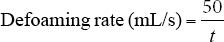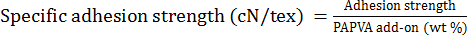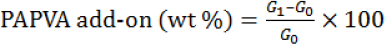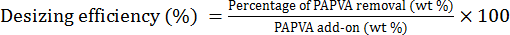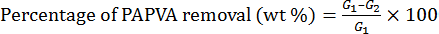Abstract
Alkyl alcohols were evaluated as defoamers to eliminate the foam produced by partially alcoholyzed polyvinyl alcohol (PAPVA) according to their defoaming ability and actual influence on adhesion to fibers, film properties, and desizability. The alcohols considered included 1-pentanol, 1-hexanol, 1-heptanol, 1-octanol, 1-nonanol, 1-decanol, isooctyl alcohol, and 2-octanol. The impact of alkyl alcohols on defoaming performance was examined and an acceptable dose of 1-octanol was determined. Results showed that defoaming ability, adhesion to fibers, and film behaviors depended on the alkyl group and dose of alcohols used. Straight alcohols were superior to the branched ones tested in defoaming ability and properties. Generally, 1-octanol was the best defoamer of the selected alcohols. Excessive increases in alkyl length and dose induced poor defoaming and damaged the adhesion and film behaviors.
Introduction
Polyvinyl alcohol (PVA) has been regarded as one of the most effective and versatile ingredients for warp sizing,1,2 since good results are always achieved and many difficulties in warp sizing can be overcome when it is used. Moreover, it can be used alone or in blends with other sizes (e.g., starches, acrylic copolymers, and polyester resins) for sizing a large variety of warp yarns.
Currently, PVA is an essential ingredient for fine, high density, and synthetic fiber warps. 3 PVA is manufactured via two steps: addition polymerization of vinyl acetate and alcoholysis of the resulting polyvinyl acetate. 4 By controlling the degree of alcoholysis of acetate pendant groups in the second step, a series of PVA grades can be obtained to meet various specific sizing needs. 5
The acetate pendants randomly distributed on partially alcoholyzed PVA (PAPVA) chains act as wedges to prevent other neighboring chains from close association via inter-molecular hydrogen bonding, and substantially reduce the regular arrangement of molecular chains, thus facilitating the water penetration needed for rapid dissolution. 4 In addition, the pendants improve the specific adhesion of PAPVA to polyester and other hydrophobic fibers, 5 so as to improve the tensile strength of sized polyester yarns due to their greater affinity arising from the similarity in chemical structures and non-polarity of the pendants and ester groups of the fibers. Furthermore, PAPVA is significantly weaker than fully alcoholyzed PVA in the tensile strength of the adhesive layers and can actually decrease the force needed to separate sized yarns at the bust and lease rods. This can result in a measurable decrease in yarn hairiness and clinging in the weaving loom, a key requirement for air-jet weaving. 4
For these reasons, PAPVA is a suitable candidate for sizing a large variety of hydrophobic fiber warps. However, the use of PAPVA in warp sizing often creates a massive amount of foam, which has negative impacts on the quality of sized yarns and the operation of the sizing process.6,7 This is due to the fact that the pendants distributed on molecular chains impart surface activity to the PVA. Consequently, PAPVA can stabilize bubbles which accumulate at the liquid surface as foam.
Although this is useful to improve the wetting, spreading, and impregnation of size mixtures, the accumulated foam in size mixtures causes many problems in the sizing operation. Once foam is present, sizing control becomes more difficult as the foam leads to incomplete mixing and incorrect dosing in size mixtures, generates overflow from size mixers, and results in non-sized area on the surfaces of sized yarns. Furthermore, the foam affects size add-on by reducing the amount of sizes applied to warp yarns, and the bubbles trapped inside sizing film on the surface of the yarns will spoil its protective qualities to sized yarns, thereby substantially decreasing the qualities of size mixtures and sized yarns. 7
It is a common practice to eliminate existing foam from size mixtures and prevent the mixtures from foaming by adding some defoamers. However, it is still a challenge to find a chemical defoamer that is available and that gives a desirable performance to get rid of foam generated by PAPVA in the sizing process. Therefore, defoamers were examined and tested for disrupting and breaking up the unwanted foam generated by PAPVA in warp sizing.
