Abstract
A spirooxazine-based photochromic dye was encapsulated by an oil-in-water emulsion, solvent evaporation method. The encapsulated dye was applied to cotton fabric by a pad-cure process with different binder types. Ultraviolet (UV) protection was increased with application of the encapsulated photochromic dye. The type of binder used affected the photochromic color build-up during UV irradiation and could modify the UV protection imparted by the encapsulated photochromic dyes on textiles.
Introduction
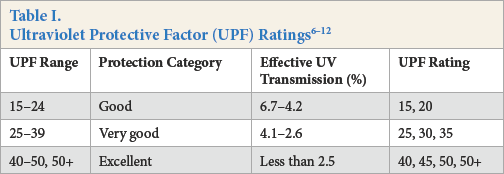
The penetration of ultraviolet (UV) irradiation leads to damage in human skin.1–3 Tese adverse effects began to increase gradually due to ozone layer depletion. Tus, the development and production of UV-protective textile materials should be pursued, since clothing is considered to be a significant step for UV protection. The UV-protection rating of textile materials is characterized by the UV Protection Factor (UPF), which is defined as “the ratio of the average effective UV irradiance calculated for unprotected skin to the average effective UV irradiance calculated for skin protected by the test fabric.”4–6 Various UPF ratings according to commonly-used standards are given in Table I.6–12
The first standard developed for UV-protection measurement was Australia/New Zealand (AS/NZ) standard 4399:19961, followed by AATCC Test Method (TM) 183-2004, ASTM D6544, and ASTM D6603 in the United States, and EN 13758-1 in Europe. Tese standards vary according to the scanning range, the position of the fabric on the device, and the erythemal action spectrum and clas-sification. For example, if the fabrics have a UPF rating of greater than 50, EN 13758-1 reports them as > 50, whereas ASTM D6603 and AS/NZS4399 report them as 50+.
The fabrics must be conditioned at a specified temperature and humidity before testing according to EN 13758-1 (and BS 7914), however AS/NZS 4399 does not require any con-ditioning. ASTM D6603 indicates that the samples should be conditioned with laundering, UV exposure, and chlorinated pool water equivalent to two years of normal use. EN 13758-1 and AATCC TM 183 specify the measurements made when the fabrics are wet and/or stretched whereas AS/NZS 4399 currently specifies testing in the dry and relaxed state only. ASTM D6603 specifies both a numeric UPF value and a description of the garment as providing good, very good, or excellent UV protection based on UPF values of 15–24, 25–39, and > 40, respectively.9–13
Recently, increased public awareness of the harmful risks of UV exposure generated demands for the use of UV-protective clothing. 14 For instance, the World Health Organization (WHO) reported that clothing and hats provide the best protection, in addition to use of sunscreens, for parts of the body that remain exposed.15,16 However, the size of UV-protective clothing market is uncertain. 14 Moreover, there are no governmental regulations for sun-protective clothing, textiles, or accessories. 17
Besides fabric constructional parameters, color is one of the most important parameters that influence UV-protection by wearing textiles.4–8 Color can have a considerable effect on fabrics’ UV protection properties. Tis protection depends on the UV absorption bands of dyes and their concentration on the fabric. 5 In general, darker colors provide better UV protection, however the use of dark-colored garments during the summer is not generally preferred. Tus, UV absorbers can be applied onto light-colored textile materials to impart UV protection.
