Abstract
Transport of cotton fiber samples to remote locations for water content testing was studied. Cotton fibers were sealed in a glass vial with a septum in an aluminum crimp cap and analyzed within three hours for water content by Karl Fischer titration. At least five days were allowed between capping and analysis for shipping from remote sites to locations with the apparatus. A tight seal was not maintained by changing the septa and crimp cap. Instead, capped vials were put in aluminum foil envelopes with minimal free space, heat sealed, and stored under diverse conditions. There was no storage effect on the water content of the cotton. The envelope effectively preserved the original water content in the fibers. An air transport trial produced similar results.
Keywords
Introduction
The standard test method for determining the total amount of water (free and bound) in lint cotton by oven evaporation combined with volumetric Karl Fischer titration (KFT) is fully automated (ASTM D7785). 1 The system includes a sample turntable. Glass vials are sealed with a septum set in an aluminum-crimp cap and lowered into a single-sample oven. A double-walled needle forced through the septum allows for dry nitrogen to transport the evolved water vapor to a dry titration cell where it is titrated with Karl Fischer reagent. Platinum electrodes monitor the titration and detect the end-point. Results are displayed as percent water. The titer of the Karl Fischer reagent is determined using a water standard and the overall system is checked using an oven standard.
Recovery of water available for titration is quantitative, > 99.99%, and the documented selectivity for water over the other materials in white lint is high. 2 Errors in the KFT method have been minimized. 2 Accuracy of the test method was confirmed by comparability studies using low temperature distillation (LTD). This weight-loss method slowly dries the specimen at a reduced temperature in a flowing stream of ultra-dry nitrogen. 2
ASTM D7785 has been successfully used in the cotton industry.3–5 More recently, the method was extended to the measurement of water content in brown cotton, fax, and rayon. 6 The water content of scoured and bleached (S&B) cotton was less than that of greige fiber from Pima or Upland cotton. One reason is that the dust and trash in the greige cotton fiber contributes to the observed water content. 3
Due to the requirements of the KFT system (i.e., initial cost plus consumables, operation in a laboratory environment, and the need for trained operators), their availability is limited in the cotton industry. An alternative is to ship the sealed vials to sites with the apparatus. This would provide industry-wide use at reduced cost. However, is the tightness of the aluminum crimp cap vials used in D7785 suitable for vial storage during transportation and in the recipient laboratory before actual analysis (see Table I)?
Terms and Nomenclature
Glatzel and Well commented that publications on the tightness of septum-capped vials (screw cap and crimp cap) are scarce. 7 No study was reported on storage of water vapor in crimped-cap vials. To evaluate septum-capped vials for storage of gas samples of nitrous oxide (N2O) during air transport, the low pressure (586 mbar inside passenger and freight airplanes) was simulated in desiccators. 7 After 10 days storage, the greenhouse gas concentration inside the vials was determined by gas chromatography. They concluded that in contrast to crimped-cap vials, when Exetainers screw-cap vials were used, no N2O loss following low-pressure storage was detectable after 10 days. Frozen storage of drug products at -60 °C was evaluated under static conditions using aluminum-crimped caps. 8 After one week of storage, vials were allowed to equilibrate to room temperature (RT) for 1 h prior to a helium leakage test and headspace analysis for other gases. Vials were found to leak. Shipping stress was not investigated. It was reported that during a bumpy food distribution process, leaks resulted in more rapid exchange of gases between the package and outside environments. 9 Laughlin and Stevens found Exetainers screw-cap vials suitable for long-term storage of N2 and N2O at RT and ambient pressure. 10 Spotl tested the same type Exetainers for changes in 13 CO2 during 12 days of storage and found no changes under ambient laboratory conditions. 11 Segschneider et al. found that, among the various seals tested (i.e., butyl, ethylene propylene diene monomer (EPDM), silicone, Teflon-faced butyl stoppers, and septa), only the butyl-rubber stopper provided an effective seal for up to seven days. 12
In this research, we divided the work into two categories: (1) basic research aimed at increasing scientific knowledge at the fundamental level, and (2) applied research aimed at using the basic research to solve the problem using crimped vials.
The objective of the study was to determine if KFT aluminum-crimped cap vials maintained the original water content (OWC) of the cotton test specimen after five-days storage in the vials. First, we investigated the fundamentals to understand and predict storage results. Next, we tested and confirmed the vial closure system integrity by different approaches: (1) aluminum-crimped cap and time series studies at 21 °C storage in high and low relative humidity (RH) chambers, (2) changed the composition of the septum and metal crimped cap, and stored in the humidity chambers for the full five days, (3) inverted the septa in the crimp cap and stored in the humidity chambers for five days, and (4) stored the aluminum-crimped cap vials in a foil envelope secondary container, and stored five days in different environments: in humidity chambers, frozen, and in a pickup truck representing ground transport. Finally, we confirmed that the secondary container concept worked with air transport of cotton specimens in aluminum-crimped cap vials.
Fundamentals
The general procedure for automated water content determination by KFT is as follows. The bulk cotton sample is conditioned in a textile testing laboratory (TTL) under standard conditions of 21 ± 1 °C and 65 ± 2% RH. Next, the test specimen is weighed, transferred to a standard Metrohm 6-mL glass vial and the entrance hole of the vial is covered with Metrohm aluminum-crimp cap. After crimping with an electronic crimper, the vial is placed on the KFT sample turntable for immediate analysis. The time interval from capping to analysis is only 1–3 h, depending on the position of a particular vial on the turntable (1 to 35). In the limited time period between crimping and analysis, the amount of water vapor that may leak across the cap-vial interface is insignificant. Container closure leaks did not present a problem in development of the KFT reference method for water in lint cotton.1,2
If the capped vial is to be shipped to another location for KFT analysis, as, for example, in inter-laboratory collaborations, the shipment may take one-to-two days. The transport process itself may be bumpy, along with undergoing temperature and humidity variations. After arrival at the test site, the samples must be scheduled for analysis; one-to-three days additional storage may be required before the analysis.
In this study, five days was the target time interval between capping and KFT analysis. The ground transport process itself was simulated by storing the capped vials in a pickup truck driven and used for local transportation for five days. These conditions may require a special vial storage procedure to prevent significant leaks.
Consider a 0.1 g cotton test specimen in a sealed 6-mL glass vial for water analysis by KFT. The total volume in the vial, including the neck of the container, is 9 mL. If the volume of the cotton itself is neglected, then the headspace volume (Table I) is 9 mL. The presence of the headspace allows for partitioning of the water in the fibers into the vapor or gaseous phase. The total mass of water in the vial can be expressed as in Eq. 1.
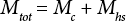
Mtot is the total mass of water in the cotton and gaseous phases sensed by KFT, Mc is the mass of water in the cotton, and Mhs is the mass of water vapor in the vial headspace.