Generally, defoamers are surface-active agents with low hydrophile-lipophile balance (HLB) values. 5 They are substances for specific use, without versatility. 7 Also, the defoamer chosen in this work must be able to eliminate foam without causing excessive side effects. For this reason, it is advisable to develop the defoamer not only based on its defoaming ability, but also according to its effects on sizing performances of PAPVA such as adhesion to fibers, film behaviors, and desizability.
Alkyl alcohols are defoamers with potential uses in fermentation, 8 drainage, 9 and the oil industry. 10 However, there are no literature studies on the effects of the defoamers on their performance on foam generated by PAPVA, as well as on the adhesion, film behaviors, and desizing ability of PAPVA in sizing applications.
Therefore, the effect of number of carbon atoms in alkyl alcohol chains on defoaming the foam produced by PAPVA during warp sizing needed studying. The organic alcohol with best performance properties (i.e., defoaming ability, specific adhesion, film properties, and desizability of PAPVA), among select alcohols with 5–10 carbon alkyl chains, was chosen. Furthermore, the dose of the alcohol used was also examined according to the impacts on the same performance properties.
Materials and Methods
Reagents and Yarns
PVA 0588 (500 in degree of polymerization and 88% mole in degree of alcoholysis) was an industrially-produced product provided by Sinopec Sichuan Vinylon Works. The alcohols tested (1-pentanol, 1-hexanol, 1-heptanol, 1-octanol, 1-nonanol, 1-decanol, isooctyl alcohol, and 2-octanol) were chemically pure reagents obtained from Sinopharm Chemical Reagent Co. Ltd. Hydrogen peroxide (30%, mass fraction), hydrochloric acid, and sodium hydroxide were of analytical grades and were supplied by Sinopharm Chemical Reagent Co. Ltd.. The JFC (Haishihua, fatty alcohol polyoxyethylene ether, 12∼18 carbon atoms in carbon chains and 15∼20 eth-ylene units in polyethethylene) used as penetrating agent in desizing trials was supplied by the Haian Petrochemical Plant. Pure cotton roving (460 tex, 112 in twist factor) was supplied by the Wuxi Talak Factory. All polyester roving (396 tex, 49.8 in twist factor) was obtained from Yizheng Chemical Fiber Co. Ltd. The average lengths and linear densities of the fibers were 29 mm and 1.72 dtex for cotton and 38 mm and 1.73 dtex for polyester, respectively. Cotton yarns (ring spun yarn, 28 tex) used for sizing and desizing trials were supplied by the Wuxi Talak Cotton Factory.
Defoaming Ability
The defoaming ability of the alcohols on PAPVA solutions was evaluated in terms of defoaming time and defoaming rate. The defoaming rate 11 is a rate of foam removal and was measured as follows: a 250-mL graduated cylinder was used to record foam height. The PAPVA solution (30 mL) used as the foaming liquid was added into the cylinder and shaken vigorously to produce 70 mL of foam (total volume of foam and PAPVA solution was 100 mL). Then, 0.45 mL of defoamer (prepared by mixing 0.5 g of the alkyl alcohol into 25 mL of 0.25% JFC solution under mechanical agitation for 8 min at room temperature) was added into the foamed PAPVA solution and time measurements were started. Time recording was ended at the time when foamed PAPVA solution decreased by 50 mL, corresponding to the removal of 50 mL of foam. Five runs were made with each solution and their mean time was used to calculate defoaming rate in accordance with Eq. 1.
t (s) is the time required for the removal of 50 mL of foam.
