Abstract
The coupler 2-
Introduction
Azo disperse dyes have been studied widely for use as coloring materials, as photo responsive molecular switches due to their photo isomerization,1-3 materials for reversible optical information storage, 4 and nonlinear optics. 5 Ease of synthesis, low-cost efficient production, high color strength, and adjustable wide color range from yellow to blue-green are the advantages of azo disperse dyes.6-10 Azo dyes derived from thiazole have been reported for polyester fiber with good washfastness.10-13 Disperse dyes derived from 2-aminothiazoles have been studied, with the 2-amino-5-ni-trothiazole based blue dischargeable monoazo C.I. Disperse Blue 339 being commercially produced.14-16 2-Aminothia-zoles are also known as intermediates used in various drug syntheses.17-20 Thus, azo dyes obtained from 2-aminothiazole were investigated in recent years.18,20-27
We have synthesized five novel heterocyclic azo disperse dyes derived from 2-
Experimental
Materials and Methods
Aniline,
Synthesis and Characterization
Dyes
Synthesis of dyes
Synthesis of (E)-N ,N- Dibutyl-5-((4-nitrophenyl)diazenyl)-4-phenylthiazol-2-amine (1)
A solution of aryl diazonium chloride was prepared by adding cold (0-5 °C) sodium nitrite solution (7.37 mmol) to a cold (0-5 °C) suspension of
