Abstract
In the production of silver-enabled textiles, the amount of silver in the produced textile is always important. The goal of the present study is to develop a simple device that can directly detect silver instead of using destructive tests and calculations. The instrument developed by our group is simple, easy to use, and portable. It gives a voltage response corresponding to the silver content. To show its capabilities, a correlation is presented between the measured voltage and the relative amount of metal (i.e., silver) on silver-enabled textiles after several washing and electrolysis steps. The results of scanning electron microscopy-energy dispersive X-ray analysis (SEM-EDX) measurements are also presented as a reference for the silver determinations.
Introduction
Expectations of clothing fabrics have changed in the modern era. It is not enough for clothes to fulfill their traditional roles (protecting, decorating, and distinguishing). Depending on their different functions, fabrics today have to meet new demands. Today's clothing is expected to be sweat managing,1,2 or water-proof,3–5 quick-drying,6,7 easy-to-use, and dirt-, 3 water-, and oil-repellent. It should also provide UV8–10 and antimicrobial protection,11–13 while home textiles need to be flame resistant. 14
The growth of microbes on textiles can be prevented with an antimicrobial coating or by mixing biocides into the synthetic fibers during extrusion. 15 Silver and nanosilver are widely applied for their antimicrobial, antifungal, anti-odor, and partially antiviral properties in various products (i.e., packaging materials,16,17 paper, 18 and textiles 19 ). It was shown that textiles coated with silver nanoparticles have long lasting antimicrobial properties 11 , 20–22 Bacterial resistance to elemental silver is exceptionally rare. 23
Numerous methods exist to apply nanoparticles to textiles (e.g., spraying, printing, dipping, steaming, and impregnation). Because of its simplicity, the most widely-used method is impregnation, where the textile is treated with a solution containing nanoparticles, then it is dried.5,24
In the production of silver-enabled textiles, silver can be incorporated into the fibers in three ways: by applying a film-like coating to the surface of the fibers, by application of silver nanoparticles to the textile surface, and by the addition of silver particles to the liquid mass during the fiber spinning process.
Silver nanoparticles are most effective for antimicrobial textile preparation because of the high surface/volume fraction, resulting in a large proportion of silver atoms in direct contact with their environment, 25 making their interaction with bacteria easier. When silver nanoparticles are dissolved in water or when they get into the cells, cationic silver is released. 26 As a result, nanoparticles have better antimicrobial properties than free silver ions, because the antibacterial properties are due to both the physical properties of the nanoparticles and the release of silver ions. 27
The widespread use of silver nanoparticles in commercially-available products, especially in textiles, will most probably lead to an unprecedented spread of silver in the environment. Many studies have focused on the effects and possible risks of silver on humans and on the environment.28–33 To determine the environmental effect of silver nanoparticles, the measurement and description of silver released from silver-enabled products is important. A model study found that biocidal plastics and textiles were responsible for 15% of the silver released into the environment. Taking into consideration the life cycle of nano products, the concentration of silver nanoparticles in the aqueous environment was 0.03 μg/L. 34
The present work aims to show the capability of our newly-developed metal detecting device to measure the relative quantity of silver on textiles in a non-destructive way. As a reference, the silver content was also characterized by scanning electron microscopy-energy dispersive X-ray analysis (SEM-EDX) and conductivity measurements. All samples in the present study were commercially-available textiles. According to their labels, the textiles contained silver-coated fibers or nanosilver. The samples were studied as received, then after silver removal by electrolysis, and after several washing steps as well.
The washing experiments were carried out to model the everyday washing load of the textiles. Electrolysis was used to detect the relative residual silver amount left after heavy use. The measurement of textile resistance was performed to study the breaks in the silver coating on the fiber, and thus the break in the electrical resistance. The manual metal detector for the textile industry was used to show the changes in the amount of metal on the textiles.
EDX analysis was used to determine the elemental composition of the single fibers forming the textile pattern. At the same time, the type of metal (silver) was also detected by EDX.
Experimental
Materials
Samples A and B are commercially-available textiles made of fibers with a continuous silver coating. Samples C and D are commercially-available nanosilver containing textiles based on the manufacturer's description. Their properties are given in Table I and Fig. 1.
Weight, Composition, and Knitting Pattern of Samples A-D


