Abstract
A series of 2-aminothiazoles is prepared in moderate-to-good yields by the direct coupling of ketones and thiourea using I2/dimethyl sulfoxide as a catalytic oxidative system. This method avoids the preparation of lachrymatory and toxic α-haloketones and the use of an acid-binding agent, thus providing a more convenient approach to 2-aminothiazoles compared to the Hantzsch reaction.

Thiazole is an important heterocycle and has found a broad spectrum of applications in fields from agriculture to medicine.1–6 In particular, aminothiazole is a useful structural motif and is present in numerous compounds possessing diverse biological properties, including anticancer, anticonvulsant, antidiabetic, antiviral, and antituberculosis, among many others.7–11 The aminothiazole backbone has been widely used as a privileged pharmacophore in the discovery of potential new chemical entities with diverse biological activities. Various drugs bearing an aminothiazole moiety with important therapeutic effects are now available, such as dasatinib, cefcapene, talipexole, meloxicam, and many others (Figure 1).12–17

Representative drugs with an aminothiazole unit.
Given these proven utilities in medicinal chemistry, considerable attention has been paid to the synthesis of 2-aminothiazoles. The Hantzsch reaction provides a practical approach to 2-aminothiazoles via the condensation reaction of α-haloketones and thioureas in a polar solvent. 18 Subsequently, various modified Hantzsch methods have been developed such as using heterogeneous catalysts, microwave heating, solid synthesis, or greener solvents including ionic liquids, PEG-400, and water.19–25 Other synthetic methods toward 2-aminothiazoles have utilized different coupling partners such as phenyl thiocyanates and propargylamines, alkyne(alkene)-substituted iodine(III) reagents and thioureas, 1,2-dicyano-3-bromo-2-phenylpropenes and thioureas, and α-SCN-substituted ketones and amines.26–29 However, the preparation of these coupling materials is generally difficult, needing multiple steps and harsh reaction conditions.
Notwithstanding, the Hantzsch method and modified reactions still represent efficient approaches to 2-aminothiazoles due to the use of readily available α-haloketones and thioureas. However, the synthesis of lachrymatory and toxic α-haloketones makes these methods inconvenient. Recently, Abedi-Jazini et al. 30 reported a more convenient synthesis of 2-aminothiazoles via the I2-mediated oxidative coupling of ketones and thiourea in the presence of an organic base. This method proceeded through the in situ formation of α-iodoketones and provided 2-aminothiazoles in high yields; however, the requirement of using stoichiometric I2 as the oxidant and an organic base (such as NEt3) as an acid-binding agent might limit its further use. In recent years, the I2/dimethyl sulfoxide (DMSO) catalytic oxidative system has been successfully applied in various types of organic transformations.31–35 In these methods, the I2 catalytic cycle was completed using DMSO as the oxidant. We focused on utilizing this catalytic oxidative system in the Hantzsch method to achieve the one-pot synthesis of 2-aminothiazoles without the preparation of α-haloketones. Herein, we report the first example of I2 catalytic synthesis of 2-aminothiazoles through the oxidative coupling of ketones and thiourea using DMSO as the solvent and oxidant.
We started our optimization studies by reacting 2 mmol each of acetophenone (
Optimization of the reaction conditions. a

Conditions:
Isolated yield of
p-TsOH (1.0 equiv.) was added to the reaction mixture.
AcOH (1.0 equiv.) was added to the reaction mixture.
CH3SO3H (1.0 equiv.) was added to the reaction mixture.
Bromine (0.2 equiv.) was used instead of I2.
Aqueous HBr (0.2 equiv.) was used instead of I2.
Sulfur suspended in the reaction mixture was isolated.
DMSO (2mL) and H2O (2mL) was used as the solvent.
