °C, 50% ± 2% RH) for 4 h. After dropping a drop of printing ink (3 μL per drop) onto the surface of the polyamide-coated polyamide fabric, the rectangular wooden frame containing the sample was quickly placed vertically in an RH box. The length of the ink on the polyamide-coated polyamide fabric was measured 4 h later, five times for each sample (two samples total; one prepared with unmodified kaolin and the other with modified kaolin), and the average value was taken. pH value of polyamide-coated polyamide fabric was investigated using GB/T 7573-2009. 11
The contact angle of the polyamide coating film removed from the glass plate was determined. The surface morphologies of polyamide coating film removed from the glass plate were investigated by SEM.
Results and Discussion
Citric Acid Modified Kaolin Standard Working Curve for Rhodamine B
The maximum absorbance determination and standard curve for rhodamine B are shown in Figs. 1 and 2, respectively. Fig.1 shows the maximum absorption wavelength of 554 nm for rhodamine B. The standard curve for rhodamine B had a linear relationship from 0 to 10 mg/L in the low concentration range (Fig. 2), the standard curve equation was y = 0.2102x + 0.0368 (R 2 = 0.9997).
Citric Acid Concentration Effect on Adsorption Capacity of Kaolin
The effect of citric acid concentration on the adsorption capacity of kaolin itself was studied, as shown in Fig. 3. The adsorption capacity of kaolin for rhodamine B dye increased with adsorption time. The adsorption rate of rhodamine B dye on kaolin slowed down at 60 min, and the adsorption reached saturation at 90 min. The adsorption rate of rhodamine B dye on kaolin and the amount of adsorption increased after citric acid treatment. At increased citric acid concentrations, the H+ concentration increased and the adsorption capacity of kaolin for rhodamine B was enhanced. The adsorption capacity increased slowly when the concentration of citric acid was 1.5-2.0 M. In this study, the concentration of citric acid used was 2.0 M. (The unmodified kaolin and modified kaolin in the subsequent tests refer to kaolin treated with 0 M and 2.0 M citric acid, respectively.)
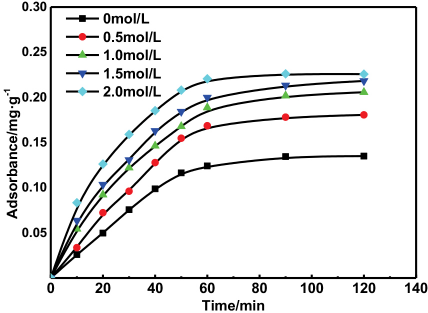
Effect of citric acid concentration on the adsorptive capacity of kaolin.
Characterization of Citric Acid Modified Kaolin
Particle Size and BET
The kaolin particle size and specific surface area before and after modification are shown in Fig. 4 and Table I. The citric acid treated kaolin showed a decreased particle size distribution; the proportion of modified kaolin with smaller particle sizes increased (Fig. 4). The average particle size of kaolin decreased from 20.620 μm to 17.939 μm, and the specific surface area increased from 4.9827 m2/g to 6.9256 m2/g. This resulted in the improved adsorption capacity of the modified kaolin.
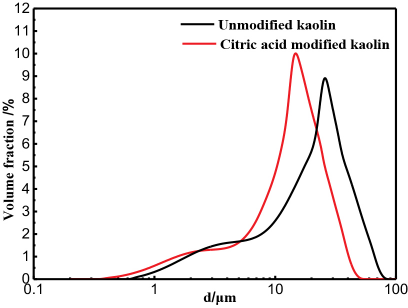
Kaolin particle size distribution.
Specific Surface Area and Volume Mean Particle Size of Kaolin

SEM
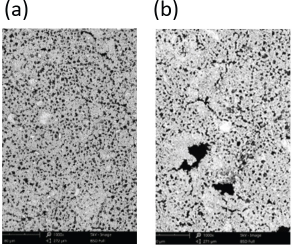
The surface morphology of kaolin before and after modification as determined by SEM analysis is shown in Fig. 5. More needle-like nicks appeared on the surface of citric acid modified kaolin. Kaolin itself has a 1:1 type layered structure. Citric acid releases H+ on the kaolin surface and the gap layer and replaces Si4+ and Al3+ in kaolin. Therefore, the layered structure of kaolin is destroyed. The anions in solution pass through these holes into the internal layer structure of kaolin, further damaging the kaolin by decreasing the kaolin particle size and increasing the specific surface area.

Surface morphology of (a) unmodified kaolin and (b) modified kaolin (×10,000).
FTIR
The FTIR spectra of the kaolin fillers are shown in Fig. 6. The chemical formula of kaolin is Al2O3•2SiO2•2H2O. The infrared spectrum of kaolin contains mainly Al-O and Si-O vibration peaks.13,14 The -OH and H2O vibrations in the interior and surface of kaolin are at 4000-3200 and 1630 cm−1, while the vibrations of Si-O and Al-O are mainly located at 400-1300 cm−1. After treatment with citric acid, the bending vibrations of Si-O shifted from 1223 to 1094 cm−1. The symmetrical stretching vibration peaks of Si-O-Si appeared at 935 and 831 cm−1, and the flexural vibration peaks of Si-O and Al-O appeared at 579 and 473 cm−1. The reason for this could be that the structure of kaolin was destroyed after modification by citric acid, so that the Si4+ and Al3+ atoms in kaolin were replaced with H+ and the surrounding environment was changed. In addition, after citric acid treatment, the C=O stretching vibration peak appeared at 1736 cm−1, indicating that citric acid complexed with Al3+ and Si4+ on the kaolin surface.
