Abstract
The aim of this work was the antimicrobial functionalization of a textile material via a host-guest system. Cotton was treated with β-cyclodextrin host molecules (BCD) via conventional exhaust application, followed by a curing process. These fabrics were then treated with an antimicrobial agent (chlorhexidine acetate). Chlorhexidine acetate adsorbtion onto these fabrics was calculated and compared to a control. Further antibacterial tests were done on the functionalized fabrics and compared to their controls as well. This work also describes the selection of an antimicrobial molecule, a spectral study done on two kinds of BCD-antimicrobial molecules, the adsorption onto BCD-fixed fabrics, and the subsequent antibacterial property of such fabrics.
Introduction
β-Cyclodextrins (BCDs) are cyclic oligosaccharides com-posed of glucose units linked by α-1,4-glycosidic bonds. Due to this chair arrangement of the glucopyranose units, cyclo-dextrins are conical cylinders, with the secondary hydroxyl groups of the glucopyranose molecules on the wide side of the cylinder and the primary hydroxyls on the narrow side of the cylinder. The inner surface of the cavity is covered with glycosidic oxygen bridges and hydrogens of the glucose residues, giving the cavity a hydrophobic property, while the outside of the cavity is hydrophilic due to the hydroxyl groups. This gives the BCD molecules the ability to complex with a range of hydrophobic molecules depending on their molecular weight, solubility, and geometrical structure. Such complexation finds applications in the food, pharmaceutical, paper, textiles, cosmetic, chemical, and agro industries due to the ability of the BCD molecule to function as a host molecule apart from its ability to increase the solubility of complexed hydrophobic guest molecules indirectly. 1
The attachment of different types of BCDs to cotton was explored in an earlier work. 2 For this work, only the unmod-ified or native BCD was selected for fixation to cotton. The attachment of BCD onto cotton was done via a crosslinker, 1,2,3,4-butane tetracarboxylic acid (BTCA).1–4 The catalyst sodium hypophosphite (SHPI) was also added into the liquor and the BCD was fixed to cotton via an esterification reaction. 2 This work describes the subsequent antimicrobial functionalization of such BCD-treated cotton (BCD-cotton).
The disinfectant chlorhexidine was selected as the antimicrobial molecule. Chlorhexidine is available as a base (CHXB) and in its various salt forms. Some of the commercially-available chlorhexidine salts are chlorhexidine dihydrochloride (CHXH), chlorhexidine diacetate (CHXA), and chlorhexidine digluconate (CHXG). CHXA was chosen as the antibacterial agent for the antimicrobial functionalization of the BCD-cotton samples. The reasons for this choice is further explained in the treatment section. In this work, “chlorhexidine” refers to the molecule in general (i.e., its base and salt forms).
Materials
Plain woven bleached cotton (with fabric density of 170 g/m2) was obtained from WFK Testgewebe.
Chemicals-General
BCD was obtained from Roquette. BTCA, sodium hydrox-ide, sodium carbonate, tris(hydroxymethyl) aminomethane (Tris), phenolphthalein, and sodium hypophosphite (SHPI) were obtained from Sigma Aldrich..
Chlorhexidine
CHXB and CHXA used in this study were obtained from Sigma Aldrich.
An ideal guest molecule for formation of a BCD complex should have a molecular weight between 100-400 g/mol and water solubility of less than 10 g/L. 3 Chlorhexidine was chosen as the guest antibacterial agent in this study on the basis of its hydrophobicity, molecular weight, and its well-known ability to form a BCD inclusion complex. 4
Chlorhexidines are bisguanides and are therefore closely related to polymeric biguanides such as polyhexamethylene biguanide (PHMB). PHMB and chlorhexidines are used for disinfection mainly in hospitals. Cationic chlorhexidines are attached to an anionic textile surface through electrostatic interaction.5,6 Chlorhexidines are known to attack bacterial cell membranes. This is said to reduce the risk of microbial resistance development in comparison to more widely-used quaternary ammonium compounds. 7
Chlorhexidine is commercially synthesized in its base and salt forms. The structure of chlorhexidine in its base form (CHXB) is given in Fig. 1. The molecular weight of this base molecule is 505.45 g/mol.
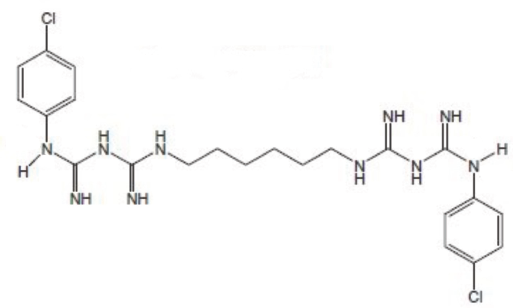
Structure of chlorhexidine base (CHXB).
