Abstract
This study is focused on optimizing fluorochemical urethane finish concentration for treatment of 100% cotton fabric. Fluorochemical urethane (FU) is a newer innovation with a shorter fluorocarbon chain (C4), is free from perfluorooctanoic acid (PFOA) and perfluorooctanesulfonate (PFOS), and thus is a safer alternative to older C6- or C8-based fluorocarbons. The finish concentration was optimized based on the liquid repellency (water and oil repellency) and its influence on selected physical properties of the fabric. In addition, the laundering durability of the repellency effect was studied as well. The results indicate that a 80 g/L concentration of fluorochemical urethane was the optimal finish concentration for 100% cotton woven fabric based on desired liquid repellency and physical properties of the fabric.
Keywords
Introduction
Background
Gautama Buddha said, “a lotus flower is born in water, grows in water, and rises out of water to stand above it unsoiled.” The phenomenon of liquid repellency exists in nature and is a much-desired property in the textile field, with several applications. There is more than half a century of research involved in this area. Since the mid-1900s, scientists have developed various technologies such as fluorinated polymers, silicones, hydrocarbons, dendrimers, and inorganic chemistry-based technologies to impart water and oil repellency properties to textiles. However, only a perfluorinated textile finishing agent offers repellency against both polar (water based) and non-polar (oil based) substances from the fabric surface.1–3
The science of surface engineering aims at modifying the chemical composition of the surface property of materials and is a branch of nanotechnology. Certain chemical finishes, such as fluorochemical finishes, create a non-polar surface on the fabric to impart hydrophobicity. It is very difficult to create a textile surface that has both hydrophobic as well as oleophobic (oil-repellent) surface properties.1,2 Theeffect of hydrophobicity and oleophobicity is usually measured by surface contact angles. The higher the contact angle, the greater the cohesion of water (or oil) molecules, and lesser adhesion to the fabric surface that imparts a unique “lotus leaf” effect on the fabric surface, and gives it a water-repellent (or oil-repellent) surface.
Durable water repellents (DWR) based on C8-10 technology result in persistent, bio-accumulative, and toxic (PBT) substances, including perfluorooctanoic acid (PFOA) and perfluorooctanesulfonate (PFOS), that have been detected around the world in the food chain, drinking water, animals, and human blood. 2 The use of C8 or greater fluorination chemistry releases PFOA and PFOS that are toxic for the environment as well as for human health. After the implication of a ban from US Environmental Protection Agency (US-EPA), researchers tried to find a more ecofriendly alternative to C8 and C6 chemistry. In that effort, a C4-based alternative was developed with a shorter fluorochemical chain that is environmentally safer and also offers a broad-spectrum liquid repellency. The C8- and C6-based fluorochemical finishes also require high temperature heat setting to impart good repellency effect. Therefore, urethane compositions, including those containing fluorine, that have high water solubility eliminate the need for extreme high temperature curing conditions.
C4-C8 Chemistry and Toxicology
C6 chemistry produces a byproduct called PFHA (perfluorohexanoic acid) which is 40 times less bioaccumulative than PFOA. 4 C6 is the closest chemistry to C8 that produces desirable repellency results without PFOA emissions in the environment. C4 chemistry produces PFBS (perfluorobutane sulfonate) as a byproduct that does not accumulate in the environment to the extent that higher perfluorinated fluorocarbon byproducts did, based on test results done by the manufacturer following US-EPA guidelines.4,5 PFBS does not remain in human and animal blood streams for long and is eliminated much more quickly than PFOA and PFHA, therefore, it is considered environmentally safe. The challenge is to use the C4 chemistry in an optimal concentration to get the desired repellency effect. As the fluorocarbon chain gets shorter, it also becomes a less effective repellent. Banning or abolition of certain chemicals with known environmental and health risks may not be sufficient to permanently eliminate the risks and may result in nearest substitution. A new invention of nearest substitute may result in switching from known to unknown risks, it is imperative that the substitutes are safer and we understand their use in correct methods and proportions for optimal performance and minimal risks.
