Abstract
A sensitive and fast method using gas chromatography-mass spectrometry (GC-MS) analysis was developed for simultaneous determination of 16 polycyclic aromatic hydrocarbons (PAHs) in functional textile samples. The method under optimized conditions exhibited good linearity with linear correlation coefficients between 0.9930 and 0.9999, with maximum recoveries within 74.0∼99.0%, and with relative standard deviation (RSD) of 0.89 to 4.94%. Quantitation and detection limits ranged from 1.3 to 12.2 üg/L and 0.4 to 3.8 üg/L, respectively. These values were less than the limits of two relative standards (Oeko-Tex Standard 100 and ZEK 01.2-08). When applied to coated denim fabric and waterproof tarpaulins, results showed that PAH concentrations in waterproof tarpaulins were greater than the limits of the two relative standards.
Introduction
Polycyclic aromatic hydrocarbons (PAHs), bearing muta-genic and carcinogenic properties, are distributed widely in the environment and pose serious health risks to all living organisms.1,2 PAHs have drawn considerable attention and are included in the priority pollutant lists of European Union (EU) and US Environmental Protection Agency (US EPA).3–6 The level of benzo[a]pyrene (B[a]P), as well as the total amount of 16 PAHs, is limited by Oeko-Tex standard 100 (2012 edition) 7 and ZEK01.2-088 (Table I), due to their potential toxicity.
Limits for Textile PAH Content based on Oeko-Tex Standard 100 (2012 edition) and ZEK01.2-08.
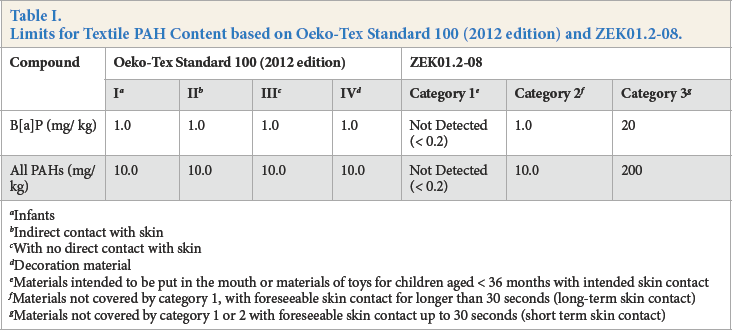
Infants
Indirect contact with skin
With no direct contact with skin
Decoration material
Materials intended to be put in the mouth or materials of toys for children aged < 36 months with intended skin contact
Materials not covered by category 1, with foreseeable skin contact for longer than 30 seconds (long-term skin contact)
Materials not covered by category 1 or 2 with foreseeable skin contact up to 30 seconds (short term skin contact)
Many methods for PAH detection have been investigated, involving high performance liquid chromatography (HPLC), gas chromatography (GC), gas chromatography-mass spectrom-etry (GC-MS), spectrophotometry, and electrophoresis.9–18 With regards to spectrophotometry, many disturbance factors affect the results, leading to false positives. Hence the method's precision and accuracy were unsatisfactory. The main problem for spectrophoto-metric multi-component analysis is the simultaneous determination of numerous PAHs in the same sample.
GC-MS and HPLC were widely applied to PAH analysis in recent years. GC columns provided better separation than HPLC columns, especially for PAH isomers in multi-component analysis, and could effectively reduce baseline interference, resulting in more accurate quantitation and limits of detection (LODs) than those from HPLC methods. GC-MS was chosen as the standard method used in many countries and was used in this study.
Most PAH reports are focused on environmental samples19–21and food matrices.22,23 Very few reports24,25 involve textiles, especially functional clothing, which are increasingly popular with outdoor recreational consumers. Customs inspections for this clothing is also increasing remarkably.
The present study investigated PAH levels in textiles, espe-cially in coated materials (e.g., coated denim fabrics and waterproof tarpaulins). In our previous studies,26,27 Soxhlet extraction was used to extract phthalate esters (PAEs) from textiles. However, Soxhlet extraction of PAHs was complicated and time-consuming, taking 24 to 72 h.28–30 By comparison, ultrasonic extraction was reported to be simple and rapid with high recovery, and was recommended by the US EPA for nonvolatile and semivolatile organic compounds, including PAHs in SW-846-3550.31–33 Solid phase extraction (SPE), widely used in the pretreatment of various samples, is a simple and reliable alternative. However, due to matrix interferences, SPE was not suitable for trace sample analysis. To reduce matrix interferences, liquid-liquid extraction (LLE) proved to be a routine and efficient sample preparation method, was introduced as the initial cleanup procedure before SPE.