Chlorhexidines have a molecular length of 28.55 Å, while the length of a BCD cavity is 7.8 Å. The entire length ofa chlorhexidine molecule cannot ft into the BCD cavity. However, the radial dimensions of the phenyl rings of the chlorhexidine molecule are small enough to fit widthwise inside the BCD cavity.8,9 Therefore, the two hydrophobic phenyl rings of one chlorhexidine molecule inside two BCD cavities results in a 1:2 complexation ratio, respectively (Fig. 2). In reality, one or more BCD molecules may additionally thread through the chlorhexidine molecule leading to a complexation ratio of greater than 1:2. 4
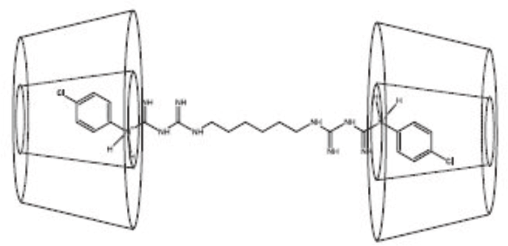
Complexation of chlorhexidine with BCD molecules. (Reproduced from Denadai, et al. 8 with permission from Elsevier).
The most commonly used chlorhexidine type in disinfec-tant solutions are the salt forms such as CHXA, CHXG, and CHXH. This is due to the improved stability and solubility of the salt forms as compared to the base molecule. Among the various chlorhexidine salt forms available, CHXA was chosen for this work due to its aqueous solubility and molecular weight. The water solubility of this salt (as compared to the base and other salts) was particularly suitable for textile exhaust applications. CHXA has a molecular weight of 625.6 g/mol. 10 The structure of CHXA is given in Fig. 3.
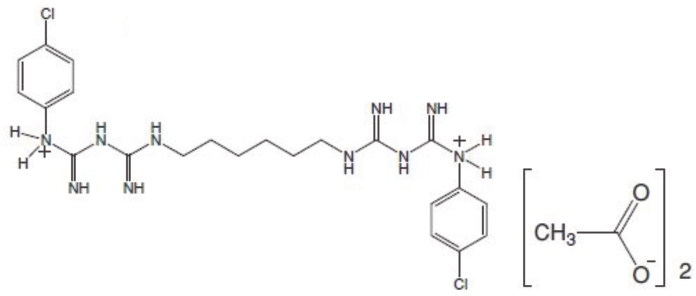
Structure of CHXA.
Methods
Quantification of Fixed BCD
Quantification of the BCD fixed onto cotton was done using the phenolphthalein method, which was described in detail in an earlier work. 2 Determination of the BCD fixation amount from the phenolphthalein method was indirect and assumed that all of BCD cavities fixed on the fabric were accessible to the phenolphthalein molecules. Any steric influence contributed by the crosslinker on the BCD-phe-nolphthalein absorbance was also not considered, since the calibration curve used for the calculations used only BCD-phenolphthalein solutions.
UV-Visible Spectrophotometric Solution Studies
UV-Visible (UV-Vis) spectrophotometric studies of BCD-guest solutions were done to study the complex's stability and binding properties. Changes in the UV-Vis maximum absorbance of the aqueous guest (CHXA) before, during, and after complexation with BCD were determined. During measurements, the CHXA concentration in the solution was kept constant, while the BCD concentration was steadily increased. As the complexation proceeded, a shift in the maximum absorbance peak was observed. 4 Complexation of the CHXA chromophores inside the BCD cavity led to this shift. The absorbance value of this shifted peak increased with the BCD concentration in solution. This continued until no further complexation took place upon the addition of (excess) BCD into the solution.
We hypothesized that due to the differences in water solu-bility and molecular weight between CHXA and CHXB, complexation between them and BCD would not be the same. This would be observable in the UV-Vis spectral studies, where complexation would result in a change in maximum absorbance.
UV-Vis spectrophotometric scans of CHXB and CHXA in aqueous solutions at neutral pH showed two absorbance peaks at 234 nm and 254 nm, respectively (Fig. 4). In this work, the 254 nm absorbance peak was used for all experi-ments, as it appeared to be sharper than the peak at 234 nm.
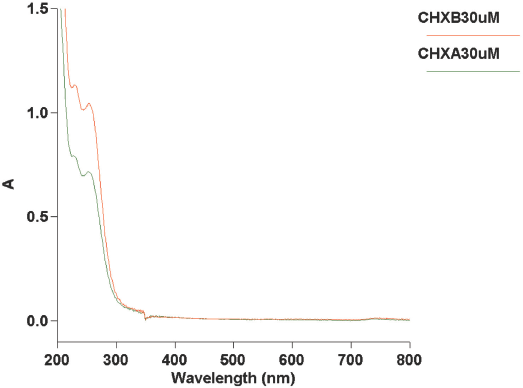
UV-Vis spectrophotometric absorbance (A) scans through 200-800 nm of aqueous solutions of CHXB and CHXA. The concentration of CHXA and CHXB is 30 μM.