A special group of dyes, called photochromic dyes, can also absorb UV radiation. Photochromism is defined as “the absorption spectrum change of a molecule or crystal [revers-ibly] when the sample is irradiated by UV light.”18,19 Tese dyes appear colorless until UV irradiation changes their structure into a colored material. They then return to the colorless form structure when the UV irradiation source is removed. Typical examples of photochromic dyes are spiroox-azines, spiropyrans, naphtopyrans, and chromenes. 18 Detailed information on the chemical structures and mechanisms of photochromic dye reactions can be found elsewhere.18–22
Photochromic dyes have been used in sunglasses and ophthalmic lenses, 23 cosmetics, 24 and apparel. 25 Fashion products, such as color changing t-shirts (e.g., SolarActive, 26 De-Facto, 27 Del Sol, 28 and Solartees 29 ) and SolarActive slippers, have appeared in the clothing market. It was also reported that photochromic dyes have a UV-protection function since they absorb UV radiation.19,30 However, there are no studies on the UV-protective properties of photochro-mic dyes, although the application of these dyes on textiles has been studied by a number of authors.30–38
Photochromic dyes can be applied to textiles by several techniques. For example, they can be blended with fiber spinning solutions or dope-dyed. Coating or other traditional dyeing methods are additional application options.39,40 However, it was reported that incorporation of photochromic dyes into a rigid matrix (i.e., between micelles of fibers or a coating resin) may restrict the configurational change of these dye molecules. 30 Application of photochromic dyes onto textile materials, especially to natural fibers, is problematic because they are neutral molecules with a balance of hydrophilic and hydrophobic properties similar to conventional disperse dyes. 35 Additionally, insolubility in water, low heat resistance, and poor washfastness are other disadvantages. To overcome these problems, photochromic dyes for textiles are often produced in encapsulated form. Microencapsulation helps to improve the application and durability properties of these dyes and protects them from environmental influences. 41
In this study, the application performance of encapsulated photochromic dyes on cotton fabrics was investigated. For this purpose, the pad-cure process was selected and binders available on the market were used. Color build-up on irra-diation properties, durability, and UV-protective properties were assessed for use of photochromic dyes in the development of UV-protective textile materials.
Experimental
Materials
Dichloromethane, polyvinyl alcohol (product #P8136), and ethyl cellulose (product #200646) were purchased from Sigma Aldrich. A spirooxazine-based photochromic dye, 5-chloro-1,3-dihydro-1,3,3-trimethylspiro[2H-indole-2,3′-(3H)naphth[2,1-b](1,4)oxazine] (product #322555) supplied by Sigma Aldrich was used. The chemical structure of the photochromic dye is shown in Fig. 1.
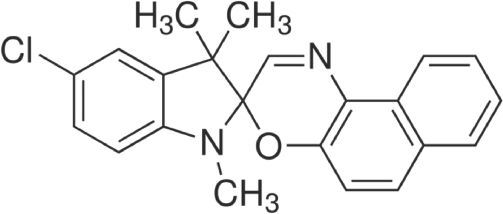
Chemical structure of 5-chloro-1,3-dihydro-1,3,3-trimethylspiro[2H-indole-2,3′-(3H)naphth[2,1-b](1,4)oxazine]. 42
A twill woven cotton fabric (290 g/m2, 59 yarn/cm warp density, 28 yarn/cm weft density, and scoured and bleached) was used in this study. Synthetic thickener (Tubivis DL-600), acrylic-based binder (Tubiprint CH 450), styrene/acrylic based binder (Tubifast AS GKB), and polyurethane-based binder (Tubicoat PUS) were supplied by CHT.
Dye Encapsulation
The spirooxazine-based photochromic dye was encapsulated with ethyl cellulose using an oil-in-water emulsion, solvent evaporation method. Ethyl cellulose (70 mg) and photochro-mic dye (30 mg) were dissolved in dichloromethane and the oil-in-water emulsion was prepared by adding this organic phase into a 1% poly(vinyl alcohol) solution. The emulsion was then sonicated with a probe sonicator at 70 W for 60 s in an ice bath. The dichloromethane evaporated during magnetic stirring for 2 h at room temperature (RT). The size of the capsules was around 220 nm. 43
Pad-Cure Process
Encapsulated photochromic dyes were applied to the fabric by the pad-cure process. Padding was carried out by a Rapid Labortex laboratory-scale padder. The wet pickup ratio was set to 100%. Tree different types of binders (acrylic, styrene/acrylic, and polyurethane based) were used with a binder concentration of 50 g/L. Encapsulated photochromic dye concentrations of 0.5, 1, and 2 g/L were selected. After padding, samples were dried at 80 °C for 3 min and cured at 150 °C for 5 min in a laboratory-scale tenter (Ataç, GK 40). Tree repetitions were carried out for each type of experiment.