In this study, an oxime-blocked polyisocyanate finish extender was used to impart repellency performance.
Fluorochemical Urethane and its Applications
Fluorochemical urethane (FU) is a patented technology that uses a short-chain fluorinated hydrocarbon and is derived from reaction with a polyfunctional isocyanate compound, fluorinated monofunctional compounds, at least one hydrophilic polyoxyalkylene compound optionally, and isocyanate-reactive silane compounds. 5 The manufacturer of the product provided FU as a standalone compound (instead of as a ready-to-use commercial product that serves as a stain repellent for carpets) for the purpose of this research. Currently, FU polymer is part of a commercial stain repellent for carpets and has several applications in soil/stain-repellent/release/resistant finish that offers a broad-spectrum durable effect against water-and oil-based stains to porous surfaces, including textiles, tiles, concrete, grout, paper, and leather. The application and uses of FU are well established; however, the optimal finish concentration that allows economical use of the finish and yet offers high water and oil repellency is unascertained. Moreover, the negative impact of the finish on durability, air permeability, and fabric hand can be reduced by using the optimal finish concentration.
Materials and Methods
Fabric
A 100% cotton ready-to-finish fabric with a 1 × 1 plain weave, 0.45 mm thickness, and 210 g/m2 was selected for this study. The fabric was procured from a weaving mill and was dyed and unfinished. It was essential to use an unfinished fabric for this research as most textile finishes alter the pH and absorption of the fabric.
Finishing Preparation
FU performance can be impaired by the presence of auxiliary residues, especially silicone and cationic softeners. To remove these and assure proper absorption, and better and uniform application of the finishing agent, the textile substrates were first pretreated. First, the substrates were soaked in an alkaline wash solution to ensure that the fabric was free of any surface impurities. For this, each fabric was soaked in a solution of 2 g/L of anionic detergent (Sigma Aldrich) containing 1 g/L of sodium carbonate (Sigma Aldrich). The pH of the solution ranged between 8-9 and the temperature of the solution was kept at 40 °C. The substrates were soaked in the above mentioned solution for 20 min and then rinsed well with water afterwards. Subsequently, the fabric was soaked in an acid wash following the same method as the alkaline wash. The 1 g/L of sodium carbonate was replaced with 3 mL/L of acetic acid (Sigma Aldrich) for the acid wash.
Determination of Substrate pH
The FU-based finish works best at slightly acidic pH values. Fabric treatments such as bleaching and dyeing change the pH of fabric dramatically, so it is important to determine the pH of the fabric before application of finish. The substrates were soaked in 100 mL distilled water, keeping the material-to-liquor ratio (MLR) at 1:20. In another beaker, 100 mL of distilled water was taken as a control. Both solutions were brought to boil and then allowed to cool. The pH of these were measured at room temperature using a digital pH meter.
Determination of Silicone Presence
Silicone forms a hydrophobic film on textile surfaces and is often applied on the fabric surface after dyeing to improve fabric softness and to keep surface impurities/dirt from sticking on the fabric surface. For this study, it was important to test for the presence of silicone, as silicone does not allow absorption of the finishing agent to the fabric. The following tests were conducted to determine the presence of silicone.
Method 1
Regular adhesive tape was stuck on the fabric surface. If the tape stuck to the fabric, the absence of silicone in the fabric was presumed.
Method 2
A 1 × 1 cm fabric swatch was dipped in 10 mL of perchloroethylene in a test tube. The test tube was shaken for few minutes. If no foam formed, it was presumed that no silicone was present in the fabric.