Stock solutions of CHXA (60 μM), CHXB (60 μM), and BCD (15 mM, which is the solubility limit of BCD in water) were made with demineralized water in 100 mL beakers. A series of test tubes containing mixtures of aqueous CHXB and BCD solutions were then prepared. The final CHXB solution concentration in the test tubes was kept constant at 30 μM, while the BCD solution concentration was varied from 0 to 7.5 mM. UV-Vis spectral measurements were then made with a UV-Vis spectrophotometer (Cary 100) at room temperature (RT) and neutral pH. This was then followed by UV-Vis measurements of CHXA in BCD solutions under the same conditions.
Cotton and BCD-Cotton Treatment with CHXA
Cotton substrates used in this work were treated with 100 g/L of BCD and 30 g/L of BTCA. 2 The amount of BCD fixed on cotton was calculated, using the phenolphthalein method, as 6.2 mg of BCD per gram of cotton.
CHXA guest antibacterial solubility was 19 g/L (1.9 % w/v) as compared to 0.08 g/L (0.008% w/v) for CHXB. 11 This higher water solubility allowed a wide range of application bulk concentrations with water as a solvent. In the case of CHXB, a solvent such as ethanol would have to be used for higher application bulk concentrations. On the other hand, CHXA was not as hydrophobic as CHXB. The other two salts of chlorhexidines, CHXG and CHXH, were not chosen due to their inability to match the application requirements. CHXG aqueous solubility was greater than 700 g/L (> 70% w/v), 11 but its molecular weight was 897.8 g/mol (outside of requirement limits). On the other hand, CHXH, with its lower molecular weight of 578.37 g/mol and an aqueous solubility of 0.6 g/L (0.06% w/v), 11 may be suitable. However, the treatment of cotton with CHXA would allow the use of higher bulk con-centrations, even in comparison to CHXH. This was expected to give fabrics with greater antibacterial activity. As a tradeoff between guest molecule hydrophobicity and its aqueous solubility, the latter was given priority. Therefore as a starting point, CHXA was chosen as the guest molecule among the various chlorhexidine salts for this work.
Cotton (control) and BCD-cotton were treated with CHXA in a beaker with an a liquor ratio (LR) of 10:1. Textile sample dimensions were 10 × 10 cm. Fabric treatments were done at 40 °C and neutral pH. Treatment duration was kept at 30 min. The bulk application concentrations of CHXA ranged from 0.01 g/L to 2.5 g/L.
After treatment of the various samples with CHXA bulk liquor, the fabrics were gently removed from the bath. The UV-Vis absorbance of the bulk liquor was then measured. The concentration of CHXA in the bulk liquor was finally determined by using a calibration curve shown (Fig. 5).
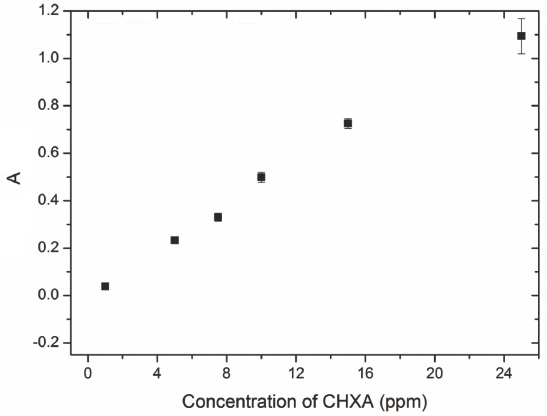
Calibration curve of CHXA. Absorbance (A) values were measured with UV-Vis spectrophotometer at 254 nm. 1 ppm = 0.001 g/L.
A linear curve was obtained between 1 to 25 ppm (0.001 g/L to 0.025 g/L) of CHXA (Fig. 5). Treatment of the fabrics with CHXA ranged from bulk concentrations of 0.001 g/L to 2.5 g/L. CHXA concentrations in the bulk liquor after treatment were much higher than the measurable limit shown in the calibration curve. The exhaust bulk solutions after textile treatment were diluted by 10× to 1000×, and the UV-Vis absorbance was measured. The concentrations in the beaker were then recalculated using the calibration curve after taking into consideration the dilution factor. From the difference between the known bulk concentration at the start of the experiment and the calculated bulk concentrations at the end of the experiment, the adsorption of CHXA onto the textile could be calculated as shown in Eq. 1.
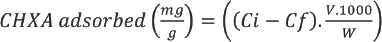
BCD-cotton treated with CHXA is referred to henceforth as BCD-cotton-CHXA. The respective control fabrics treated with CHXA are the cotton-CHXA fabrics.
Antibacterial Activity of CHXA-Treated Fabrics
Chlorhexidine is a broad spectrum antibacterial agent effective against Gram-negative and Gram-positive bacteria. The cationic biguanide groups of chlorhexidine have an affinity for the negatively-charged bacterial cell wall. These anionic sites stabilize the divalent cations on the membrane surface. Fig. 6 shows the chlorhexidine antibacterial mechanism.
