Abstract
Using previously determined optimized reaction conditions, an ultrasound-enhanced bioscouring process was scaled to ten-gallon capacity and a system of rollers was added, which allowed for continuous fabric feed and equipment operation. UV-Vis spectrophotometric data from bioscoured fabric samples confirmed pectin removal and provided evidence that the combination of ultrasound and enzymes was an environmentally sustainable alternative to the present-day caustic scouring method.
Introduction
Over the past several decades, little effort has been made in traditional textile processing to reduce or eliminate the caustic scouring agents used to remove pectins from greige cotton. The tons of alkaline wastewater generated in scouring greige cotton textiles were simply neutralized and discarded, costing valuable time, money, and water resources. In the present age of greater environmental awareness, there is a desire to develop greener processes that eliminate these hazardous waste streams. To mitigate the environmental impact of textile processes, researchers at the US Department of Agriculture (USDA), Southern Regional Research Center (SRRC) have recently made progress towards the development of a large-scale ultrasound-enhanced bioscouring reactor. The reactor combines the acoustic energy of ultrasound with the highly-specific chemistry of enzymes to remove pectin and, subsequently, wax from greige cotton without the need for caustic substances. This research is a progression from earlier published work on a smaller scale in which the four process factors of power, time, enzyme concentration, and ultrasonic frequency were optimized. 1 In the present work, these four optimized process factors were applied to a ten-gallon ultrasonic tank, equipped with a system of rollers which permitted continuous fabric feed into the bioscouring reactor.
Enzymes are currently used in many textile processes for desizing (amylase), scouring (pectinases/cellulases), bio-finishing (cellulases), peroxide breakdown (catalase), bio-bleaching (lacases), and laundry washing (proprietary mixtures).2–12 The benefits of enzymes are many, including ease of use, low environmental impact, and highly-specific reaction products. The drawbacks include slow reaction rates and increased material costs, when compared to the conventional caustic method. Our research has found that the combination of enzymes and ultrasound has a synergistic effect when removing pectin from greige cotton fabric. 1 This synergistic effect is due to the improved transport of enzymes to and from the greige fabric surface and the corresponding removal of enzyme products away from the fabric surface. When ultrasound and enzymes are applied separately, pectin removal was lower as measured by Ruthenium Red staining techniques. Although wax material was removed from the textile substrate during ultrasound-enhanced bioscouring, the present research focused solely on the removal of pectin. 13
The improvement in enzyme transport is explained by the turbulent effect created by ultrasonic cavitation in the buffer medium. 14 When a piece of fabric is submerged in a buffer solution without ultrasound, the enzyme must rely upon laminar flow, which describes the fluid transport and molecular diffusion, to contact the surface of the fabric. This is inefficient and results in poor pectin removal. With ultrasound applied, the laminar flow is disrupted by turbulence created by the growth and sudden collapse of cavitation bubbles, causing greater accessibility of enzyme to the substrate. In a buffer solution containing no fabric, cavitation bubbles are evenly distributed throughout the medium. However, when a piece of fabric is introduced, the heterogeneity created causes the cavitation bubbles to form preferentially on the fabric surface in a process called acoustic streaming, resulting in improved transport of enzyme to the substrate and an increase in enzyme efficiency. 7
The formation of cavitation bubbles is somewhat analogous to the formation of a crystalline lattice. In a homogeneous, saturated solution, the formation of a crystal will not always occur. It takes a slight heterogeneity such as a speck of dust, or a variation in the medium's density for the formation of a seed crystal. In this same way, ultrasound cavitation requires the heterogeneous presence (substrate introduction) and, like a crystal, the cavitation bubble develops around the heterogeneity. The size and number of cavitation bubbles is influenced by the frequency and power applied.7,14-15 Generally speaking, the more power applied, the more cavitation bubbles; the higher the frequency, the smaller the size of cavitation bubbles.
The objectives of the present research were 1) to determine whether engineering process controls can be maintained and optimized at the larger scale, 2) to examine ultrasound-enhanced bioscouring performance over time as fabric is continuously fed into the bioscouring reactor, and 3) to provide recommendations for improvements in experimental procedure and equipment operation and design. The results are reported and discussed herein.
