Abstract
A kinetics and thermodynamics study of turmeric extract dyeing on
Introduction
Silk is a well-known fiber for use in clothing and apparel. It is a soft, delicate, textured, and very smooth fiber originating from silkworm cocoons as a continuous filament of protein-aceous polymer resembling synthetic fibers. Among many varieties, the Mulberry silk (
Efforts have been made to improve the physical properties of silk,1–7 including dyeability and colorfastness with different classes of dyes (e.g., natural, reactive, acid, basic, and metal complex dyes).8–12
Earlier studies of turmeric extract dyeing of silk were successfully performed at dyeing temperatures between 60 °C to 100 °C using different mordants (e.g., FeSO4, alum, tartaric acid, and CuSO4).13,14 The color strength increased with the depth of shade, with the best results at pH 7. 14 The washfastness and crockfastness of turmeric dyed silk were observed to be nearly similar to that of cotton dyed with the same dye, in most cases between the ratings of 2–3 to 3–4. 14
Kinetic models are very important for a dye house manager or dyeing specialist as it shows how various experimental conditions affects the speed of chemical reaction and thus helps to select preferable conditions for dyeing. But in fact, research on silk dyeing adsorption kinetics was performed for very few dyes up to now.
The adsorption kinetics and thermodynamics of lac dyeing was reported by several researchers.15–17 Furthermore, equilibrium and kinetic modeling of indigo carmine, sodium copper chlorophyllin dye, and acid dye on silk were also investigated.18–20 However, the dyeing kinetics and thermodynamics of silk dyed with turmeric from the
Turmeric is rich in curcuminoids and belongs to the diaroylmethane group named diferuloylemethane. 22 This natural dye is environmentally friendly and has antibacterial properties.23–28 The active coloring component in turmeric rhizome is curcumin (C.I. number 75300, C.I. Natural Yellow 3). 29 It has a molecular weight of 368.38 g/mol and molecular formula of C21H20O6 . 30 The chemical structure of curcumin is shown in Fig. 1. 22
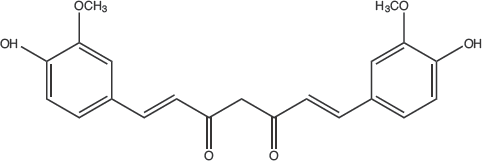
Chemical structure of curcumin (keto form).
This research focuses on regression analysis for two different kinetic models of dyeing woven silk fabric with turmeric extract. An investigation of the thermodynamic aspects was also made, adopting kinetic rate constants from the best ft kinetic model.
Experimental
Material
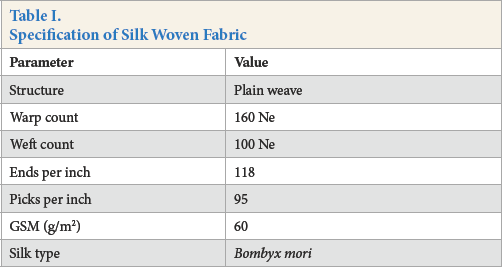
In this work, degummed and bleached 100% plain woven silk fabric was supplied by Rajshahi Silk Industries. The fabric specifications are listed in Table I.
Specification of Silk Woven Fabric
Mordanting
Before dyeing, the fabric was mordanted with 0.5 g/L FeSO4 at 70 °C at a 1:20 material liquor ratio (MLR) for 10 min.
Extraction of Turmeric Dye
Turmeric powder was supplied by Square Food and Beverage Ltd. The dye was obtained by extracting 1 g/L powder with deionized water at 95 °C and pH 7 for 90 min. The solution had a yellowish color.
Dyeing Process
Dyeing of silk fabric by turmeric extract was done in a Mathis Labomat laboratory dyeing machine equipped with a programmed temperature controlling system using infrared (IR) heating and a combined air-water cooling unit. Tree different temperatures (70 °C, 85 °C, and 100 °C) were used for dyeing at pH 7 with a 1:20 MLR in a 500 mL dyebath. The dyeing process was continued for 100 min with 1.0 mL of dye solution removed every 2 min for the first 20 min, every 5 min from 20 to 40 min, and every 20 min from 40 to 100 minutes, for the spectroscopic absorbance measurements. After dyeing, a hot wash was carried out at 80 °C in water and then a cold wash was done at room temperature (28 °C). Each fabric was soaked in water at a 1:50 MLR, stirred gently for 5 min, and then taken out for squeezing. Finally, the samples were dried in a dryer for 10 min at 60 °C.