Consecutive laundry washings were applied in a Wascator washing machine for 5 and 10 cycles using the TS 5720 EN ISO 6330 standard for testing application durability. Laun-dering processes were carried out at 40 °C for 1 h with 60 g of washing agent, followed by drying at RT.
Characterization and Performance Testing
Color measurements were carried out according to the CIELab color space using a portable spectrophotometer (Gre-tag Macbeth Eye-One) in a UV2/PCR UV cabinet (UVP) with a UV light bulb (USHIO G25T8 UVA type bulb, 25 W, maximum wavelength 365 nm) (Fig. 2). The samples were placed into the UV cabinet (Fig. 2a) and irradiated with UV light for 2 min (Fig. 2b). After irradiation, the UV light was switched of and color measurement were carried out as immediately as possible (∼3 s) (Fig. 2c–d). The color value of untreated sample that was not irradiated was regarded as the standard. Color build-up during sample irradiation was determined by the difference between the standard and irradiated colored samples by using L* (lightness/darkness of the color) and DE (color difference) values.

Color measurement equipment: (a) UV cabinet; (b) UV bulb, and (c) and (d) portable spectrophotometer.
To test the color build-up rate (maximum color formation time), samples were exposed to UV light for a certain time and color measurements were carried out immediately after the removal of the UV light.
Color build-up of the fabrics under UV irradiation decreased after a series of irradiation on and of cycles. Tus, the fatigue resistance properties of encapsulated photochromic dyed cotton fabrics under repeated exposure to UV irradiation were also investigated. For this purpose, encapsulated photochromic dye and polyurethane binder applied fabric samples were exposed to UV light for 2 min every day followed by immediate color measurement. Fatigue resistance tests were carried out for 18 days.
UPF values of the samples were measured using an SDL Atlas M284 device according to standard AS/NZ 4399:1996. Samples were irradiated during the measurement within the device. At least four specimens were taken from each sample submitted for testing according to the standard. The samples were first placed in the measurement compartment and the compartment lid was closed (Fig. 3). Ten, sample scanning was started and the UV transmittance of the specimen from 290 to 400 nm, with logging of transmittance data at least every 5 nm, was recorded. 9

UV transmittance measurement compartment.
Results and Discussions
Color Build-Up
Color build-up on UV irradiation of the samples padded with 0.5 g/L encapsulated photochromic dye using different binder types were investigated by determining L* and DE values (i.e., color difference between untreated/non-irradiated and treated/ irradiated samples). Figs. 4 and 5 give L* and DE values of the samples, respectively, after padding and after consecutive laundering tests. The photochromic capsule treated samples were colorless (white) in the absence of irradiation. A reversible blue color developed after irradiation (Fig. 6). The L* values of irradiated photochromic capsule treated fabrics were lower than those of untreated greige fabrics, which confirmed the color build-up under irradiation. It was shown that the binder type did not have a significant effect on the L* values. On the other hand, DE values varied depending on the binder type, due to the differences in the resulting a* and b* values (e.g., a* and b* values for the sample with acrylic binder were –12.26 and –7.7, for styrene binder were –6.52 and –4.75, and for polyurethane binder were –8.34 and –6.94, respectively). Tese data confirm that the type of the binder had an effect on the nuance of the photochromic color build-up under UV irradiation.
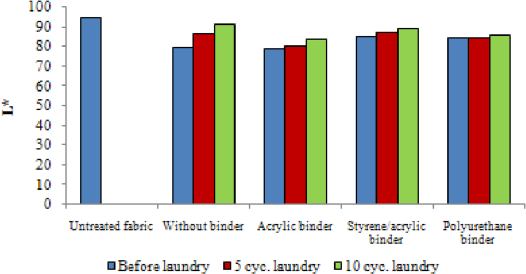
L* values of the samples before and after laundering.