Experimental
Ultrasound Equipment and Preparations
The ultrasound equipment used in this series of experiments consisted of a seven frequency multiSONIK-2 Ultrasonic Generator (Blackstone-Ney Ultrasonics), a programmable controller, a specially designed ten-gallon stainless steel tank equipped with heat exchange coils and a drain plug, and a programmable temperature-controlled circulating bath. A removable system of nine stainless steel rollers was fabricated, which could process an 11” × 60” greige cotton woven fabric sample within the ultrasonic tank. The complete experimental setup is shown in Fig. 1.

(a) Overall view of equipment setup, (b) overhead view of ultrasonic Ftaign.k1 with system of rollers and greige fabric, and (c) view of circulating bath, roller speed controller, ultrasonic power source, and programmable controller.
As part of experimental preparations, 40 L of McIlvaine's buffer solution (pH = 3.8) were prepared from sodium phosphate dibasic dihydrate (0.2 M) and citric acid (0.1 M), both purchased from Sigma-Aldrich, and added to the ten-gallon ultrasonic bath. Triton X-100 (20 mL, Acros) and MultiFect pectinase FE (12.4 mL, Genencor) were added to the bath and stirred until completely mixed. MultiFect Pectinase FE contained 1–5% of pectinase enzyme according the manufacturer's MSDS. The ultrasonic bath was then heated to 43 °C and the system of rollers placed inside the ultrasonic tank. As an example of one experiment, a print cloth leader sewn to a desized length of greige cotton fabric (Schott Textile, 7 oz/yd 2 , 140 g) was wound through the system of rollers and collected on a catch roller. The system of rollers was controlled by a variable speed motor that was adjusted to restrict fabric residence time within the tank to 60 min. Initial dimensions of greige fabric used in the experiments were 7.5” × 89.0” and each experiment lasted approximately 2 h and 15 min. Later experiments occurred over an eight-hour period and involved 38 ft of desized greige fabric continuously fed into the bioscouring reactor.
All scaled-up experiments were run with the following previously optimized set of reaction conditions: ultrasonic power = 100% (980 W), ultrasonic frequency = 220 KHz, time = 60 min residence time for fabric in bath, and enzyme concentration = 31 μL/100 mL buffer (12.4 mL/40 L of buffer in scale-up). 1
The desized greige fabric was positioned just above the buffer solution, allowing for its leading edge to enter the ultrasonic tank buffer once the experiment commenced. Over the course of 1 h, the leading edge wound its way through the system of rollers and emerged on the other side where it was collected onto a catch roller. Once the ultrasound-enhanced bioscouring was complete, the fabric was removed from its leader and placed into a 4-L beaker of boiling water (3.0 L) for 15 min to quench enzymatic activity. The fabric was then rinsed of any residual buffer solution, padded, and dried in a domestic clothes dryer on high heat for 30 min. The bioscoured fabric was reconditioned overnight.
Ruthenium Red Staining
The bioscoured fabric was then cut into 7.5” × 12” sections and from 1–3 2” × 2” smaller pieces were then cut from the lower left, middle, and upper right from every 12” length of the bioscoured fabric. These smaller samples were placed in separately-labeled organza bags (3” × 4”) and placed into a preheated (50 °C) 4-L beaker containing Ruthenium Red and McIlvaine's buffer (pH = 6.0). The volume of McIlvaine's buffer and the weight of Ruthenium Red were based upon the weight of the fabrics to be stained. The ratio of fabric, buffer, and Ruthenium Red was 50 mg Ruthenium Red/100 mL buffer/g fabric.1,16,17 The weight of the organza bags was not included in the fabric weight and the Ruthenium Red stain did not affect the nylon organza bags. It is important to note that the Ruthenium Red crystals needed to be crushed prior to addition to the buffer solution to ensure complete dissolution in the buffer solution. After stirring at 50 °C for 30 min, the fabric samples were transferred to a 4-L beaker of boiling deionized (DI) water. After 15 min in the boiling bath, the sample fabrics were removed, rinsed with DI water, and dried at room temperature (RT) overnight.
