Abstract
New antimicrobial textile finishes with enhanced action against microbes are needed for improved protection in critical applications, including hospital infection control and biodefense. This study compares fabric treated with a novel quaternary ammonium compound-based antimicrobial treatment with untreated fabric, using a new method for determining the antimicrobial efficacy of fabrics and textiles. The effects of contact temperature, contact time, and bacterial load were demonstrated. The lowest concentrations of bacterial cells were killed very quickly. Increasing contact time resulted in inactivation of larger numbers of cells, as did elevating temperature from 24 °C to 37 °C. Important physical properties of the fabrics were retained. The simplicity, and good reproducibility, of the test procedure supports its use for the rapid screening and assessment of fabric specimens for antimicrobial activity.
Introduction
Concern about microbial attachment to surfaces has spurred increasing activity in the research and development of textile and other surfaces that are actively antimicrobial. By inhibiting or preventing microbial attachment and proliferation on such surfaces, problems such as infection, contamination, malodor, and biofilm formation can be prevented, or at least significantly reduced.1–4 For example, the costs associated with hospital-acquired infections are increasing each year, and a larger proportion of infections are being caused by antibiotic-resistant bacteria. The role of good hygiene and infection control practices in preventing the transmission of pathogens through hospital and extended care facilities is universally recognized. However, laboratory and clinical studies have shown that bacteria can survive for weeks and even months on items found in hospital patient rooms, including privacy curtains, computer keyboards, and keypads on medical equipment. Viable, opportunistic pathogens and antibiotic-resistant bacteria have been recovered from stethoscope covers, privacy curtains, white coats, surgical scrubs, and other frequently-touched items such as beds, bedrails, and bedside equipment.2,4–14 Neely and Maley published a study of the survival times of several Gram-positive bacteria on fabrics and plastic films commonly used in hospital settings. 6 All the microorganisms survived for at least one day, and several for more than 90 days. Technologies that prevent the proliferation and transmission of microbes on inanimate objects without placing additional manpower burdens on clinical and janitorial staff could play a significant role in complementing current infection control practices.
Antibacterial textile surfaces can be created by incorporating chemical agents into the material or fabric. 15 This can be accomplished by blending agents such as silver, Triclosan, 16 or copper compounds, with known antibacterial activity onto or into the polymer, fiber, or fabric. 17 These agents can then leach out of the fibrous matrix at a slow rate and can inhibit or even kill microbes in the local environment.
The antimicrobial properties of silver ions have been known for centuries. Silver ions kill microbes by crossing the cell membrane to enter the cell, where the ions bind to and denature proteins that are key to cellular function. Although effective and active against a broad-spectrum of microorganisms, it can take hours or even days to achieve cell death. Another limitation in the use of silver is its relatively high cost. Copper works in a similar manner to silver, and its fun-gicidal properties have been used commercially for decades. 18 Other compounds, such as antimicrobial peptides 19 and N-halamine 20 compounds have also been incorporated into fabrics to control microbial colonization, but these approaches have found limited commercial application.
Triclosan is a chlorophenol, widely used in consumer products, such as toothpaste and hand soaps, and has also been incorporated into household items such as chopping boards and shower curtains. Triclosan works intracellularly, and must migrate out of the item or product, enter the cell, and disrupt normal cellular function. However, as the use of Triclosan has increased, questions have been raised regarding its efficacy and potency.16,21–23 Additionally, concerns about its environmental impact have emerged, resulting in restrictions on its use. 24 The State of Minnesota has enacted a ban on Triclosan starting on January 1, 2017. Because silver, copper, and Triclosan work on intracellular processes, one of the more significant limitations of these antimicrobials is that they cannot be bonded to a material, but instead must be blended onto or into it. Since the agent needs to be able to migrate from the treated material into the cell, migration into and contamination of the broader environment is inevitable.
