Abstract
With frequent oil spills occurring worldwide, oil/water separation technologies using materials with special surface wettability have attracted attention. In this study, a polyacrylamide hydrogel was synthesized and crosslinked onto polyester fabric via UV-initiated polymerization. The modified fabric showed superhydrophilicity and underwater superoleophobicity, which allowed water passage and inhibited oil permeation. Different oils (including corn, diesel, and pump oils) could be easily separated from water with high efficiency. By rinsing with water, the fabric could be easily cleaned and recycled, while maintaining good separation performance over ten uses. A textile-based material for oil/water separation with high flux, easy-cleaning ability, and long-term efficiency was developed.
Introduction
During the past few decades, development of oil/water separation materials attracted much attention due to the problem of industrial oily wastewater cleanup from dyeing, printing, metal smelting, leather treatment, and so forth.1–7 Research is needed on materials that can effectively separate oil and water. As is widely recognized, oil/water separation is an interfacial challenge. Designing novel materials with special wettability is a facile and efficient way to achieve this.8–13 Such surfaces could be obtained by appropriate chemical composition design and surface construction.14–16
“Oil-removing” materials with both superhydrophobic and superoleophilic properties are commonly used for filtering or absorbing oil from oil/water mixtures. 17 As an absorbent material, superhydrophobic calcium carbonate powder treated with a fatty acid, reported by Shen et al., separated more than 99% of the original oil from a mixture of water and diesel or crude oil. 18 Chen et al. fabricated superhydrophobic and superoleophilic polyurethane sponges for oil/water separation, which were modified with Fe3O4 particles (improving the surface roughness) and hexadecyltrimethoxysilane (reducing specific surface energy). 19 Meng et al. prepared an ultra-light (0.01 g/cm3) carbon aerogel from microfibril cellulose, which was used as a highly-porous oil absorbent material (99% porosity). 20 Cao et al. prepared a ultra-light weight carbon nanotube (CNT) sponge, which could quickly absorb oil at eight hundred times its own weight.21-22
However, these oil adsorbent materials were unsuitable for the removal of oils with high viscosity, or as oil separators for industrial sewage treatment. 23 In this respect, the separation of oil/water mixture using a filtration method is a facile and high-throughput process. For example, cotton fabric coated with polyaniline and fluorinated alkyl silane was used for oil removal under severe environmental conditions, such as high temperatures, high humidity, and the presence of corrosive substances. 24 Using a nitric acid solution as etchant and grafting with 1-hexadecanethiol (HDT) on the copper surface, Wang et al. fabricated an oil/water separation material with great stability under acidic and basic conditions, and in the presence of salts. 25 However, materials with these oil-loving properties might be fouled and blocked by oils, negatively impacting the separation efficiency, service life, and reusability.26-27
Recently, a breakthrough, inspired by nature, helped to solve this problem. 28 The surface of fish scales shows superhydrophilicity in the air and superoleophobicity under water (with an oil contact angle of 156.4 ± 3.0°), which allowed water to pass, while preventing the membrane on the scales from being fouled by oil. The fish scales are composed of hydrophilic hydroxyapatite, protein, and a thin layer of mucus. Scientists have prepared a series of bio-inspired materials for long-term oil/water separation. For example, Zhang et al. grafted poly(2-vinylpyridine-b-dimethylsiloxane) onto a polyurethane sponge to achieve underwater oleophobicity. 29 The material could release oils at low pH, and therefore could be regenerated and recycled many times due to its smart oil-spill cleanup ability.
In this study, a polyacrylamide (PAM)-modified polyester fabric with superhydrophilic and underwater superoleophobic properties was fabricated via UV-initiated polymerization. Polyester fabrics possess excellent properties such as low cost, good elastic recovery ability, and high strength. The bio-inspired hydrogel-like coating covered the polyester fabric with a layer of highly-hydrated film, which could block oil penetration and offer good oil-water separation performance. 30 Due to its underwater superoleophobicity, PAM-modified fabric showed excellent anti-fouling properties.
