Abstract
The aim of this study was to determine whether dendrimer-based products can replace classic fluorocarbon-based products in water repellent, woven cotton fabrics. Furthermore, the effect of adding a dendrimer to a fluorocarbon product on the resulting water and oil repellency was studied. For that purpose, fluorocarbon, dendrimer, and dendrimer-fluorocarbon mixture-based commercial products were applied to cotton fabrics using the pad-dry-cure method. Repellency performance and wash resistance of these chemicals were investigated. It was found that the same water repellency values could be obtained in lower concentrations or under milder curing conditions using the dendrimer-fluorocarbon mixture-based product compared to that of the fluorocarbon-based product.
Introduction
Today, people expect their clothes to provide additional functions besides simply covering and beautifying. Consumers frequently desire water and oil repellency.
Historically, the major share of the repellency market was held by fluorocarbon (FC) compounds. The efficiency of FC compounds is due to the structure of the bond between the F and C atoms. While the length of a C-F bond is 1.35 Å, that of a C-C bond is 1.54 Å. Because a C-F bond is shorter, the movement of fluorinated alkyl groups is less, as the C-F bond is a stronger bond than a C-C bond. Tis causes fluorocarbon com pounds to have a very low boundary surface tension. 1 Fluoropolymers owe their special properties to their low surface energy, which means that they not only repel water but also oil-based substances. Fluoropolymers suited for textile finishing generally consist of a polymeric backbone (e.g., acrylate or polyurethane) with fluorinated side chains. Their effectiveness for each application varies with chain length, the shape of the chain, and the type of fluorinated side chain end groups. 2 Conventional fluorocarbons (C8) are polymers that consist of 8 carbon and 17 fluorine atoms. Tese C8 fluorocarbons release the bio-persistent and toxic component perfluo-rooctane sulfonate (PFOS) and perfluorooctanoic acid (PFOA). 3 PFOS has very serious effects on health and the environment, consequently, PFOS and related substances are being phased out. To replace C8 fluorocarbons, several manufacturers have moved towards other fluorinated compounds that have similar technical properties. 4 After extensive R&D efforts, chemists have succeeded in replacing C8 chemistry with C6 (i.e., six fluorinated carbon chain) chemistry, 3 which is classed as non-bioaccumulative, since C6 fluorocarbons are more easily broken down in the environment than their C8 counterparts. 2
A recent approach to water repellency is based on the use of dendrimer nanoparticles. Dendrimer, having derived from the words “dendron” (tree) and “meros” (part) in Greek, means tree-like branched compounds. 5 Dendrimeric chemistry was developed as a spinoff of conventional polymer chemistry. Dendrimers are gaining importance due to their versatile applications in materials science. In contrast to linear polymers, dendrimers are highly branched, fractallike macromolecules of well-defined, three-dimensional structure, shape, and topology. 6 Their structure is built of branch cells, which include a core, interior cells, and surface cells with chemically reactive or inert terminal end groups. 7 Many dendrimer chemical and physical properties, such as state of aggregation, reactivity, stability, and solubility are closely related to the nature of the end groups. 8 For example, dendrimers terminating in hydrophilic groups are soluble in polar solvents, while dendrimers having hydrophobic end groups are soluble in non-polar solvents. 9
The repellency effect of dendrimers is based on their forming nano-sized crystalline structures that impart wash resistance through water repellency and high abrasion resistance. In her study, Sancaktaroğlu investigated changes in water and oil repellency values after treating cotton and cot-ton/polyester fabrics with products containing a dendrimer, a fluorocarbon, and a dendrimer-fluorocarbon combination. The dendrimer-fluorocarbon treatment gave the best oil and water repellency effect. 10
Atav et al. researched the effects of dendrimer, fluorocar-bon, and dendrimer-fluorocarbon product treatments on the color of reactive dyed, knitted cotton fabrics. Water and oil repellencies of these commercial product-treated fabrics were also compared. The effects of all three products on color change were acceptable and the dendrimer-fluorocarbon product treatment was the most effective in terms of water and oil repellency. 11
Namlιgöz et al. applied polymeric dendrimer-containing fluorocarbon, nano-sized fluorocarbon polymer, nano-silica acid, and conventional agents (e.g., a paraffin emulsion containing zirconium salt and a conventional fluorocarbon compound) to cotton fabrics. After treatments, the results were compared. In general, the water, oil, and stain repel-lency results showed that the dendrimer-fluorocarbon combination gave the best results. 12
Colleoni et al. treated cotton/polyester fabrics with den-drimer, dendrimer-fluorocarbon, and fluorocarbon compounds. The best water and oil repellency values were obtained using the dendrimer-fluorocarbon treatment. 13
Minimizing the amount of fluorocarbons used in fabric treatments would be environmentally beneficial. 2 Consequently, the aim of this study was to determine whether dendrimer-based products could replace classic fluorocar-bon-based treatments for producing water repellent, undyed woven cotton fabrics. The effect of adding a dendrimer into a fluorocarbon product on the resulting water and oil repel-lency and its permanence was also studied.