Kinetics Experiments
A UV-Visible spectrophotometer (UV 1800, Shimadzu Corp.) was used for absorbance measurements with quartz cuvette cells of 1-cm path length. Dye concentrations were determined at time zero and at subsequent times from the absorbance values at λmaxof 419 nm. The concentration of dye in liquor was calculated by the Beer-Lambert equation (Eq. 1).
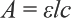
The amount of dye (mg/g) absorbed on silk at any time
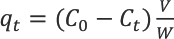
Examination of Fiber Cross Sections
To avoid false equilibrium and confirm that the dyes fully penetrated the inside of the fiber upon reaching equilibrium, cross sections of dyed silk fabrics were observed under a digital microscope (Micros MCX100LED). Only the samples at equilibrium were considered and compared with an undyed silk sample. The images are shown in Fig. 2.
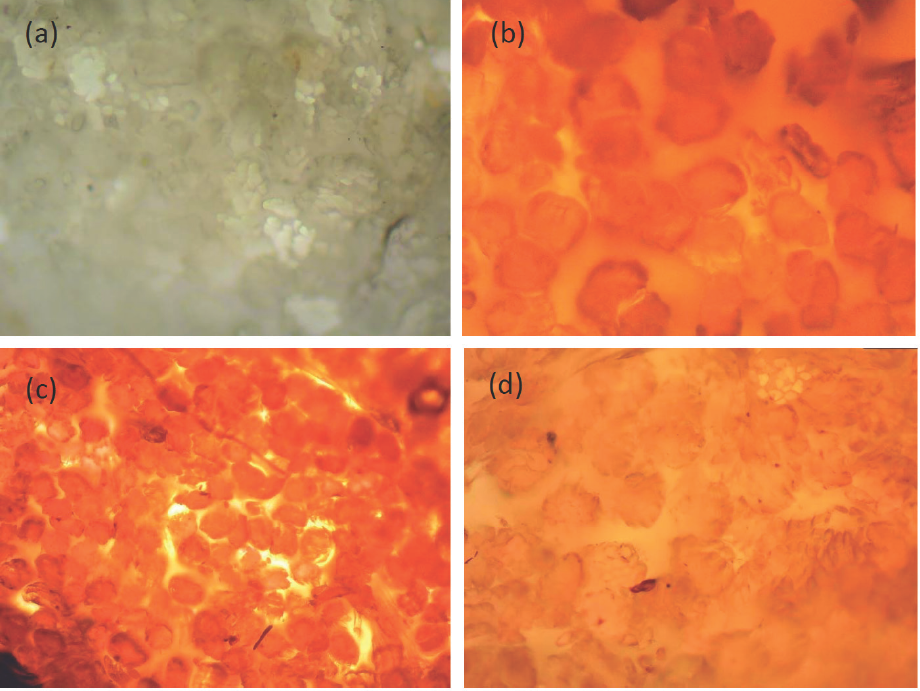
Cross-sectional view of (a) greige silk, (b) silk dyed at 70 °C for 40 min, (c) silk dyed at 85 °C for 20 min, and (d) silk dyed at 100 °C for 10 min.
Results and Discussion
Temperature Effect on Turmeric Extract Adsorption
The initial dye adsorption rate (
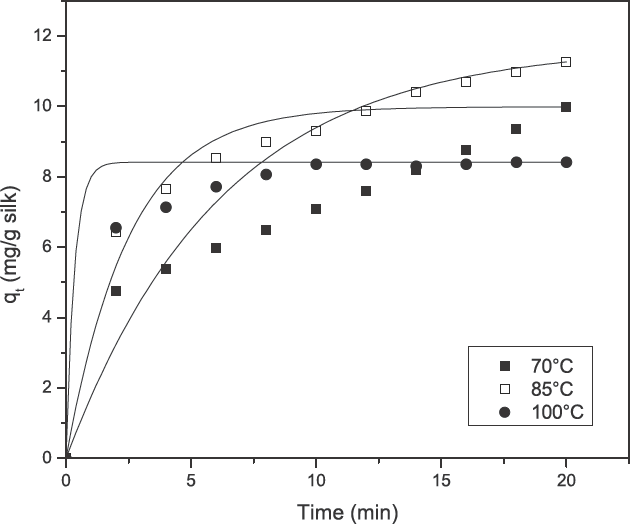
The effect of contact time and temperature of turmeric dyeing on silk (0–20 min) at an initial dye concentration of 1 g/L, MLR 1:20, and pH 7.0.