Specific Adhesion Strength
Specific adhesion strength of PAPVA to cotton and polyester fibers were measured according to FZ/T 15001-2008, 12 a legal method regulated by the China Cotton Textile Association. Briefly, 9 g (dry base) of PAPVA was dissolved in 900 mL of distilled water under mechanical stirring at 95 °C for 1 h. The PAPVA solution obtained was transferred into a stainless-steel container and kept at 95 °C. The roving that was carefully wound onto a frame was impregnated with the solution for 5 min. Afterwards, the impregnated roving was dried in air at room temperature (RT) and collected for mechanical measurement. Mechanical measurement was performed on a HD206 Electronic Fabrics Strength Tester (Nantong Hongda Experimental Instrument Co. Ltd.), and operated at a crosshead speed of 50 mm/min with an initial chuck distance of 100 mm, under standard conditions (65 ± 3% relative humidity (RH) and 20 ± 1 °C) after the roving had been equilibrated under the same standard conditions for 24 h. In each case, 30 samples were measured and their average values were reported after outlying values had been rejected by statistical analysis. 13
Specific adhesion strength was defined as a standard of adhesion strength produced by 1 wt % of PAPVA add-on (i.e., the adhesion strength divided by percent PAPVA addon, as given by Eq. 2. 14
Mechanical Properties of PVA Film
PVA film was cast from PAPVA solution (400 mL, 6% w/w) by dissolving PAPVA in water under mechanical stirring at 95 °C for 2 h) onto a polyester film (650 × 400 mm) spread on a glass plate and dried under standard conditions. The film cast was cut into 10 × 200 mm strips and kept under standard conditions as mentioned previously for 24 h. Then, the strips were mechanically tested on a Zwick/Z2.5 Tensile Tester running at a stretching speed of 50 mm/min with a clamping distance of 100 mm. For each set of data, 20 samples were measured to obtain their mean tensile strength, breaking elongation, and work-to-break. 15
X-ray diffraction patterns of PAPVA film were recorded using a D8 X-ray diffractometer (Bruker AXS GmbH). The sample was exposed to Ni-filtered Cu Ka radiation with an X-ray generator running at 40 kV and 30 mA. The data were collected over the 2θ angular range from 3° to 60° with a speed of 4 °/min and an angular step of 0.01°.
Hank Sizing and Desizing
Hank sizing and desizing were performed to evaluate the influence of alcohol defoamers on the desizability of PAPVA from sized cotton yarns. The sizing operation included two steps: PAPVA dissolving and yarn sizing. Nine parts (dry basis) of PAPVA were dispersed into 141 parts of distilled water. The dispersion was heated to 95 °C and mechanically stirred at the temperature for 2 h. Tree parts (dry basis) of hank yarns were immersed in the PAPVA solution for 20 min. Then, the immersed yarns were placed in an open-meshed fabric bag and excessive PAPVA solution was squeezed of manually. Finally, the squeezed yarns were hung up and dried in air. The PVA add-on was calculated using Eq. 3.
G0 and G1 are the weights of the yarns before and after sizing, respectively.
A desizing trial was performed by cooking sized yarns in desizing liquor at 100 °C for 30 min at a liquor ratio (LR) of 1:50. Doses of the chemicals in the desizing liquor were 2 g/L of NaOH, 4 mL/L of H2O2, and 2 g/L of JFC penetrating agent. After cooking, the yarns were washed twice with hot distilled water and dried in air. Two individual tests were carried out and their mean values were used to calculate the desizing efficiency of PAPVA using Eqs. 4 and 5. 16
G2 is the weight of desized yarns.
The time required for breaking the PAPVA film in water was determined to assess the impact of alcohol defoamers on the desizability of PAPVA. The time denoted the period required to break the PAPVA film in hot water.13,17 The film was cast and then was cut into strips (100 × 20 mm) and the strips were kept under standard conditions for 24 h before being tested. A dead weight of 0.5 g was fixed at one end of each strip to create a tension of 490 dynes for stretching the film in water during the test. Half of the strip with dead weight was saturated with hot water at 80 °C. 13 The time required for breaking PAPVA film was determined by measuring a period beginning from the time when one end the strip was put into water and ending at the time when the strip was broken. The average values reported were 10 successful tests, with outlying values rejected by statistical analysis.
Results and Discussion
Effect of Alkyl Alcohol
The effect of the alkyl chain length of the alcohols tested on their defoaming ability in the PAPVA solution was measured, as shown Table I. The defoaming time decreased as the alcohol chain length initially increased, reached a minimum of 2.57 s with 1-octanol, and then sharply increased. The length of the alkyl chains played an important role in their defoaming rate. In addition, branched alcohols were inferior to the straight chain ones tested in defoaming ability. This suggested that 1-octanol was the best defoamer of the alcohols tested. Furthermore, for alcohols containing 5-10 alkyl carbon atoms, no foam occurred on the PAPVA solution after it had been mechanically agitated at RT for 30 min.
Effect of Alkyl Alcohol Chain Structure on Defoaming Ability in PAPVA Solutions a

Mass fraction of the alcohols was 0.5 wt % (vs. PAPVA).