Therefore, a sensitive and fast method based on ultrasonic extraction, LLE and SPE purification, and subsequent GC-MS analysis, was investigated for simultaneous determination of 16 PAHs in functional textile samples.
Experimental
Instruments and Reagents
A 7890A-5975C GC-MS (Agilent) instrument was equipped with a high pressure-5 mass spectrometry (HP-5MS) GC column (30 m × 0.25 mm × 0.25 μm). An ultrasonic cleaner was obtained from ELMA. A nitrogen blowing instrument was supplied by Organomation.
HPLC grade reagents (n-hexane, cyclohexane, dichloro-methane, and dimethylformamide (DMF)) were purchased from Tianjin Guangfu Fine Chemicals. Anhydrous sodium sulfate, commercially available from Tianjin Dengzhong Chemical, was stored at 400 °C for 4 h prior to use. Mixed standard solutions of 16 PAHs in dichloromethane, with each component at a mass concentration of 100 μg/mL, were purchased from Supelco.
Procedures
Preparation and Extraction of Textile Samples
The cotton samples containing PAHs were crushed into a 0.5 × 0.5 cm size. The crushed sample (1 g) was added into a conical flask with a stopper containing 50 mL of cyclo-hexane, which was then treated in an ultrasonic bath under 650 W at 45 °C for 30 min. The mixture was transferred to a 250-mL Teflon separatory funnel and extracted three times with a 15-mL mixture of DMF and water (v/v = 9:1). The water phase was collected in a 500-mL separatory funnel, diluted with 90 mL of distilled water, and re-extracted with 135 mL of cyclohexane, into which 1 g of NaCl was added for demulsification. The cyclohexane phase was collected and concentrated to 20 mL by nitrogen blowing, and then to 2 mL under a gentle stream of nitrogen.
Sample Purification
A borosilicate glass column (18 cm, 1.5 mm ID) was packed to a height of ∼10 cm with ∼7 g of silica gel. The silica gel in the column was covered with ∼1 cm of anhydrous sodium sulfate, to remove water left in the extracts from the textile samples, and the top of the column was sealed with degreasing cotton.
The column was conditioned with n-hexane prior to use. The extract was loaded quantitatively onto the extraction borosilicate glass column and subsequently eluted with 20 mL of n-hexane. The column was finally eluted with 30 mL of hexane/dichloromethane (v/v = 1:1) mixture. The collected effluent was concentrated to 1 mL under a gentle stream of nitrogen.
Standard Solution Preparation
Working solution concentrations of PAHs (12.5, 25, 50, 100, and 200 μg/mL) were prepared, from which 1 μL was drawn out respectively for GC-MS testing, with retention time used for qualitative analysis and peak area used for quantitative determinations.
GC-MS Analysis
The GC oven temperature program was started at 60 °C for 1 min, increased to 210 °C at a rate of 10 °C/min, increased to 310 °C at a rate of 5 °C/min, and then was maintained at 310 °C for 8 min. The mass spectrometer was run in full scan mode (m/z range was 50 to 500). The ionization voltage was set at 70 e V.
Results and Discussion
PAH Separation and Determination
Purified sample (1 μL from 1 mL of the concentrated efflu-ent) was injected to the GC-MS system. 16 PAHs were identified by GC-MS run in full scan mode and then run in selected ion monitoring (SIM) mode, to enhance the sensitivity for quantitation.