The container closure system consists of the glass vial, rubber septum, and aluminum-crimp cap. The equilibrium between Mc and Mhs is sensitive to a leaking vial seal. If water vapor in the headspace leaks out into the surrounding environment, then the actual mass of Mc and Mhs may change. The end result could possibly be a significant variation in the mass of water Mtot sensed by KFT.
Leak Rate Reduction
Direct Method—Modified Septum and Metal Cap
The time dependency of water vapor leakage from a KFT glass vial loaded with a cotton test specimen and sealed with the standard Metrohm septum and aluminum-crimp cap has not been modeled. The critical pressure to maintain an adequate seal over the five-day storage period is unknown.
Other factors, like deformation of the crimped cap on the residual sealing force, may contribute to vial leakage. Thus, changing the rubber septum (e.g., to a different hardness) and metal cap (e.g., aluminum to steel) may not prevent seepage. The leak rate may increase.
Indirect Method—Aluminum-Crimp Cap Vial in Secondary Leak-Free Container
Under Textile Testing Conditions
Suppose that under textile testing conditions the aluminum-crimp cap vial with cotton inside is placed in a leak-free aluminum foil envelope with a minimum excess or free volume of air (herein referred to as free space, Table I). Since the free space has been minimized, the rate of water vapor that may diffuse across the vial seal will rapidly slow down further diffusion due to the reduction in the concentration gradient inside and outside the vial.
Eq. 2 describes the correction (ΔMfs) to the total mass of water in the vial (Mtot), where ΔMfs is the change in the mass of water in the free space due to the vial seal, which does not remain tight during the five-day period.
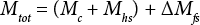
Note that ΔMfs may be positive or negative, depending on the compartment (headspace or free space) with the higher initial water concentration. The correction is a function of the temperature, headspace and free space volumes, and water vapor concentration difference. If the correction is not significant, Eq. 2 reduces to Eq. 1 and the OWC of the cotton in the vial is maintained after five-days storage.
Frozen
Now consider that the aluminum-crimped cap vial with cotton inside is placed in the leak-free foil envelope with minimal free space and the temperature lowered. The new temperature of the envelope is about –15 °C (approximate temperature in a refrigerator's freezer). This is above the glass transition temperature of commonly-used rubber septa between -55 °C and -65 °C and, therefore, should not influence the vial seal. Additionally, –15 °C is above the glass transition temperature (< -100 °C) of the ultrathin polymer coating on the inside of the envelope. The open end of the envelope is heat sealed (the polymer film melts and solidifies) to provide the watertight seal.
The water vapor in the vial headspace is converted to ice. This phase transition prevents the water molecules from diffusing out of the vial. Similarly, the water vapor in the envelope's free space is converted to ice and is prevented from diffusing into the vial. Thus, on both sides of the vial's septum, the frozen state exists and no water vapor is present to diffuse across a leaky vial seal.
Eq. 3 describes the frozen state, where Mtoti is the total mass of ice in the vial, Mci is the mass of frozen water in the cotton, and Mhsi is the mass of frozen water in the vial head-space (ice coating on the inside glass wall).
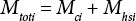
Upon thawing the frozen vial to RT, Eq. 3 reverts back to Eq. 1, since Mci = Mc and Mhsi = Mhs. The OWC of the cotton in the vial is maintained after five-days frozen storage. The aluminum-crimped cap vial that is put in the secondary leak-free container and frozen provides the ideal state to maintain the OWC over extended storage.
However, to maintain -15 °C during shipment, the envelope must be placed in an insulated container with a large amount of an appropriate frozen gel pack (for example, those available at shipping/delivery services) to last up to five days. Compared to regular shipments under ambient conditions, the frozen shipment of the envelope would be expensive. In the context of this paper, the frozen state is limited to the envelope placed in a refrigerator freezer to provide for control samples with a high probability of maintaining the OWC in the cotton fibers.
Ground Transport
Ground transport is a necessary component of any air shipment. In the context of this paper, the crimped vials in the foil envelopes take the following one-way route: by ground transportation from the authors’ lab in New Orleans, LA, USA to the airport; by air shipping to the collaborator in Florida; and finally, by ground hauling to the collaborators’ lab in Riverview, FL, USA.
Eq. 4 describes the correction (ΔMfsgt) to the total mass of water in the vial after ground transport (Mtotgt). Note that ΔMfsgt is the change in the mass of water in the free space due to the vial seal, which may not remain tight during the bumpy five-day ground transport period.
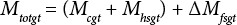
Note also that ΔMfsgt may be positive or negative, depending on the compartment (headspace or free space) with the higher initial water concentration. The correction is a function of the headspace and free space volumes, and water vapor concentration difference. The net effect of the bumpy transport 9 should be more rapid mixing of the gases in the headspace and free space. If the secondary container remains leak-free, then the correction should not be significant. Consequently, Eq. 4 reduces to Eq. 1, since Mcgt = Mc and Mhsgt = Mhs, and the OWC of the cotton in the vial is maintained after five-days ground transport.
Air Transport
Eq. 5 describes the correction (ΔMfsat) to the total mass of water in the vial (Mtotat) after air transport. In more detail, ΔMfsat is the change in the mass of water in the free space due to the vial seal, which does not remain tight during the air transport period
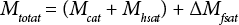
The correction is a function of the headspace and free space volumes, and water vapor concentration difference. The effect of the air transport may be possibly significant expansion of the secondary container volume at reduced pressure. In this paper, this effect is reduced by using two foil envelopes and minimizing the free space volume to provide a stiff secondary container wall that is resistant to an increase in dimensions.
If the increase in the free-space volume is minute and the secondary container remains leak-free then the correction should not be significant. Consequently, Eq. 5 reduces to Eq. 1, since Mcat = Mc and Mhsat = Mhs, and the OWC of the cotton in the vial is maintained after air transport.
Experimental
Cotton Fibers
Three different cottons were used in the research including an S&B, an unknown Pima, and an unknown Upland cotton. The S&B cotton had been mechanically cleaned prior to wet cleaning and is available in retail stores as cotton balls. A bag of cotton balls was chosen at random from the large supply in a retail store. The other two cottons were unprocessed ginned lint (greige cotton) and were selected at random from the pool of similar kinds of samples in our laboratory.
Ten different cottons were used in the confirmation (air transport trial) research. The cottons came from the Agricultural Marketing Service (AMS) in Memphis, TN, USA and were selected to represent different crop years, areas grown, and maturity. Five were raw lint from cotton bales and the remaining five from other bales that had been mechanically cleaned.