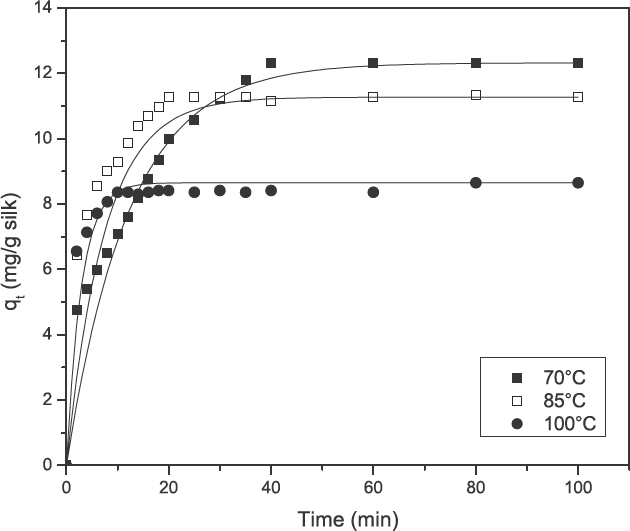
The effect of contact time and temperature of turmeric dyeing on silk (0–100 min) at an initial dye concentration of 1 g/L, MLR 1:20, and pH 7.0.
Kinetics of Adsorption
Pseudo first-order and second-order kinetic models were applied to the experimental data to represent the adsorption kinetics of turmeric yellow dye on silk. The linear form of the pseudo first-order equation (Lagergren equation) is given in Eq. 3.
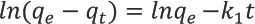
The pseudo second-order kinetic model based on the adsorption equilibrium can be expressed in linear form in Eqs. 4 and 5.31,32
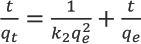
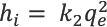
Kinetic data, obtained from turmeric yellow dye adsorption in the current study, was analyzed using the pseudo first-order kinetic model (Eq. 3) as well as the pseudo second-order kinetic model (Eq. 4) and is shown in Figs. 5 and 6. The results are depicted in Table II. The correlation coefficients (
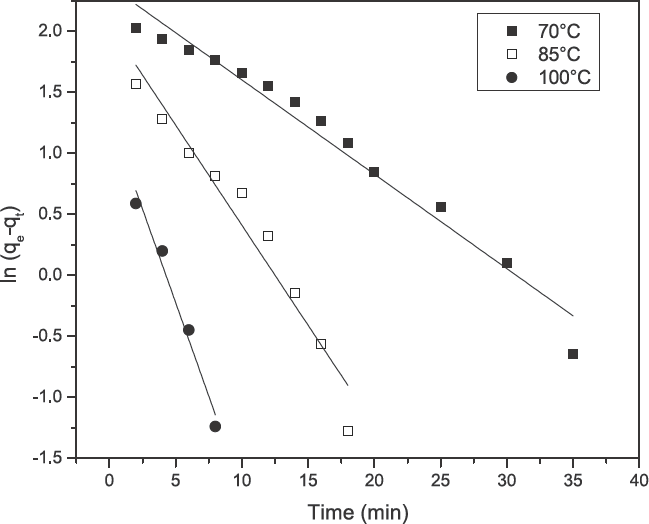
Plot of the pseudo first-order equation at various temperatures for the adsorption of turmeric yellow on silk.
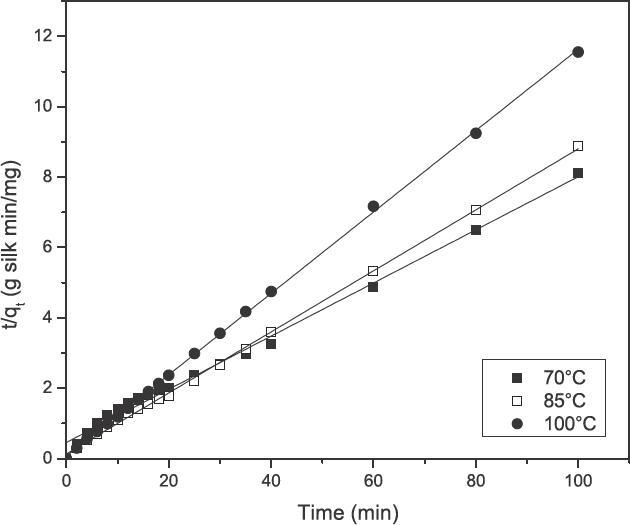
Plot of the pseudo second-order equation at various temperatures for the adsorption of turmeric yellow on silk.