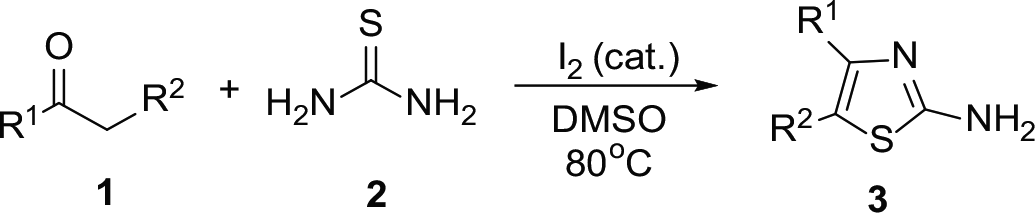
With optimized conditions in hand, we proceeded to investigate the scope of the ketones. As shown in Table 2, a range of substituted acetophenones bearing various substituents (F, Cl, Br, CH3, OCH3, CH3SO2, and OH) on the phenyl moiety were suitable for the reaction, providing the corresponding thiazoles in moderate-to-good yields. Substituted acetophenones bearing neutral (H) or electron-withdrawing groups (F, Cl, Br, CH3SO2) at the ortho-, meta-, or para-position of the phenyl moiety reacted smoothly under the optimized conditions, giving the corresponding products in 40%–79% yields (
Reaction scope of the ketones. a
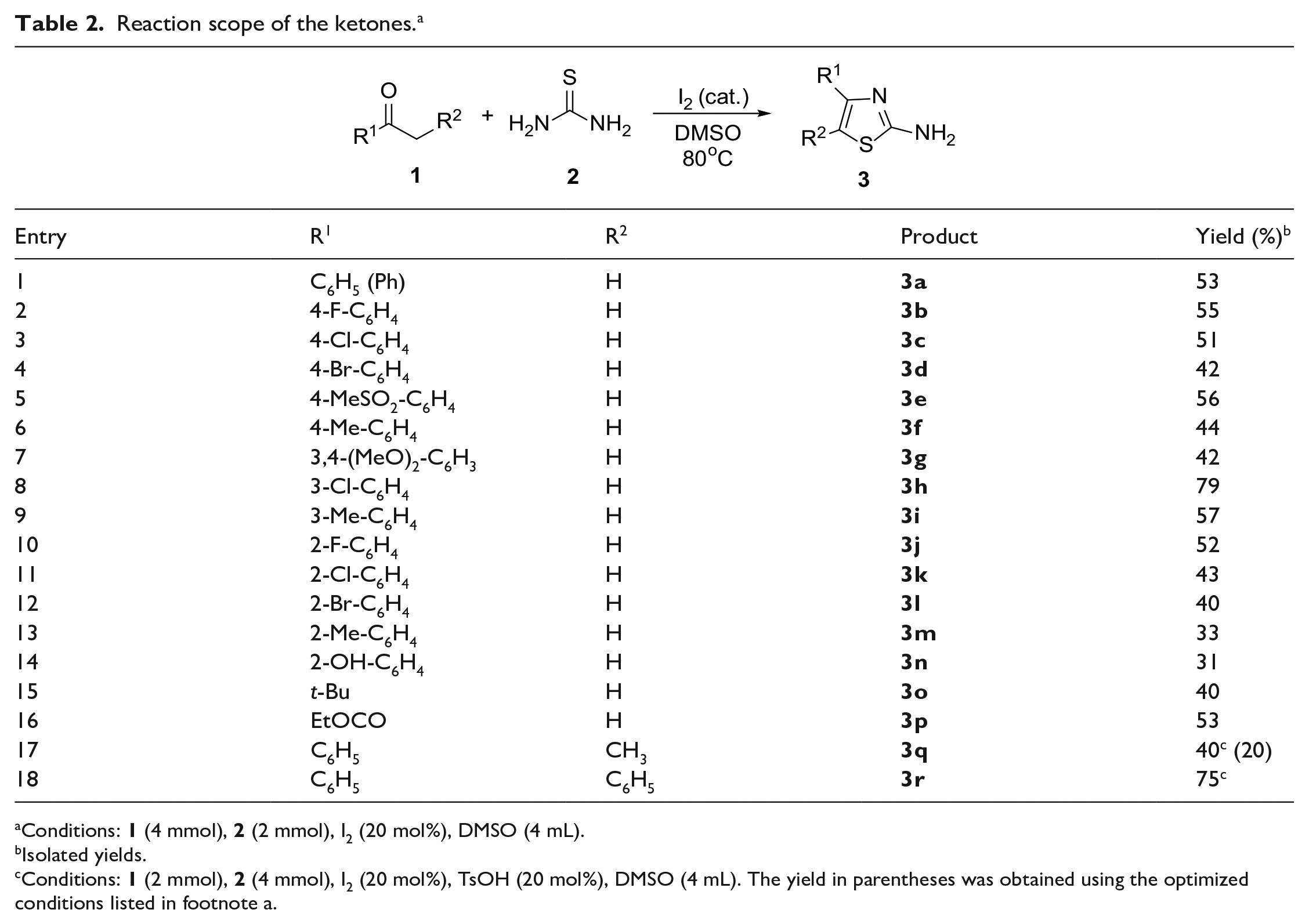
Conditions:
Isolated yields.