Finish Application
A set of finish solutions at various FU-finish concentration levels (50, 60, 70, 80, and 90 g/L) each containing 10 g/L of isopropyl alcohol (Sigma Aldrich) and 10 g/L of oxime-blocked polyisocyanate (Huntsman Chemicals) were prepared for finishing the 100% cotton fabric. Isopropyl alcohol acted as a wetting agent and helped with uniform and better application of the finishing agent, whereas, the oxime-blocked polyisocyanate improved the laundering durability of the finish. The pH of the finish solution was maintained at 5.5 as the FU application works at acidic pH only. Each finish solution was applied on a textile substrate using the lab model padding mangle with a wet pickup of 85%. After the finishing treatment, the textile substrates were dried at 130 ± 5 °C for 8 min using a lab model oven and cured at 170 ± 5 °C for 5 min. The curing treatment ensured the longevity of the finish on the fabric. The finish concentration was optimized based on the air permeability and water and oil repellency, and the effect of finish concentration on selected physical properties of the finished textiles was studied.
Textile Substrate Testing
A series of tests were conducted on the treated and untreated textile substrates to study the effect of finish concentration on fabric hand, durability, and air permeability. Fluoro-chemicals are known to affect the strength and pliability of treated fabrics negatively.3,4 Additionally, liquid repellency and laundering durability tests were conducted to study the performance and longevity of the finish.
Water Repellency (Drop Test)
Water repellency is the ability of the substrate to resist wetting by water or water-based liquids. The water repellency of the substrates was tested by the water/alcohol drop test. 6 The drops of standard test liquids consisting of specified proportions of deionized water and isopropyl alcohol (reagent grade) by volume were used as test liquids to evaluate the water repellency of the substrates.
The substrates were cut to 20 × 20 cm each for the test. The sample was put on a fat and horizontal surface. Using a dropper, a drop of ∼5 mm in diameter was put gently at three different places, keeping the dropper at a distance of 1 cm from the fabric. Care was taken that the dropper did not touch to the fabric. This drop was allowed to stand undisturbed for 10s. After 10s, if two of the three drops were still visible as spherical to hemispherical, the substrate passed that particular test liquid. Substrates were rated as pass or fail for the appropriate test liquid that remained visible. The water repellency of the control and all the substrates treated at various concentrations of fluorochemical finishing agent were tested in this manner.
Water Repellency (Spray Test)
The spray test was conducted to measure the resistance of fabrics to wetting by water. The test was conducted as per AATCC Test Method (TM) 22-2001. 7 The size of the substrate used for the test was 20 × 20 cm. To carry out the test, 250 cm3of water at 27 ± 2 °C was poured steadily into the spray funnel. A standard distance of 6 in. from the bottom of the spray funnel to the center of the fabric was maintained. The fabric was placed and stretched in a 6-in. diameter embroidery hoop and fixed on the platform of an angle of 45°. After spraying, the substrate holder was removed and the surplus water was removed by tapping the frame six times against a solid object, with the face of the substrate towards the solid object. The tapping was done in two stages, three taps at one point on the frame and then three times at a point diametrically opposite. The assessment of the fabric water repellency against spray was examined visually by matching against the standard rating chart. Water repellency against spray of the control and all the substrates treated at various concentrations of fluoro-chemical finishing agent were tested in this manner.
Oil Repellency
Oil repellency is the ability of a textile fiber, yarn, or leather to resist wetting by oily liquids. It was determined using AATCC TM 118-1992. 8 The drops of standard test liquids, consisting of a selected series of hydrocarbons with varying surface tensions were used to perform the test.
A 20 × 20 cm substrate was used to test oil repellency. The test substrate was placed on a smooth horizontal surface. Using a dropper, a drop ∼5-mm in diameter was put gently at three different places, keeping the dropper at a distance of 1 cm from the fabric. Care was taken that the dropper did not touch the fabric. This drop was allowed to stand undisturbed for 30s. After 30s, if two of the three drops were still visible as spherical to hemispherical, the substrate passed that particular test liquid. Substrates were rated as pass or fail for the appropriate test liquid which remained visible. The oil repellency of the control and all the substrates treated at various concentrations of fluorochemical finishing agent, different pickups and at different curing temperatures, were tested in this manner.