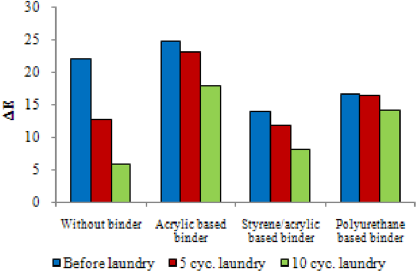
D E values of the samples before and after laundering.

Images of the samples before and after UV irradiation.
Color build-up on irradiation decreased after consecutive laundering, as expected. The use of binders increased the washing resistance of encapsulated photochromic dye. Polyurethane-based binder, which is used for textile coating applications, was more resistant to washing compared to other binder types.
Color Build-Up Rate
Fig. 7 illustrates the color build-up rate of photochromic cotton fabrics under UV irradiation. For these experiments, a 1 g/L encapsulated photochromic dye concentration and 50 g/L of polyurethane-based binder was selected. Samples reached their maximum color build-up value within ∼2 min. After maximum color build-up, if the samples were irradiated, the color difference decreased especially for the samples without binder before laundering (Fig. 7a). L* values gave similar results as shown in Fig. 8. After maximum color build-up, L* values of the samples with polyurethane-based binder did not show any significant change during extended irradiation. The results show that the use of binder improved color durability.
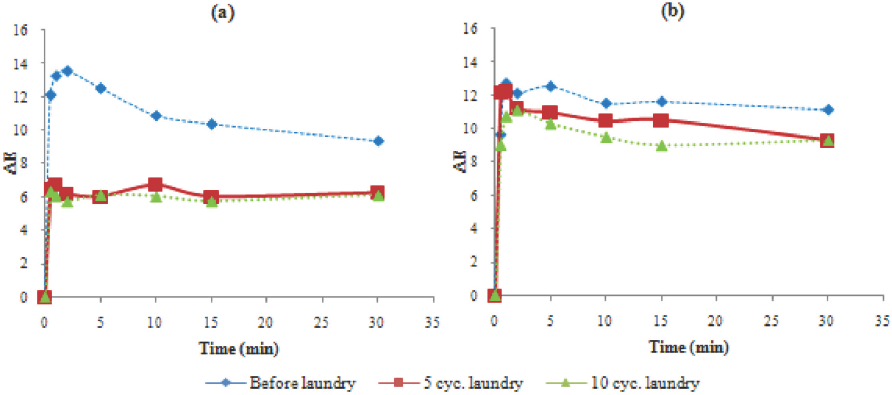
ΔE values of the samples as a function of UV exposure time: (a) without binder and (b) with polyurethane based binder.
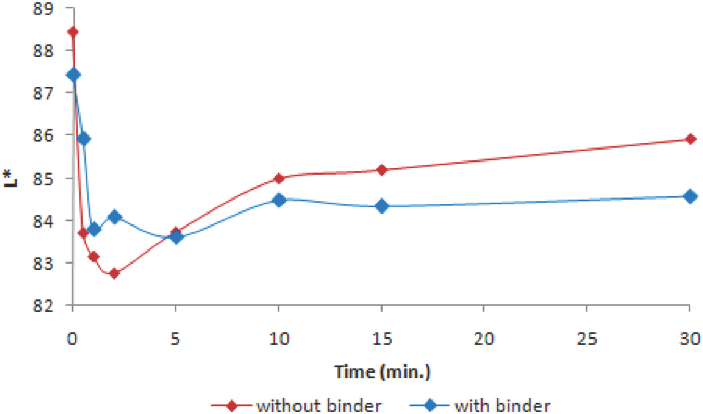
L* values of the samples as a function of UV exposure time.
Fatigue Resistance
The L* values of the samples as a function of time are shown in Fig. 9. The fatigue resistance properties of encapsulated dye decreased with time after repeated irradiation. Tis decrease was more pronounced for samples without binder.