Alkyl alcohols with one hydroxyl as hydrophilic group and five to ten carbon atoms in alkyl chain were used in this study. The hydroxyl group is able to associate with the hydrogen-bonding network in water. However, when it is attached to a hydrophobic group, its effect is diminished, or even eliminated, if the hydrophobic group is large enough.18 The alcohols considered in this work were relatively lipophilic and formed oil droplets in aqueous liquid since their HLB values were in the range of 3 to 6. 5
Chemical defoamers usually work by breaking the bubble film of foam to release the trapped air. 7 It was suggested that for efficient defoaming, the oil droplets must enter air-water interfaces and spread as a duplex film on each side of the original film. 9 This acts to drive out the original liquid film and leaves an oil film which is unstable and can easily break.19,20 The defoaming ability of the alcohols are related to their solubility in water. The alcohols with shorter alkyl chains are miscible with water, where fewer alcohol molecules can be accumulated as oil droplets to enter the air-water interface and spread onto the liquid film of the foam. As the hydrophobic alkyl group becomes longer, water solubility declines and more alcohol molecules can form oil droplets for driving out foam film, thereby improving the foam removal. On the other hand, the alcohols are practically insoluble if the number of carbons is greater than 8. 18 For this reason, alcohols with more than eight carbons had a lower ability to disrupt and break the foam produced by PAPVA. This is consistent with Zhang's observation when he disrupted the foam produced by sodium dodecyl sulfate aqueous solution using alkyl alcohols. 21
The influence of alkyl chain of alcohols on specific adhesion strength of PAPVA to cotton and polyester fibers is demonstrated in Table II. The presence of the alcohols within the PAPVA reduced its adhesion to these two kinds of fibers. Regardless of the fiber type, the adhesion was sensitive to the alkyl chain length. It decreased consistently as the number of alkyl carbon atoms increased, with the exception of isooctyl alcohol on polyester. This observation suggests that the increase in length of the alkyl chain was deleterious to the adhesion of PAPVA to cotton and polyester fibers.
Effect of Alkyl Alcohol Chain Structure on Specific Adhesion Strength of PAPVA to Cotton and Polyester Fiber s a
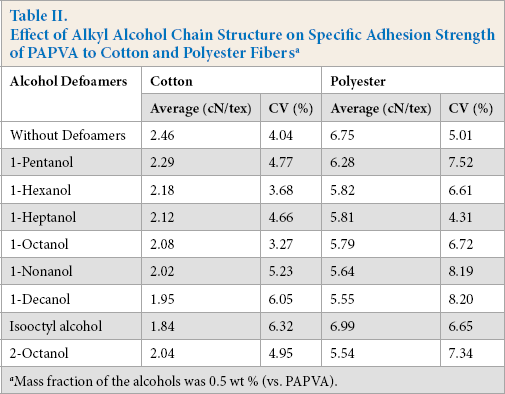
Mass fraction of the alcohols was 0.5 wt % (vs. PAPVA).
Alcohols with five to ten carbon atoms in the alkyl chain are lipophilic and can be adsorbed onto the surfaces of hydrophobic substrates. Polyester fibers have hydrophobic surfaces, and raw cotton contains hydrophobic wax on fiber surfaces3,4 and thus, in both cases, impart a hydro-phobic nature to the fiber surfaces. The hydrophobic alcohols adsorbed not only retard wetting, wicking, and adsorption of PAPVA solution onto fiber surfaces, but also subsequently induce weak boundary layers (i.e., the regions of low cohesive strength between fiber substrates and PAPVA adhesive layers).22,23 This concept was first proposed by Bikerman, 24 based on the presence of impurities and low molecular weight ingredients, which give rise to weak boundary layers. In many cases, a weak boundary layer is probably an accurate description of the cause of adhesive failure. 23 Weak boundary layers are deleterious to adhesion because it prevents compact contact between the adhesive and fiber. Therefore, it reduces intermolecular forces on adhesive layer-fiber interfaces, leading to adhesive failure at lower stresses, and thus adversely influencing the adhesion of PAPVA to fibers. As alcohol alkyl chains become longer, the water solubility of the alcohol decreases and more alcohol molecules may be adsorbed onto fiber surfaces, thereby creating weak boundary layers between adhesive and fiber, and thus reducing adhesion.