Laundering Durability
Laundering durability is defined as the ability of a protective finish to continue to perform its function after being subjected to repeated launderings. The laundering durability test was determined on a top loading automatic washer recommended by AATCC using AATCC TM 43-1995. 9
The size of substrate used for the test was 20 × 20 cm. The substrates were put into the washer, which was filled to the high water level with water at 41 ± 3 °C. Standard detergent (20 g) was put into it and substrates were washed using a 12-min “Normal” wash cycle. After the completion of a specified number of wash cycles, the substrates were taken out and dried in a forced air circulating oven at 65 ± 6 °C. The performance of these substrates was then tested.
Tensile Strength
The tensile strength of the samples was determined on a computerized universal tensile strength tester (Instron).10 Themean and standard error values for tensile strength and elongation at break were measured in the fabric warp and weft directions using 325 × 60 mm samples. Yarns were raveled from both sides to obtain a substrate of uniform width. Prior to the test, the specimens were conditioned for moisture equilibrium. For testing, a specimen was held between the two clamps of the tensile strength testing machine such that the same set of yarns was gripped by both clamps. A continually-increasing load was applied longitudinally to the specimen by moving one of the clamps until the specimen ruptured. The value of breaking strength of the test specimen was read from the machine. In this manner, the tensile strength of the control and all the treated samples was tested five times each, and then the mean and standard deviation values were calculated.
Bending Length and Flexural Rigidity
The bending length of the samples was determined with the help of a stiffness tester (Model 112, Paramount Instruments Pvt. Ltd.) using the ASTM D1388-96 method. 11 A 6 × 1 in. rectangular strip of fabric was mounted on a horizontal platform. It was supported by two side pieces made of plastic. A mirror was attached to the instrument that enabled the viewer to view both of the index lines from a convenient position. The scale of the instrument was graduated in centimeters of bending length. The specimen was put on a platform in such a way that it overhung like a cantilever and bent downwards. The length of the overhanging portion, when depressed under its own weight, and the angle between the lines joining the tip of the edges of the platform were measured. Using this method, three warp-way and three weft-way specimens were tested. Each specimen was tested four times with the front face and again with the strip turned over for the back face. The mean values and standard deviation for the bending length in warp and weft directions were calculated for the values of flexural rigidity.
Crease Recovery
Fabric crease recovery, measured quantitatively by the crease recovery angle (CRA), was tested using an AATCC wrinkle recovery tester as per AATCC TM 66-1998. 12 A 2 × 1 in. wrinkle free specimen was folded gently end-to-end in half and compressed under a specified load for a specified time. The load was then removed and the specimen was allowed to recover for the specified time. After the load was removed, the specimen was transferred to the instrument clamp. For this, one end of the specimen was held in the tweezers and the other was placed in the clamp. As the specimen recovered, the dial was rotated to keep the free edge of the specimen in line with the knife edge. Ten the CRA in degrees was read on the dial. The procedure was repeated for five specimens each in warp and weft direction and the mean and standard deviation values were calculated.
Air Permeability
The air permeability of the fabrics was determined by ASTM D737-1996. 13 The test specimen was placed on the air permeability tester (SDL Atlas) between the top and bottom of the column. The fabric should be placed right side down with the air pressure is lower at that side. Using a vaccum, the air pressure was different on one side of the fabric. Airflow occurred from the side with higher air pressure, through the fabric, to the side with the lower air pressure. From this rate of air flow, the air permeability of the fabric was determined and displayed on the screen of the tester. The readings were noted and ten specimens of each fabric were tested. Thereafter, mean values of each fabric were calculated.
Results and Discussion
Finish Concentration Optimization Based on Liquid Repellency
The liquid repellency of the FU-finished textile substrates was studied three ways. The results of water repellency (drop test), water repellency (spray test), and oil repellency drop test as indicators of laundering durability are discussed as follows.