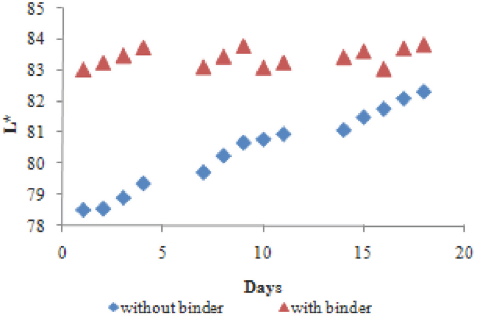
L* values of the samples as a function of time.
UV-Protective Properties
It is known that greige or bleached (white) textile materials give a lower degree of UV protection. In addition, the UV-protection capability can be increased by coloration. Since photochromic dyes lead to a color build-up during UV irradiation, the UPF of the dye treated fabric was greater than that of the untreated fabric samples.
The change in UPF values after 5 and 10 laundering cycles are shown in Fig. 10 for 0.5 g/L encapsulated photochromic dye-treated fabric. To see the effect of binder on UV protection, only binders were applied on fabric—the results show that the binders used in these experiments had no effect on UV protection.
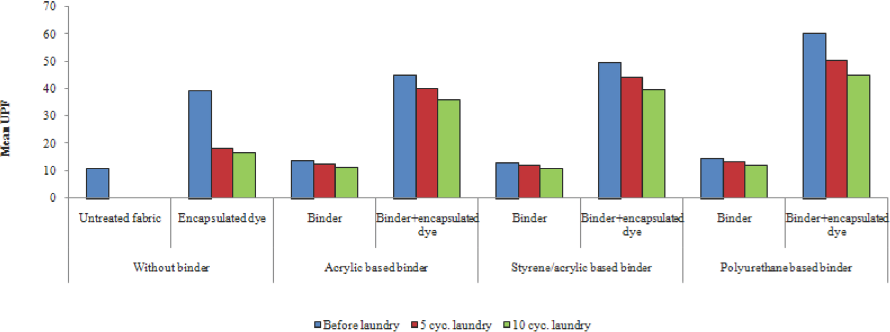
UPF values of the samples before and after laundering.
UPF values increased with the application of encapsulated photochromic dye to the cotton fabric. The binder type modi-fed the UV protection of encapsulated photochromic dyes for the binders tested. The results show that photochromic textiles using the polyurethane-based binder maintained good UPF values even after 10 laundering cycles. Therefore, application of encapsulated photochromic dyes onto textile materials was feasible for the production of UV-protective textiles.
Polyurethane-based binder was used to investigate the effect of encapsulated photochromic dye concentration on UV protection (Fig. 11). UV protection properties of the samples improved with increasing concentration of encapsulated photochromic dye. In addition, a 1 g/L encapsulated photo-chromic dye concentration was quite adequate to ensure an excellent UPF value for the chosen experimental method.
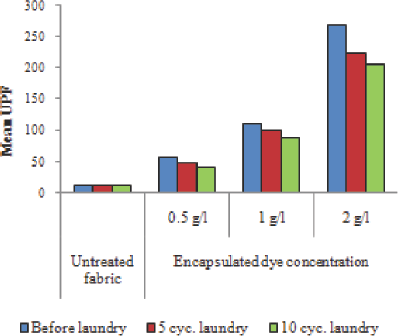
UPF values of the samples as a function of encapsulated photochro-mic dye concentration.
Conclusion
A pad-cure process was used to apply encapsulated photo-chromic dyes onto cotton textiles. Binders were included in the process to bond the dye capsules to these fabrics. The type of binder affected the nuance of the photochromic color build-up during UV irradiation, and could modify the UV protection imparted by encapsulated photochromic dyes on textiles.
Repeated UV exposures decreased the color build-up during irradiation of encapsulated photochromic dye applied cotton fabrics. The fatigue resistance properties of encapsulated dyes under UV irradiation decreased with time. Photochro-mic dye functionalized textile materials exhibited excellent UV protection even after 10 laundering cycles.
Footnotes
Acknowledgement
The authors would like to gratefully acknowledge the financial support for this research received through Project No. 109M363 of The Scientific and Technical Research Council of Turkey (TUBITAK).