The effect of the alkyl chain of the alcohols on tensile properties of the film cast from PAPVA is shown in Table III. The presence of the alcohols was harmful to the film properties, depending on the length of alkyl chain. Tensile strength, breaking extension, and work-to-break of the film decreased as the length of the alkyl chain increased, thereby reducing the film properties. Table III also compares individual tensile strength, breaking extension, and work-to-break of the films blended with straight and branched octanol, with 1-octanol having the best film properties and the most effective defoaming ability.
Effect of Alkyl Alcohol Chain Structure on Mechanical Properties of Film Cast from PAPVA a
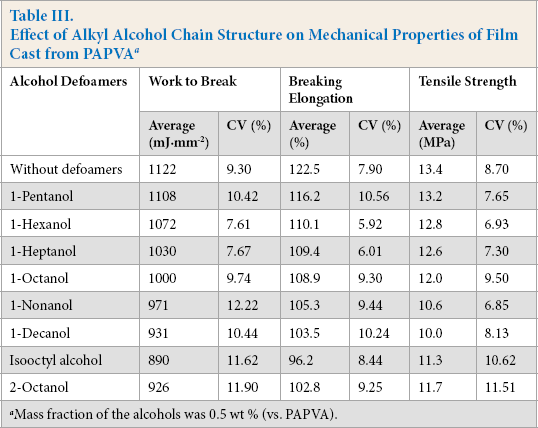
Mass fraction of the alcohols was 0.5 wt % (vs. PAPVA).
X-ray diffraction analysis of PAPVA films was performed to find out how the decreases in tensile properties of PAPVA film blended with the alcohols occurred (Fig. 1). It was surprising to find that the diffraction patterns were almost the same no matter what alcohols were present. The degrees of crystallinity for the films cast from pure PAPVA solution, and the solution blended with 1-hexanol and with 1-octanol were 8.92%, 8.87%, and 8.84%, respectively. This confirmed that, for such small amounts of alcohols (0.5 wt % vs. PAPVA) did not significantly alter the degree of crystallinity of the film. Additionally, alkyl alcohols and PAPVA are incompatible and will form an island-in-sea microstructure when blended together before casting. Due to their lower content, the alcohols turn into a dispersed phase, whereas PAPVA forms a continuous phase.25 The two ingredients blended together are incompatible and have little degree of segmental interdiffusion. Such interdiffusion of two ingredients at phase boundaries in blending films cannot support large load and extension. Accordingly, tensile properties of the film decrease after the alcohols are included within the films. With the increase in alkyl chain of the alcohols, the domain of the dispersed phase becomes greater due to increased hydrophobicity and reduced dispersibility of the alcohols in water. Therefore, the mechanical properties of the film decrease as the alkyl chain becomes longer.
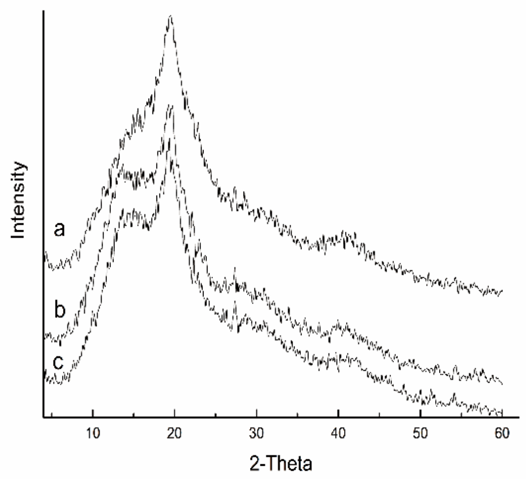
X-ray diffractograms of the films made from PVA 0588 incorporated with (a) no defoamer, (b) 1-hexanol, and (c) 1-octanol. (Mass fraction of the defoamers was 0.5 wt %, vs. PAPVA).
PVA sized onto warp yarns must be completely removed before dyeing, printing, and finishing. Accordingly, the effect of alkyl alcohols on the removal of PAPVA must be evaluated. Table IV shows the effect of the alkyl chain of the alcohol on the desizing efficiency of PAPVA. Alkali-oxidant desizing demonstrated that the alcohols did not present much effect on the desizing efficiency of PAPVA. The efficiency was relatively insensitive to the structure of the alkyl chain.