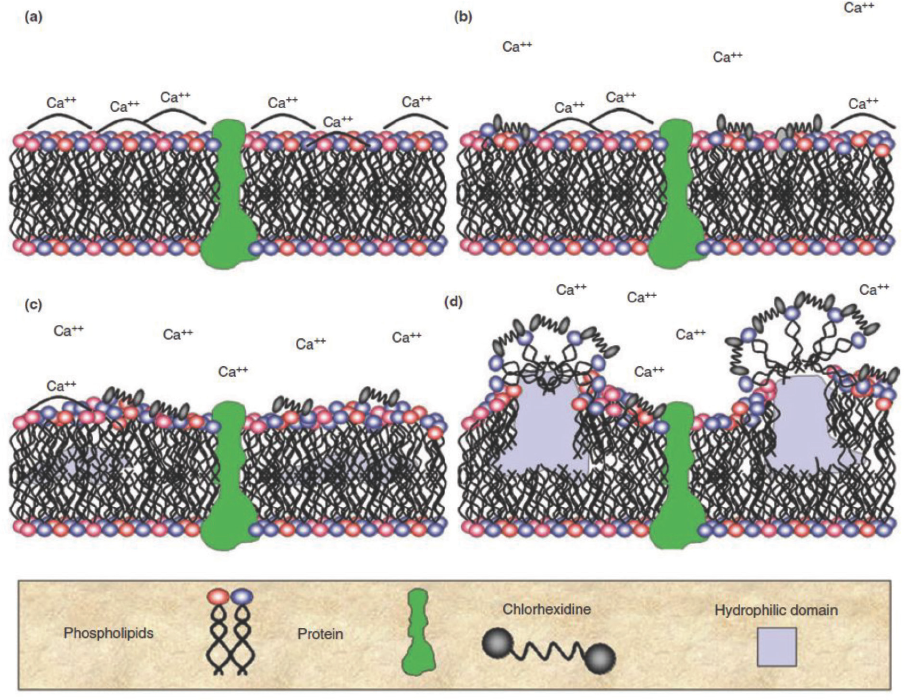
Attack of bacterial cell membrane by chlorhexidine. (Reproduced from Gilbert, et al. 7 with permission from John Wiley and Sons).
The first step of the attack is destabilization of the divalent cations on the membrane surface and the interaction of the end groups of chlorhexidine on the two phospholipid heads of the membrane bilayer. The hexamethylene structure is relatively inflexible, due to being only six carbons long, and therefore does not enter the phospholipid bilayer. The antibacterial molecule then proceeds to solubilize the hydrophobic membrane leading to cellular leakage, followed by structural disintegration of the bacterium. 7
Antibacterial tests were done on BCD-cotton-CHXA and cotton-CHXA samples following the JIS L 1902 standard.12 Thetests were done using the
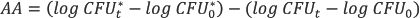
Release of CHXA from CHXA-Treated Fabrics
Antibacterial test results showed that BCD-cotton-CHXA samples had less antibacterial activity when compared to cotton-CHXA samples for the same bulk concentration treatments. This was attributed to the slow release phenom-enon of CHXA from the BCD complex.
CHXA release experiments were done to confirm the slow release of CHXA from the BCD complex. The CHXA-treat-ed samples were soaked in water and the release of CHXA from the samples into solution was spectrophotometrically measured. BCD-cotton-CHXA and cotton-CHXA fabrics treated with 1.5 g/L of CHXA bulk concentration were chosen for the release experiments. The release of CHXA into solution was spectrophotometrically measurable with the BCD-cotton-CHXA samples treated only at concentrations greater than or equal to the CHXA bulk concentration. Samples treated at lower CHXA bulk concentrations (less than 1.5 g/L) showed very little release of CHXA into the solution, making spectroscopic measurements inconsistent.
BCD-cotton-CHXA and cotton-CHXA samples measuring 2.5 × 2.5 cm were placed in a beaker containing 20 mL of demineralized water. The water in the beaker was kept at RT and neutral pH, and was magnetically stirred. The duration of the experiment was 60 min. The UV-Vis absorbance of the solution was measured at 254 nm. The bulk liquor (1 mL) was pipetted out for each measurement, after which it was replaced back into the bulk solution. This measurement was performed every minute for the first 10 min, followed by every two minutes for the next 10 min, and finally for every five minutes up to 60 min. The release of CHXA into the solution was calculated from the calibration curve in Fig. 5.
Results and Discussions
UV-Vis Measurements
UV-Vis scans were done on solutions containing a mixture of BCD and respective guest molecules. As described earlier, the concentration of the guest molecule was kept constant at 30 μM, while that of BCD was increased steadily in these solutions. No shift in the CHXA/CHXB absorbance peak at 254 nm was observed in the guest-BCD solutions at molar ratios from 1:1 to 1:40. The maximum absorbance peak of CHXA-BCD and CHXB-BCD solutions (Figs. 7 and 8) shifted from 254 nm to 258 nm at BCD concentrations equal to or greater than 1.25 mM.