Dendrimer, fluorocarbon, and dendrimer-fluorocarbon products were applied to undyed woven cotton fabrics using the pad-dry-cure method. Water and oil repellency performance and wash resistance of these treated fabrics were investigated. Although some literature studies of these treatments are available, there is no comprehensive study of undyed woven cotton fabric where optimized application conditions (e.g., concentration, fixation tem-perature, and time) of these products were determined. The resistance of fabric water and oil repellency to successive washings, with and without ironing between each wash cycle, was determined according to ISO 4920 and ISO 14419 standards, respectively.
Experimental
Fabric
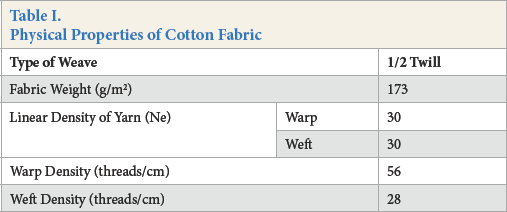
All experiments were carried out with desized, scoured, and bleached 100% cotton woven fabric. Cotton fabric was kindly supplied by Söktaş-Turkey. The physical properties of the fabric are given in Table I.
Physical Properties of Cotton Fabric
Chemicals
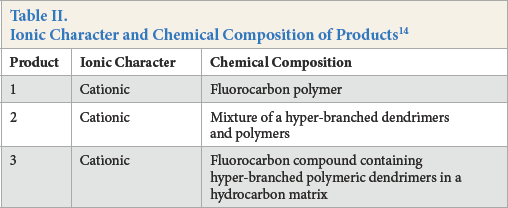
In this study dendrimer, fluorocarbon, and dendrimer-fluorocarbon mixture-based commercial products were used. All of them were kindly supplied by Rudolf Chemie. In Table II, ionic characters and chemical compositions of these products are listed.
Ionic Character and Chemical Composition of Products 14
Finishing Treatment
Cotton fabrics were impregnated with each of the three different finishing chemicals on a laboratory-scale padding machine, model FY350, made by Ataç. Aqueous solutions (adjusted to pH 5-5.5 with acetic acid) of these products were applied to cotton fabrics at three different concentrations (20, 40, and 60 g/L) with a pickup value of 80%. After impregnation, cotton fabrics were dried (85 °C for 4 min.) and subsequently cured on a laboratory stent er, model GK40, made by Ataç. To determine the optimal curing conditions for each product, the curing process was carried out at three different temperatures (140 °C, 150 °C, and 160 °C) and three different times (1, 2, and 3 min).
After the application processes were completed, the samples were divided into four groups. The first group of fabrics were not washed, while the second, third, and fourth groups were washed 1, 5, and 10 times, respectively. In practice, users generally iron the textile products after each washing before using it again. For this reason, successive washings were split into two groups: those ironed and those not ironed after each wash cycle. The laundering process was carried out according to BS EN ISO 26330 standard. Each wash step was done using 4 g/L of soap at 30 °C for 40 min, then all samples were dried.