Effect of Alkyl Alcohol Chain Structure on Desizing Efficiency of PAPVA a
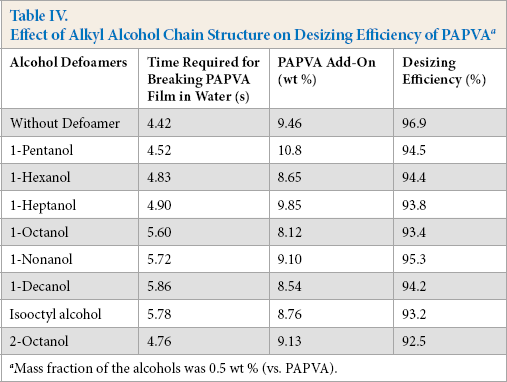
Mass fraction of the alcohols was 0.5 wt % (vs. PAPVA).
Oxidative desizing in textile pretreatment is based on the degradation of molecular chains by oxidation. The process uses oxidants to break the backbones of PAPVA as well as alkyl chains in the alcohols in a random manner. The oxidation reduces the length of both PAPVA and degrades alkyl alcohols. As a result, the alcohols did not present much effect on desizing efficiency of PAPVA. In fact, the degraded products can be easily washed away with hot water, thereby achieving relatively high desizability. This result allows desired alcohols to be chosen for their defoaming ability, as well as their end-use ability, such as adhesion-to-fibers and film properties.
Effect of 1-Octanol Dose
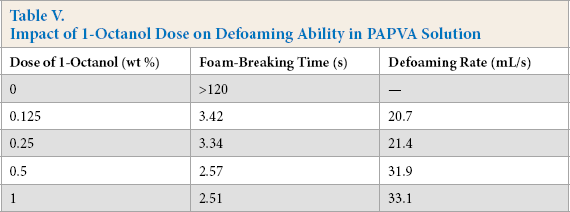
Based on the previous experimental results in this work, 1-octanol was found to be the best defoamer of the alcohols considered and did not present a significantly adverse influence upon the desizing of PAPVA. The alcohol could be added into the PAPVA solution or blended with PAPVA, although it had some adverse influences upon adhesion and film properties. The defoaming and end-use abilities of the alcohol defoamer are not only dependent on its alkyl structure, but also on the amount of the alcohol applied. Accordingly, the impacts of the 1-octanol dose on its defoaming and end-use abilities were also examined in this work. The dose range was from 0.01 wt % to 1.0 wt % (based on weight of dry PAPVA solids).
The influence of 1-octanol alcohol dose upon defoaming ability in PAPVA solution is shown in Table V. The defoaming time and defoaming rate were dependent on the dose. The time decreased, while the rate increased as the dose initially increased. This was mainly attributed to the amount of oil droplets formed in PAPVA solution. As the dose increases, the amount of 1-octa-nol droplets increases. Accordingly, there are more defoamer droplets that can enter the air-water interface of the foam and spread as a duplex film on liquid film of the foam, thus making the foam unstable and breaking the foam. Results suggested that the time and rate did not vary greatly when the dose exceeded 0.5 wt %. A relatively efficient removal of the foam could be achieved at the dose of 0.25 wt % to 0.5 wt %.
Impact of 1-Octanol Dose on Defoaming Ability in PAPVA Solution
The effect of 1-octanol dose on specific adhesion strength of PAPVA to cotton and polyester fibers is depicted in Fig. 2. Regardless of the fibers used as substrates, the strength depended on the dose of 1-octanol applied. A lower dose favored adhesion to cotton and polyester fibers. Diminished adhesion at higher doses was mainly attributed to the enhanced weak boundary layers on adhesive-fiber interfaces. An increased amount of the alcohol was able to increase area and thickness of the boundary layers, and thus prevent compact contact of the adhesive with fibers, reducing intermolecular forces on the interfaces, and adversely influencing the adhesion. Therefore, the dose of the defoamer must be as small as possible in warp sizing for achieving sufficient adhesion to fibers when 1-octanol is used as defoamer.