Experimental
Materials
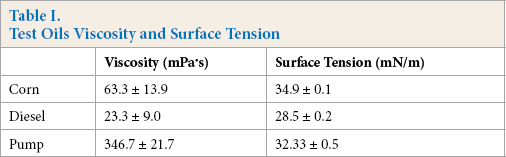
Knitted polyester fabrics, with a mean pore size of ∼400 μm, were purchased from Zhejiang Furun Printing & Dyeing Co. Ltd. Before experiments, the fabric was pretreated with methanol to remove impurities. Acrylamide (AM), N, N'-methylene bisacrylamide (MBA), 2,2′-diethoxyacetophenone (DEOP), PAM (Mn = 8000 KD), and Oil Red dye (CAS number 1320-06-5) were purchased from Aladdin Company. All chemicals were analytical grade and used as received without further purification. Corn oil was purchased from the local market. Diesel and pump oil were purchased locally. Oil viscosity and surface tension are listed in Table I.
Test Oils Viscosity and Surface Tension
PAM-Modified Polyester Fabric Fabrication
PAM-modified polyester fabrics were prepared via UV-initiated polymerization. The pre-polymer solution (50 mL) was prepared by dissolving AM (monomer), MBA (crosslinker), DEOP (initiator), and PAM (thickening agent) in distilled water at a mass ratio 50:2.5:1:3, with an AM concentration of 50 mg/mL. The polyester fabrics (2 g) were then carefully immersed into the above solution for 5 min, and padded with a padder at varying pressures (0, 0.05, 0.10, and 0.15 MPa). The fabrics were irradiated with UV light (Shimadzu UV-2600, 365 nm) for 1 h. After polymerization, these modified fabrics were soaked in distilled water (250 mL) for 24 h to remove thickening agent and excess monomer and then dried at 60 °C for 6 h.
Characterization
Surface morphology of modified fabrics was characterized using scanning electron microscopy (SEM, JEOL JSM-5610LV). The surface chemistry of the resulting fabrics was characterized by Fourier transform infrared spectroscopy (FTIR, Bruker Vertex). Measurement of water contact angle (WCA) was carried out using a contact angle measuring instrument (DSA10, KRUSS) with 3 μL water droplets at ambient temperature. The underwater contact angle was measured by the captive bubble method. 31 Smoothness, softness, stiffness, and drape properties were measured using a fabric handle tester (FAST, CSIRO, Australia).
Oil-Water Separation Capability Measurement
Mixtures of water and various oils (i.e., corn, diesel, and pump oils) were prepared in a volume ratio of 2:1. Before the oil/water separation experiment, the prepared fabrics (10 × 10 cm) were wetted with water. Ten, the above oil-water mixtures were poured onto the fabrics and allowed to gravity filter. The water passed through the fabric and was collected in a measuring glass. The oil contained in the water was extracted by petroleum ether and the amount of oil was measured by UV-Vis spectrophotometry. The absorbance of petroleum ether solutions with different concentrations of oil was recorded. A standard equation was obtained to calculate the content of oil in water collected. The oil/water separation efficiency was calculated according to Eq. 1.
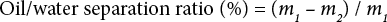
m1 and m2 were the total oil amounts (g) in the original oil/water mixture and in the collected water after separation, respectively. Oil/water separations were repeated for 10 times to evaluate the reusability of the PAM-modified fabric. After each experiment, the oil-stained fabrics were rinsed with running water for 5 min.