Comparison of Pseudo First- and Second-Order Adsorption Rate Constant of Turmeric Dyeing on Silk a
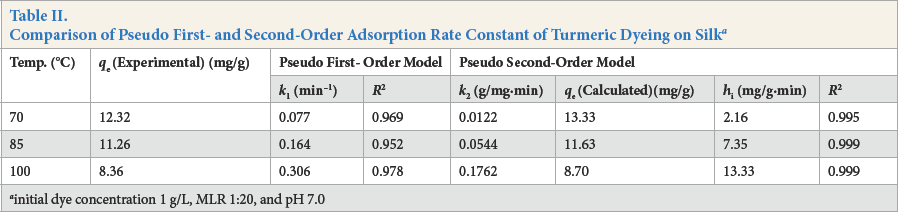
initial dye concentration 1 g/L, MLR 1:20, and pH 7.0
Activation Parameters
From the rate constant of pseudo second-order kinetics
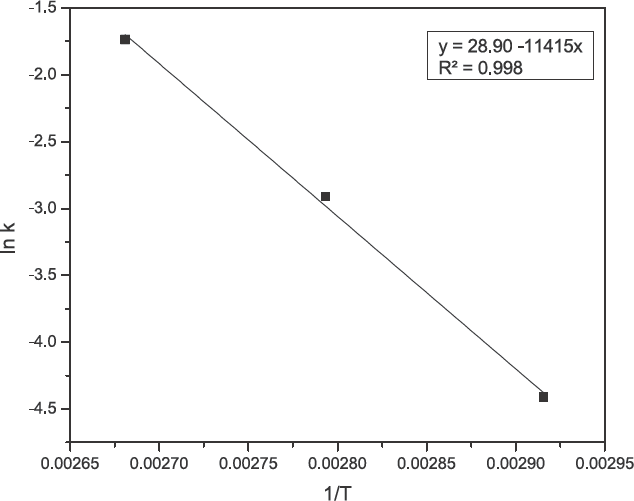
Arrhenius plot for the adsorption of turmeric yellow on silk.
Activation Parameters for the Adsorption of Turmeric Yellow on Silk a
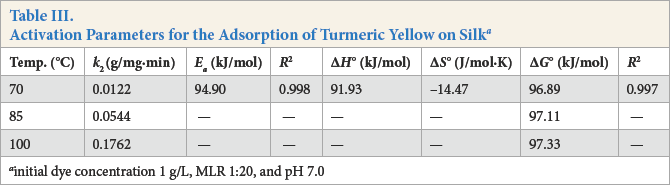
initial dye concentration 1 g/L, MLR 1:20, and pH 7.0
The enthalpy
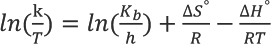
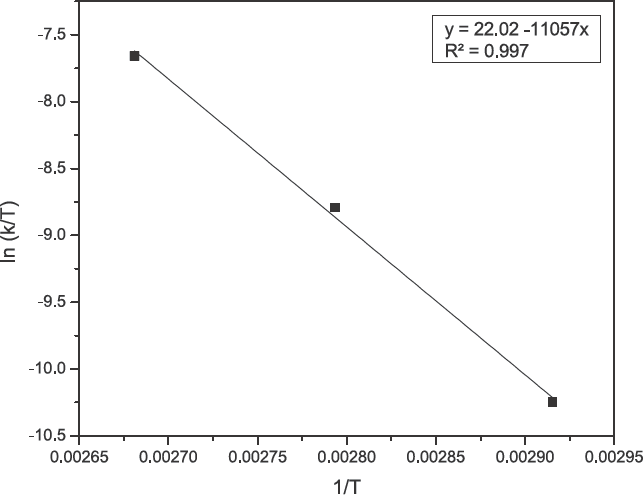
Eyring plot for the adsorption of turmeric yellow on silk.
The Gibbs energy of activation (Δ
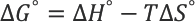
The calculated values are listed in Table III. The
Conclusions
The kinetics of turmeric dyeing on silk fabric ft well with the pseudo second-order kinetic model in this study. Initial dye adsorption rates (