Synthesis of (E)-N ,N- Dibutyl-5-((4-chlorophenyl) diazenyl)-4-phenylthiazol-2-amine (2)
A diazonium chloride solution of
Synthesis of (E)-N ,N- Dibutyl-5-((4-methoxyphenyl) diazenyl)-4-phenylthiazol-2-amine (3)
A solution of
Synthesis of (E)-N ,N- dibutyl-4-phenyl-5-(p-tolyldiazenyl) thiazole-2-amine (4)
The procedure for synthesis of
Synthesis of (E)-N ,N- Dibutyl-4-phenyl-5-(phenyldiazenyl) thiazole-2-amine (5)
Aniline oil (10.95 mmol) was mixed in 2.3 mL of conc. HCl at 0-5 °C. A cold (0-5 °C) solution of sodium nitrate (11.05 mmol) was added dropwise to the amine solution at 0-5 °C and the mixture was stirred for 30 min at 0-5 °C. The 2-
Dyeing Procedure
Disperse dyeing of polyester and nylon fabrics with 1.5% synthesized dyes
Results and Discussion
Synthesis of Azo Dyes
Novel 2-
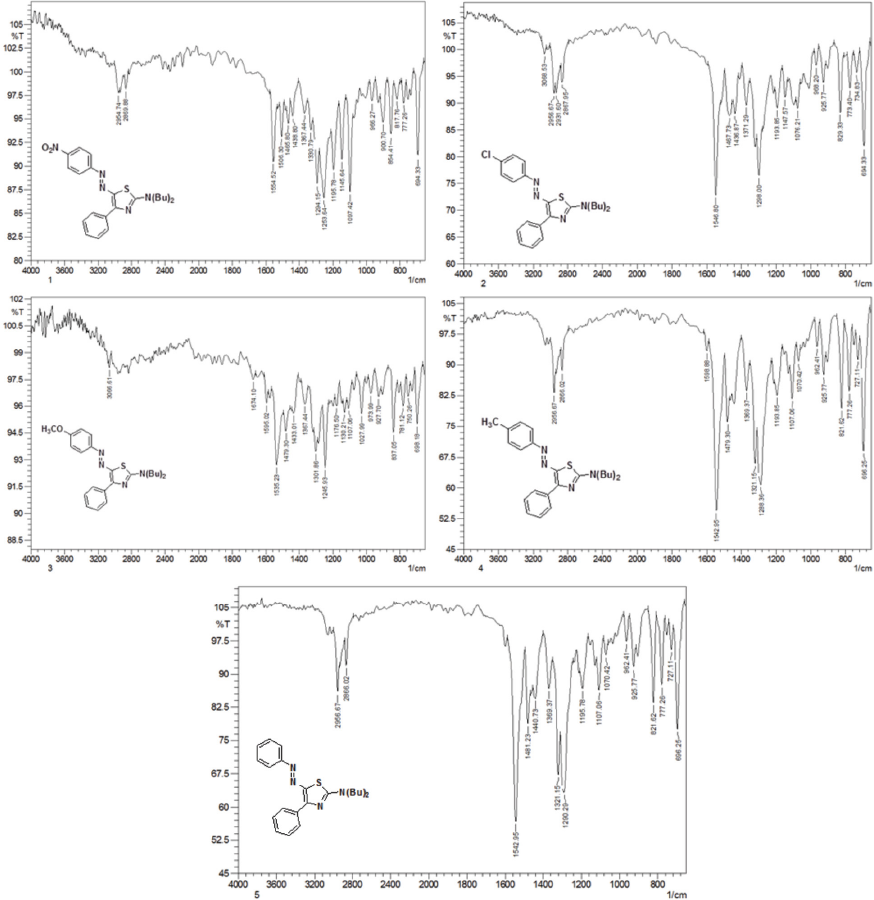
FTIR spectra of dyes
Photophysical Properties
The photophysical properties of 2 μM solutions of disperse dyes
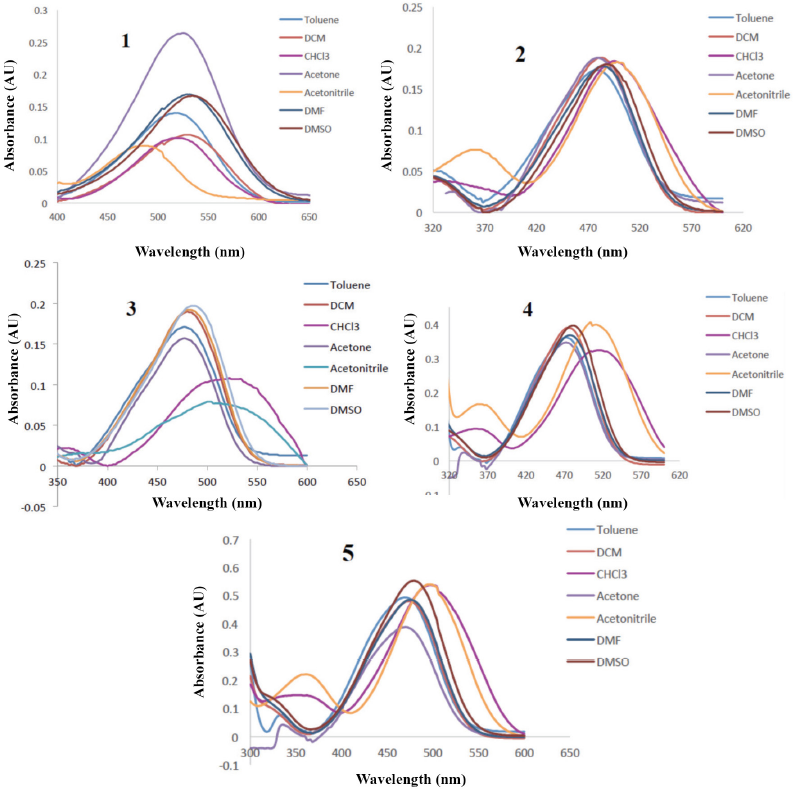
UV-Vis absorption spectra of 2 μM solution of dyes
Photophysical Properties of Dyes 1-5 in Different Solvents
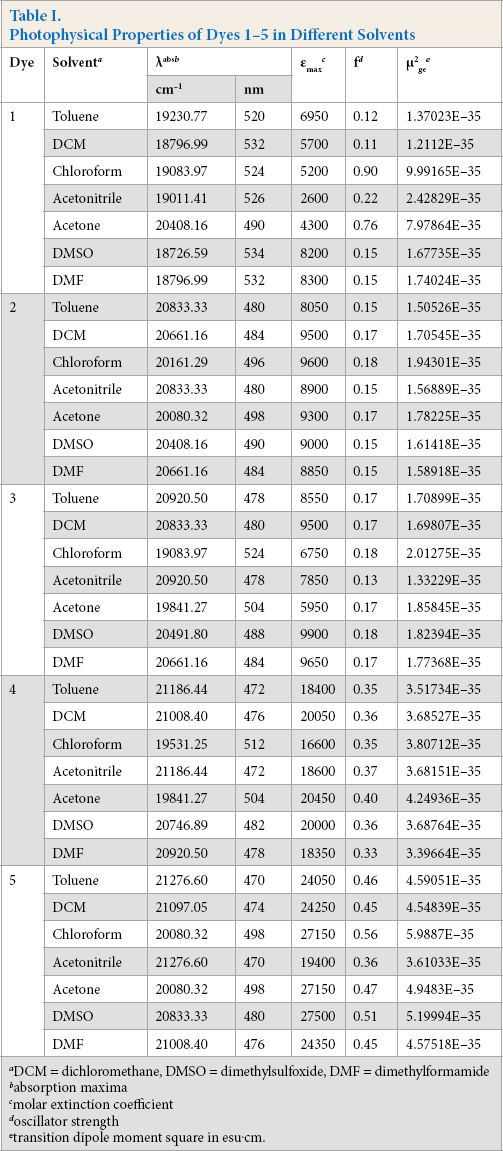
DCM = dichloromethane, DMSO = dimethylsulfoxide, DMF = dimethylformamide
absorption maxima
molar extinction coefficient
oscillator strength
transition dipole moment square in esu.cm.