Results and Discussion
PAM-Modified Polyester Fabric
To introduce homogenous and thick PAM layers onto polyester fabric, high molecular weight PAM was used as a thickener. It increased the viscosity of the AM monomer solution and was expected to make the fabric carry a larger amount of monomer on it. In this study, the AM amount on the fabric was controlled by padding pressure. Under UV irradiation, the ketone groups in DEOP molecules transit from singlet state to triplet state, which then capture hydrogen from C-H bonds in polyester molecules. In this way, free radicals were produced on the surface of polyester fabric and surface graft polymerization was initiated. In the presence of monomer (AM) and crosslinker (MBA), a crosslinked PAM layer was covalently introduced onto the fabric. As shown in Fig. 1, the polyester fabric weight growth rate gradually decreased with the increase of rolling pressure. When the rolling pressure was 0.05 MPa, the weight growth rate was 1.8%. Under a higher rolling pressure, lager amounts of AM monomers were removed from the fabric by the padder. Although a higher rolling pressure resulted in a lower weight ratio of PAM, it was hoped that the resulting PAM coating would be more homogenous by driving more of the PAM into the fabrics internal pores.
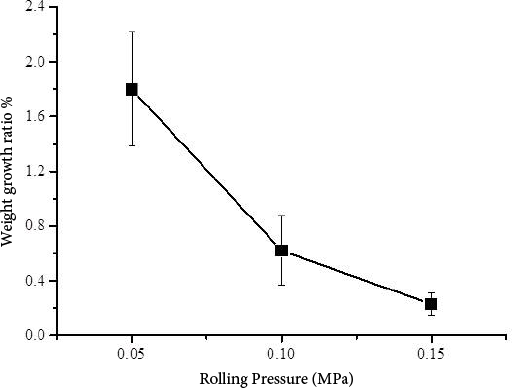
PAM weight growth ratio of polyester fabric at various rolling pressures.
Chemical Composition Analysis
FTIR was used for surface chemistry characterization of PAM-modified polyester (Fig. 2). For unmodified polyester fabric, peaks at 730, 1050, and 1730 cm-1 were attributed to C-C, C-O and C=O groups, respectively. After PAM modification, the peaks of amide II, amide I, and C-H groups appeared at 1500, 1625, and 2900 cm1. Since the weight ratio of PAM was low, these characteristic peaks in the FTIR spectrum were weak, but suggested that PAM was successfully crosslinked to the polyester fabric.
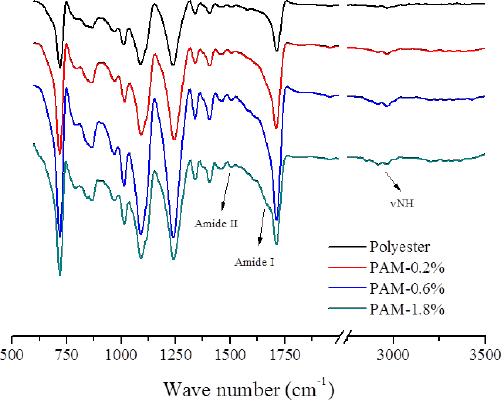
FTIR spectra of polyester fabric and PAM-modified polyester fabrics. The weight ratio of PAM on polyester fabric was 0.2%, 0.6%, and 1.8%, respectively.
Surface Wettability
The wetting behavior of water droplets on fabric was studied by measuring the water contact angles (WCA) at different times. Unmodified polyester fabric was relatively hydrophobic, with a WCA of about 120°. For the PAM-modified polyester fabric, the WCA gradually decreased over time due to the wicking effect. With an increased amount of PAM, the fabric WCA measured at t = 0 decreased from 120° to nearly 40°. The wicking time (the time that the WCA decreased to 0°) also decreased from nearly 2700 ms to almost 0 ms (Fig. 3). The results suggest that the fabric with greater amounts of PAM had smaller WCAs and shorter wicking time, and thereby, better surface hydrophilicity.