Non-leaching antimicrobial surfaces can be created by chemically bonding certain compounds to the desired polymer, fiber, or fabric.1,25–27 For antimicrobial activity to be retained, successful compounds are typically antibacterial agents that work via disruption of the microbial cell membrane. Compounds include quaternary ammonium compounds (Quats, QACs, Fig. 1), and certain antimicrobial peptides. Quats have been used for decades as sanitizers and disinfectants in hospitals. They possess broad-spectrum activity against bacteria, molds, and fungi, killing cells by membrane disruption that leads to cell lysis. Their activity against enveloped viruses results in disruption of the viral envelope, thereby potentially interfering with the ability of the virus to bind to its host cell. Quats have been used for decades in hospital disinfectant formulations, as preservatives in personal care products, and in antibacterial hand soaps. Quaternary ammonium compound dendrimers are highly-branched polymers (dendrimers or hyperbranched polymers) that have quats immobilized at the chain ends (QAC-Ds). QAC-Ds have been shown to be much more potent that linear quats. 28 QAC and QAC-D molecules can be modified to include chemical groups for bonding and immobilization.
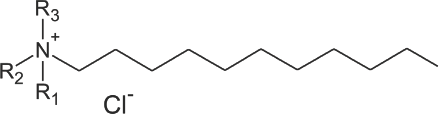
Chemical structure of quaternary ammonium compounds (quats, QACs).
The development of new technologies and capabilities for textile and materials requires the appropriate development of test and evaluation methods for both performance and function. Ideally, laboratory tests should be efficient with respect to time and materials used, efficient to perform, and produce reliable and consistent results that allow the effects of certain experimental variables to be assessed. Below, we describe a procedure developed to assess the antimicrobial efficacy of fabrics treated with a new antibacterial finish.29,30We examine the influences of contact time and temperature on the effectiveness of antimicrobial-treated textile finish to either kill, or inhibit, the growth of bacteria. Microbial survival after contact with untreated fabric was also assessed, serving as an experimental control.
Materials and Methods
Antimicrobial Fabric Preparation
All fabrics used in this study were sourced by the College of Textiles, North Carolina State University, and scoured and bleached prior to use. Quaternary ammonium compound dendrimers (QAC-D) antimicrobial finishes were applied to all fabrics, which were subject to a range of mechanical and physical tests.
Antimicrobial Finish Preparation and Application
QAC-Ds were synthesized and prepared as reported previously. 29 Briefly, the dendritic molecule was dissolved in solvent and dried using rotary evaporation. A two-step reaction was then performed to convert the terminal groups on the dendritic molecule to immobilized QAC-Ds, with a proportion of silane groups to provide chemical coupling to the fabric. The antimicrobial QAC-D compounds were immobilized on the cotton/polyester blend fabrics described above. The compounds were dissolved in a minimal amount of ethanol. A ∼1.5 L aqueous bath was set up consisting of ∼85% distilled water, 15% ethanol, and acetic acid, to achieve a pH of 4-6. Fabric swatches ∼6 in. square were placed in the bath. The QAC-D/ethanol mixture was then added, achieving a target concentration of 0.2–0.5 wt% of the antimicrobial compound, with gentle agitation at room temperature (RT). After 24 h, the fabric swatches were removed, triple-rinsed in distilled water, and dried in an oven at about 50 °C.
QAC-D Concentration on Fabrics
QAC-Ds will form a complex with bromophenol blue indicator, which provides a very rapid and easy method to determine their presence.31–33 Treated and untreated fabric samples were incubated in an excess of aqueous bromophe-nol dye for a minimum of 20 min. The specimens were then triple rinsed with distilled water to remove unbound dye from the surface. As shown in Fig. 2, the untreated fabric samples did not retain any of the bromophenol blue dye after rinsing. In contrast, the QAC-D-treated fabrics were stained a uniform deep blue, confirming that quaternary ammonium compounds were present on the surface of the fabric. Releasing the dye using a fixed aliquot of sodium dodecylsulfate solution, followed by spectrophotometric measurement of the color intensity at a frequency of 590 nm, provided a means to quantitatively determine the concentration of QAC on a fabric. All fabrics used in this study had a minimum of 500 nmoles of QAC-D/in 2 .

Bromophenol blue retention by untreated cotton (left) and cotton treated with QAC-D compounds (right).
Scanning Electron Microscopy
Specimens measuring about 0.5 cm2 were cut at random from the treated and control fabrics and mounted on aluminum stubs using conductive tape and coated with gold/ palladium using a Hummer 6.2 Sputter Coating System to obtain an average uniform coating of 100 Å thickness. The specimens were coated five times, each time depositing a thickness of 20 Å at different angles. Images were acquired from a JEOL JSM 5900-LV scanning electron microscope (SEM) using an accelerating voltage of 15 kV and spot size 20. Multiple random micrographs were obtained by digitally scanning at magnifications of 100× and 1000× using the optimal black and gain settings for clarity and the JEOL Digital Scan Generator V 2.00 software interfaced with the microscope. Fig, 3 shows the photomicrograph images of the polyester and cotton fibers at 1000 × magnification before and after QAC-D microbiocidal treatments.

