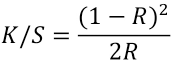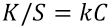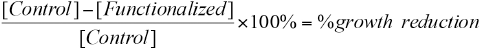Abstract
This investigation aimed to determine which of two surface activation methods, N,N'-carbonyldiimidazole (CDI) or 2,2,6,6-tetramethylpiperidine-1-oxyl (TEMPO), allowed more effective bonding of Cys-LC-LL-37, an antimicrobial peptide (AMP), onto cotton fibers and which one presented greater antimicrobial activity. The antibacterial effect of the TEMPO-activated cotton functionalized with Cys-LC-LL-37 was more effective against Staphylococcus aureus, while the CDI-activated cotton covalently bonded to Cys-LC-LL-37 was more effective against Klebsiella pneumoniae. CDI-activated cotton retained a higher percentage of the AMP, making this bioactive strategy the choice for development of AMP antimicrobial textiles.
Keywords
Introduction
Microorganism growth in textiles can adversely affect the wearer, therefore it is important to apply an antimicrobial finishing or incorporate the antimicrobial agent into synthetic fibers during extrusion. 1 The increasing demand for a more hygienic lifestyle by consumers stimulates the research and development of new antimicrobial textiles. 1
Antimicrobial agents must exhibit sufficient antimicrobial activity with a wide spectrum of action against bacteria and fungi, low toxicity, and good skin compatibility. 2 A variety of synthetic antimicrobial agents are in use, including silver, quaternary ammonium salts, polyhexamethylene biguanide, triclosan, chitosan, dyes, and regenerable N-halamine compounds and peroxyacids. 1 The problem with these agents are side effects, activity against non-target microorganisms, and environmental pollution. 3 Hence, antimicrobial textiles based on safer agents, such as antimicrobial peptides (AMPs), are a good alternative antimicrobial finishing for textiles since they have excellent activity against a broad spectrum of microorganisms.
Cysteine (Cys)-containing peptides are very important biomolecules due to the inherent reactivity of the thiol group, allowing them to participate in an array of processes ranging from redox reactions, metal ion binding, posttranslational modifications, and disulfide and thioether bond formation, among others. 4 Thus, Cys-LC-LL-37, a modified antimicrobial peptide (C-LC-LLGDDFFRKSKEKIGKE-FKRIVQRIKDFLRNLVPRTES), derived from an LL-37 altered with a LC as a 6-carbon linker to bind the Cys amino acid, was used in this study.
Wiradharma et al. modified the end-terminal(s) of the peptides with Cys residues. The results showed that the incorporation of Cys at the terminal end of AMPs significantly broadened their antimicrobial activity to Gram-positive and Gram-negative bacteria, and yeasts. 5
AMPs are often intended to work at local surfaces in most of their natural applications, therefore, immobilizing AMPs onto a biomaterial can be advantageous. 6 Covalent immobilization of an AMP can increase its long-term stability, while decreasing its toxicity, as compared to incorporation using leach- or release-based systems. 6 Furthermore, the proper orientation of the peptide may result in enhanced activity.
In this work, N,N'-carbonyldiimidazole (CDI) was used to form reactive carbonyl groups from the hydroxyl groups of cotton cellulose. 7 An AMP was then attached to the activated cellulose. CDI activates the hydroxyl groups to form the highly reactive imidazolyl carbamate. At elevated pH, nucleophilic attack of free amino groups in peptides on these reactive intermediates occurs to immobilize the peptide onto cotton. This results in the addition of one carbon to the peptide length and the formation of a stable N-alkyl carbamate linkage. 7
Ionic attachment of an AMP onto cotton cellulose was also attempted, using the stable nitroxyl radical 2,2,6,6,-tetra-methylpiperidine-1-oxyl (TEMPO) in combination with a non-metallic co-catalyst. This approach has been explored for the oxidation of alcohols using molecular oxygen as a terminal oxidant, 8 thus converting hydroxyl groups at C-6 of cellulose into carboxylates. This surface activation provides a negative charge to the cotton fibers, which facilitates AMP binding. 8
This research proposes two surface activation modification strategies, namely use of CDI and TEMPO radical, of cotton textiles, followed by functionalization with the AMP, Cys-LC-LL-37 (Fig. 1). A comparison of the antibacterial efficacy of both functionalized textiles against two bacterial strains was performed.
