Abstract
Large quantities of pineapple peel waste are discarded every year that can be used for extraction of useful textile functionalization chemicals. In the present work, pineapple peel extract (PPE) was used for simultaneous dyeing and multifunctional finishing of wool fabric. The PPE was used for dyeing wool fabrics, with and without mordants. The dyed fabrics were characterized and evaluated for color values and fastness properties. The functional properties of the dyed fabrics viz. antibacterial activity, UV protection, and antioxidant activity were evaluated. It was confirmed that wool fabric can be dyed using pineapple peel extract without mordants; however, additional color strength can be gained using pre-mordanting. The dyed fabrics showed light shades with good fastness properties along with excellent functional properties.
Introduction
Recently, coloration of textiles using natural dyes has gained momentum, resulting in researchers investigating various sources and methods of natural dyeing. The functional-ization of textile materials using natural dyes is of recent interest in research. There are various reports available in the literature regarding simultaneous coloration and functional-ization of textiles using natural dyes.1-6
Use of waste materials for the extraction of chemicals, like natural biomolecules, offer dual benefits in terms of waste material management as well as profits from the extracted chemicals. There is a huge quantity of waste generated from fruit peels that can be used for the extraction of functional biomolecules. Some sources of natural dyes include fruit peels (e.g., pomegranate and orange peels).
Pineapple (Ananas cosmosus) is a popular fruit which is rich in antioxidants, namely flavonoids, and vitamins A and C. Pineapple processing generates large quantities of solid waste (∼75% of the fruit), the majority of which (almost 35% of the total pineapple weight) consists of pineapple peels. Such peels are generally discarded, which may lead to serious environmental issues and bioresource waste problems. This peel waste is a source of important biomolecules of high value and exploitation potential, making it an interesting material for research. The peels are well known ingredients in ethnomedicine.7-12
When extracting useful biomolecules from pineapple peels, researchers have focused primarily on the extraction of the bromelain enzyme, and secondarily on the use of the waste as a low-cost raw material for the production of phenolic anti-oxidants, organic acids, bioethanol, biogas, and fiber.9,13-16 The other alternative use for pineapple peel waste is in biopolymer production, such as polyhydroxy-alkanoates (PHA), by fermentation. 17 Pineapple peels showed moderate levels of protection against the spread of pathogenic microorganisms. 18
There are a number of reports available on the use of pomegranate peels,19,20 orange peels,21,22 coconut shells, 23 and banana peels for dyeing. 24 Pineapple peel contains functional components, like polyphenolic compounds, which can provide coloration and functionalization of textile materials. Application of pineapple peels in functional coloration of textiles has not been previously reported.
In the present work, pineapple peel waste was used for simultaneous dyeing and multifunctional finishing of wool fabric. The dyed fabrics were evaluated for color values and fastness properties. The efficacy of such fabrics as multifunctional textiles was explored. The novel source of pineapple natural dye for simultaneous dyeing and functional finishing of wool was studied using pineapple peel extract (PPE).
Materials and Methods
Materials
Ready-for-dyeing (RFD) wool fabric (woven, GSM = 140, EPI = 67/2, PPI = 52/2) was supplied by the Wool Research Organization, Mumbai. Ferrous sulfate and other chemicals were purchased from Sigma Chemicals. Pineapple peels (PP) were collected from a local juice shop and dried at 50 °C for 24 h. The dried peels were powdered using a mortar and pestle.
Methods
PP Dye Extraction and UV-Visible Analysis of PPE
The PP powder (2 g) was added to 100 mL of distilled water and the mixture was heated to 90 °C for 60 min. The obtained extract was filtered using nylon filter cloth. The volume of the extract was made up to 100 mL with distilled water to get a 2% stock solution. The UV-Visible spectrum of PPE was recorded using a UV-Visible spectrophotometer (Shimadzu).
Exhaust Pre-Mordanting and Dyeing
The wool fabrics (samples 4-6) were mordanted using ferrous sulfate (10% owf, pH 4.16) mordant at 90 °C for 60 min. A material to liquor ratio (MLR) was maintained at 1:10 and mordanting was carried out in Rota Dyer (Texlab). The un-mordanted (samples 1-3) and mordanted (samples 4-6) samples were dyed separately using PPE (10, 20, and 30% owf) at 90 °C for 60 min. The dyed samples were washed with cold water and dried.