(a) Polyester fibers showing a typical round cross section and smooth surface morphology before treatment, (b) polyester fibers from the treated sample showing smooth surface morphology and a lack of inter-fiber adhesion after treatment, (c) cotton fibers showing a typical twisted and wrinkled surface morphology before treatment, (d) cotton fibers from the treated sample showing a twisted and wrinkled surface morphology and lack of inter-fiber adhesion after treatment.
Physical Property Testing
Treated and untreated fabric samples were also subjected to a range of physical tests including fabric thickness and tensile mechanical behavior. Prior to testing, all fabric samples were pre-conditioned for at least 8 h in a standard atmosphere to bring them to uniform temperature and moisture equilibrium based on standard practice.34,35 Standard test methods were followed to measure each of the physical characteristics as follows.
Fabric Thickness
The fabric thickness of each sample was measured using a dead-weight type thickness gauge with a circular presser foot (SDL 94 thickness gauge, Shirley Developments Ltd.) measuring 1.129 in. (28.7 mm) in diameter, and applying a pressure of 0.60 psi (4.14 kPa) as described in ASTM Standard Test Method D1777-96. 35 Five measurements were taken from randomly-selected locations on each sample and their means and standard errors were calculated.
Tensile Strength and Elongation
The mean and standard error values for tensile strength and elongation at break were measured in the fabric warp direction using the 1 inch (25 mm) ravel strip method described in ASTM Standard Test Method D5035-06. 36 Five specimens measuring 6 × 1.5 in. were cut parallel to each other in the warp direction of each fabric sample and the warp yarns were raveled down to exactly 1 in. width. The tests were performed to failure on a Uniaxial Instron Mechanical Tester, Model 2712-864 using a 3 in. (75 mm) gauge length, with pneumatic clamps at 80-psi grip pressure, and a rate of extension of 12.0 in./min (300 mm/min). The values for the peak force and elongation at the maximum tensile force were recorded at 50% break sensitivity statistically.
Antimicrobial Activity
Staphylococcus aureus, ATCC 6538, was obtained from the American Type Culture Collection. Test organisms were grown overnight at 35 °C in Trypticase Soy Broth (TSB). After incubation, the bacteria were washed twice in Phosphate Buffered Saline (PBS). The resulting pellets were re-suspended in 1 mL of PBS without calcium and magnesium and re-centrifuged. The final pellet was re-suspended in 1 mL of PBS and used as the stock bacterial suspension for the remaining antibacterial tests.
To determine the total number of organisms applied to the test surfaces, serial 1/10 dilutions of the stock bacterial suspension were made using the first six wells of a 24-well microtiter plate. Bacterial numbers, as colony forming units (CFUs), were determined by plating 10 μL aliquots from each well onto Trypticase Soy Agar (TSA) plates and incubating overnight at 37 °C. Plates containing 30–300 colonies were counted and used to determine the number of organisms present in the stock suspensions.
An inoculum consisting of 20 μL of the stock bacterial suspension was applied to 0.5 in. 2 swatches of fabric held in sterile Petri plates. Care was taken to ensure that the inoculum was evenly distributed and completely absorbed into the fabric. Bacterial stock suspension (20 μL) was also added to control (untreated) fabric swatches. To avoid drying, which can stress the microbes and contribute to loss in viability, 37 the Petri plates with the inoculated surfaces were placed into gasketed chambers containing a water reservoir to maintain humidity. The chambers were then transferred directly into an incubator at either 24 °C or 37 °C for a series of preplanned times. For each set of conditions, three replicates were used.
After incubation, the swatches were placed onto TSA plates and incubated at 37 °C. At 24 and 48 h, the swatches were observed, and bacterial survival (as indicated by the observation of S. aureus colonies) was assessed in terms of growth (G), reduced growth (RG) or no-growth (NG).
Results
Fabric Thickness
Microbiocidal treatment resulted in no significant changes in fabric thickness, but caused up to 3% fabric shrinkage in either the warp or weft direction of the three cotton/polyester blended fabrics. Such dimensional changes are typical for textile finishing operations and can be limited by appropriate control of mechanical, thermal, and chemical finishing conditions.