Testing
After conditioning the fabrics for 24 h under standard atmospheric conditions (temperature: 20 °C ± 2 °C, relative humidity (RH): 65% ± 2%), water and oil repellency tests of fabric samples were carried out according to ISO 4920 (spray test) and ISO 14419 standards respectively. For water repellency tests, the SPR01 model (Ataç) test device was used. Optimal conditions for each product were determined from the water and oil repellency tests. Samples that had been treated under these optimal conditions were subjected to Martindale abrasion tests (TSE EN ISO 12947-1 standard) by using an ATC-MD04-06-09 model (Ataç) test device.
Analysis
Scanning electron microscopy (SEM) analysis of the optimally treated and untreated samples was performed. Tese images provided a visual assessment of the resulting repellency treatment process on the surface structure of the treated cotton fibers. A JSM-6060LV scanning electron microscope (JEOL) was used for imaging the fabric samples at 2500× magnification with accelerating voltage of 5 k V. To determine the changes in chemical structure of cotton fiber after application of water and/or oil repellency products, Fourier transform infrared (FTIR) spectra of treated (under optimal conditions) and untreated samples were recorded over the 650 to 4000 cm−1 range on a Spectrum 100 ATR-FTIR spectrometer (Perkin Elmer).
Results and Discussion
Water Repellency Tests
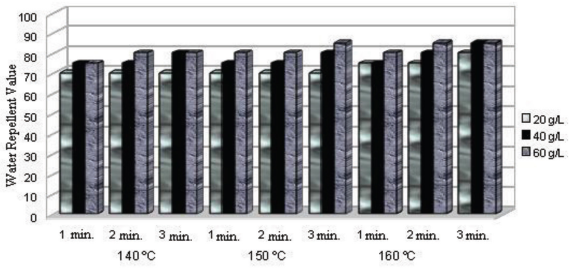
The results of water repellency tests related to the fluoro-carbon, dendrimer, and dendrimer-fluorocarbon fabric treatments are given in Figs. 1–3.

Water repellency test results for fluorocarbon treated fabric under various processing conditions.
Water repellency test results for dendrimer treated fabric under various processing conditions.

Water repellency test results for dendrimer-fluorocarbon treated fabric under various processing conditions.
The products giving the greatest water repellency after fabric treatment, from the most effective to the least effective, were dendrimer-fluorocarbon > fluorocarbon > dendrimer. For the fluorocarbon-based water repellency agent, optimal conditions were 20 g/L concentration and 1 min curing at 150 °C. Increasing the curing temperature and time did not provide a significant improvement in water repellency value (Fig. 1). A value of 80 (out of a 100 maximum) was achieved using a 20 g/L concentration and 3 min curing at 160 °C for the dendrimer fabric treatment. However, the water repellency value was still less than that for the fluorocarbon treatment.
When the dendrimer-fluorocarbon product was used for treatment, a repellency value of 100 was obtained even at a low concentration (20 g/L), curing temperature (140 °C), and time (1 min). In the presence of the dendrimer, the orientation of fluorocarbon chains on the fabric surface increased. 10 Tis is why the same effect was obtained using the dendrimer-fluorocarbon product treatment under milder curing conditions compared to use of the product treatment containing only fluorocarbon.
Not only are the initial water repellency features of a textile product important to consumers, but also the resistance of treatments to successive laundering. For this reason, the water repellency values of samples were also tested after 1, 5, and 10 washings (Figs. 4–6).