Evaluation of Color Values and Fastness Properties
Spectral reflectance values (taken between 400 and 700 nm wavelengths) of the dyed fabric were measured using a Gretag Macbeth Color Eye 7000. The color values were evaluated using the Kubelka-Munk equation. Dyed wool fabrics were evaluated for washing (ISO 105-C06:2010, A1M), 25 rubbing (ISO 105-X12), 26 and light (ISO 105-B02) fastness properties. 27
Functional Properties
Radical scavenging activity of dyed fabric was determined by measuring the decrease in absorbance of 2,2-diphenyl-1-pic-rylhydrazyl radical (DPPH) at 517 nm as per the procedure reported in the literature. 28 The dyed wool samples were evaluated for the UV protection factor (UPF) as per standard AS/NZS 4399:1996 using a Shimadzu UV-2600 spectrophotometer. 29 The antibacterial activity of dyed wool fabrics was evaluated as per AATCC TM100. 30
FTIR-ATR Analysis of Dyed Wool
Dyed wool fabrics were characterized using a Fourier transform infrared-attenuated total reflectance (FTIR-ATR) spectrometer (Nicolet iS50).
Results and Discussion
Color Measurements
PPE contains various biomolecules that can be complexed using metal ions inside the fiber structure to achieve colored and functional wool fabric. The mechanism of dyeing in such cases is very similar to acid-mordant dyeing of protein fibers, where the fiber is a treated separately with mordant and dye (the sequence is dependent on the particular type of acid-mordant dyeing) to form a water insoluble complex inside the fiber. The pre-mordanted wool fiber contained iron (Fe) ions, which interacted with various functional groups on wool fiber. Dyeing of such pre-mordanted wool with PPE, containing various polyphenolic compounds and flavonoids, resulted in a wash fast complex formed inside the wool fiber to give coloration. Reports regarding the presence of ferulic acid, catechin, gallic acid, epicatechin, isomeric N,N′-diferuloylspermidines, hydroxycinnamoyl(di)glycer-ides, mono- and di-feruloylspermidine, hydroxycinnamic acid amides, hydroxycinnamoylspermidines, and other compounds in PPE are available in the literature.31-38
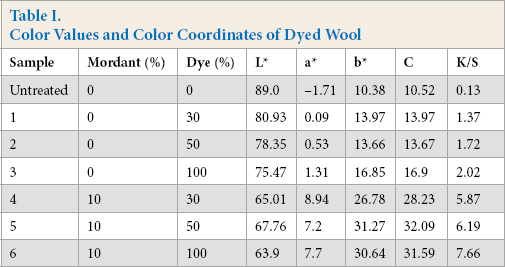
To study the coloration of wool using PPE, the color values and color coordinates of the dyed wool were measured and the results are presented in Table I. The color values of the samples dyed using PPE without pre-mordanting were less than that for mordanted samples. This was expected because for un-mordanted wool, the attachment of the dye to wool fibers was due to interaction with amino groups, while for mordanted wool, the presence of ferrous ions facilitated more dye pickup. Hence, the higher color values were a combined effect of contribution of ferrous ions in imparting color and the enhanced attachment of active biomolecules. However, dyeing with PPE showed great promise in wool dyeing without mordant. The samples dyed using pre-mordanting however, were brighter than un-mordanted fabric samples (as indicated by higher values of C). The shades became darker on increasing the shade (%), irrespective of absence or presence of mordant. This was expected as the availability of active macromolecules increased with increase in shade (%); the more dye that got attached to wool fiber led to an increase in darkness of shade (as indicated by a decrease in the L* value and an increase in K/S value). For the pre-mordanted samples (samples 4-6), the increase in shade (%) resulted in the decrease in redness and the increase in yellowness of the dyed wool.
Color Values and Color Coordinates of Dyed Wool
Color Fastness
Evaluation of color fastness to color change and color stain was performed and the fastness ratings are tabulated in Table II. All samples subjected to washing fastness showed good color fastness to color change and good to fairly good color fastness to color staining. The wool samples dyed without any mordant also displayed a satisfactory level of fastness, indicating the firm attachment of the biomolecules extracted from pineapple peels with the fiber. Wool, being a protein fiber, can be protonated, and thus offers sites for the attachment of anionic chemicals. The affinity of biomolecules for the wool fiber, as well as their ability to bind with wool keratin, was thus confirmed. The pre-mordanted samples showed relatively poor fastness ratings compared the un-mordanted samples, especially at higher shade (%). However, the fastness ratings for all dyed wool samples remained satisfactory.