Water Repellency (Drop Test) Analysis
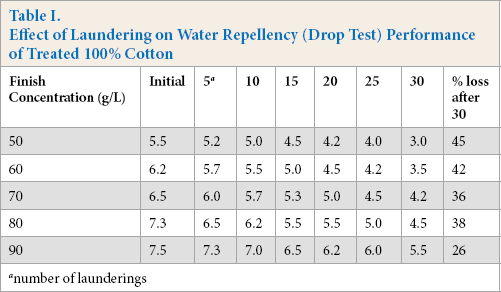
At the initial stage, the difference between the mean values of the substrates treated at 50 (5.5), 60 (6.2), and 70 g/L (6.5) FU-finish concentrations (Table I) was not significant (p ≤ 0.05). A significant increase in water repellency among textile substrates finished at the 80 g/L concentration was observed. Nevertheless, a non-significant difference was observed between the 80 (7.3) and 90 (7.5) g/L finish concentrations. A similar trend was observed after 30 laundering cycles.
Effect of Laundering on Water Repellency (Drop Test) Performance of Treated 100% Cotton
number of launderings
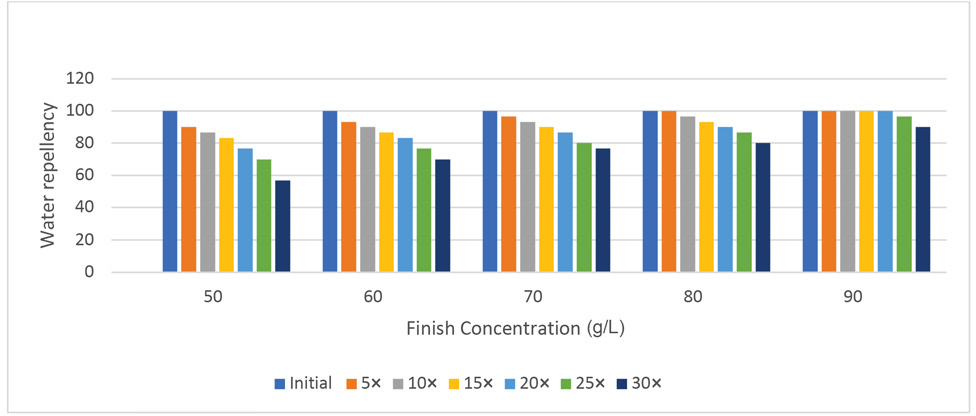
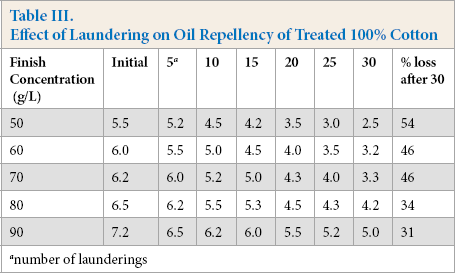
The percent loss of finish performance was calculated from the water repellency data by comparing initial and final water repellency values after 30 laundering cycles. The loss of hydrophobicity is common in fabrics treated with fluorochemicals after washing (Table I). The FU-treated textile substrates in this study showed fair to very good hydrophobicity even after 30 laundering cycles depending on the finish concentration used. The use of oxime-blocked polyisocyanate as a finish extender aided in the laundering durability as well as increased the absorption of the finish. Therefore, the durability of this finish was significantly better than that of older fluorochemical finishes that wore of after 5–7 laundering cycles. As the number of laundering cycles increased (Fig. 1) a decrease in the water repellency values was observed at all finish concentration levels. The loss of water repellency after 30 laundering cycles ranged between 51 and 30% at finish concentrations of 50 g/L and 90 g/L, respectively. The higher the finish concentration was, the lower the repellency loss was after laundering. There was no statistically significant difference in the percent loss of finish performance among fabrics treated at 50, 60, and 70 g/L finish concentration. However, the percent repellency loss was significantly less using 80 g/L of the finish concentration.