Conditioning Environments
Cotton samples for this study were conditioned in (1) the TTL under the previously described standard conditions and (2) humidity chambers placed in the TTL. Heat transfer through the walls of the chambers allowed for temperature equilibrium with the larger TTL. Tree acrylic conditioning chambers, with dimensions 9” × 10 ½” × 16”, housed the pans with the different salt solutions used to produce different RH conditions. The chambers were available online as Stackable Acrylic Desiccator with Sliding Trays (www.coleparmer.com, T-08933-10). The sliding tray for solid desiccant was replaced with aluminum-foil bake pans for the various salt solutions. The pans were available online (www.webstaurantstore.com, 9991155), with dimensions (inches): top 7 ⅞ × 7 ⅞, bottom 6 ⅞ × 6 ⅞, and depth 1 5/16.
Deionized water was used to generate the 100% RH environment in one of the chambers. The other two chambers contained (1) a saturated aqueous salt solution and (2) sodium nitrite-magnesium chloride solution that provided 65% and 35% RH conditions, respectively. 13 Two pans were used per chamber. Empty pans were placed on a paddle board, filled to 1” depth, and carefully pushed into the acrylic chamber.
KFT Vials and Caps
Aluminum, stainless steel, and 2-leg bromobutyl-rubber stoppers were used to cap the vials (Fig. 1). The standard aluminum-crimp cap (with septum) was provided by the KFT instrument manufacturer. The steel-crimp cap (without septum) (www.chromres.com, 320608, 20-mm Magnetic Crimp Cap with 8-mm hole) and the 2-leg bromobutyl-rubber stoppers (www.wheaton.com, W224100-406, 20 mm) were available online. In addition, the Teflon-coated silicone septa in the standard aluminum-crimp cap were used with the silicone side facing the vial (the recommended position) as well as inverted (i.e., facing the cap in other experiments, for both aluminum and steel caps) as described in Table I.

KFT test specimen vials for five-day leakage tests. From left: Silicone/ Teflon combination septum in aluminum- and steel-crimp caps, respectively. Third from left: vial plugged with 2-leg bromobutyl-rubber stopper. Inverted septa and stoppers show thin Teflon layer and the two legs. Black lines on aluminum crimp cap are about 4-mm long; compare to the 10-mm center hole.
Preparing KFT Test Specimens
Bulk cotton samples and KFT vials and caps were conditioned for a minimum of 24 h in the TTL. Test specimens were then prepared in the TTL by pulling tufts at random from the larger samples, weighed to 0.1000 ± 0.0005 g, and placed in the preconditioned KFT vials. Vials were capped accordingly for the various experiments; three replicates of each capping option for each humidity and cotton were used. Sample vials were then placed in the chambers to be conditioned for five days. For comparative purposes, the controls consisted of test specimens that remained in open vials in both the TTL and humidity chambers. Control vials were sealed just before KFT analysis.
Aluminum-Foil Envelopes
Aluminum-foil envelopes provided a heat-sealable moisture barrier for the capped vials during the storage experiments. This secondary container was available online (www.protectivepackaging.net, MST5X12, MIL-PRF-131C1MS360, 5” × 12”) along with the Crimper Sealer 6” Hand Held #A1E-HHCW heat sealer.
To ensure a good seal, a double bagging system was used. For the presumptive experiments, three capped vials (replicates) of the same cotton were placed in the aluminum envelope and heat sealed with a tight ft, of approximately 4” × 3 ”, to minimize the free space (Table I) in the envelope. The filled packet was then placed in a second aluminum bag and the process repeated; a tight ft of the secondary container limited the space between the two envelopes. For the confirmation experiment, five capped vials (replicates) of the same cotton were placed in the envelope.
Water Content Determination KFT
For all presumptive experiments, all water contents were determined by KFT (ASTM D7785). 1 All hardware, software, and reagents were supplied by Metrohm. The KFT apparatus consisted of a fully-automated Metrohm 774 oven sample processor and micro oven held at 150 °C and dry nitrogen to transport released water vapor into the titration cell. A 35-glass vial carousel, 841 Titrando system with 800 Dosino dosing electronic buret, an 801 stirrer, an 803 Ti stand for the titration cell with platinum electrode, and the Tiamo 1.2 titration software all worked seamlessly to measure water content. One buret was for the addition of the titrant; a complete setup required a pump to add fresh solvent. Hydranal composite 5K was used as the titration reagent and Hydranal medium K was the solvent in the titration cell.
For the confirmation (air transport trial) experiments, capped vials in the aluminum-foil envelopes were air shipped to Orlando, FL, and ground transported to the collaborator's lab in Riverview, FL. Water content was determined by KFT (ASTM D7785) 1 within 3 h of removing the capped vials from the aluminum-foil envelopes. Ten replicates were run per cotton. Storage time of the vials in the envelopes at under ambient conditions the collaborators'lab before analysis ranged from analysis on the day of arrival to three days.
Near Infrared (NIR) Spectroscopy
NIR reflectance spectra (Bruker Model MPA FT-NIR instrument with OPUS 5.0 software) taken through the bottom of capped vials was used to confirm an increase in water content in the fibers due to water vapor leaking into vials stored in the 100% RH chamber. 2 The peak height at 1940 nm was measured.
LTD
For the confirmation experiments, the control samples with known QWC were analyzed in our New Orleans laboratory by LTD, an oven-drying method at 75 °C that uses dry nitrogen to purge the moisture from the fibers. 2 Half-gram samples had been conditioned in open vials in both the TTL and 65% RH humidity chamber. Samples were oven dried at 70 °C in nitrogen that was dried using a built-in purifier in the compressed gas tank. Water content in the cottons was measured by sample weight loss. There were six replicates per cotton. The certificate of analysis of the nitrogen gas showed a moisture content < 20 ppb (w/v).
Statistical Tests
Mean and standard deviation of the results were calculated, including water content by KFT, NIR peak height at 1940 nm, and capped vial weights, before and after storing in the humidity chambers. The statistical software JMP was used to determine one-tailed t-tests to test for significant difference (set at p < 0.05) between storage condition mean water content and control mean water content.
Results and Discussion
Presumptive and Confirmatory Tests—Cotton Samples and Controls
In the context of this research, presumptive tests were the initial experiments conducted to determine feasibility of specific capped vial storage treatments. These studies excluded air transport, and instead, looked for promising treatments to maintain the OWC of the cotton specimen in the vial for five days. The presumptive results relied on cottons (one each), selected at random from three cotton types: Upland, Pima, and S&B. The control samples provided the known OWC; experimental results were compared with the control OWC to determine which vial storage treatments showed potential. All results, OWC and after vial storage, were measured in our lab by KFT. We should emphasize that the presumptive outcomes did not apply to air transport and the population of cottons within a cotton type.