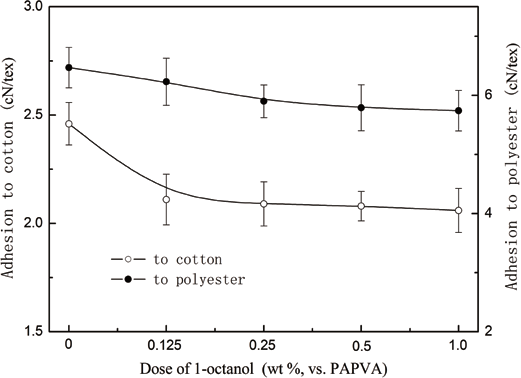
Effect of 1-octanol dose on specific adhesion strength of PAPVA to fibers.
The impact of 1-octanol dose on tensile properties of the film cast from PAPVA solution is presented in Table VI. In general, the dose determined the tensile strength, breaking elongation, and work-to-break of PAPVA film. The increase in the alcohol dose was unfavorable to the properties of PAPVA film. This was probably due to the increased amount and volume of dispersed phase of low molecular weight oil droplets in the continuous phase of the PAPVA major component. The amount and volume of dispersed phase increased with the increase in the dose, and thus enhancing the risk of interfacial failure between PAPVA and 1-octanol phases within the film. For this reason, tensile properties of the film decreased. Therefore, the alcohol used as defoamer for eliminating the foam produced by PAPVA solution must be minimized during warp sizing.
Influence of 1-Octanol Dose on Tensile Properties of Film Cast from PAPVA
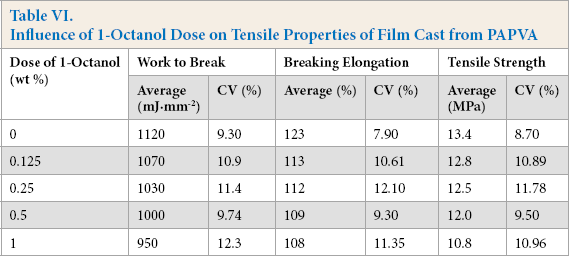
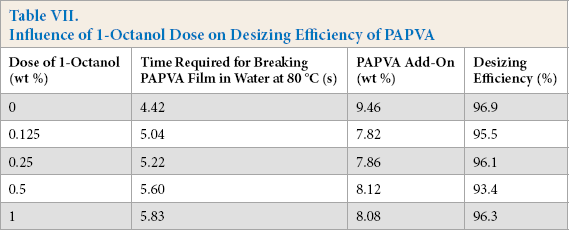
Table VII shows the influence of 1-octanol dose on PAPVA desizing efficiency. No dose effect was observed on the efficiency, although the time required for breaking PAPVA film in water at 80 °C decreased as the dose increased. As a result, it is possible to choose a required amount of the alcohol for effective defoaming without limitations to desizing.
Influence of 1-Octanol Dose on Desizing Efficiency of PAPVA
Conclusions
Based on the defoaming ability, adhesion of PAPVA to fibers, film properties, and desizability, 1-octanol was chosen as a suitable defoamer to be used in PAPVA size mixtures, since it gave the best results of the alcohols tested with the least detrimental defoamer-related effects. The length of the alkyl chain (5–10 carbon chains) in the alcohols had marked effects on defoaming ability, and showed substantial impact on the adhesion of PAPVA to fibers and the mechanical properties of PAPVA film. The defoaming ability of the alcohols increased as length of the alkyl chain initially increased, reached a maximum for 1-octanol, and then decreased. Straight chain alcohols were superior to the branched ones tested. This suggested that 1-octanol possessed the strongest defoaming ability of the alcohols considered in this work. In addition, the increase in length of the alkyl chain impaired adhesion of PAPVA to cotton and polyester fibers and reduced the tensile strength of PAPVA film. However, alkyl length did not show significant impact on the desizing efficiency of PAPVA. Furthermore, the amount of 1-octanol used also played an important role in defoaming ability, adhesion of PAPVA to fibers, and mechanical properties of PAPVA film. At higher amounts of 1-octanol, the defoaming ability increased, whereas the adhesion became weaker and the sizing film was less extensible and more likely to break. Therefore, the amount of 1-octanol used as defoamer was minimized. The desizing efficiency of PAPVA was completely insensitive to the amount of 1-octanol used. The preferred dose of 1-octanol as defoamer to remove the foam generated by PAPVA was found to be in the range of 0.25 wt % to 0.5 wt % (vs. PAPVA).