A total ion chromatogram was obtained (TIC, Fig. 1) was obtained. The 16 PAHs could be quantitated by comparing the ratio of the SIM ion peak abundance for each PAH in the sample to that of a standard substance using the external standard method. Each analytical test took just 36 min, which was nearly the same as that reported by Wei 25 and much faster than that reported by Mao. 24
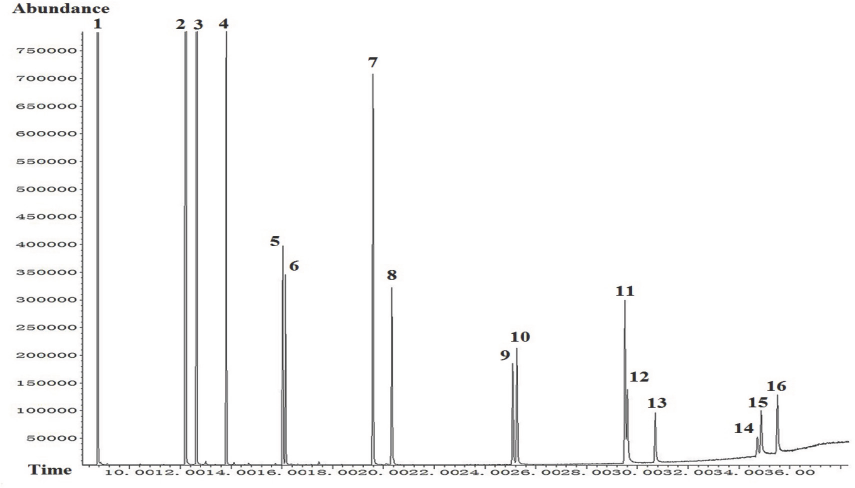
Total mass spectra of 16 PAHs: 1. naphthalene, 2. acenaphthylene, 3. acenaphthene, 4. fluorene, 5. phenanthrene, 6. anthracene, 7. fluoranthene, 8. pyrene, 9. benz[a]anthracene, 10. chrysene, 11. benzo[b]fuoranthene, 12. benzo[k]fluoranthene, 13. benzo[a]pyrene (B[a]P), 14. indeno [1,2,3-cd] pyrene, 15. dibenzo[a, h]anthracene, 16. benzo[ghi]perylene.
Limits of PAH Quantitation and Detection
The standard working solution concentrations showed a good linear relationship with the peak abundances, where the correlation coefficients varied from 0.9930 to 0.9999. The limits of quantitation (LOQ) for the analytics ranged from 1.3 to 12.2 μg/L on the basis of a signal-to-noise (S/N) ratio of 10, with limits of detection (LOD) between 0.4 and 3.8 μg/L on the basis of an S/N ratio of 3, which were low enough to meet the 10 mg/kg limit of Oeko-Tex Standard 10 and ZEK01.2-08 (category 2 (10 mg/kg) and category 3 (200 mg/kg)).
Ultrasonic Extraction Temperature Optimization
An extraction temperature of 60 °C was used in ZEK01.2-08. However, it would take a long time for the ultrasonic extraction instrument to increase the temperature from room temperature (RT) to 60 °C. In addition, a longer extraction time was needed for the complete extraction of PAHs at lower temperatures. In view of these circumstances, the ultrasonic extraction was performed at 45 °C in this study.
Ultrasonic Extraction Time Optimization
The extraction time is an important factor, influencing the extraction efficiency. In this study, extraction efficiencies were investigated and compared at 10, 20, 30, 40, 50, and 60 min (Fig. 2). The extraction efficiency increased with the extraction time in the beginning, and stayed at nearly the same level after 30 min. To shorten the testing time and guarantee a relatively high extraction efficiency, the extraction time was set at 30 min in the following experiments.
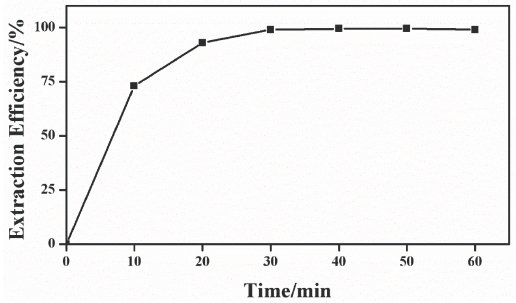
The effect of the ultrasonic extraction time on the extraction time.
Re-Extraction Solvent Selection
Since the functional textile samples were complex, indirect extraction would introduce interferents, such as lipids and non-aromatic chemicals, influencing the analysis. There-fore, this work intended to re-extract the extract for the initial cleanup.