Confirmatory tests were used for further analysis of a specific storage treatment—an actual air transport trial. The scope of the confirmation setup ranged from sample conditioning and vial capping in our lab in New Orleans to ground transport to the airport, air shipment to Orlando, and ground transport to the collaborator's lab in Riverview. The water content of the shipped fibers was measured at the collaborator's lab by KFT. In this first air transport trial, we included a number of Upland cottons (raw and mechanically cleaned). Compared to Pima cottons, most commercial cottons are Upland. To provide a more rigorous confirmation, the control samples were measured in the New Orleans lab for OWC by oven drying in nitrogen. 2
Presumptive, Part I—Time Series Study with Standard Aluminum-Crimp Cap
The three cotton samples for this study were conditioned at 21 °C and in two different humidity chambers (100% RH and 65% RH). Note that the humidity gradients were in opposite directions for the 100 and 35% RH environments relative to the standard 65%: ΔRH = 100% - 65% = 35% and ΔRH = 35% - 65% = -30%. Thus, if the capped vials were not tight, moisture will leak either into or from the vials and closure integrity was lost.
Table II lists the daily storage results by KFT for vials capped with the standard aluminum-crimp cap. There was inconsistency in the tightness of vial seals. Some of the capped vials remained tight while others leaked. Similar findings were reported in helium leakage tests after extended storage of drug products in crimped-cap vials. 8
Time Series Mean Storage Data at 100% and 35% RH a
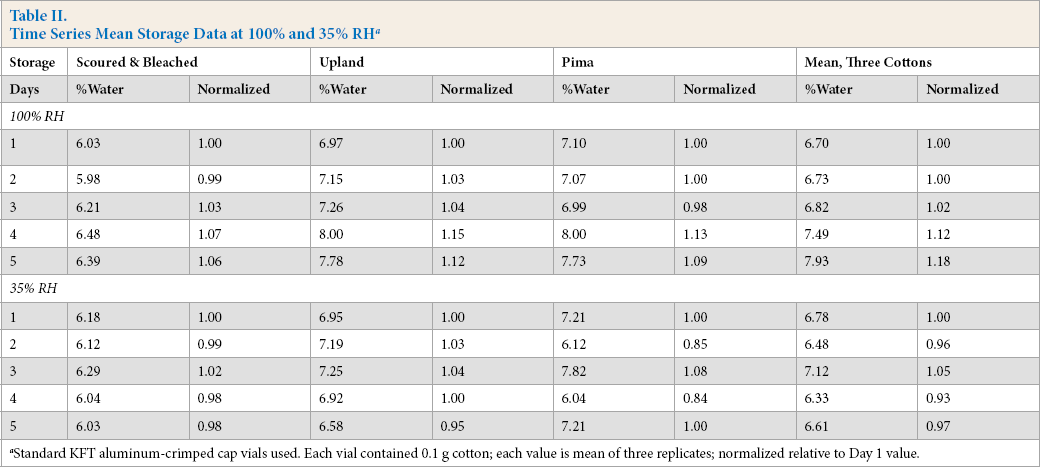
Standard KFT aluminum-crimped cap vials used. Each vial contained 0.1 g cotton; each value is mean of three replicates; normalized relative to Day 1 value.
The normalized water levels (Table II) simplified the interpretation of results. For the 100% RH study, the increases in mean water content, relative to the day 1 value, were as follows (day 4 and day 5): S&B, 7% and 6%; Upland, 15% and 12%; Pima, 13% and 9%; and the average of the three cottons was 12% and 18%, respectively. For the 35% RH study, smaller changes were observed.
An independent confirmation of water vapor leaking into the capped vial from its surroundings in the 100% RH chamber was demonstrated using an NIR sensor (Fig. 2). Water in the cotton absorbs the 1940 nm source radiation; the NIR peak height increased with storage time.
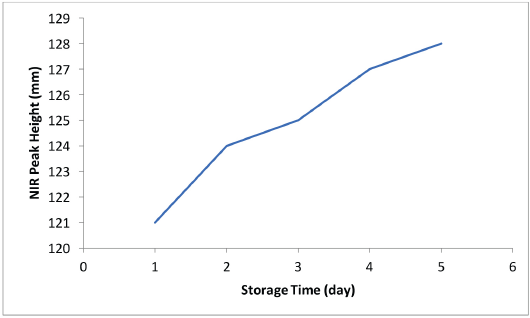
NIR peak height at 1940 nm showing ingress of water into the 0.1 g cotton in KFT vial sealed with aluminum-crimp cap and stored at 21 °C in 100% RH chamber. The spectra were taken through the bottom of the glass vial.
We believe the time series data presented is sufficient to demonstrate that over five days, the KFT vials, with standard aluminum-crimp cap, were not tight to water vapor in cotton. This finding is in agreement with other findings that crimped vials were not tight to other gases such as N2O and helium.7,8
Presumptive, Part II—Day-Five Study with Different Septa and Metal-Crimp Caps
The experimental data is presented in Tables III and IV, and plotted in Figs. 3 and 4. Fig. 3a illustrates the trends at high and low humidity. Note that the control bar represents the OWC in the control cottons (stored open in the TTL at 65% RH). The dashed horizontal lines show the expected five-day concentrations at 100 and 35% RH in gas-tight aluminum-capped vials. For all varieties of cotton, the three vertical bars would show the same water content with the non-leaking vials. Fig. 3a shows there was water vapor diffusion into the vials stored in the high-humidity environment, and also, leakage from the vials into the low-humidity surrounding.
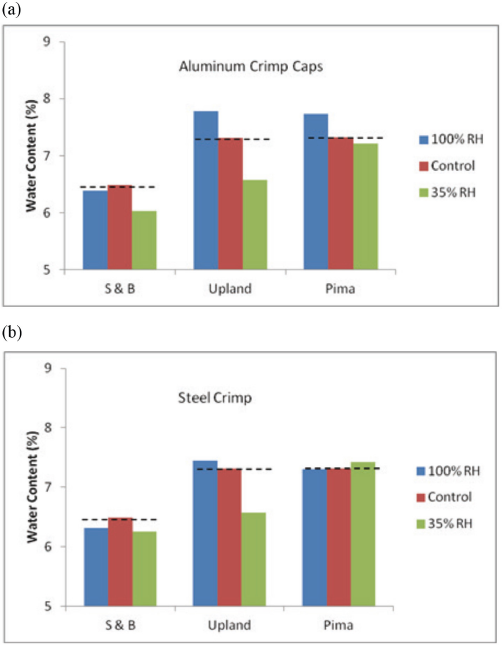
Day-five mean water content of 0.1 g cotton in capped KFT vials stored at 21 °C in 100% and 35% RH chambers. The vials were sealed with: (a) aluminum-crimp caps and (b) steel-crimp caps with septa taken from the aluminum caps. Cottons: S&B, Upland (Up), and Pima.