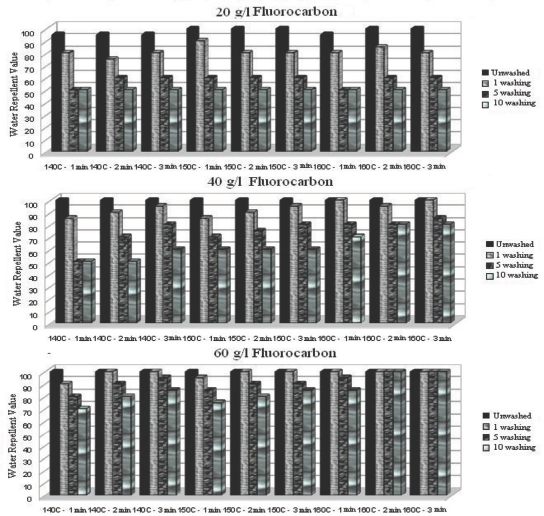
Water repellency values after 1, 5, and 10 washings of fluorocarbon treated fabric under various conditions.
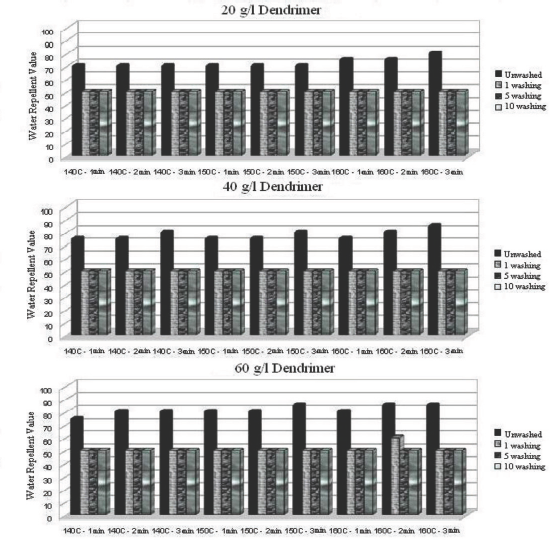
Water repellency values after 1, 5, and 10 washings of dendrimer treated fabric under various conditions.
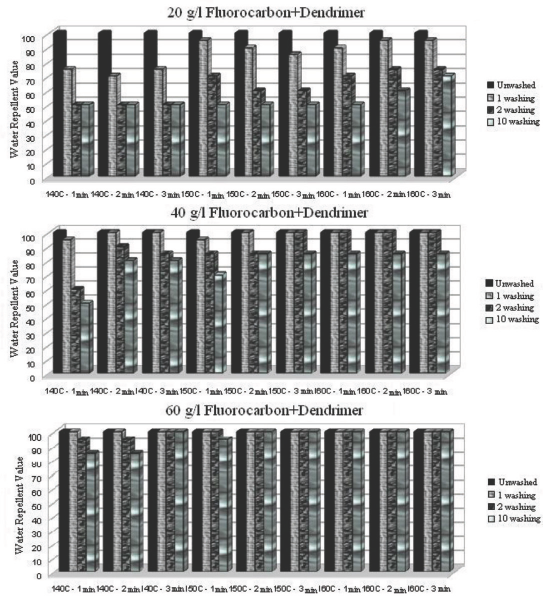
Water repellency values after 1, 5, and 10 washings of dendrimer- fluorocarbon treated fabric under various conditions.
Water repellency values of fluorocarbon treated fabric decreased gradually with increased number of wash cycles (Fig. 4). Although adequate curing is provided, textile products treated with fluorocarbon finishings are generally exposed to a large loss of water repellency after washing. 13 The reason for this is that fluorocarbon polymers are subjected to unfavorable wash conditions. Surface-active agents, and especially water (that provides a polar medium), together with temperature and mechanical effects lead to loss of fluorocarbon chain orientation, with the chains becoming buried in the material's surface away from their edges. 13 This explains why water repellency decreases with an increase in the number of wash cycles when fluorocarbon treatment is used.
For the dendrimer treatment, a sharp decrease occurred after the first wash cycle, but water repellency values were not affected by further successive wash cycles (Fig. 5). On the other hand, when the dendrimer-fluorocarbon treatment was used at a 40 g/L concentration, much smaller decreases in water repel-lency occurred as the number of wash cycles increased (Fig. 6). Furthermore, when a 60 g/L concentration of the dendrimer-fluorocarbon treatment was used under proper treatment conditions, a water repellency of 100 was achieved and no water repellency was lost even after 10 washings.