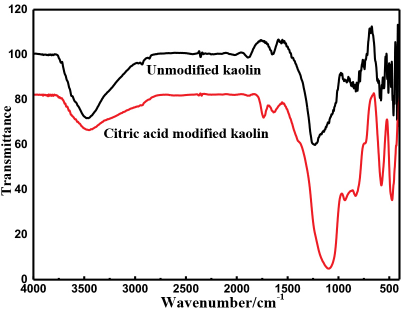
FTIR spectra of the kaolin fillers.
X-ray Powder Diffraction
The XRD diagram of kaolin before and after modification is shown in Fig. 7. The kaolin crystal structure is a 1:1 type layered structure in the triclinic system, connecting silicon- oxygen tetrahedra and aluminum-oxygen octahedra by oxygen. The structure of functional groups on the surface of the kaolin are -Si(A1)-OH, -Si-O-Al-, and -Si(A1)-O.15,16 As shown in Fig. 7, at 2θ = 10∼85°, compared with the unmodified kaolin, there were several diffraction peaks (2θ = 25°, 2θ = 40°) in an obviously weakened spectrum of modified kaolin, and the 13° and 17° near diffraction peaks disappeared. This shows that kaolin treated with citric acid affected the kaolin structure.
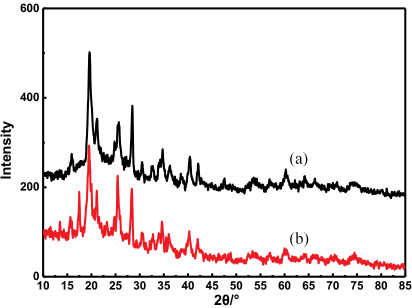
X-ray diffraction of the (a) citric acid modified kaolin and (b) unmodified kaolin.
Characterization of the Polyamide Wet Coating
The ink length of polyamide-coated polyamide fabric, and the contact angle and SEM images of polyamide-coated film from the glass slides were studied to investigate the effect of kaolin modified by citric acid in the polyamide wet coating. The pH value of the polyamide-coated polyamide fabric was used to judge whether the modified polyamide coating product meets Oeko-Tex Standard 100 requirements.
Ink Absorbency
The effect of kaolin on the ink absorption of polyamide-coated test materials was studied, as shown in Table II and Fig. 8. The contact angle (35.16°) for the modified polyam-ide-coating film removed from the glass plate and the length of the ink (8.67 mm) for the modified kaolin polyamide coating on polyamide fabric were less than that for the respective coatings using unmodified kaolin. Kaolin modi-fed by citric acid increased, and improved the speed of, ink absorption for the polyamide coating on polyamide fabric.
Effect of kaolin on the surface morphology of polyamide coating membrane using (a) unmodified kaolin and (b) modified kaolin (×1000).
Effect of Kaolin on the Ink Absorption of Polyamide Coating
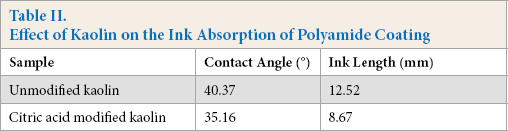
Meanwhile, the surface pore size distribution of the coated film removed from the glass plate was consistent in the poly-amide coating containing kaolin modified by citric acid (Fig. 8). The particle size of the citric acid modified kaolin was reduced, allowing easy dispersion into the coating slurry, and increased the specific surface area. The ink adsorption was thereby improved.
pH Value
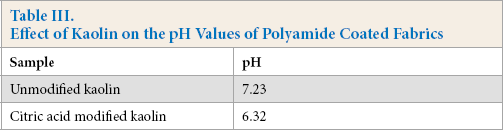
Table III showed that, for the citric acid modified kaolin polyamide-coated polyamide fabric, the pH value changed from 7.23 to 6.32. This meets Oeko-Tex Standard 100 requirements for the polyamide-coated polyamide fabrics, due to the modified kaolin and citric acid chemical interaction. The carboxyl groups from citric acid were now present on the surface of modified kaolin as shown by FTIR.
Effect of Kaolin on the pH Values of Polyamide Coated Fabrics
Conclusion
By comparing the adsorption capacity of rhodamine B with different citric acid concentrations, the effect of the 2.0 M citric acid solution modification on kaolin used in polyam-ide wet coating was optimal, and the modified kaolin had strong adsorption capacity.
Based on the modified kaolin particle size, BET, SEM, FTIR, and XRD analyses were performed to investigate the nature of citric acid modified kaolin. Citric acid released H+ on the kaolin surface and in the gap layer where kaolin and citric acid interact. The interlayer structure of kaolin was destroyed and the anions in the solution entered the interior of kaolin through surface holes by interacting with Al3+ and Si4+ to adsorb on the surface. Furthermore, the layered structure of kaolin was destroyed, the particle size decreased, and the specific surface area increased.
The citric acid modified kaolin was applied to polyamide wet coating on both glass plates and polyamide fabric. The ink absorption was improved and the pH value of polyamide wet coated polyamide fabrics decreased, while still meeting Oeko-Tex Standard 100 requirements.
Footnotes
Acknowledgments
This research was funded by the Huzhou Xinli Label Belt Co. Ltd. The author would like to thank the Key Laboratory of Advanced Textile Materials and Manufacturing Technology, Hangzhou, China.