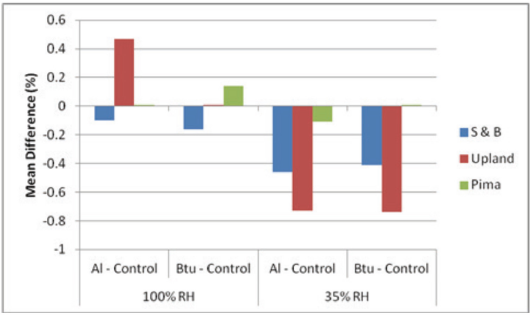
Side-by-side comparison of the mean difference in day-five water content between KFT vials sealed with aluminum-crimp caps and sealed only with the 2-leg bromobutyl-rubber stoppers. There was 0.1 g cotton in the vials, which were stored at 21 °C in the 100% and 35% RH chambers. The rubber stopper was replaced with the aluminum-crimp cap immediately before KFT analysis. Cottons: S&B, Upland (Up), and Pima.
With no obstruction in the vial aperture, the five-day mean water content for the control cottons stored in open vials in the humidity chambers and capped immediately before analysis (Table IV) shows a wide range of water content. For example, for the S&B cotton, the values at 100% and 35% RH were, respectively, 19.3% and 4.64%. In contrast, when the barrier in the aperture was an aluminum-crimped cap, the water contents (Table III) at 100% and 35% RH were, in that order, 6.39% and 6.03%. The ranges without and with the barrier were: without, 19.3% – 4.64% = 14.7%; and with, 6.39% – 6.03% = 0.36%. If the aluminum-crimped cap had been a tight seal, the expected range would have been 0%.
Day-Five Water Content (%) of 0.1 g Cotton in KFT Vials with Various Caps at Various RH Values a
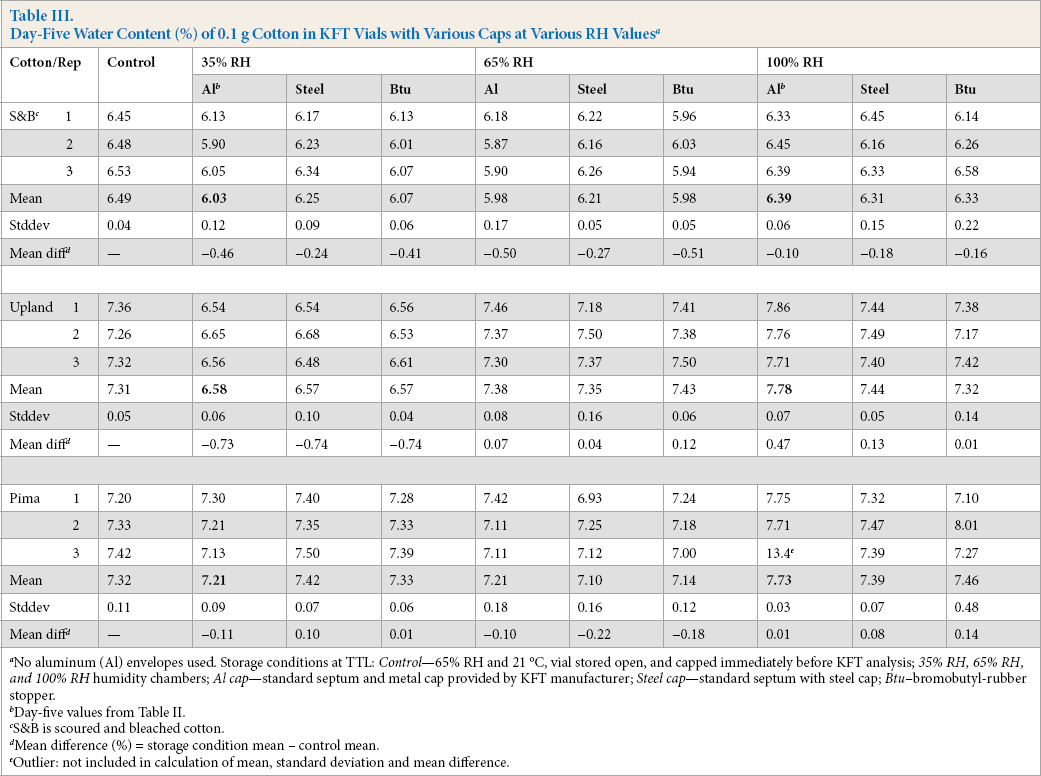
No aluminum (Al) envelopes used. Storage conditions at TTL: Control—65% RH and 21 °C, vial stored open, and capped immediately before KFT analysis; 35% RH, 65% RH, and 100% RH humidity chambers; Al cap—standard septum and metal cap provided by KFT manufacturer; Steel cap—standard septum with steel cap; Btu–bromobutyl-rubber stopper.
Day-five values from Table II.
S&B is scoured and bleached cotton.
Mean difference (%) = storage condition mean – control mean.
Outlier: not included in calculation of mean, standard deviation and mean difference.
Summary differences with respect to the control mean for open and capped vials are given at the bottom of Table IV. The open differences do not correlate well with the corresponding capped differences. We should note that in Glatzel and Well's paper, even with four to eight vials for each treatment in evaluation of septum-capped vials, differences were marked, yet not significant, due to variability in extent and frequency of leakage. 7 This is a characteristic of aluminum-crimped cap vials over extended time. In the present study with three vials for each specific treatment, one vial may have been leak free, the second, perhaps a small leak, and the third, perhaps a large leak. This leads to variability in results; increasing the number of vials for a specific treatment will not do away with variability if leaks do occur with a particular type of crimp cap.
Day-Five Mean Water Content (%) of 0.1 g Cotton in KFT Open Vials and Stored at 21 °C at Various RH Values
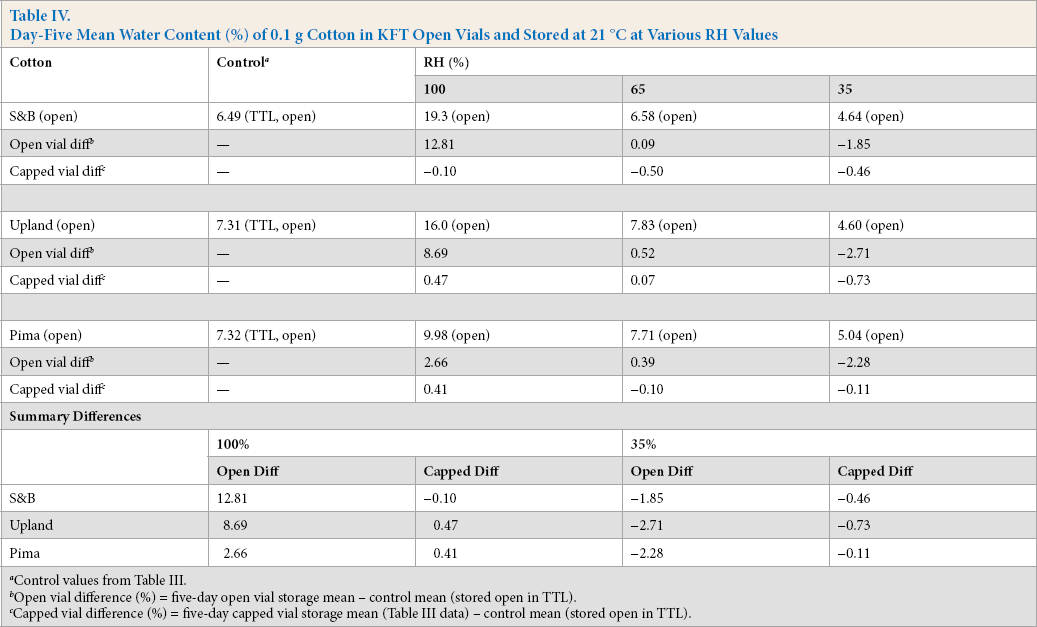
Control values from Table III.