Optimal application conditions (concentration, curing temperature, and time) of fluorocarbon and dendrimer-fluo-rocarbon treatments were 60 g/L at 160 °C for 2 min, and 60 g/L at 140 °C for 3 min, respectively, if the water repellency value after the first wash was to be maintained after 10 successive wash cycles (Figs. 4–5).
Successive washings were also performed with ironing applied after each wash cycle. Tese results are given in Figs. 7–9.
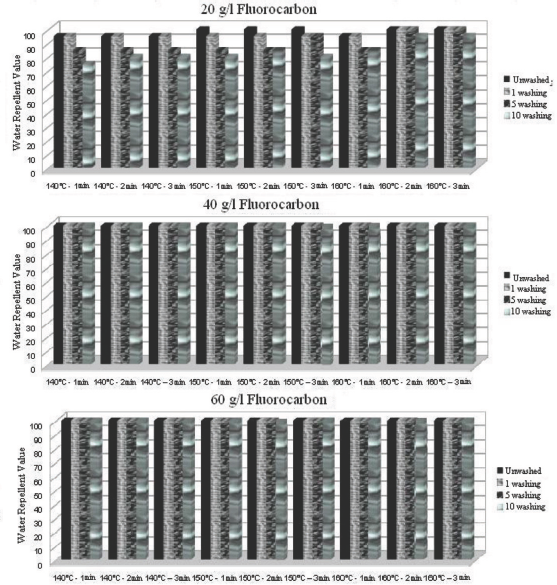
Water repellency values after 1, 5, and 10 washings of ironed (after each wash cycle) fluorocarbon treated fabric under various conditions.
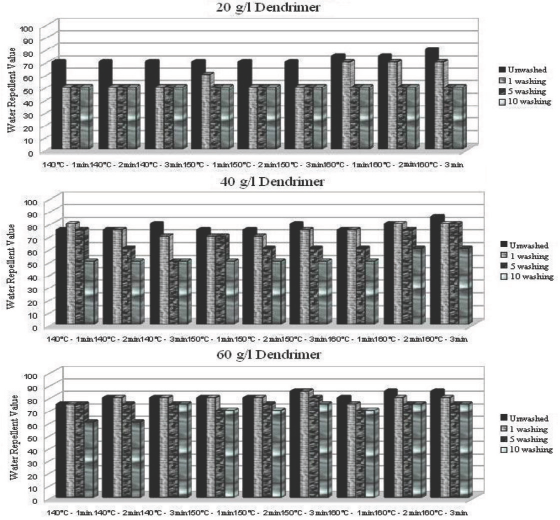
Water repellency values after 1, 5, and 10 washings of ironed (after each wash cycle) dendrimer treated fabric under various conditions.
Water repellency values after 1, 5, and 10 washings of ironed (after each wash cycle) dendrimer-fluorocarbon treated fabric under various conditions.
For all three fabric treatments, the water repellency values obtained after successive washings with ironing in between each wash cycle were greater than the ones obtained without ironing (Figs. 7–9). The ironing process, in which the textile material is heated over the melting temperature of the fluorocarbon chains, is sufficient to reorient fluorocarbon chains and recover the fabric's previous water repellency levels. 15
Ironing between each successive wash cycle for fabrics treated with optimally applied fluorocarbon (40 g/L at 140 °C for 1 min) and dendrimer-fluorocarbon (20 g/L at 140 °C for 1 min) maintained water repellency values of 100 even after 10 successive wash cycles (Figs. 7 and 9). Tese results indicate that the use of less finishing product on the fabric maintained the same water repellency values for up to 10 wash cycles if ironing was applied between each successive wash cycle. Additionally, a water repellency value of 75 after 10 washes could be obtained with the dendrimer treated fabric if the treatment was applied at a 60 g/L concentration.