Paschke used DMF as a re-extraction solvent. 9 Grimmer compared the extraction effects of DMF, dimethyl sulfoxide (DMSO), and nitromethane (NM). These results suggested that DMF and DMSO performed much better than NM and the recovery for PAHs would increase in the presence of less than 10% water (v/v). 10
Based on the above, DMF and cyclohexane were selected as the PAH re-extraction solvents in this study. The extraction procedure was carried out as previously described, with an additional 90 mL of water added to change the polarity of the mixed solution of DMF and water. This forced the nonpolar PAHs out of the polar phase and into the cyclohexane phase.
Solid Phase Extraction Solvent
The following solvents were tested as potential solid phase extraction solvents: 20 mL of n-hexane, n-hexane/ dichloromethane (v/v = 2:1), n-hexane/dichloromethane (v/v = 1:1), n-hexane/dichloromethane (v/v = 1:2), and dichloromethane. The results showed that only aliphatic hydrocarbons were detected in the component eluted by n-hexane. For n-hexane/dichloromethane (v/v = 2:1 and v/v = 1:2), no target PAHs were found in the analysis. Unfortunately, the elution efficiency of dichloromethane was nearly zero. However, the target PAHs were completely eluted by n-hexane/dichloromethane (v/v = 1:1), which was used in the following work.
Elution Solvent Volume
The volume of the elution solvent also affects the accuracy and efficiency of this analytical test. Various elution volumes of n-hexane/dichloromethane (v/v = 1:1) were investigated using GC-MS analysis (Fig. 3). The PAHs were completely eluted using 20 mL of solvent. Therefore, the elution volume was set to 20 mL in this study to ensure maximal PAH recovery.
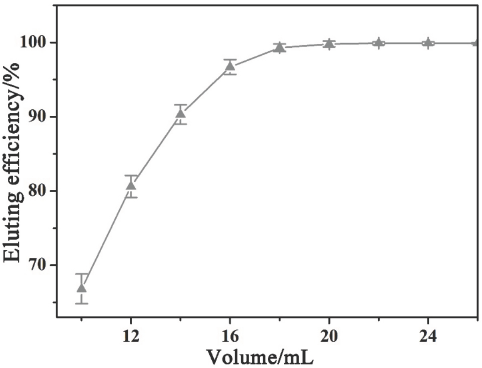
Influence over total extraction efficiency of PAHs from elution solvent volume.
Validation
The feasibility of the proposed method was studied as described in this section. The PAH-containing coated cotton samples were prepared as previously described and spiked with each target compound at the concentration of 1 mg/L (n = 6). The testing results were collected in Table II. It was concluded from the data that the proposed method per-formed well. The recoveries of 16 PAHs in the samples were between 74.0 and 99.0%, with corresponding relative standard deviations (RSD) varying from 0.89 to 4.94%, which performed better than those in the reported literature.15,16,24 Thecalculated LODs for PAHs were in the range of 0.4 to 3.8 μg/L, complying with the criteria requirements of Oeko-Tex Standard 100 and ZEK 01.2-08.
Detection Results for PAHs in Coated Cotton Samples a
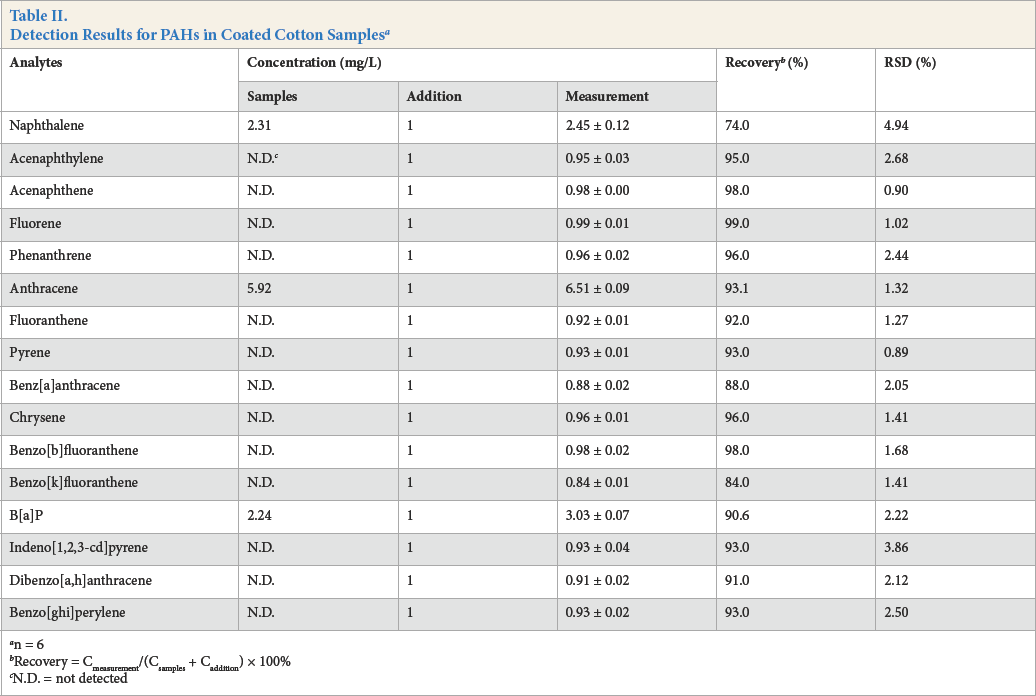
n = 6
Recovery = Cmeasurement/(Csamples + Caddition) × 100%
N.D. = not detected
Comparison with other Methods