Open vial difference (%) = five-day open vial storage mean – control mean (stored open in TTL).
Capped vial difference (%) = five-day capped vial storage mean (Table III data) – control mean (stored open in TTL).
Next, we hypothesized that we could stop the day-five leakage by changing the metal-crimp cap from aluminum to steel. The steel cap is stronger than the aluminum and should maintain tightness of the septa seal to a greater degree. The experimental results are given in Table III and Fig. 3b. Generally, the extent of leakage decreased with the steel crimp cap, but four mean differences remained that were greater than 0.10%: 35% and 100% RH for S&B and Upland cottons.
We then hypothesized that, rather than a septum (see nomenclature, Table I) to seal the vials, a rubber stopper (tapered plug) would be sufficiently tight to prevent leakage during storage in the humidity chambers. A 2-leg bromo-butyl-rubber stopper was used. Immediately before the measurement of the water content, the stopper was replaced with the standard aluminum-crimp cap. The results are given in Table III and displayed in Fig. 4. The figure shows mean differences relative to the control values, comparing the aluminum-crimp cap and bromobutyl-rubber stopper side-by-side. A significant reduction in leakage (p < 0.05) was found with the stopper, but only for the Upland cotton stored at 100% RH.
Presumptive, Part III—Day-Five Study with Inverted Septa
The common septa used in the aluminum- and steel-crimp caps consist of a blue silicone disk bonded to a white Teflon disk (Fig. 1). The Teflon disk faces the vial opening and is harder than the silicone rubber. Thus, inverting or flipping the two-component septum in the metal cap results in the softer silicone rubber pressing against the glass vial. Presumably, this would extend seal integrity over the five-day storage period in the humidity chambers.
Table V and Fig. 5 (bar clusters 1a and 1b, 2a and 2b, 3a and 3b, and 4a and 4b), present the side-by-side results for the standard and flipped septa placement in the metal caps. Generally, the rate of leakage increased with flipping, for both kinds of metal caps. Since these results were unexpected, we also compared—flipped septa only—differences in vial weight and KFT water content between the 100 and 35% RH chambers after five-days storage (Fig. 6).
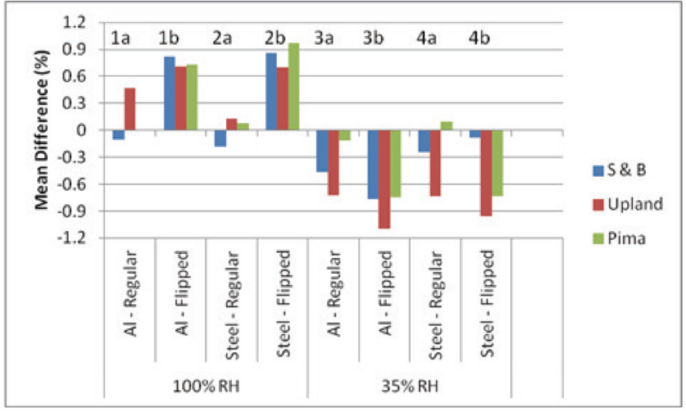
Side-by-side comparison of the mean difference in day-five water content between KFT vials sealed with the regular and the inverted position of the septum in the metal cap. There was 0.1 g cotton in the vials, which were stored at 21 °C in the 100% and 35% RH chambers. Aluminum and steel caps were used with common septa. Cottons: S&B, Upland (Up), and Pima.
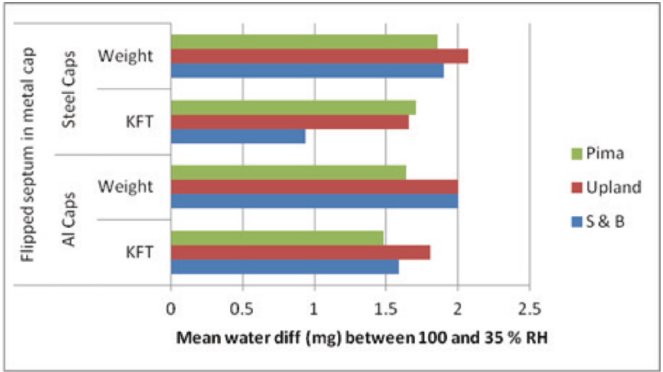
Side-by-side comparison of the mean difference in day-five amount of water (mg), measured by KFT and a balance, between the capped vials stored in the 100% and 35% RH chambers. There was 0.1 g cotton in the vials, water ingress at 100% RH, and egress at 35% RH. Aluminum and steel caps were used with common septa. Cottons: S&B, Upland (Up), and Pima.
Day-Five Mean Water Content (%) of 0.1 g Cotton in KFT Vials Capped with an Inverted Septum a
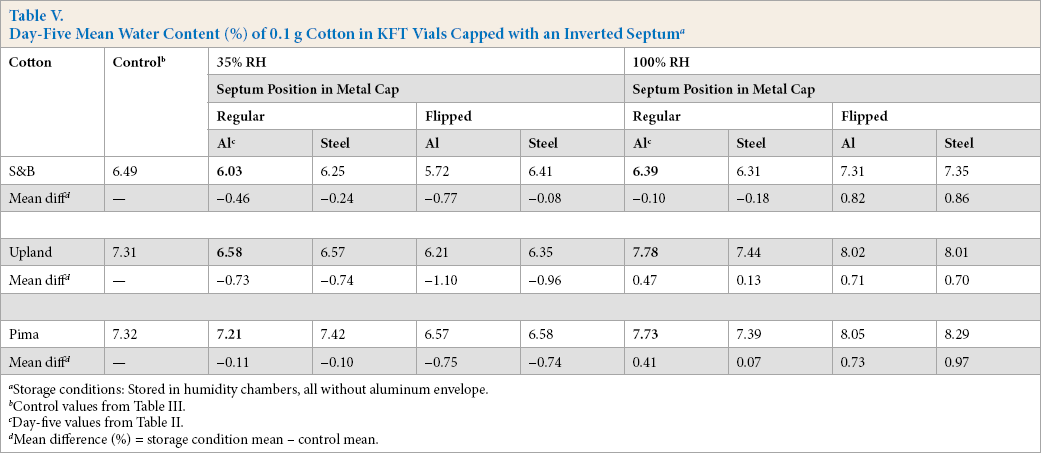
Storage conditions: Stored in humidity chambers, all without aluminum envelope.