Oil Repellency Tests
Results showed that the dendrimer treated fabric did not demonstrate an oil repellency effect. Tis might be due to the higher critical surface energy of the dendrimer product in comparison with oil molecules (20-35 mN/m). 10 The results of oil repellency tests on the fluorocarbon and dendrimer-fluorocarbon treated fabrics are given in Figs. 10 and 11.
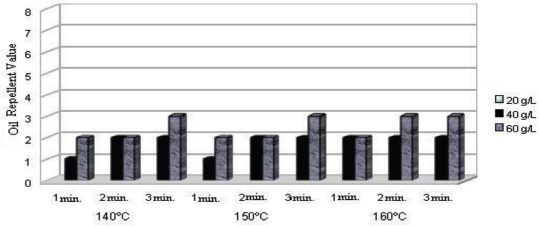
Oil repellency test results for fluorocarbon treated fabric under various processing conditions.
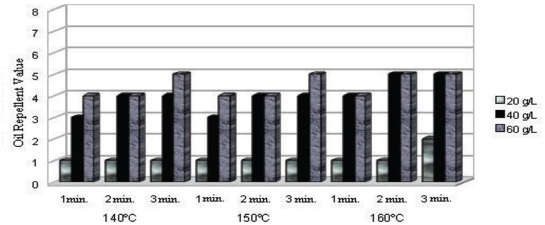
Oil repellency test for dendrimer-fluorocarbon treated fabric under various processing conditions.
Higher oil repellency values were obtained for dendrimer-fluorocarbon treated fabric compared to fluorocarbon treated fabric. The optimal oil repellency value obtained for the fluo-rocarbon treated (40 g/L, 1 min curing at 140 °C) fabric was 2. The corresponding value for dendrimer-fluorocarbon treated (40 g/L, 1 min curing at 140 °C) fabric was 4. When combined with fluoropolymers, dendrimers force them to co-crystallize leading to self-organization of the whole system and an enrichment of the fluoro polymers on the outermost textile l a y e r. 13 Tis could be the reason why a higher oil repellency value was obtained for the dendrimer-fluorocarbon treated fabric than for the fluorocarbon treated fabric.
According to these results, the optimal concentration of both products used on the fabric was 40 g/L. Therefore, the resistance of oil repellency to successive washings was made on fabric treated with a 40 g/L concentration of each of the two products. The results can be seen in Figs. 12 and 13.
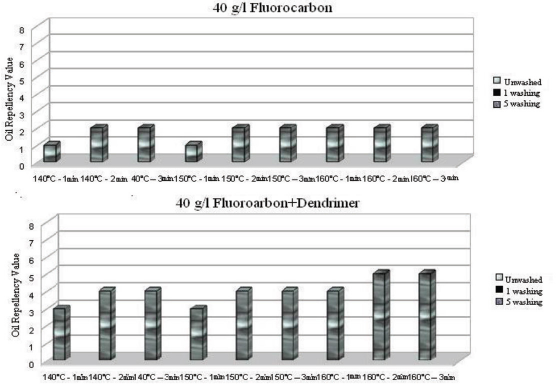
Oil repellency values after 1 and 5 washings of fluorocarbon and dendrimer-fluorocarbon treated fabrics under various curing conditions.
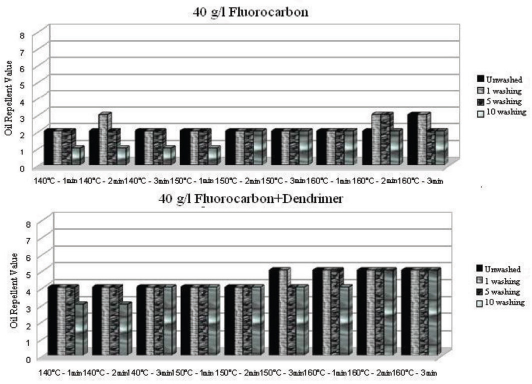
Oil repellency values after 1, 5, and 10 washings of ironed (after each wash cycle) fluorocarbon and dendrimer-fluorocarbon treated fabrics under various curing conditions.