Fastness Ratings of Dyed Wool Samples
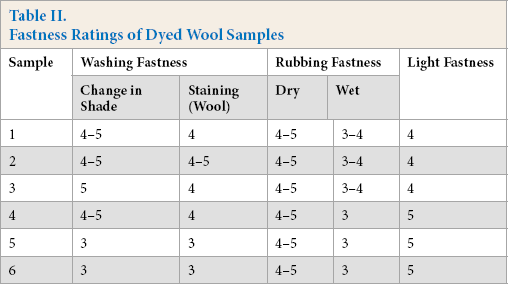
Dry rubbing fastness for all samples was in the excellent range, which indicated the resistance of dye removal during dry abrasive rubbing. However, wet rubbing fastness ratings were relatively poor, but remained in the acceptable range. Wet rubbing as per ISO 105-X12 resulted in removal of surface color, leading to staining. Wet rubbing fastness ratings of pre-mordanted samples were poor (lower by 0.5 scale) as compared to the un-mordanted samples. This may be attributed to the higher color values of the pre-mordanted samples and the presence of more surface color, which was partially removed during wet rubbing. For natural dyeing, the dye-mordant complex might also form on the surface, resulting in dye removal during wet rubbing. For acid dyeing of wool, boiling conditions are generally used. The temperature of dyeing, in this case, was lower than boiling, therefore, dye penetration may have been hindered.
Light fastness of samples dyed without pre-mordanting showed satisfactory levels of light fastness, which was improved when pre-mordanted samples were used. The pre-mordanted samples showed higher color values, indicating the presence of more biomolecules, in the complex form with mordant, which resulted in better resistance to color removal under exposure to light. The aggregated and complexed forms with metal ions generally gave better light fastness.
Functional Properties
Functional properties of the dyed wool samples were evaluated and results are summarized in Table III. The dyed samples showed significant bacterial colony reduction against both Gram-positive and Gram-negative bacteria. This was due to the presence of active flavonoids and tannins, which act on bacterial cells by disrupting the cyto-plasmic membrane and inhibiting enzymatic activity. Better antibacterial activity was evident against Escherichia coli when compared to that against Staphylococcus aureus.
Functional Properties of Dyed Wool Fabrics
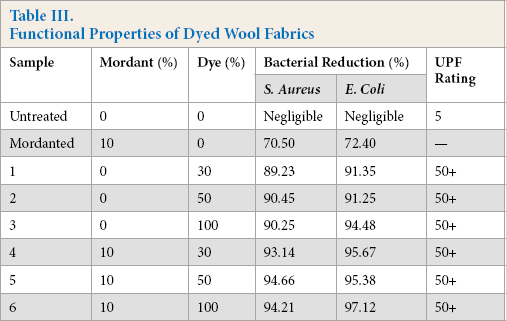
Pineapple peels are reported to contain ferulic acid, which is known to possess antimicrobial, anti-inflammatory, antioxidant, and UV absorber activities.31-35 The presence of gallic acid, catechin, and epicatechin, along with ferulic acid, in pineapple peels was reported. 36 The antibacterial activity showed only a marginal increase in the presence of the ferrous mordant. The mordanted wool (without dyeing) showed a bacterial reduction of greater than 70%, which, after washing, was reduced to unsatisfactory levels (less than 40%). After dyeing of mordanted samples, an increased level of bacterial reduction was achieved. The wool fabric dyed without pre-mordanting also displayed excellent antibacterial activity. This indicated the superior role of biomolecules in imparting antibacterial properties to the dyed wool fabrics. However, for dyed samples with pre-mordanting, the dye-mordant complex also helped to impart superior functional properties to the fabric.
Results in Table III also indicated the excellent UV protection offered by the dyed wool fabrics, both with and without mordant. This might be attributed to the presence of a number of phenolic compounds in PPE. The transition between pineapple peel and pulp is not clearly defined; compounds from pulp might be present in peel extract. However, the presence of several biomolecules in the peel extracts was reported earlier. The important ones are isomeric N,N′-diferuloylspermidines and hydroxycinnamoyl(di)glycerides. Mono- and di-feruloyl-spermidine, as well as several other hydroxycinnamic acid amides, were previously identified as constituents of pineapple plants. 37 The hydroxycinnamoylspermidines, which were reported as phenolic constituents of pollen and reproductive organs in various plant species, may be involved in the UV protection of the nucleic acids. 38 The hydroxycinnamo-ylspermidines in the pineapple peel also contribute to the UV protection of the fruit. Apart from these, the presence of flavonoids, which generally show absorption maxima in the UV region, impart photo-protection. The UV-Visible spectrum of PPE extract (Fig. 1) showed strong absorption in UV region (λmax = 305 nm), which also confirmed the excellent UV protection offered by dyed samples.