Tensile Strength and Elongation
For fabrics that contained at least 50% polyester fiber, the micro-biocidal treatments resulted in no significant changes in the tensile strength for Fabrics A and C (Fig. 4). However, a significant tensile strength loss of 7–8% was observed for the fabric containing the greatest proportion of cotton fibers, Fabric B.
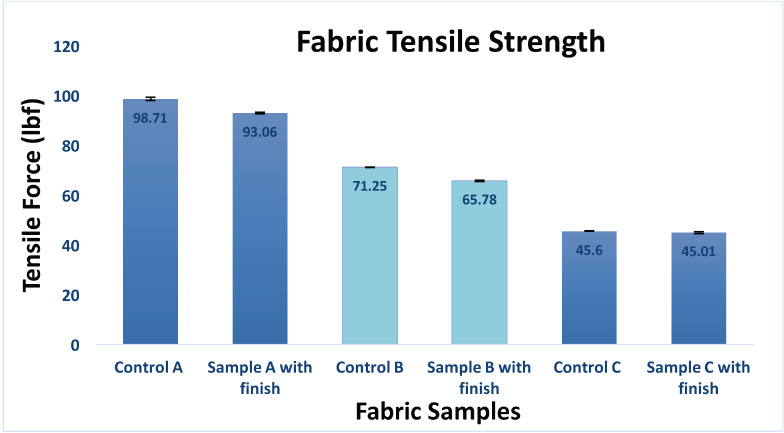
Changes in tensile strength for untreated and treated fabrics.
Fabric elongation increased significantly for each of the three samples (Fig. 5). All fabric samples had a higher elongation after treatment, which is likely due to the silicone component of the antimicrobial formulation.
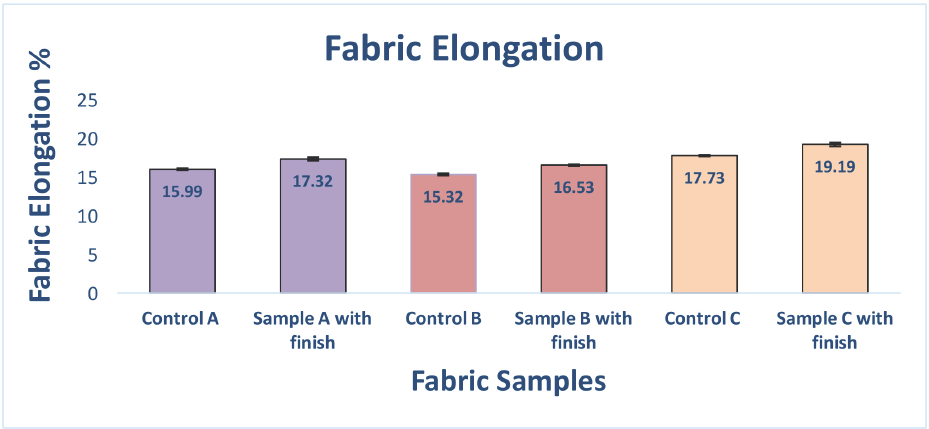
Changes in fabric elongation for untreated and treated fabrics.
SEM
SEMs of treated and untreated fabric samples are presented in Fig. 3. The morphological appearance of the fibers before and after finishing were compared; there appeared to be minimal surface degradation of the fabric. The smoothness of both cotton and polyester fibers was retained, and there was no evidence of fibers or fibrils being damaged by the application of the antimicrobial treatment. Additionally, there was no evidence of fiber-to-fiber bonding or inter- fiber adhesion following the antimicrobial finish that could have resulted in a stiffer or rougher fabric with inferior aesthetical appeal.
Bacterial Survival Studies
Bacterial survival studies were performed, using a range of incubation conditions with different contact times, contact temperatures, and microbial cell concentrations. Test pieces were evaluated by the visual assessment of inoculated microorganism growth. A summary of the test conditions examined, and the outcomes are reported in Table I.