Conditions:
Based on the results of our experiments and previous literature,18–25 we propose the following reaction pathway (Scheme 1). Briefly, acetophenone (
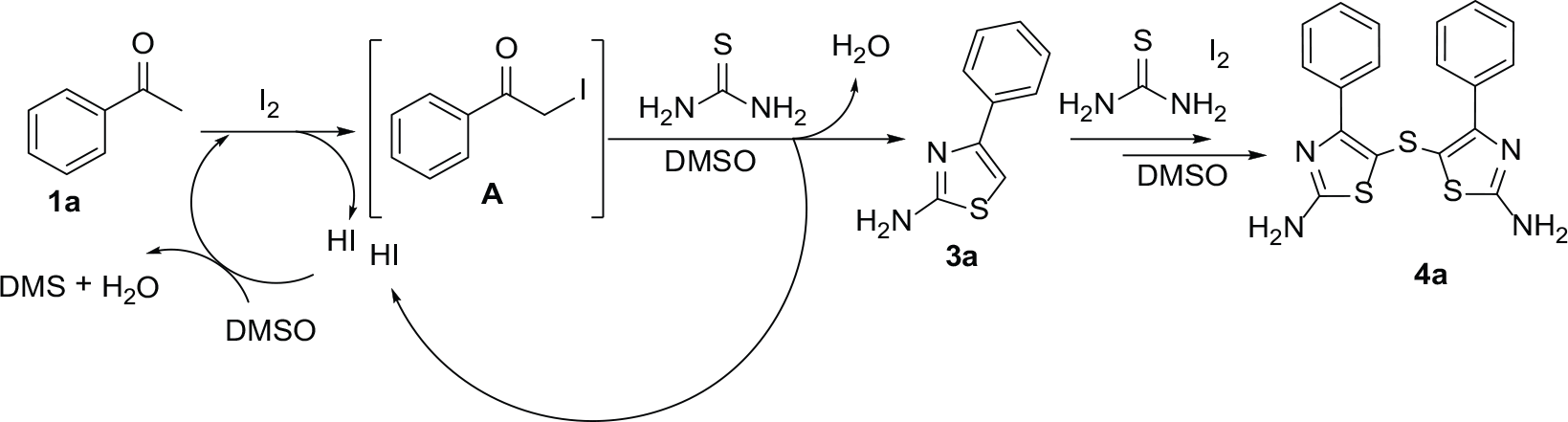
A plausible reaction pathway.
In conclusion, we have developed the I2-catalyzed synthesis of 2-aminothiazoles via the direct oxidative coupling of ketones and thiourea. This reaction proceeds through the formation of α-iodoketone and employs DMSO as the oxidant to complete the I2 catalytic cycle. This method avoids the preparation of α-haloketones and the use of an acid-binding agent, thus providing a convenient approach to 2-aminothiazoles.
Experimental
NMR spectra were obtained on a Bruker Ascend TM600 or a Bruker AVANCE III 500 MHz spectrometer in CDCl3 or DMSO-d6 using TMS as the internal reference. Chemical shifts (δ) are given in parts per million (ppm), and coupling constants (J) are given in Hz. All products were isolated by short-path chromatography on Qingdao Haiyang silica gel (200–300 mesh) eluting with a mixed solvent of petroleum ether (60–90 °C)/ethyl acetate (5/1, v/v). Melting points were recorded on a SGWX-4B melting point apparatus. Commercially obtained reagents and DMSO were used without further purification.
I2-catalyzed coupling reaction of ketones and thiourea: general procedure
A Schlenk tube (20 mL) was charged with ketone
2-Amino-4-phenylthiazole (
2-Amino-4-(4-fluorophenyl)thiazole (
2-Amino-4-(4-chlorophenyl)thiazole (
2-Amino-4-(4-bromophenyl)thiazole (
2-Amino-(4-(4-methylsulfonyl)phenyl)thiazole (
2-Amino-4-(4-methylphenyl)thiazole (
2-Amino-4-(3,4-dimethoxyphenyl)thiazole (
2-Amino-4-(3-chlorophenyl)thiazole (
2-Amino-4-(3-methylphenyl)thiazole (
2-Amino-4-(2-fluorophenyl)thiazole (
2-Amino-4-(2-chlorophenyl)thiazole (
2-Amino-4-(2-bromophenyl)thiazole (
2-Amino-4-(2-methylphenyl)thiazole (
2-Amino-4-(2-hydroxyphenyl)thiazole (
2-Amino-4-(t-butyl)thiazole (
Ethyl 2-Aminothiazole-4-carboxylate (
2-Amino-5-methyl-4-phenylthiazole (
2-Amino-4,5-diphenylthiazole (
5,5′-Thiobis(4-phenylthiazole-2-amine) (
Supplemental Material
SI – Supplemental material for A one-pot synthesis of 2-aminothiazoles via the coupling of ketones and thiourea using I2/dimethyl sulfoxide as a catalytic oxidative system
Supplemental material, SI for A one-pot synthesis of 2-aminothiazoles via the coupling of ketones and thiourea using I2/dimethyl sulfoxide as a catalytic oxidative system by Qian Zhang, Jiefei Wu, Zexi Pan, Wen Zhang and Wei Zhou in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.