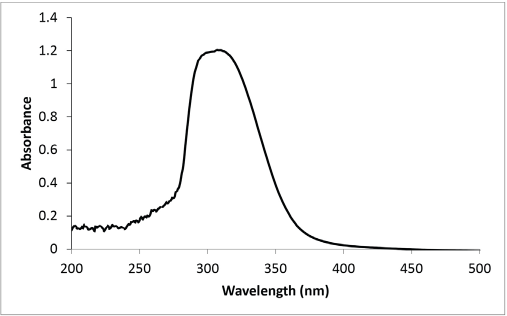
UV-Visible spectrum of PPE.
For the pre-mordanted samples, the complex formed between ferrous ion and PPE biomolecules resulted in enhanced UV protection. The values for all dyed samples remained in the excellent range.
Antioxidant activity of dyed wool fabrics was evaluated using radical scavenging activity against DPPH radical. Results are summarized in Fig. 2. In general, all dyed samples displayed excellent antioxidant activity. The samples dyed without mordant showed distinctly better antioxi-dant activity when compared to that of samples dyed using ferrous mordant. This may be due to the availability of un-complexed biomolecules for radical scavenging for samples dyed without ferrous mordant. Although samples dyed with ferrous mordant showed higher color values, the biomol-ecules were complexed with ferrous ion, affecting their availability for radical scavenging. The biomolecules, especially flavonoids and the various polyphenolic compounds, are known for their antioxidant activities. The results obtained in the present research validated the capability of such biomolecules in imparting various functional properties to wool fabric.
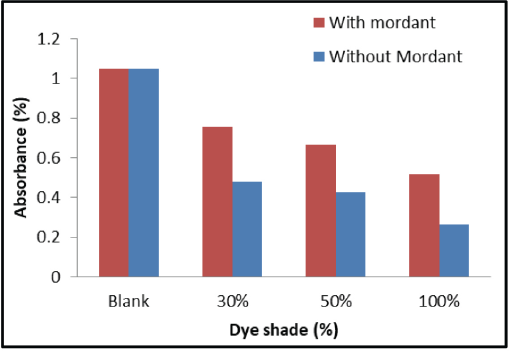
Absorbance of DPPH radical in presence of wool samples.
Characterization
To confirm the presence of biomolecules from PPE on linen fabric, FTIR-ATR spectra of undyed and dyed wool fabrics were recorded (Fig. 3). The FTIR-ATR spectrum of the dyed sample showed introduction of distinct peaks in the fingerprint region. The polyphenolic compounds are rich in hydroxyl groups and are present in the dyed wool, mainly complexed with mordant. The introduction of a peak at 3276 cm−1 indicated the rich presence of hydroxyl groups. A distinct peak at 1712 cm−1 identified in dyed wool indicated the presence of carboxylic acid groups. This confirmed the introduction of acidic biomolecules (e.g., ferulic acid) and polyphenolic compounds in the dyed fabrics. The FTIR-ATR spectrum of dyed wool showed low intensity peaks along with the shifting of some peaks, thereby indicating the interaction between PPE, wool, and mordant. Introduction of new peaks at 867 and 719 cm−1 were assigned to the out-of-plane deformation of aromatic phenolic compounds.
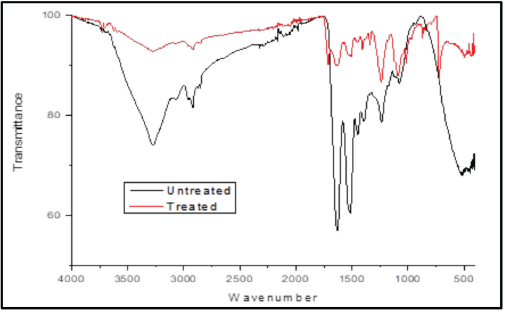
FTIR-ATR spectra of undyed and dyed wool fabrics.
Conclusion
The biomolecules extracted from pineapple peel waste were successfully used for functional coloration of wool fabric. The dyed wool displayed satisfactory coloration along with acceptable fastness ratings and efficient functional properties. The excellent functional properties, including antibacterial, antioxidant, and UV protection were correlated with the introduction of biomolecules using FTIR-ATR spectroscopy. The first successful application of pineapple peel waste material was reported for functionalization of wool protein fabrics.
Footnotes
Acknowledgement
The authors gratefully acknowledge the Science and Engineering Research Board (SERB, India) for ECRA funding (project file no. ECR/2017/001041).