Photos of samples A–D.
Methods
Washing
The samples were tested after 0, 5, and 10 washing cycles. Washing was performed according to the MSZ EN ISO 6330:2001 standard; 35 the washing was done in a vertical drum machine, using a delicate program at 30 ± 3 °C for 30 min, and detergent without optical brightener (Tomi Color (Henkel)) (to avoid introduction of extraneous metals) was used. The samples were air dried, and they were conditioned at 20 ± 2 °C and 65 ± 4% RH before the tests. During the washing, no hydrogen peroxide, peracetic acid, or chlorinated detergents were used, as these can accelerate the release of silver. 36
Silver Removal by Electrolysis
Electrolysis was used for the removal of silver from the silver-coated fibers. Electrolysis simulated the silver residue after heavy textile use. The electrolysis cell contained an electrolyte solution of distilled water and vinegar (the concentration of the solution was 270 ppm, which was equivalent to a conductivity of 0.5 mS/cm), into which two electrodes were put 2 cm from each other. A power source of 4.5 V was applied, and a silver-coated fabric was placed on the anode. The sample was removed from the electrolysis cell after 12 h, was rinsed with water, and then tested after drying and conditioning.
Resistance of Silver-Enabled Textiles
The metal coating on the surface of the fibers forms a continuous layer; thus, the fibers become conductive, and can be used for making e-textiles, portable electronic devices, or smart clothing. If the silver-coated fibers are knit or woven into textiles, or the textiles are sewn with such a thread, these textiles will also have conductivity, and their resistance is measurable until the fibers or the metal coating on the fibers is broken. The coating can break during washing, use, or wear, resulting in the textile losing its conductivity. 37
The resistance of the textiles was measured using an Ohm meter, after laying and pressing the textile between two plexiglass plates on a 15 × 15-cm stretch. The measurements were repeated 10 times on 10 different stretches, and an average was calculated.
Metal Detection Testing of the Silver Enabled Textiles
For the detection of metal particles present in the textiles, an absorption-based metal detector was used (Fig. 2). The metal detector was based on an LC blocking oscillator that operates on the frequency of about 1 MHz. The operating current of the oscillator was reduced until the limit of vibration was reached. Approach of a metal object to the detection coil drew energy from the resonant circuit and the amplitude of the vibration was reduced. This energy loss was significant because of the eddy currents induced in the metal objects. The LED light and beeper connected to the switching and amplifier circuit indicated the weakening in the operation of the oscillator by strengthening the light and sound intensity; in addition, the change was also measurable numerically with the connected voltmeter.

Photo of the new metal detector.
The porous structure of the metal-coated textile prevented the formation of high intensity eddy currents, but they still distorted the magnetic field lines around the coil, so the amplitude of vibration of the oscillator was measurably reduced. 37
The measurements were performed on textile samples placed between two plexiglass plates. Each sample was measured four times by rotating the samples after each measurement, and an average was calculated.
Scanning Electron Microscopy with Energy Dispersive X-ray Analysis (SEM-EDS)
SEM-EDX is a coupled technique used for investigating the elemental composition of the sample by detecting the characteristic X-ray photons of the atoms created by the incident electron beam. This method is commonly used for detecting metal particles.25,38–40
SEM-EDX analysis of the samples was performed using a JEOL JSM-5500LV instrument. To prevent charging, the samples were coated with a gold-palladium (Au-Pd) conductive layer (∼9-nm thickness). A secondary electron detector was used for the microscopic images. EDX measurements were performed on three different sections of three different portions on each sample (i.e., the composition of nine single fibers was tested for each textile). The average of the nine results was then calculated.
Results
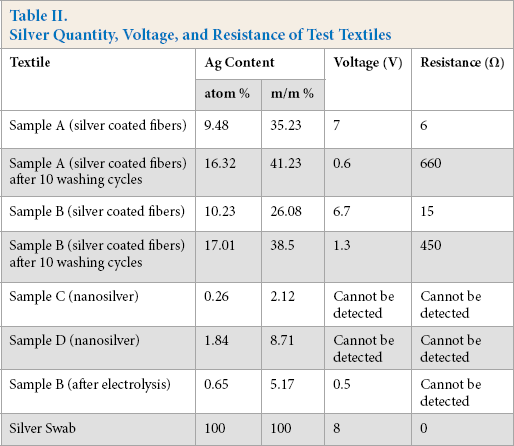
Resistance and metal detection tests were performed on the test textiles that contained fibers with a continuous silver coating (samples A and B, Table II).
Silver Quantity, Voltage, and Resistance of Test Textiles
The SEM image of the silver coated fibers of sample B (Fig. 3) shows the cross section of the fibers. Due to the continuous silver coating on the surface of the fibers, clear contours can be seen.