UV-Vis Spectrophotometry
A Varian Cary 100 spectrophotometer, fitted with a Labsphere integrating sphere diffuse reflectance accessory, was used for percent reflectance measurements at 540 nm of the Ruthenium Red stained fabrics. The diffuse reflectance accessory was run inside the sample compartment holder of the spectrophotometer and the instrument and computer were controlled using a Varian Scan software program. The spectrophotometer also used a photomultiplier tube detector with a spectral wavelength range of 200 to 800 nm. To perform a baseline correction, a Spectralon (PTFE) certified reflectance disk was placed over the open 70-mm port of the accessory, followed by scanning at the specified wavelength range in reflectance mode. Similarly, to collect spectral data on the bioscoured fabrics, the Spectralon disk was replaced with the individual bioscoured fabric samples over the open port, followed by scanning the same wavelength range in reflectance mode.
Results and Discussion
Maintaining appropriate noise levels and temperature controls are challenges in any scale-up. In small-scale experiments, it was previously reported that the noise level directly above the ultrasound bath was measured at 76 dBA. 1 In large-scale experiments, the noise level directly above the ultrasound tank was measured at 73 dBA (Fig. 2a). The reduction in noise level was likely due to the attenuation of sound by the introduction of a system of rollers. A difference of 3 dBA is the minimum reduction in sound that is detectable by human hearing. The sound level in scale-up experiments at 2 ft from the ultrasound tank was measured at 65 dBA (Fig. 2b). This is essentially the same value recorded in smaller-scale experiments (66 dBA). Taken together, these two measurements provide evidence that the combination of ultrasound and enzymes, when operating under prescribed optimal conditions, does not pose a threat to worker safety. By analogy, 65 dBA is equivalent to the noise level of normal human conversation at a distance of 3 ft. 73 dBA is approximately equal to twice the noise level of that same conversation. Additionally, 73 dBA is well-below the 85 dBA threshold, wherein the US National Institute for Occupational Safety and Health (NIOSH) restricts noise exposure to 8 h (the US Occupational Safety and Health Administration (OSHA) restricts noise level exposure to 8 h at 90 dBA). 18 Consequently, no hearing protection is required for worker safety, nor is it necessary to build sound-attenuating barriers around the equipment. This can mean considerable cost savings when the equipment is first installed.

(a) Sound level measurement of 73 dBA directly above operating e quipment and (b) sound level measurement of 65 dBA at 2 ft from operating equipment.
Maintaining proper temperature control is a crucial parameter for optimal enzyme activity. Overall, with the equipment used in these experiments, it was moderately difficult to maintain reactor tank temperature control within the narrowly-prescribed range of 45–50 ± 3 °C over a period of 9 h. The 12-L capacity of the circulating bath and the system of heating/cooling coils mounted on the inside walls of the ultrasonic bath did maintain optimal operating temperature during the entire experiment, but it became necessary to add ice once per hour to the circulating bath that, although set to 10 °C, was never able to effectively cool to below ∼25 °C. With the external circulating bath temperature operating in the above manner, the heat generated by the ultrasound generator operating at 980 W was off-set sufficiently to maintain a bath temperature within the optimal range for enzymatic activity. Typically, the starting temperature was in the 43–45 °C range and increased ∼2 °C during the experiment, ending at ∼45–47 °C. The open bath environment provided excellent ventilation of excess heat. The diameter of the cooling coils (Fig. 3a and b.) should be increased in subsequent equipment designs to allow for increased surface area, better fluid circulation, and heat exchange. This would facilitate initial bath heating and more easily maintain temperature control during experimental operation. Additionally, the system of coils should be extended higher on the walls of the tank, providing more efficient heat exchange.

(a) Interior view of ultrasound bath with heating coils along wall and (b) zoomed in view of heating coils.