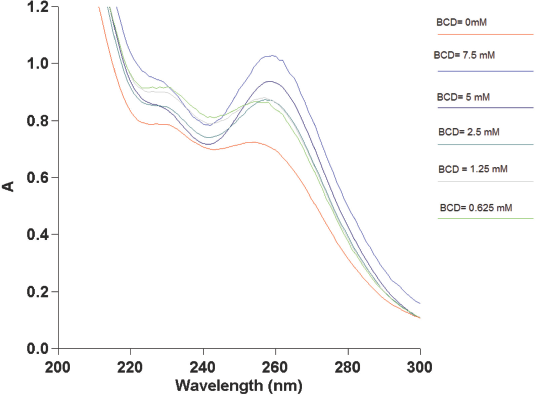
UV-Vis spectrophotometer absorbances (A) of 30 μM CHXB with varying concentrations of BCD.
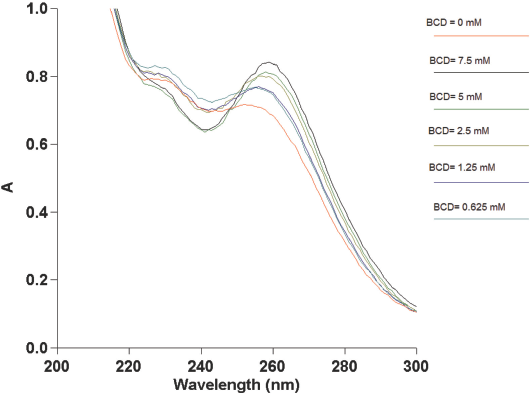
UV vis spectrophotometer absorbances (A) of 30 μM of CHXA with varying concentrations of BCD.
These results indicated that a BCD molar excess of about 40× that of CHXA/CHXB was needed for complexation to start under these experimental conditions. This implies that the complexation starts when there are 40 or more molecules of BCD in the solution for every CHXA or CHXB molecule. The absorbance values of the shifted peak increased with the increase in BCD concentrations in the mixed solutions. This was an indication of increased complexation between the BCD and the guest molecules, despite the total number of CHXA/CHXB molecules added to the mixture remaining constant
BCD concentrations in solution required for the start of complexation with CHXA, and then with CHXB, were identical. The onset of BCD-CHXA complexation was expected to occur at greater BCD concentrations (therefore at greater guest to BCD ratios) when compared to BCD-CHXB. This was due to the reduced hydrophobicity and the higher molecular weight of CHXA in comparison with CHXB. It was also hypothesized that this delay in the onset of complexation with CHXA would be observable in UV-Vis spectra. However, in this spectral study, except for the relative differences in absorbances, no differences were seen in the BCD concentrations required for the onset of complex-ation between these two types of chlorhexidine. The same set of experiments were repeated in the presence of a 2.5 × 2.5 cm cotton fabric (in test tubes containing the BCD-guest solutions). As observed earlier, the shift in the maximum absorbance of CHXA and CHXB remained at a BCD concentration of 1.25 mM.
Chlorhexidine contains two hydrophobic
However, on closer observation of the pharmaceutical industry protocol, it appeared that this conclusion was incorrect. The industry procedure followed the addition of solid CHXA/ CHXB to the aqueous BCD solution. In such cases, complex-ation was reported to start with a BCD molar excess of 0.5 to 0.75. 15 In this work, CHXA/CHXB was dissolved in water prior to the addition to BCD solution. A concentration of 30 μM was low enough for these materials to dissolve in water easily. Therefore, such a situation was not conducive for com-plexation with BCD where the guest molecules were already solubilized in water. This is due to the fact that complexation would require the breaking of already established hydrogen bonds between CHXA/CHXB and water in favor of hydro-phobic interactions of CHXA/CHXB with the BCD cavity.
According to the literature, the presence of BCD has minimal influence on the solubility of already soluble drugs. 16 If CHXA or CHXB were added directly to BCD solutions in excess of their solubility limit, complexation with BCD would start immediately (i.e., at lower guest to BCD mixture ratios). This would be due to the simultaneously available hydrophobic interactions (for the apolar moieties of the guest molecules) and hydrogen bonds from water (for the polar groups of the guest molecules). In this situation, the onset of complexation was significantly influenced by aqueous solubility limits of the two forms of chlorhexidine.
No new differences (with regard to the shifts in the peak) were observed in spectra obtained during the BCD com-plexation with two guest molecules in the presence of cotton fabrics. This implies that the CHXA or CHXB electrostatic interaction with cotton and their complexation with the BCD molecules were not influenced by each other under current experimental conditions.