All dyes showed less solvatochromism due to the presence of the
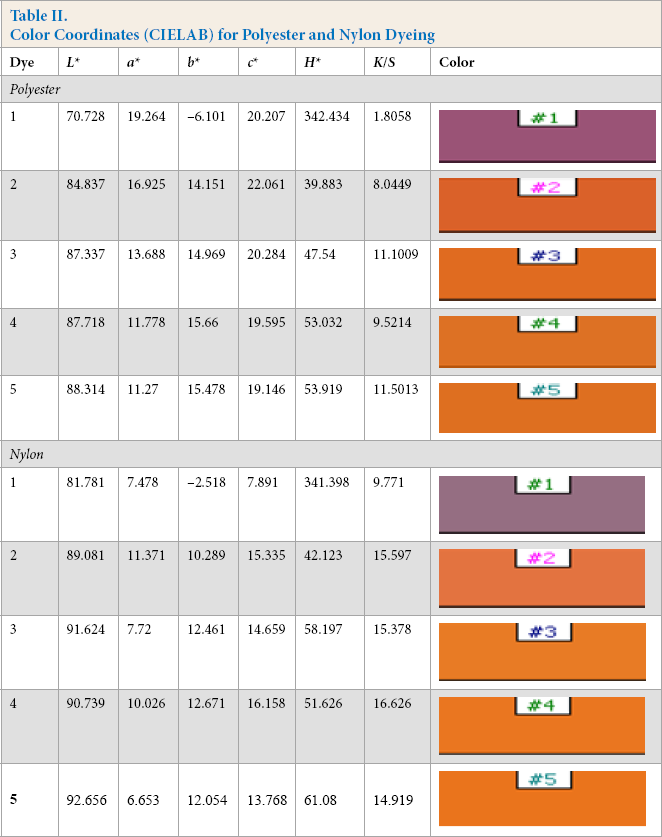
Color Assessment of Polyester and Nylon Fabrics
The color assessment of the dyed samples was performed using a Spectrascan 5100+ instrument, and
Color Coordinates (CIELAB) for Polyester and Nylon Dyeing
Lightfastness
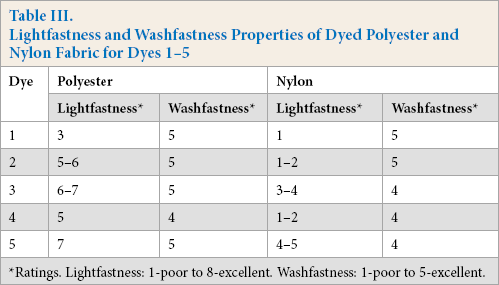
The lightfastness of the dyed fabrics was evaluated. A sample specimen was cut in half, with one half covered by a black sheet and the remaining half exposed to a Xenon lamp continuously for 17 h on a Q-SUN lightfastness tester. The samples were compared with a Blue Wool Scale to rate the lightfastness. The polyester fabrics showed better lightfastness properties for all dyes when compared to the nylon fabrics (Table III). For polyester fabrics, the sample dyed with dye
Lightfastness and Washfastness Properties of Dyed Polyester and Nylon Fabric for Dyes 1-5
Ratings. Lightfastness: 1-poor to 8-excellent. Washfastness: 1-poor to 5-excellent.
Washfastness
The dyed polyester and nylon samples using dyes
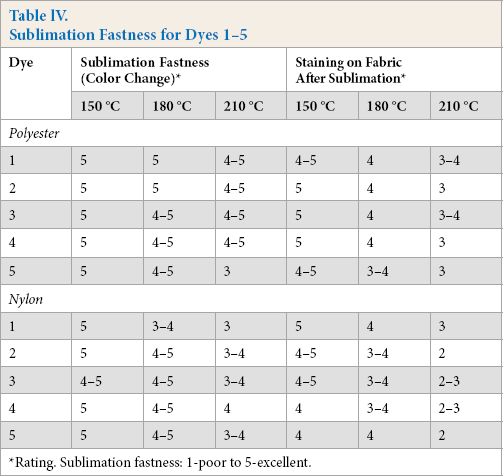
Fastness to Sublimation
Composite specimen dyed samples were made by sandwiching dyed samples between two white polyester fabrics, and were then exposed to different temperatures (150 °C, 180 °C, and 210 °C) for 30 s, and subsequently evaluated for change in color and staining of adjacent fabric by comparison with a gray scale. Sublimation fastness and staining of the polyester fabric was slightly better than for nylon fabrics (Table IV). The least color change was observed for dyes
Sublimation Fastness for Dyes 1-5
Rating. Sublimation fastness: 1-poor to 5-excellent.
Conclusion
We have successfully synthesized five novel monoazo thiazole disperse dyes
Dyeing on polyester and nylon fabrics for all dyes tested was carried by the HTHP method under suitable dyeing conditions.
Both dyed polyester and nylon fabric samples dyed with dyes
Supporting Information
The recorded 1 H and 13 C NMR spectra for all the synthesized dyes in this research are available from the authors upon request.