Cross section of the silver-coated PA fibers of sample B at 700× magnification.
On samples A and B, resistance tests were performed to monitor the decrease in silver content after various washing steps. These were tested after 10 and 20 washing cycles. The metal detector indicated the presence of metal and the alternating voltage on the voltmeter indicated the change in the quantity of silver on the textile.
The absorption-based metal detector was used for the detection of metal particles in textiles. It is of simple construction and it can detect any kind of small metallic particles. The operating principle of the absorption-based (blocking oscillator) metal detector is based on the eddy current induced in metals and the magnetic energy loss created in the search coil. The signal of the search coil is provided by a low-power, high-frequency oscillator. The field created by this coil produces high-loss eddy currents. As a result of the energy withdrawal, the amplitude of the vibration is reduced, or if a big piece of metal is too close, detection is stopped. After filtering out external interfering radio signals, the reduced vibration amplitude is detected by a voltage comparator IC, the output of which gives a signal in case there is a deviation.
Based on measurements of resistance and voltage vs. the number of washings, a polynomial trend line can be fitted to the voltage data series and a linear trend line to the resistance data series of both samples A and B (Figs. 4 and 5).
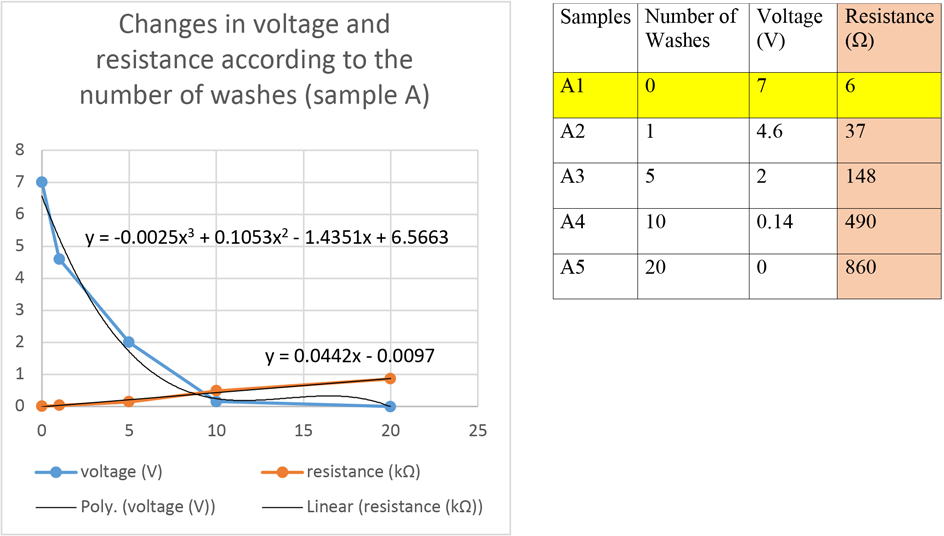
Results of voltage and resistance measurements on sample A after various washing steps.
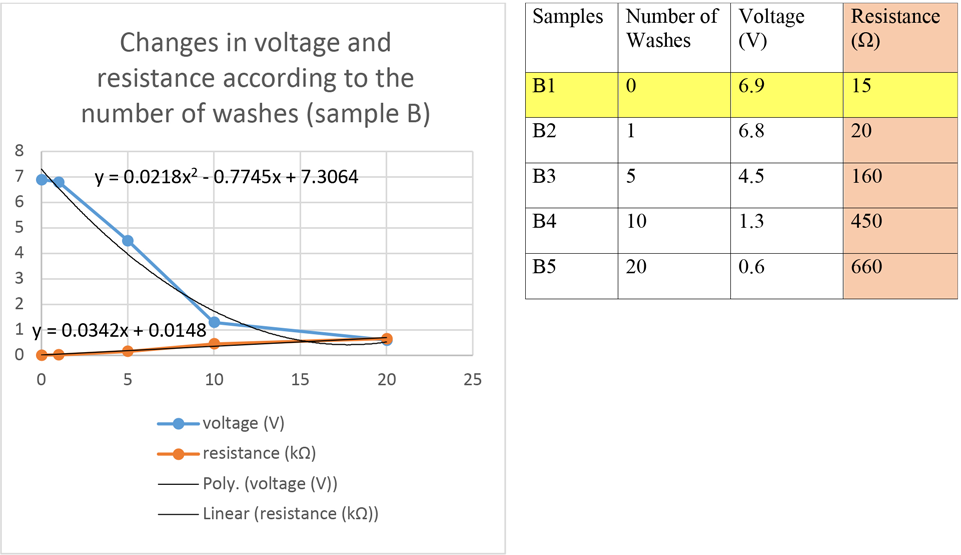
Results of the voltage and resistance measurements on sample B after various washing steps.
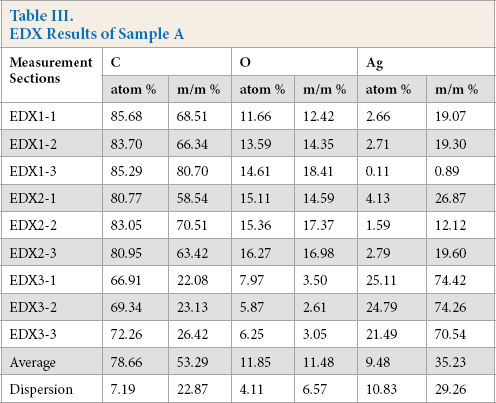
The continuous silver coating can be seen on the silver-coated polyamide (PA) fiber on the SEM image of sample A (Fig. 6). EDX analysis was used to determine the elemental composition of the single fibers forming the textile pattern, by performing measurements on three different sections of three different portions of each sample, thus testing the composition of nine single fibers. The average of the nine results was then calculated, characterizing the silver containing pattern (Fig. 7, Table III).
EDX Results of Sample A