The solubility or polarity of the guest molecules is known to also influence the stability of a complex, 17 as well as the release of the guest molecule from the complex. 18 It is not known if this is applicable to the complexes of CHXA and CHXB, which were only partially included into the BCD cavities. Determination of the binding rate constants of both CHXA and CHXB with BCD and the study of release of these molecules from the BCD complex needs to be performed.
Fabric Adsorption of CHXA
Attachment of chlorhexidine to cotton is via electrostatic interactions, according to the literature. This occurs between the two cationic biguanide groups of chlorhexi-dine and the carboxylate groups of cotton. It was reported that at higher bulk concentrations, a combination of electrostatic interactions and hydrogen bonding exists between cotton and chlorhexidine. Computer models verified that hydrogen bonding existed between the hydroxyl groups of cotton, and the
Binding of CHXA to cotton depends on the number of carboxylate groups on cotton at lower bulk concentrations and, additionally, on the number of hydroxyl groups on the substrate at higher bulk concentrations. The binding of CHXA to BCD-cotton was dependent on the number of car-boxylate groups on cotton and the hydrophobic interactions of CHXA with BCD molecules (at lower CHXA bulk con-centrations), 19 and additionally with the available hydroxyl groups of BCD-cotton at higher bulk concentrations. The number of the free hydroxyl groups in BCD-cotton were expected to be much less than that of cotton due to some of them being esterified with BTCA.
The adsorption curves of CHXA fixation on BCD-cotton and cotton are shown in Fig. 9. Up to a bulk concentration of 1 g/L, no significant difference was seen between CHXA adsorption on BCD-cotton and cotton (control). The CHXA adsorption on BCD-cotton steadily increases thereafter in comparison with cotton. This increase in adsorption was by ∼1.5 times (at CHXA bulk concentrations of 1-2.5 g/L). Table I shows the numerical values and the differences in adsorption of CHXA on the BCD-fixed samples in comparison to cotton at each bulk concentration.
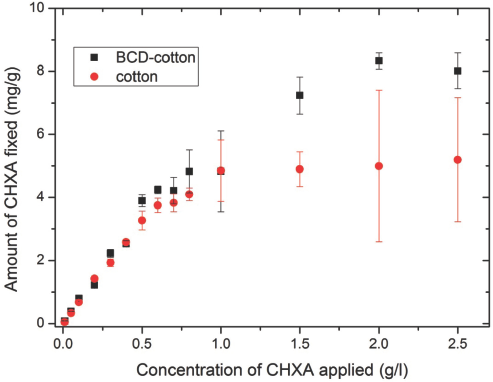
Adsorption values of CHXA (mg/g) for cotton and BCD-treated cotton materials.
Amount of CHXA Fixed on BCD-Cotton and Cotton Control
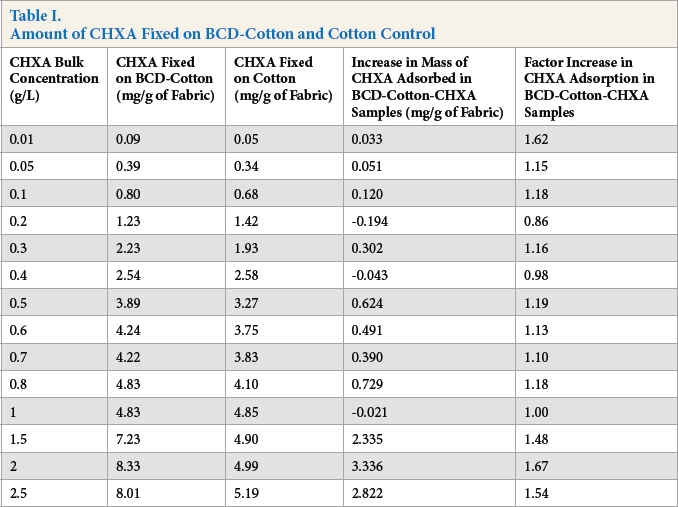
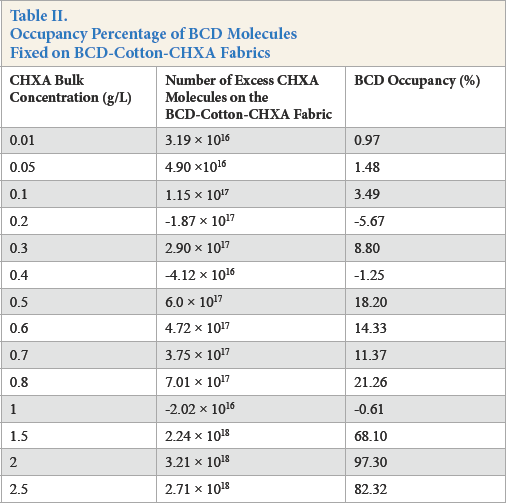
From the difference in the mass of CHXA adsorbed onto BCD-cotton-CHXA samples in comparison with cotton-CHXA samples, the number of CHXA molecules adsorbed per gram of fabric can be calculated. If a 1:1 complexation ratio between BCD and CHXA is considered, then the number of CHXA molecules would equal the number of BCD molecules. As stated earlier, the amount of BCD fixed (and number of BCD molecules) on the sample was also known. The occupancy percentage of BCD molecules (percentage of the fixed BCD molecules on the fabric that are occupied by guest molecules) therefore was further calculated at each CHXA bulk concentration.