A total of ten experiments were performed in this series of experiments. After each length of fabric was bioprocessed in the ultrasound reactor, individual pieces were cut from different sections along the fabric length and stained according to the standard Ruthenium Red procedure. The samples were then analyzed for pectin content by examining percent reflectance (pctR) at 540 nm.
In experiments using conventional desizing and caustic scouring methods, samples had a pctR value of 45.73, corresponding to 100% pectin removal. 1 In previous work involving smaller scale experiments, greater than 98% pectin removal with a pctR value of 45.15 was achieved. The first scaled-up experiment involved 8 ft of greige fabric and the results showed a slightly lower average pctR value of 42.00 (92% pectin removal). It was thought at the time that a few runs were necessary to familiarize the equipment operator with the new larger scale. After two more runs, the average pctR value of the bioscoured samples equaled that from earlier smaller-scale experiments, confirming that the optimized reaction conditions from smaller-scale experiments could be applied to scaled-up experiments.
Fig. 4 shows the average pctR data for three trials of scaled-up experiments using 8 ft of greige fabric. As shown by the graph, as the equipment operator became more familiar with operations at the larger-scale, results comparable to the smaller-scale experiments were obtained. The blue trial was the first scaled-up attempt, showing a lower average pctR of 42.00; however, in trials two (red) and three (green), the average pctR values were 45.10 and 45.32, respectively, in agreement with the earlier smaller-scale experiments.
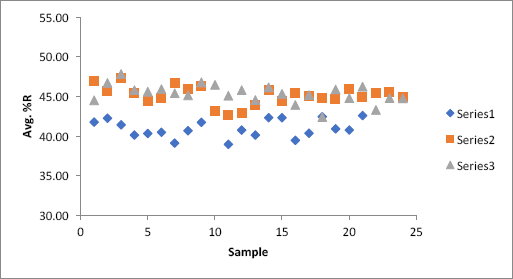
Graph of 8-ft fabric trials. Blue graph = initial 8-ft fabric experiment. Red graph = second trial. Green graph = third trial.
After obtaining consistent results with 8 ft of greige fabric, the experiments were scaled to 20 ft of greige fabric before scaling to 36 ft of fabric. The experiments with 20 ft of greige fabric produced inconsistent results, mainly stemming from a batch error in the Ruthenium Red staining. After (b) bioscouring was completed, these fabrics were treated with Ruthenium Red dye in two separate dye baths using two separate 10-f sections. This batch error resulted in an inconsistent UV-Vis analysis of the average pctR value. Having two separate dye baths resulted in the staining of the fabrics in two distinctly different environments. To correct this, all bioscoured samples were thereafter stained in a single dye batch.
The performance of the ultrasound reactor was further tested on a larger scale using 36 ft of greige fabric. The fabric length was wound onto the system of rollers and allowed to continuously pull through the ultrasound bath over 8 h, restricting each 7.5” × 60” area of fabric to 1 h of exposure in the ultrasound-enhanced enzyme bath. After bioscouring was complete, for staining purposes on the larger-scale sample, only one 2” × 2” section of fabric was cut from each foot of fabric, alternating between left, middle, and right. Fig. 5 shows the plotted average pctR data obtained from UV-Vis analysis after the Ruthenium Red dye treatment. In this case, each sample of fabric was dyed in the same batch to ensure each fabric was exposed to the same environment, eliminating any batch error. As shown in Fig. 5, there was a steady decrease in average pctR over the length of the fabric. There was a clear drop of in effective pectin removal beginning at 13–15 ft and continuing for the entire length of the remaining fabric. This was visibly evident from the fabric samples themselves (Fig. 6). In sample #15 (middle row, far left), there was a slight pink/red coloration of the fabric, which progressively got darker with each foot of fabric.
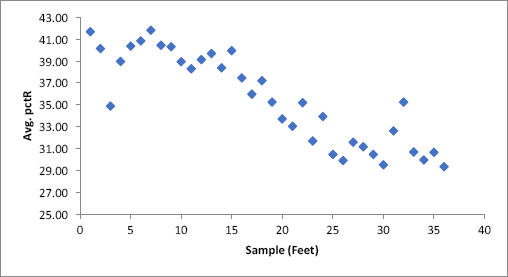
Average pctR for a 36-ft fabric showing a steady decline in performance over time.