Resulting Bacterial Growth on Antimicrobial Treated and Untreated Fabric Swatches Inoculated with S. aureus a
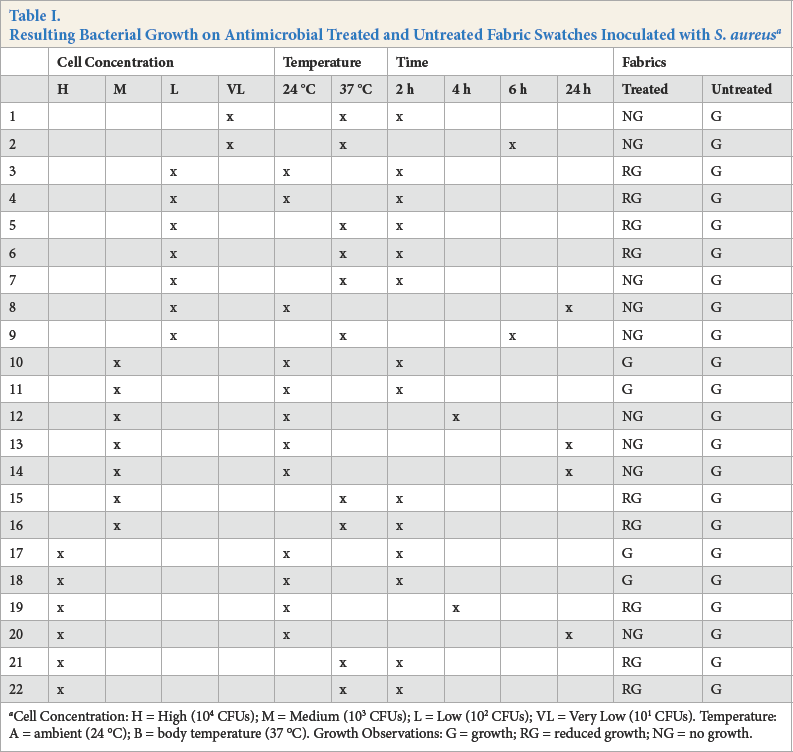
Cell Concentration: H = High (104 CFUs); M = Medium (103 CFUs); L = Low (102 CFUs); VL = Very Low (101 CFUs). Temperature: A = ambient (24 °C); B = body temperature (37 °C). Growth Observations: G = growth; RG = reduced growth; NG = no growth.
Discussion
Many factors play a role in the ultimate growth or inhibition of test organisms. We considered the numbers or concentration of the organism—designated as H (High), M (Medium), L (Low), or VL (Very Low)—the temperature (ambient, 24 °C or body, 37 °C), as well as the time (2, 4, 6, or 24 h). We found that the temperature to which the cells were exposed as well as the exposure time were important in determining the effect on microbial survival following contact with the test surfaces. As expected, the lowest numbers of colony forming units (CFUs) were most vulnerable to contact temperature and time.
When VL concentrations of the organism were applied to the treated test fabrics, no growth was observed at 37 °C after 2 h of exposure (Run 1) or after 6 h of exposure (Run 2) at the same temperature.
When an L concentration of the organism was applied to the treated test fabrics, reduced growth was observed after 2 h of contact at both 24 °C (Runs 3 and 4) and at 37 °C (Runs 5 and 6). After 24 h of exposure at 24 °C (Run 8) and 6 h at 37 °C (Run 9), no growth of the organism was observed.
When the M concentration of cells was applied, the effects of exposure time were observed. Growth was observed after 2 h of exposure at 24 °C (Runs 10 and 11), but no growth was observed after 4 h (Run 12), or after 24 h (Runs 13 and 14). The effect of temperature was confirmed by noting that reduced growth was observed after 2 h at 37 °C (Runs 15 and 16).
Similar trends were seen when the H concentration of cells was applied. When exposed to 24 °C for 2 h (Runs 17 and 18) growth was observed, whereas after 4 h of exposure, growth was almost eliminated (Run 19). By 24 h, no growth was observed (Run 20). At 37 °C, reduced growth was observed within 2 h (Runs 21 and 22).
In all cases, abundant growth was observed on the untreated control surfaces. Fig. 6 shows representative plates of triplicate samples of fabric assessed as Growth (left) and No Growth (right).