For both treatments used, the oil repellency effect on the treated fabric completely disappeared regardless of curing conditions even after the first wash cycle (Fig. 12). When ironing was applied between successive wash cycles, the oil repellency values remained at the initial value even after 10 washings, due to re-orientation of the fluorocarbon chains during heat application from ironing. 15
Martindale Abrasion Tests
To determine water and oil repellency treatment resistance to abrasion, fabric samples that were treated under optimal conditions were subjected to the Martindale abrasion test. Results are given in Figs. 14 and 15.
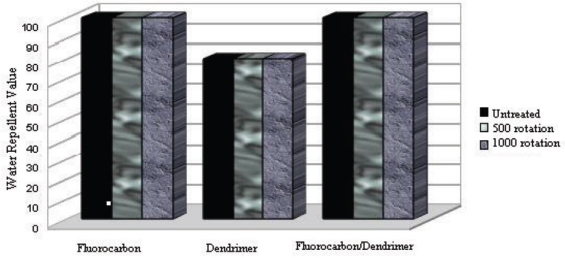
Change in water repellency values after abrasion tests.
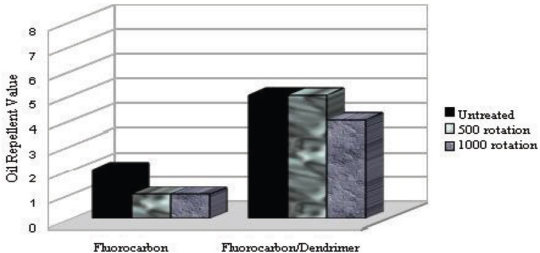
Change in oil repellency values after abrasion tests.
Water repellency values were stable, even after 1000 rotations of abrasion, for fabrics treated with all three products (Fig. 14). Oil repellency values for fabric treated with the fluorocarbon product decreased from 2 to 1 after 500 rotations (Fig. 15). In comparison, fabric treated with dendrimer-fluorocarbon product maintained the initial oil repellency value of 5 even after 500 rotations, but after 1000 rotations, it decreased to 4. Tese results demonstrate that resistance of water and oil repellency to abrasion was greater for dendrimer-fluorocarbon treated fabric than for the other treated fabrics.
SEM Analysis
Results of SEM analysis of treated and untreated cotton fabrics are given in Fig. 16. No significant difference was observed between untreated and treated samples. According to these results, it can be said that the physical structure of the cotton fiber was nearly unaffected by water and/or oil repellency treatments.

SEM photographs of cotton fabrics (a) untreated, treated with (b) fluorocarbon, (c) dendrimer, and (d) dendrimer-fluorocarbon based commercial products.
FTIR Analysis
Results of FTIR analysis of treated and untreated cotton fabrics are given in Figs. 17–20. Fabrics treated with fluorocarbon and dendrimer-fluorocarbon products showed peaks at 1260 cm−1 and 1739 cm−1, which can be attributed to C-F and C=CF2 bands, respectively. The results obtained here were very similar to those carried out by Namlιgöz et al. 12
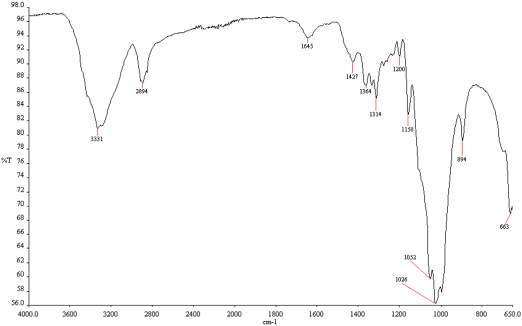
FTIR spectrum of untreated cotton fabric.
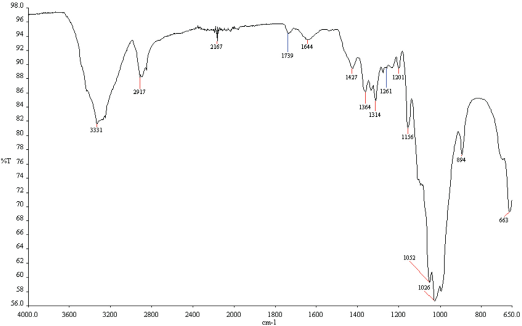
FTIR spectrum of cotton fabric treated with fluorocarbon based commercial product.