Control values from Table III.
Day-five values from Table II.
Mean difference (%) = storage condition mean – control mean.
Capped vial weights (mg) were measured after five-days storage and the mass difference between the higher (100% RH) and lower (35% RH) humidity compartments computed. Similarly, water content (%) was measured by KFT at each humidity, the difference calculated, and converted to the change in mg water titrated. By and large, Fig. 6 shows that the weight differences associated with the inverted septa in the crimped metal caps confirm the KFT results. The inverted septa did not maintain a tight seal during the five-day storage test. Vial closure system integrity was compromised.
Presumptive, Part IV—Day-Five Studies with Aluminum-Crimp Cap and Sealed in Secondary Container
We hypothesized that an indirect, special packaging approach—sealing the capped vials in a secondary container—was needed for five-day storage. This second wrapping, closed with minimum free space, would rapidly reduce the rate of leakage across the vial septum, and allow for bumpiness or movement in ground transport along with temperature and humidity variations.
We used a commercially-available aluminum-foil envelope as a secondary storage container. After insertion of three KFT capped vials (replicates from the same cotton) into the envelope, it was trimmed to minimal size and heat sealed. Experiments to test the hypothesis involved vials containing 0.1 g of fibers that were sealed with the standard aluminum-crimp cap, placed in the envelope, the filled envelope heat sealed, and stored five days in different environments: ideal (frozen), extreme (ground transport), and intermediate (stationary humidity chambers).
Table VI shows the experimental results for the wide range of storage conditions. Note that all vials were placed in two rather than only one envelope, for two reasons. First, this was our first experience with heat sealing the thin flexible sheets of foil. And second, we did not have on hand sophisticated leak-testing equipment to test integrity of the heat sealing process.
Day-Five Water Content (%) of 0.1 g Cotton in Standard Aluminum-Capped KFT Vials a
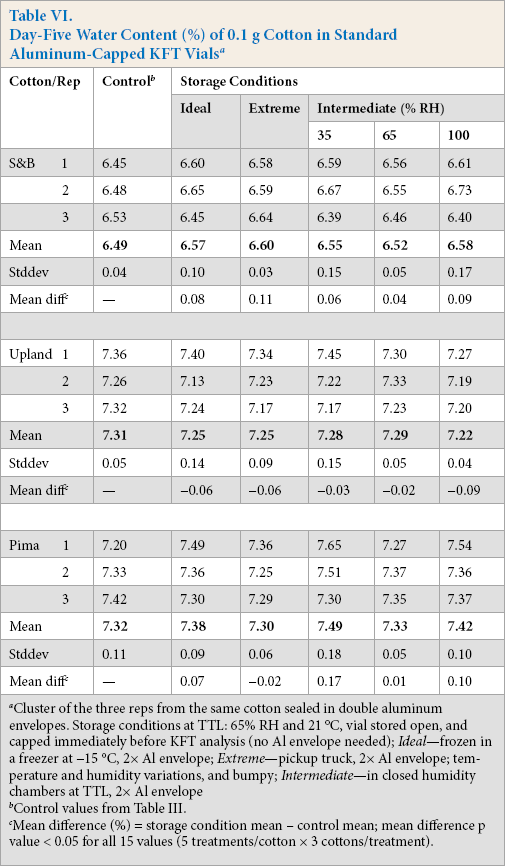
Cluster of the three reps from the same cotton sealed in double aluminum envelopes. Storage conditions at TTL: 65% RH and 21 °C, vial stored open, and capped immediately before KFT analysis (no Al envelope needed); Ideal—frozen in a freezer at -15 °C, 2× Al envelope; Extreme—pickup truck, 2× Al envelope; temperature and humidity variations, and bumpy; Intermediate—in closed humidity chambers at TTL, 2× Al envelope
Control values from Table III.
Mean difference (%) = storage condition mean – control mean; mean difference p value < 0.05 for all 15 values (5 treatments/cotton × 3 cottons/treatment).
The acceptance criterion for the Table VI results was defined as follows: the mean of three replicate vials showing a water concentration difference between any storage condition treatment and the corresponding control > 0.1% was not considered to be tight. While not statistically significant, the mean differences in Table VI are an indicator of the experimental design. The three ideal storage results (S&B, 0.08%; Upland, -0.06%; and Pima, 0.07%) were expected to demonstrate tightness (≤ 0.1%) because the water vapor levels on both sides of the septa seal was reduced by freezing.
Two of the three extreme storage results (Upland, -0.06%; and Pima, -0.02%) demonstrated tightness; the remaining cotton did not (S&B, > 0.1%). Again, while not statistically significant, ground transportation stress on the septa and foil envelopes should possibly be taken into consideration. The foil packets had been placed inside the cab of the pickup truck—on the floor facing the front seat. If any packet moved during transportation, it was pushed back in place. Since this experiment was conducted in New Orleans in June, the high temperature in the vehicle in parked mode may have exceeded 125 °F. When the air conditioner was on, the humidity plummeted.
As for the intermediate storage results, all three cottons showed the smallest mean differences in the 65% RH environment (S&B, 0.04%; Upland, -0.02%; and Pima, 0.01%). In the stationary humidity chambers, the only driving force to move water into or out of both envelope and vial was the water concentration gradient. Since the open vials before capping had been conditioned in the TTL at 65% RH, the water concentration gradient was minimized for capped vials stored in the 65% RH chamber. The mean differences for the 65% RH results did not statistically differ from 0%.
Note the intermediate storage result at 35% RH for the Pima cotton (0.17% mean difference). Although not statistically different from the control value, the result was greater than the acceptance criterion of > 0.1% and occurred in the low-humidity environment.
The ability of the aluminum-foil envelope to prevent a change in the OWC of the cottons in the KFT capped vials after five-days storage is depicted in the histogram (Fig. 7). The day-five mean differences in water content (%) were divided into ranges (e.g., 0.00 to 0.10, 0.11 to 0.20, -0.10 to 0.00, and -0.20 to -0.11). Frequency data with envelope was compiled from Table VI (15 results: 5 treatments/cotton × 3 cottons/treatment) and without envelope from Table III (27 results: 9 treatments × 3 cottons).