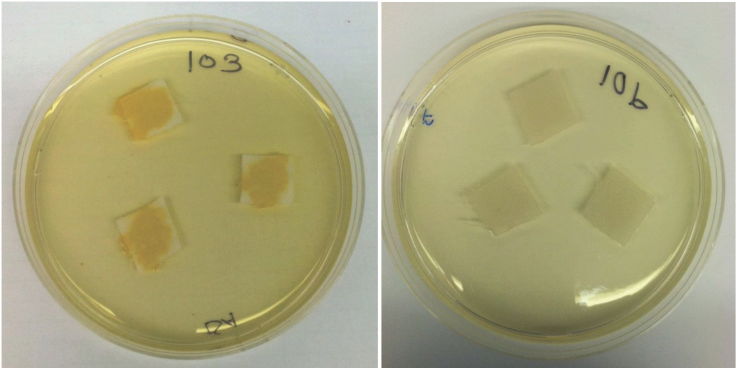
Growth of bacteria following contact of S. aureus with treated (right) and untreated fabric (lef) followed by incubation on TSA.
A significant feature of this microbial assay is that the test organisms were applied to individual fabric swatches, which are then incubated under controlled test conditions. After incubation, each swatch with its associated bacteria was placed onto a Petri plate containing TSA growth medium. Following overnight incubation, any test organisms that were not killed grow into colonies that are easily counted. Since assessment of microbial survival in this experiment did not rely on removing the inoculated bacteria from the test pieces for separate culture and enumeration, experimental variability and error were reduced. This method also did not rely on diffusion of the antimicrobial compound through agar, which can be dependent on factors such as molecular weight of the compound and test conditions.38,39Diffusion methods are also inappropriate for assessing the activity of bonded antimicrobials. Assaying the entire swatch with the inoculum yielded results with a high degree of consistency and reproducibility.
An additional feature of this test procedure was that fabric specimens of small size can be used. This specimen size, which can be as small as 0.25 × 0.25 in., reduced the quantity of fabric needed for assessment. Furthermore, three or four samples can be placed on a standard agar plate at the incubation step prior to assessing CFU growth, thus reducing the amount of materials and consumables required. Not only does this reduce the cost of performing such tests, but it also allows a larger number of materials to be tested at the same time. A similar method was also used by some of the authors to determine the antimicrobial efficacy of polymer films and hard surfaces. 40 Although the bacterial suspension was not absorbed into these types of materials, test coupons can be inverted onto the agar medium with the inoculum so that any surviving microbes can grow into colonies for subsequent observation. The simplicity and good reproduc-ibility of the method developed for this study supports use of this procedure as a rapid screening method for assessing the microbiocidal activity of textiles and fabrics.
Conclusions
QAC-D antimicrobial treatment of the test fabric resulted in at least a 2-log reduction of S. aureus within 2 h. The extent of the microbial biocidal effect increased with contact times up to 24 h. After 24 h of contact, 104 CFUs (the most concentrated inoculum tested in this regimen) of S. aureus were destroyed on a 0.5 × 0.5 in. piece of fabric (equivalent to more than 50 million CFUs/yd 2 ).
Use of higher temperatures (37 °C vs. 24 °C) also reduced the survival of the microbes in contact with the treated fabrics. Allowing the fabric swatches to dry as the microorganisms sat in contact with the treated fabric also increased the extent of microbial destruction (data not shown).
SEM images and physical tests confirmed that the microbio-cidal treatment caused no changes in the surface appearance of either cotton or polyester fibers in the three test fabrics at the microscopic level. There was also no evidence that the treatments led to inter-fiber adhesion or bonding that would cause greater stiffness, inferior hand, or lower tearing strength for treated cotton/polyester blended fabrics. In addition, no deleterious changes were measured in tensile stress/strain behavior.
This study introduced a new rapid screening and assessment method for determining the antimicrobial efficacy of fabrics, textiles, and other surfaces. The method yielded data with good consistency and reproducibility, while using a small test specimen and limited quantities of microbes, test materials, and consumables. The QAC-D finish was shown to be effective in killing S. aureus bacteria, in short time frames. Furthermore, this method was used to show the effects of time, temperature, and bacterial load on the survival of microbes in contact with the antimicrobial fabric. Increased temperature and contact time promoted microbial destruction by the treated fabric and, not surprisingly, higher bacterial loads took longer to kill. The QAC-D fabric produced at least a 2-log bacterial reduction on a 0.25 in 2 fabric sample within 2 h, and at least a 4-log bacterial reduction within 24 h, at the highest concentration of bacteria tested.
Footnotes
Acknowledgements
The authors would like to thank the National Science Foundation for support through Award 0711927.