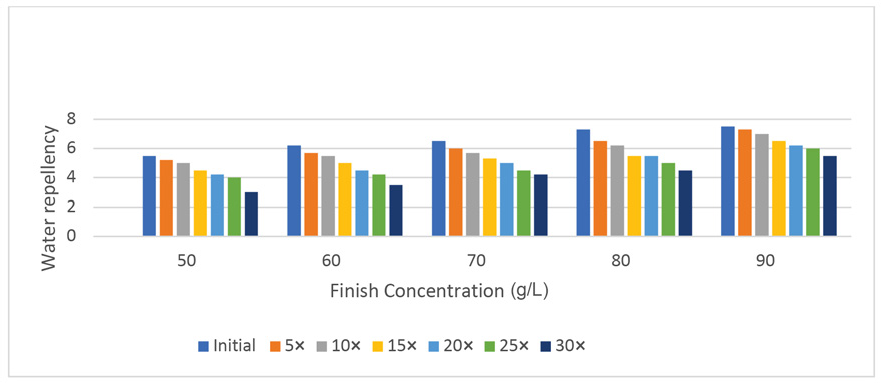
Effect of laundering on water repellency (drop test) performance of 100% cotton.
Water Repellency (Spray Test) Analysis
Based on the water repellency spray test results, the laundering durability of the FU-finish improved with higher finish concentrations. All values obtained at the initial stage and after five launderings were statistically consistent at various finish concentration levels. There was no significant decrease in the finish performance after five launderings. The water repellency spray test results were in line with the water repellency drop test results. At the 50 g/L finish concentration, there was a 50% of repellency loss after 30 launderings. Use of 90 g/L or greater finish concentrations reduced this repellency loss by 33.3% (Table II and Fig. 2). Further increases in finish concentration did not lead to a statistically significant difference in finish performance. Although, the high finish performance is greatly desirable, the use of an optimal concentration can result in cost reductions and is environmentally preferable. There was statistically no significant difference in the percent repellency loss at 80 g/L and 90 g/L finish concentrations after 30 launderings.
Effect of laundering on water repellency (spray test) performance of 100% cotton.
Effect of Laundering on Water Repellency (Spray Test) of Treated 100% Cotton
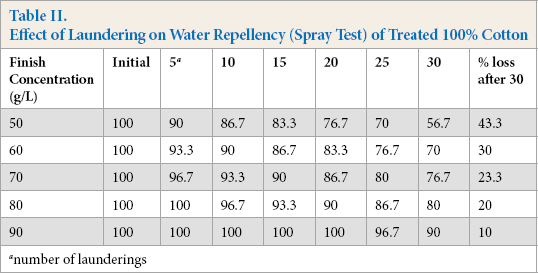
number of launderings
Oil Repellency Analysis
Initially, the oil repellency values at 50 (5.5), 60 (6.0), and 70 g/L (6.2) FU-finish concentration differences were not statistically significant. However, the difference in values of 6.5 at the 80 g/L finish concentration and 7.2 at the 90 g/L finish concentration were statistically significant. Before laundering, the oil repellency of the treated fabric was significantly greater at the 90 g/L finish concentration when compared to lower finish concertation levels. The mean oil repellency values after five laundering cycles were 5.2 and 5.5 at 50 and 60 g/L finish concentrations respectively (Table III, Fig. 3). However, differences in the mean oil repellency values of 6.0, 6.2, and 6.5 at 70, 80, and 90 g/L finish concentrations, respectively, were statistically significantly (p ≤ 0.05) among each other. After 10 laundering cycles, mean oil repellency values at 50, 60, and 70 g/L finish concentrations were 4.5, 5.0, and 5.2, respectively. Differences in the mean oil repellency values of 5.5 at the 80 g/L finish concentration and 6.2 at the 90 g/L finish concentration, were statistically significant.
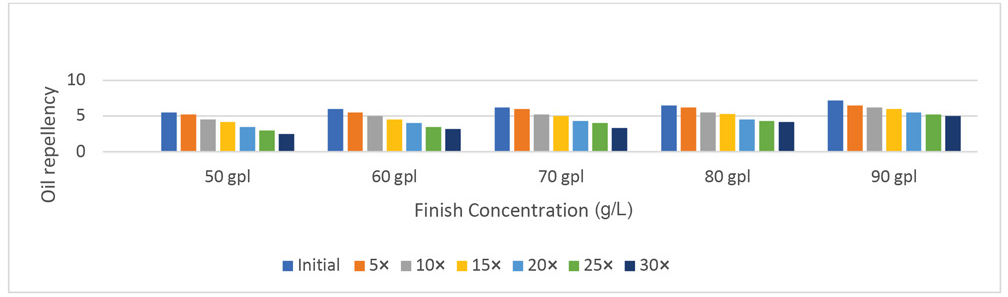
Effect of laundering on oil repellency performance of 100% cotton.