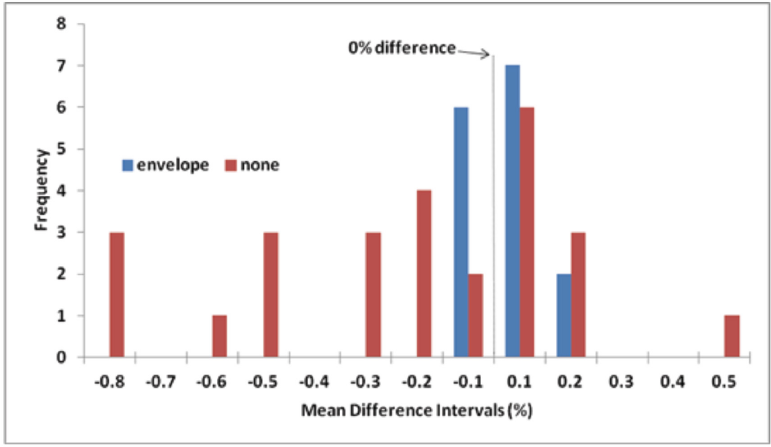
Histogram of day-five mean difference in water content between storage condition mean and control mean. All vials contained 0.1 g cotton, capped with aluminum-crimp caps and analyzed by KFT. Storage conditions: in 2× aluminum envelope–ideal, extreme, and intermediate (Table VI); without envelope (Table III).
Confirmatory—Complete Air Transport Trial with Aluminum-Crimped Cap Vials and Sealed in Secondary Container
The final prelude to an air transport trial was to demonstrate that the packed secondary container of two aluminum envelopes would not expand significantly during air transport. The low pressure in the freight space of an airplane was simulated in a desiccator as reported in the literature. 7 One filled secondary container at a time was maintained at 586 mbar for 3 h, equivalent to a one-way transport of about 1600 miles. The experiment was conducted in triplicate.
Envelope thickness was observed through the glass wall of the desiccator with the aid of a magnifying glass. A slight expansion (1 to 2 mm) did occur. However, when the pressure was raised to atmospheric, the envelope returned to its original thickness, indicating no rupture of the envelope seal. Apparently, the two foil envelopes and minimum free space volume provided a stiff secondary container wall that was resistant to a substantial increase in dimensions.
The complete airport experiment consisted of ten Upland cottons, conditioned, weighed, and capped in the vials and sealed in the double aluminum foil envelopes in New Orleans. The packets were ground transported to the airport, shipped to Orlando, ground transported to the collaborator's lab in Riverview, the vials removed from the secondary container and analyzed by KFT. The control samples from the same cottons were analyzed in the New Orleans laboratory by LTD.
Table VII details the water contents of the shipped and non-shipped (control) samples. The mean difference between the shipped and control values for all ten cottons was < 0.09%; mean difference for the sample set was –0.02%. The mean water contents were not significantly different to that for the controls. Thus, the aluminum-crimped cap vials in the secondary container resulted in maintaining the original water content of the test specimens during the air transport study.
Consistency of Water Content (%) of 0.1 g Cotton in Standard Aluminum-Capped KFT Vials During Air Transport Trial a
Cluster of the five reps from the same cotton sealed in double aluminum envelopes, and shipped from New Orleans, LA to Riverview, FL. Storage conditions and analyses: Shipped–vials with cotton test specimens conditioned open in textile testing laboratory (TTL) at 65% RH and 21 °C, capped, five reps from the same cotton sealed in double aluminum foil envelopes, ground transportation to airport, air shipping, ground transportation to collaborator's lab in Riverview, FL and analyzed there by KFT immediately after opening the envelopes. Control–0.5 g samples stored in textile testing laboratory at 65% RH and 21 °C, transferred to 25 mL screw-cap vials with rubber septum and analyzed in the New Orleans lab by LTD under ultra-dry nitrogen.
R is raw (ginned lint) cotton; Cl is mechanically cleaned cotton; sorted in descending order of mean water content in column 2. The five raw and cleaned samples are different cottons.
Each value mean of: Shipped, 10 replicates; Control, 6 replicates.
Mean difference (%) = shipped mean – control mean. Sample set mean difference = -0.02
Conclusions and Outlook
Vial closure integrity over an extended time was approached in this study by first developing the fundamentals. Equations were developed to explain and predict the effect of various factors (i.e., humidity, temperature, bumpiness in ground transport, and reduced pressure in air transport) on original water content (OWC) of the cotton specimen in the vial. It was assumed that direct vial closure integrity was not possible with crimp-cap vials over extended time. This led to the prediction that an indirect method was needed to control a leaky vial. The fundamental work predicted that by placing the leaky vial in a sealed secondary container with minimum free space, water vapor equilibrium would be rapidly established between the vial headspace and the free space. The resultant change in the OWC in the vial would not be significant.
Next, presumptive experiments (excluding air transport) were conducted. The various factors that may affect water content in a crimped-cap vial were investigated independently of each other over a five-day period (e.g., 100% RH in a static humidity chamber at constant temperature and atmospheric pressure). The goal of these independent tests was to establish promising approaches to vial storage treatments of crimped vials over time. However, the various factors were not linked together as in air transport, where each exerts its influence on consistency of water content in a specific order along with different time periods allocated. Thus, the presumptive experiments were performed with a small number of cottons and cotton replicates.
The presumptive experiments showed that various combinations of septa and metal-crimp caps did not prevent leaks and produced inconsistent water content levels. By placing the crimped-cap vials in double aluminum foil envelopes, stresses could be applied to the filled packet without a change in the OWC of the test specimens.
Finally, an actual air transport validated the fundamental and presumptive work. Ten cottons were air shipped to a collaborator in Florida in aluminum crimped cap vials. Multiple vials from the same cotton had been placed in double aluminum foil envelopes with minimum free space. The collaborator analyzed the cottons for water content and in Louisiana our lab provided the control values. The Florida and Louisiana results were not significantly different.
Use of the aluminum foil envelope as a secondary container has broad implications for KFT analysis. Since placing the crimped cap vials in the secondary container extends the OWC in test specimens for several days, user groups without the testing equipment could ship the samples to sites with the apparatus. This would provide for industry-wide access to this accurate method at low cost. Another application is direct calibration of samples for water content by KFT and transportation of the reference material in an aluminum-foil envelope. In this case, the primary and secondary containers could be aluminum-foil envelopes, with minimum free space between the two layers. A third container, made of rigid plastic or metal, would protect the foil from damage during transport.
Footnotes
Acknowledgments
The authors express appreciation to James Rodgers, Chanel Fortier, and Michael Santiago Cintron of the Southern Regional Research Center (SRRC) in New Orleans, LA, USA, and Frederick Fiddler, Metrohm USA, Riverview, FL, USA, for review of the draft manuscript. Special appreciation to Scott Segro, Metrohm USA, Riverview, FL, USA for KFT analysis of air transported test specimens in capped vials put in foil envelopes