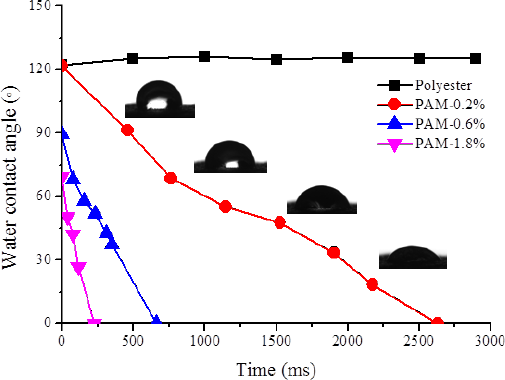
The WCA of PAM-modified polyester fabric at various times. The weight ratio of PAM on polyester fabric was 0.2%, 0.6%, and 1.8%, respectively.
The underwater oil adhesion ability of the material was studied (Figs. 4a and b). The untreated polyester fabric showed a high affinity for the corn oil droplet. When the oil droplet contacted the surface of fabric, significant interaction between the oil droplet and the fabric occurred (Fig. 4a). By moving the needle away from the surface, the droplet was stretched to deformation due to the large affinity, and finally, strongly adhered to the fabric undersurface with an underwater oil contact angle of 130.4°. The PAM-modified fabric showed an ultra-low affinity to oil. The oil droplet did not adhere onto the fabric, and was easily removed. The superoleophobicity of PAM-modified polyester fabric was attributed to the water-loving properties of PAM molecules. Water had a favorable association with the hydrophilic polymers to form a hydration layer, which could prevent oil adhesion and penetration.

Underwater corn oil contact angle of (a) untreated polyester fabric and (b) PAM-modified fabrics (PAM weight ratio: 1.8%). The arrow points to the direction of needle movement.
Morphology
Using padding and UV-initiated polymerization, a polyester fabric coated with PAM was successfully fabricated. PAM covered the fiber homogenously and a layer of polymeric film was also formed between the fibers. It was found that the film gradually thickened with an increase in the PAM weight ratio (Fig. 5).

SEM images of (a) untreated polyester fabric, and PAM-modified polyester fabrics with (b) 0.2%, (c) 0.6%, and (d) 1.8% weight ratios.
Oil/Water Separation
The process of oil/water separation is shown in Figs. 6a–d. The PAM-modified polyester fabric was used as filtration material, which was pre-wetted before the experiment. Water molecules were trapped in the PAM network structure, forming a composite water-solid interface. The resulting hydrophilic layer would prevent oil penetration. A mixed solution of oil and water was poured onto the fabric from the top of the funnel (Figs. 6a and b). Water passed through the porous fabric quickly, whereas the oil was held back (Fig. 6c). Nearly 100% of the oil remained above the fabric and water was collect in the beaker. No visible oil existed in the water (Fig. 6c), showing a very high separation efficiency. The whole separation process was driven by gravity.
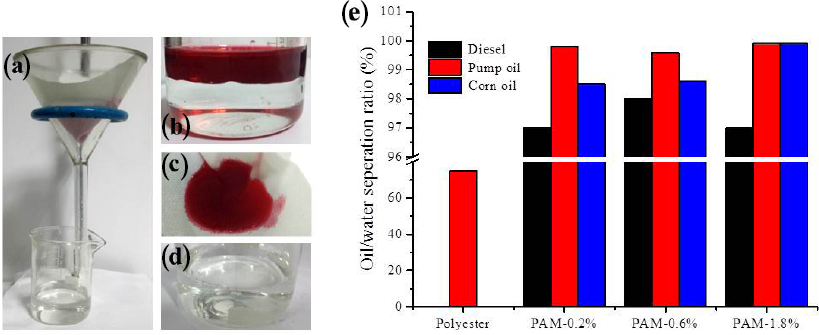
Oil-water separation process. (a) the device for oil/water separation, (b) mixture of corn oil (dyed with Oil Red) and water, (c) the oil after separation, (d) the water collected after separation, and (e) the separation efficiency of a series of oil/water mixtures using fabric with various PAM weight ratios.