It was stated earlier that a complexation ratio between BCD and CHXA was 1:2 in homogenous solutions. This complex-ation ratio does not seem plausible when BCD molecules are immobilized onto the surface of the textile fibers. In this case, the fixed BCD molecules are limited in their movement and orientation and, therefore, limited in ability to complex with guest molecules as compared to free movement in a homogenous solution. Therefore a 1:1 complex was assumed for the calculations of the occupancy percentage (Table II).
Occupancy Percentage of BCD Molecules Fixed on BCD-Cotton-CHXA Fabrics
During the exhaust treatment of the BCD-cotton with CHXA, CHXA showed an affinity towards the carboxylate groups of cotton over complexation with BCD. This is cor-roborated by the small increase in CHXA mass adsorbed onto to BCD-cotton as compared to the cotton control (Table I). As stated previously, complexation of a guest molecule with BCD was not immediate when it was completely solubilized because BCD has little influence in the case of highly-soluble guests. In the same set of experiments, it was also observed that presence of cotton did not influence the onset of complex-ation of CHXA with BCD in any way. It was also concluded that complexation was not influenced by the presence of electrostatic interactions that CHXA and CHXB have with cotton. It can be similarly concluded that the electrostatic interactions between CHXA and cotton and the hydropho-bic interactions between BCD and CHXA were parallel processes and did not significantly interfere with each other under these experimental conditions.
After the saturation of the electrostatic bonds and hydrogen bonds (as observed from the fattening of the curve in Fig. 9 at greater than 1 g/L CHXA concentrations on cotton), CHXA continued to be solely adsorbed onto the BCD-cot-ton fabrics (i.e., into the BCD cavities on the substrate) via the only prevailing forces present, which were the hydro-phobic interactions between CHXA and the BCD cavities. It can be observed that CHXA adsorption did not significantly increase with the CHXA bulk concentration (nor did the occupancy percentage) at this later stage. This was due to the limitation in the number of cavities available for further complexation. This can be seen in the high occupancy percentage of BCD molecules at these bulk concentrations. From Table II, it appears that over 80% of BCD fixed on the cotton fabric was occupied by CHXA.
The two assumptions in this work are, first, that the com-plexation ratio between the immobilized BCD molecule and the CHXA molecule was 1:1 (while in solutions it was 1:2 or greater). In reality, it may be a mixture (if a BCD to CHXA complexation ratio of 1:2 was considered, then the occupancy percentage would be double the values present in Table II). The second is that calculations pertaining to the occupancy percentage of BCD cavities assumes that all of the excess CHXA molecules contributed to the presence of BCD molecules were complexed within the BCD cavity. It is possible that some of the guest molecules were situated between the various BCD cavities (called non-inclusion complexes) within the BTCA network.
Antibacterial Activities
Antibacterial experiments were done on cotton-CHXA and BCD-cotton-CHXA fabrics. All the fabrics were treated with a CHXA bulk concentrations of 0.1 g/L to 1 g/L. A range of antibacterial activity were seen only between these application concentrations.
The results of the antibacterial tests are presented in Fig. 10. This figure shows the amount of live bacteria (CFUs) eluted from the fabrics after the required incubation time period. As expected, there was a decrease in the log CFUs with the increasing CHXA bulk concentration. The amount of CHXA fixation on BCD-cotton-CHXA was increased by a small amount in most of the treatments up to 1 g/L of CHXA bulk concentration (Tables I and II). However, the number of live bacteria extracted from BCD-cotton-CHXA fabrics was consistently greater than the number eluted from cotton-CHXA fabrics for treatments at the same bulk concentration.
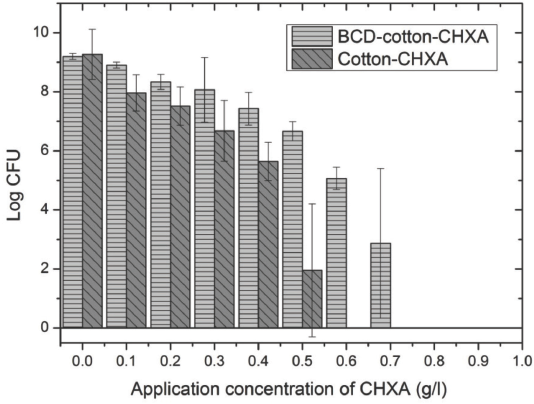
Amount of bacteria eluted (in Log CFU) of various CHXA treatments on plain cotton in comparison with BCD-treated cotton.