SEM images of sample A, number of washes: 0.

SEM images of sample A, number of washes: 0. The squares indicate the location of the EDX test points.
The silver coating from the fibers in sample A was removed by electrolysis. Fig. 8 shows the fibers after 12 h of electrolysis. After electrolysis, the smooth, continuous silver coating on the fibers of sample A was broken and silver particles remained on the surface. Fig. 9 and Table IV show the nine sections where the composition of the sample A fibers were analyzed by EDX. The poles of the electrodes in the electrolysis cell were swabbed.
EDX Results for Sample A after Electrolysis
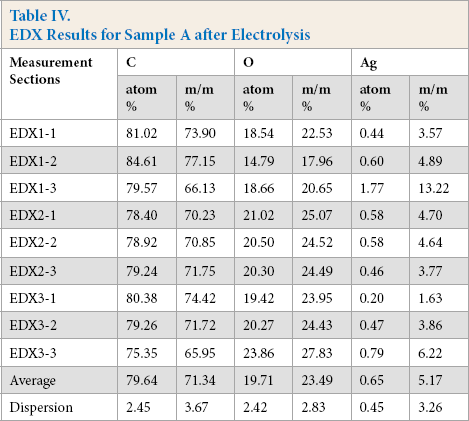

SEM images of sample A after 12 h of electrolysis.

SEM images of sample A after 12 h of electrolysis, the squares indicate the sections of EDX analysis.
The swab was analyzed by EDX and the metal content was determined to be 100% silver for all nine portions tested (Fig. 10).

SEM images of Ag swab of the electrode after sample A electrolysis.
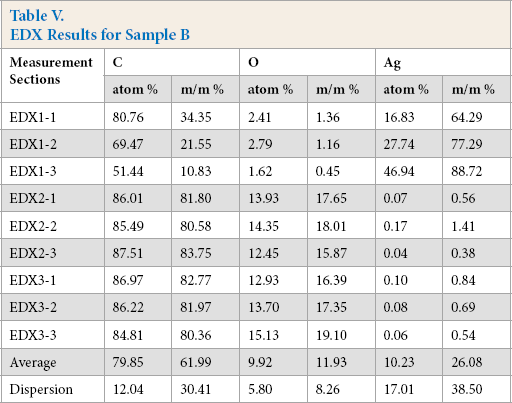
On the SEM image of sample B, the continuous silver coating can be seen on the PA fiber (Fig. 11). The sample was tested at nine different sections using SEM-EDX (Fig. 12, Table V). Sample B was tested after 10 and 20 washing cycles. (Figs. 13 and 14). The silver coating was significantly damaged and there were particles on the fibers that did not have silver coatings.
EDX Results for Sample B

SEM images of sample B at 150×, 1100×, and 3500× magnification, number of washes: 0.