Effect of Laundering on Oil Repellency of Treated 100% Cotton
number of launderings
A similar trend was observed in the data after 15, 20, 25, and 30 laundering cycles (Table III, Fig. 3). There was a loss of 54 to 31% oil repellency after 30 launderings of the treated fabrics. The loss of the oil repellency of the finish increased as the finish concentration decreased, however, this decay in performance was not statistically significant when <70 g/L of the finish concentration was used. There was a statistically significant percent loss in oil repellency between 70 and 80 g/L finish concentration levels. The oleophobicity loss was greater compared to the hydrophobicity loss in both drop and spray tests. It is only possible to achieve an oleophobic textile surface with fluorochemical-based finishes due to their non-polar surface properties and even more difficult to retain the oleophobic performance, especially after laundering.
Hybrid fluorochemical type block copolymers such as FU are effective stain repellents against water, as well as oil-based stains, even with successive washing cycles. The liquid repellency effect dropped gradually with subsequent launderings of the treated textile substrates due to the surface movement of fluorochemical segments resulting from the effects of polarity and surface tension on polymer chains.14 Thealkaline pH of the detergent used in the laundering durability test may also be a factor. Fluorochemicals are cat-ionic in nature and work best under slightly acidic pH. They lose their liquid repellency effect under alkaline conditions, which may be another cause of gradual loss in liquid repellency of treated substrates upon laundering.
Effect of Finish Concentration on Physical Properties of the 100% Cotton Fabric
Tensile Strength
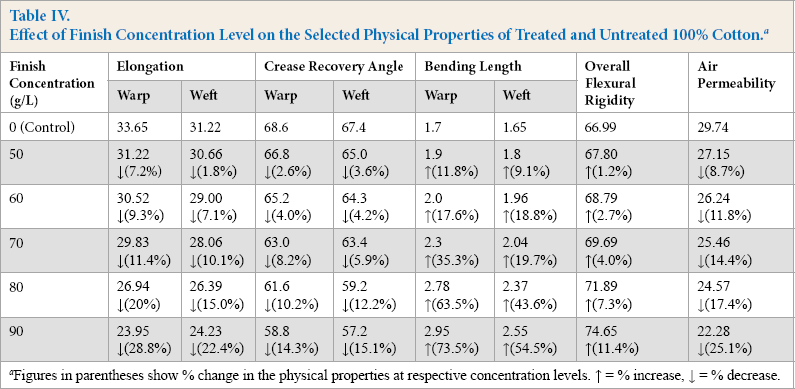
It is evident from Table IV that textile substrate elongation decreased in both warp and weft directions with an increase in the concentration of the FU-finishing agent. Fluorochemical finishes in general increase fabric stiffness/ brittleness, which lowers the fiber elongation and decreases the tensile strength of the fabric. Loss of elongation in weft were more impacted than warp yarns in this study. Even though there was a gradual decrease in tensile strength (warp direction) of the test substrates with increased finish concentration, the difference in elongation values was not statistically significant until 70 g/L of the finish concentration was used. At 80 g/L, elongation values of treated fabric in warp and weft directions decreased by 20% and 15% respectively compared to elongation of control fabric. This was a significant decrease (p ≤ 0.05) and any further increase in the finish concentration affected the tensile strength of the fabric negatively. Fluorochemical finishes applied at higher concentrations decreased the tensile strength of the treated fabrics due to increased tensile linearity and resistance to extension.6,7 The curing temperature can also contribute to the decreased tensile strength, especially in case of cellulosic fibers. These results were in concurrence with previous studies3,14–16 that used fluorochemical treatment on textiles.
Effect of Finish Concentration Level on the Selected Physical Properties of Treated and Untreated 100% Cotton. a
Figures in parentheses show % change in the physical properties at respective concentration levels. ↑ = % increase, ↓ = % decrease.