Large scale (36-ft experiment) fabric samples after being dyed with Ruthenium Red. Note the visible, gradual increase in red color beginning around 14 ft (sample #14).
At this point, two possible explanations for the decrease in enzyme performance were proposed: 1) The enzyme was degrading and becoming less active over time during the 8-h process due to exposure to ultrasound, or 2) drag out of the enzyme from the bath was occurring at the end of the 60-min residence time as the bioscoured fabric exited the bath and was collected on the catch roller.
To test these possible explanations, an experiment was performed to investigate enzyme activity under ultrasonic conditions over an 8-h time period. However, instead of winding the fabric onto the system of rollers and allowing it to transit the bath over 8 h, three 1-ft sections of fabric were cut and each 1-ft section of fabric was allowed to hang in the bath for 1 h at a time after an initial 5 h of experimental time had passed. Upon completion of 1 h, the first piece of fabric was pulled out of the bath and the second piece was then placed within the bath for 1 h, and so on, until all three sections of fabric had been exposed to the ultrasound bath for 1 h and the total experimental time equaling 8 h had elapsed. If enzyme degradation due to ultrasound exposure was occurring, it should be reflected in a lower enzyme activity as measured by Ruthenium Red staining for each 1-ft section of fabric. On the other hand, if no enzyme degradation was occurring and the loss of enzyme performance was due to drag out, then there should be little difference between 1-ft sections of fabric as measured by Ruthenium Red staining.
From each 1-ft section of fabric, three separate 2” × 2” pieces of fabric were cut to be dyed and analyzed. The results of the average pctR for each 2” × 2” inch piece are shown in Fig. 7. No decline in enzyme activity was observed over time, with an overall average pctR of 43.15. Therefore, the decline in performance from the 36-ft scale-up was due to enzyme drag out as the fabric was removed from the bath onto the catch roller. In the experimental set up, there was no device to wring the water out of the fabric as it exited the ultrasound bath. This inevitably decreased the concentration of enzyme present within the bath, leading to the decrease in average pctR over time. For this process to be more effective, it would be advisable to install a padder to remove buffer-containing enzymes from the bioscoured fabric as it exits the ultrasound bath to keep the concentration of enzyme constant throughout the duration of the experiment.
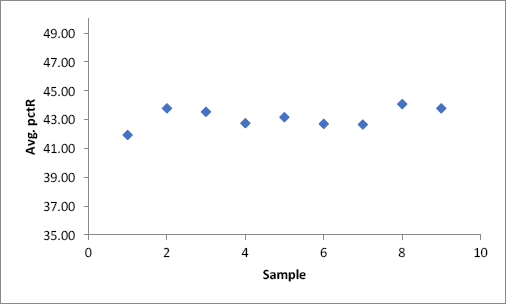
Average pctR value of investigation into enzyme degradation versus drag out.
Conclusion
Using previously optimized bioscouring process conditions, a scaled-up ten-gallon ultrasonic bath was used to evaluate bioscouring performance and process engineering controls. A system of rollers allowing continuous fabric feed was investigated. Running 8 ft of fabric through the system of rollers into the ultrasound bath gave consistent results, resulting in an overall pctR average value of 43.94 over three trials, corresponding to a greater than 96% pectin removal. Incorporation of a larger length of fabric (36 ft) resulted in a decrease in enzyme performance over time. Further investigations revealed the decrease in performance was caused by drag out of the enzyme as the fabric exited the ultrasound bath onto the catch roller. Implementation of a padder would eliminate drag out and improve performance. Noise levels were monitored during all experiments and found to be within acceptable NIOSH and OSHA limits for worker exposure. Temperature control was properly managed throughout all experiments, but a redesign of cooling coils would facilitate temperature control. Overall, the scale-up of ultrasound-enhanced enzymatic bioscouring proved successful as an effective and sustainable alternative method to traditional caustic scouring of greige cotton fabric.