Antibacterial activity (AA) was calculated using Eq. 2. Table III shows the AA values calculated for various CHXA bulk concentrations (g/L).
Antibacterial Activity of CHXA-Treated Textiles
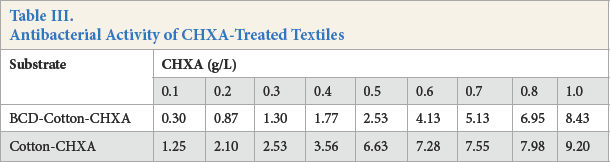
An AA value of 2 is considered to be a requirement for a textile to be considered antibacterial. 20 An AA of 2 was achieved at 0.2 g/L CHXA application concentration for the cotton-CHXA sample. The same was achieved at 0.5 g/L CHXA application concentration for BCD-cotton-CHXA samples and BCD-APES-CHXA samples. As explained earlier, this can be attributed to the slow release of CHXA from BCD complex. Other studies have also reported this reduced antibacterial activity of BCD-antibacterial agent complexes.21–23 The slow release phenomenon is known to be dependent on the binding constant between the guest molecule and the BCD molecule. 24
The slow release phenomenon of BCD treated cotton was proven with the simple CHXA release experiment described earlier. From Fig. 9, it is already known that the amount of CHXA adsorbed by the BCD-cotton-CHXA sample treated at 1.5 g/L of CHXA was 1.5× that of the cotton-CHXA sample treated with the same bulk concentration.
As seen in Fig. 11, the BCD-cotton-CHXA sample released CHXA into water at a much lower rate than the cotton-CHXA sample. This lower rate of release of CHXA from the BCD-cotton samples supports the theory that the lower antibacterial activity of the BCD-cotton-CHXA samples was due to the slow release of CHXA complexed within the BCD cavities. It is also possible that the BTCA network within the BCD-cotton hinders the release of CHXA from the fabric to an extent. However, if this was the case, the crosslinking would have inhibited or hindered the adsorption of the CHXA onto the BCD-cotton as well. The slow release phenomenon can further be confirmed by antibac-terial tests of the homogenous BCD-CHXA solutions in comparison with CHXA solutions. However, diffusion (of the agent from the fabric) plays a role in heterogeneous tests and this is absent in the homogenous tests.
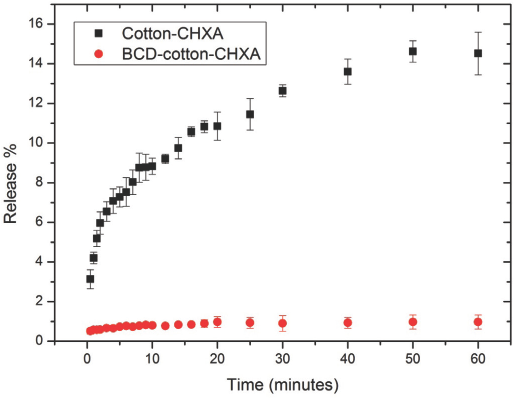
Release percentage of CHXA for cotton-CHXA and BCD-cotton-CHXA samples.
The sustained or prolonged release of an antibacterial agent from the BCD cavity could be advantageous in applications such as wound healing dressings. Optimal wound healing requires dressings with sustained release of the antibacterial agent to prevent cell cytotoxicity and skin sensitivity issues (both of which are associated with quick release of anti-bacterial agents in such dressings). 25 Such slow release also contributes to prolonging the shelf life of the fabrics.
Conclusion
Unmodified or native β-cyclodextrin (BCD) was success-fully attached to cotton via crosslinking with 1,2,3,4-butane tetracarboxylic acid (BTCA). The amount of BCD fixed on the various samples was determined using the phenol-phthalein method. These samples were then treated with an antimicrobial agent. Chlorhexidine base (CHXB) and chlorhexidine diacetate (CHXA) were considered for use in the antimicrobial functionalization step as guest molecules. UV-Vis spectrophotometric measurements were done with CHXB-BCD and CHXA-BCD solutions to study the differences in the BCD concentrations required for the onset of complexation with two guest molecules and BCD. The onset of complexation started only after the addition of a molar excess of BCD of about 40× that of CHXA/CHXB. The complexation pattern of CHXA with BCD appeared identical to that of CHXB with BCD despite the differences in their solubility and polarity. This was explained by the fact that the guest molecules were entirely solubilized in the water prior to the spectral study. Complexation of a guest molecule with BCD cavities was expected to be immediate when the guest was added to the solvent above its solubility limit.
CHXA was chosen to functionalize cotton and BCD attached to cotton (BCD-cotton). This choice was made based on its higher water solubility (as compared to CHXB), which would allow the use of a wide (and higher) range of bulk concentration treatments in aqueous media. Antibac-terial activity tests were done on the treated fabrics against