Mixtures of water and three types of organic oil (i.e., diesel, pump, and corn oils) were tested for separation. The results showed that the modified polyester fabric was an efficient filtration material with universal applicability. The PAM-modified fabric showed high separation performance for various kinds of oil, with the separation efficiency of pump oil being the greatest (> 99%) (Fig. 6e). The unmodified polyester fabric separated nearly 75% pump oil from water, with lower separation capabilities for mixtures of water and the other oils. This effect may be due to the viscous nature of the pump oil, which was easily resisted by the pre-wetted polyester fabric.
Easy-Cleaning Function and Reusability
Easy-cleaning ability is required for long-term reusability of oil/water separation materials. 32 As shown in Fig. 7, most of the oil could be removed from the fabric surface by simply rinsing with running water. This easy-cleaning ability of modified fabrics was achieved due to the superhydrophilicity and underwater superoleophobicity of the PAM coating. The corn oil/water separation was repeated 10 times with the same fabric. The separation efficiency of each experiment was greater than 99.9%, and the residual oil content was lower than 200 ppm, showing the excellent and durable performance of PAM-modified polyester fabric (Fig. 8).

The residual oil (dyed by Oil Red) on the fabric was washed away easily by rinsing with water. The PAM weight ratio was 1.8%.
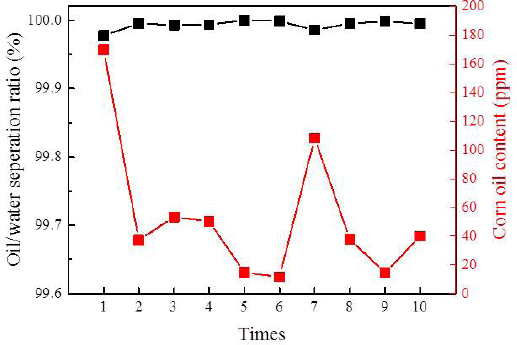
The oil/water separation efficiency (black line) and residual corn oil content in water (red line) in the repeating oil/water separation experiment of the same fabric 10 times. The PAM weight ratio was 1.8%.
Fabric Handle Analysis
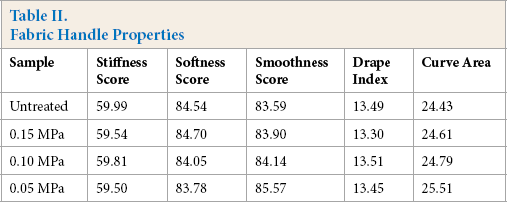
The fabric handle, affecting the fabric's oil/water separation use, is a comprehensive reflection of the physical and mechanical fabric properties. 33 Stiffness, softness, and smoothness scores, drape index, and curve area were obtained by the fabric handle tester. A fabric with a soft handle had low stiffness and high softness scores, a high drape index, and a small curve area. As shown in Table II, the PAM layer had an insignificant influence on fabric softness and drapability. Curve area increased slightly as the amount of PAM increased. The smoothness also increased with an increase in PAM, which may be due to the decreased surface roughness caused by PAM coating. In general, the PAM fabric kept a good handle after PAM modification. Excellent softness allowed the fabric to be used as an oil collecting bag for efficient oil/water separations applied to ocean oil leakage treatments, which cannot be achieved by stiff materials.
Fabric Handle Properties
Conclusion
A new, efficient oil/water separation fabric with a high flux and outstanding recyclability was fabricated in this study. The PAM-modified fabric showed superhydrophilic and underwater superhydrophilic properties, which resulted in rapid separation of oil/water mixtures. It was applicable for the separation of water and various kinds of oils with separation efficiencies greater than 97%. This treated fabric could separate water from a highly viscous oil (e.g., pump oil). Meanwhile, due to its low oil adhesion affinity and easy-cleaning function, the functionalized polyester fabric was a durable and effective material for multi-cycle oil/water separations. In addition, polyester fabric is an inexpensive, soft, and sustainable material, which possesses advantages for industrial production and use in large-scale oil/water cleanup applications.
Footnotes
Acknowledgement
This study was financially supported by the Natural Science Foundation of China (Grant No. 51303161).