Compared to other methods in the literature,24,25 the proposed method was not the most sensitive, but it was applicable for the simultaneous detection of 16 PAHs levels in functional textiles stipulated by Oeko-Tex Standard 100, ZEK 01.2-08, and similar standards. In this work, ultrasonic extraction was applied as suggested by ZEK 01.2-08 and the US EPA. With this method, the extraction time was shortened dramatically to 30 min from 1 h.8,24 n-hexane was used instead of the highly toxic8,25 toluene, acetone, 24 or ethyl acetate 15 as the extraction solvent without reducing the recoveries in this work. Meanwhile, an initial purification procedure was performed with DMF as a re-extraction solvent, which made the further purification simpler and shortened the GC-MS analysis time.
The investigated method had an advantage over those reported in literature,12,17,18 as it was capable of detecting 16 PAHs simultaneously. However, the sample pretreatment process still seemed time-consuming and tedious. Future work will include improvements in sample pretreatment.
Analysis of PAHs in Textile Samples
The proposed method was used in the determination of the 16 PAHs from coated denim fabric (including waterproof tarpaulins). The results, summarized in Table III, showed that naphthalene and acenaphthylene were found in coated denim fabric at 1.15 and 0.17 mg/L respectively. For the waterproof tarpaulins samples, 2.31-mg/L naphthalene, 5.92-mg/L anthracene, and 2.23-mg/L B[a]P were detected. The PAHs concentration in waterproof tarpaulins (including levels of B[a]P and the sum levels of PAHs) were greater than the limits set by Oeko-Tex Standard 100 (2012 edition) and ZEK01.2-08 (category 1 and 2) (Table I), which indicated a potential threat to human health. The coated denim fabric in this study, however, did not pose such a risk based on the results.
Concentrations of PAHs in Textile Samples
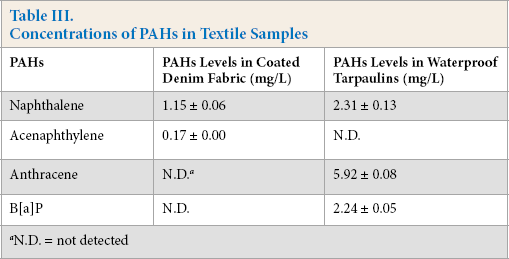
N.D. = not detected
Conclusions
The validation results for the proposed method of PAH detection in textiles was simple, fast, highly sensitive, and gave precise quantitative analysis, meeting the requirements of daily inspection methods. Compared to the previous studies (involving Soxhlet extraction or toluene extraction solvent), the new procedure enabled significant time reduction throughout the entire analytical process, and was capable of simultaneous determination of 16 PAHs in textiles.
Practical testing using the developed method under optimized condition in this work showed that the concentrations of the PAHs in the tested waterproof tarpaulins exceeded the limits of relevant standards. This result suggested that greater attention should be paid to the manufacturing of waterproof tarpaulins and that these items should be routinely monitored by the government for human health threats.
Footnotes
Acknowledgements
The authors gratefully acknowledge the Key Laboratory for Green Chemical Technology of Ministry of Education, Tianjin University. The authors also sincerely acknowledge the Tianjin Entry-Exit Inspection and Quarantine Bureau for their technical assistance.