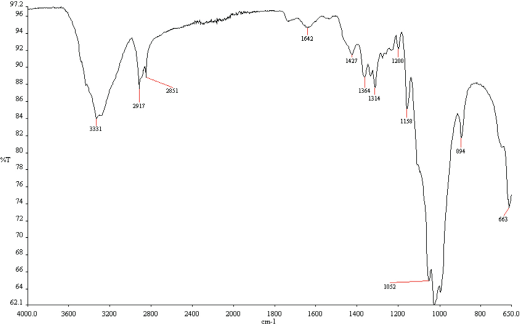
FTIR spectrum of cotton fabric treated with dendrimer based commercial product.
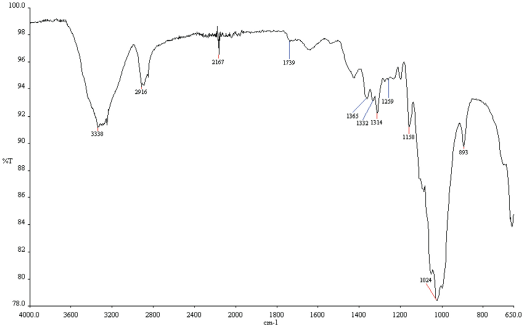
FTIR spectrum of cotton fabric treated with dendrimer-fluorocarbon mixture based commercial product.
Conclusion
The water repellency of the treated cotton fabrics tested in this study gave the following results (from most effective to least effective): dendrimer-fluorocarbon treated > fluorocarbon treated > dendrimer treated. The optimal application conditions (i.e., concentration, curing temperature, and time) of the fluorocarbon and dendrimer-fluorocarbon treated fabrics were 60 g/L of treatment agent at 160 °C for 2 min and 60 g/L of treatment agent at 140 °C for 3 min, respectively. Tese treated fabrics gave water repellency values of 100 that could be maintained even after 10 successive wash cycles. The dendrimer treated fabric gave a water repel-lency value of 50 after 10 washings after 60 g/L of the treatment agent was applied.
For the dendrimer-fluorocarbon treated fabric, less treatment agent (40 g/L, instead of 60 g/L) was suf-ficient for obtaining high water repellency values resistant to 10 wash cycles if ironing was applied between each successive wash cycle.
The dendrimer treated fabric did not demonstrate an oil repellency effect in this study. When the oil repellency performances of fluorocarbon and dendrimer-fluorocarbon treated cotton fabrics were compared, higher oil repellency values were observed for the dendrimer-fluorocarbon treated fabric compared to the fluorocarbon treated fabric. An optimal value obtained for the fluorocarbon treated (40 g/L treatment agent, with 1 min curing at 140 °C) fabric was 2. For the dendrimer-fluorocarbon treated (40 g/L treatment agent, with 1 min curing at 140 °C) fabric, an oil repellency value of 4 was obtained.
The results of this study are of interest to a textile industry seeking alternatives to fluorocarbon use for imparting water and oil repellency to fabrics. In combining fluorocarbons with dendrimers, it would be possible to reduce the amount of fluorocarbons used and/or the curing temperature. Den-drimer-fluorocarbon mixture-based products could take the place of conventional fluorocarbon-based products in both water and oil repellent finishes as economic and ecological considerations are increasingly gaining importance in today's competitive market.
Footnotes
Acknowledgements
We would like to thank Namık Kemal University for supporting this study within NKÜBAP.00.17.YL.10.30 coded Msc project. Furthermore we kindly would like to express our gratitude to Çerkezköy Türk Tekstil Vakfı Mesleki ve Teknik Eğitim Merkezi for giving us the opportunity to use their laboratory facilities.