SEM images of sample B. The squares indicate the sections of EDX analysis, number of washes: 0.

SEM images of sample B after 10 washing cycles at 150×, 1100 ×, and 2500× magnification.

SEM images of sample B after 20 washing cycles at 150× and 1100 × magnification.
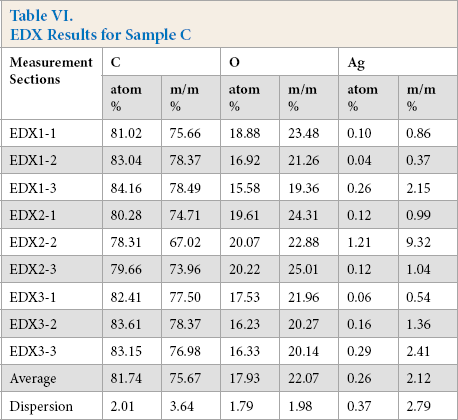
On the SEM image of sample C, no continuous silver coating was detected (Fig. 15). On the nine sections indicated in Fig. 16, the composition of the sample C fibers were tested with EDX. The results are shown in Table VI.
EDX Results for Sample C

SEM image of sample C at 150× and 1100× magnification.

SEM images of sample C. The squares indicate the sections of EDX analysis.
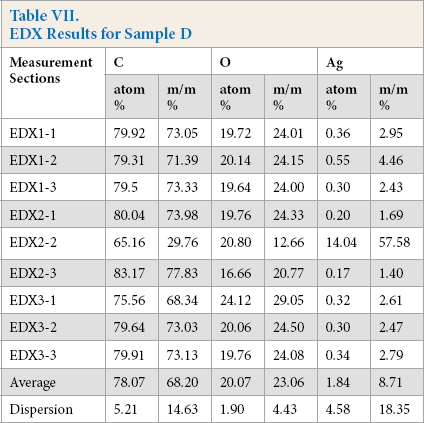
On the SEM image of sample D, no continuous silver coating was detected (Fig. 17); the typical image of cotton, resembling a twisted ribbon, can be seen. Sample D was also analyzed with EDX. The results are shown in Fig. 18 and Table VII.
EDX Results for Sample D

SEM images of sample D at 300×, 1100×, and 1500× magnification.

SEM images of sample D. The squares indicate the sections of EDX analysis.
For both samples C and D, the amount of silver present on these nanosilver containing textiles were below the detection limit of the newly-developed metal detector.
Conclusion
Although use of electrolysis was intended to remove the continuous silver coating from the silver-coated fibers, silver still remained based on the EDX analysis. After 12 h of electrolysis, the quantity of silver in weight percent was about the same as the quantity of silver in any nanosilver textile.
Based on our measurements, we found that the textiles with continuous silver coatings tested contained more silver after 20 washings than nano silver textiles at the moment of purchase. SEM-EDX can show and measure the uneven distribution of the silver particles on the whole surface of the textile. After 10–20 washing cycles, the continuous silver coating wore of and its continuity was broken. The metal detecting method can show the decrease of voltage and increase of resistance. Thus, the reduction in the quantity of silver was detectable by this method.
The measurement range of the newly-developed metal detector was 0 to 9, with 9 indicating pure metal. Textiles with a continuous silver coating give a ∼7, while 1 means 15 atom % and 35 mass %. These tests show that our metal detecting method can show the change in the quantity of metal. It is a portable device that can be easily used even under industrial conditions without sample destruction.
Footnotes
Acknowledgements
An OTKA-PD-109129 grant is acknowledged. I. M. S. is grateful for receiving a János Bolyai Research Fellowship of the Hungarian Academy of Sciences.