Fabric Stiffness
For the crease recovery data, a statistically significant (p ≤ 0.05) increase in fabric stiffness was observed, in both warp and weft directions of the textile substrates, when >70 g/L of the FU-finish concentration was used (Table IV). Likewise, the percent increase of bending length did not show statistically significant differences among the 50, 60, and 70 g/L finish concentrations, but there were statistically significant increases (10.2% and 12.2%) in warp and weft directions, respectively, at the 80 g/L finish concentration. Application of a fluorochemical finish improves the dimensional stability and affects fabric hand negatively.3,16-19 Theoverall flexural rigidity of textile substrates gradually increased from 1.2% at 50 g/L finish concentration to 11.4% at the 90 g/L finish concentration (Table IV). As the finish concentration increased, the stiffness of the treated fabric also gradually increased. This increase was statistically significant at the 80 g/L finish concentration. Any further increase in finish concentration led to high stiffness in the fabric that made it inappropriate for apparel use; this increased stiffness also lessened the fiber elongation, thereby decreasing the tensile strength of the treated textiles. The mean values of substrates were statistically (p ≤ 0.05) consistent below the 80 g/L finish concentration.
Air Permeability
The air permeability results showed reduction in the air porosity of the fabric as the FU-finish concentration increased (Table IV). After statistical analysis of the data, there were no significant (p ≤ 0.07) decreases in air permeability values among substrates finished at 50 to 70 g/L finish concentrations. Therefore, a higher finish concentration that resulted in better repellency was more desirable as the air permeability of the fabric finished at lower concentrations was statistically the same as those finished at higher concentrations up to a 70 g/L finish concentration. However, a statistically significant decrease in air permeability was observed at the finish concentration level of 80 g/L and above. These results were in line with previous studies,3,16,20–22 where it was reported that a reduction in the air permeability of the fluorochemically-treated fabrics occurred at higher finish concentration levels. Since fluorochemicals change the critical surface tension of the finished material at the nano level, this did not result in a change of fabric air porosity unless a finish concentration greater than 80 g/L was used to treat the fabric.
Conclusion
Versatile application of fluorochemical finishes make it desirable for outdoor, weatherproof, protective, automotive, household textiles, antibacterial, and several other applications. Excellent hydrophobicity and oleophobicity can be achieved with the use of fluorochemical finishes as opposed to its alternatives, which provide good hydrophobicity, but not oleophobicity.16,23 However, the downside of fluoro-chemical finishes is that they impact the tensile strength and drapeability of the finished fabric negatively when used at higher concentration levels. High temperature curing that is required for proper fixation of the finish partially adds to this problem. Nonetheless, their liquid repellency is unbeatable and it plays a vital role in the DWR industry.
It is imperative to optimize fluorochemical finishing conditions to minimize the negative effect of the finish on finished textiles and achieve the best possible results. Based on the liquid repellency data of this study, textile substrates finished at 80 g/L with fluorochemical urethane (FU)-finish performed significantly better compared to lower concentrations even after 30 laundering cycles. In regard to the impact of the finish on physical properties of 100% cotton fabric, the fabric stiffness increased significantly in textile substrates finished at 70 g/L or higher. The tensile strength, overall flexural rigidity, and air permeability deteriorated significantly at a finish concentration greater than 80 g/L. Therefore, a finish concentration of 80 g/L was considered optimal for 100% cotton fabric, as the liquid repellency, laundering durability, and tensile strength were the important properties affected by textile finishing. However, a finish concentration of 70 g/L could be considered optimal where fabric pliability was more desirable. The cost-effective and superlative liquid repellency of FU finishes can be expected when used in an optimal concentration.
Limitations and Future Research
This study was focused on finding an optimal FU finish concentration for 100% cotton fabric only. A further study on optimizing the FU concentration for other fiber contents will be conducted and the results will be reported at a later date.
A future study will also be focused on application of the finished fabric to develop personal protective equipment (PPE).