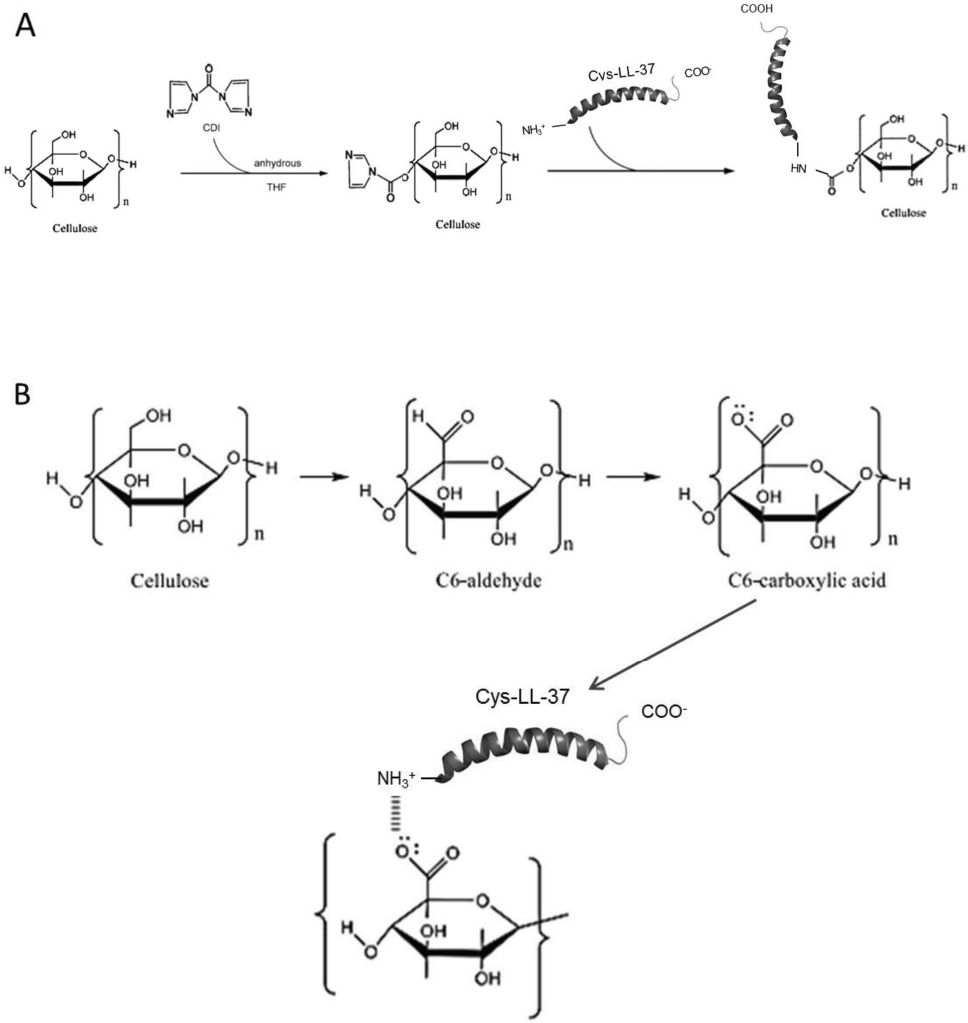
Schematic representation of the preparation of cotton functionalized with Cys-LC-LL-37 through CDI-mediated activation (A) and through TEMPO-mediated activation (B).
Materials and Methods
Chemicals
Cys-LC-LL-37 was purchased from Eurogentec. Nutrient agar and nutrient broth were obtained from Cultimed. Sodium hypochloride and methanol were purchased from Panreac and fetal bovine serum (FBS) from Biochrom. All other reagents were obtained from Sigma-Aldrich.
Antibacterial Testing of AMPs
The minimal inhibitory concentration (MIC) of Cys-LC-LL-37 against Staphylococcus aureus (ATCC 6538) and Klebsiella pneumoniae (ATCC 4352) was determined using the broth microdilution method, adapted from National Clinical and Laboratory Standard, NCLS M7-A6. 9 Cys-LC-LL-37 stock solution was prepared in sterile, deionized water (pH of 5.5) to a concentration of 20 μg/mL. Serial dilutions of AMP stock solutions were made in Mueller-Hinton Broth (MHB) with concentrations ranging from 10 to 0.156 μg/mL.
Inocula were prepared from colonies of fresh cultures that were incubated for 24 h. The bacterial turbidity was adjusted to 0.5 McFarland units with sterile water. Afterwards, bacterial work suspensions were made by diluting 500 μL of the 0.5 McFarland unit suspension in 4500 μL of MHB. Bacterial work suspensions (50 μL), 50 μL of the AMP dilutions, and 1 mL of the stock solution were added to the wells in a 96 multi-well plate. The multi-well plates were incubated for 24 h at 37 °C. For each AMP concentration, the turbidimetry of bacterial growth was compared with the controls—all determinations were performed in triplicate.
Cytotoxicity Assay
AMP cytotoxicity was evaluated by an MTT (3-[4,5-dimethylthiazol-2-yl]-2,5-diphenyltetrazolium bromide) viability assay 10 using normal human dermal fibroblasts (NHDF), since the textile material was to be in contact with the human skin. Cells were routinely maintained at 37 °C in a humidified atmosphere containing 5% CO2 and cultured in RPMI (developed at Roswell Park Memorial Institute, hence the acronym) medium supplemented with 10% fetal bovine serum (FBS), HEPES buffer (0.01 M), 1-glutamine (0.02 M), sodium pyruvate (0.001 M), and 1% antibiotic/antimycotic. Cells were used on the 20th passage.
The cells were seeded in 24-well plates (2 × 104 cells/well) in culture medium containing FBS. After 48 h adherence, some of the wells were treated with different concentrations of Cys-LC-LL-37 (2.00 and 5.00 μg/mL), and incubated at 37 °C in a 5% CO2 atmosphere for 48 h. Two concentrations were chosen: the MIC values against S. aureus and K. pneumoniae and 40% of those MIC values. Untreated cells were used as control. Afterwards, the liquid content of the wells was removed and was replaced with 200 μL of MTT solution (1 mg/mL in PBS buffer). The multi-well plates were incubated for 4 h at 37 °C with a 5% CO2 atmosphere in the dark. Next, the content of the wells was removed and 200 μL of dimethylsulfoxide (DMSO) and 20 μL of 10% glycylglycine buffer were added to dissolve the formazan crystals and to stabilize the color, respectively. The absorbance in each well was measured at 570 nm using a Biochrom Anthos 2020 microplate reader. The extent of cell viability was expressed as the percentage of viable treated cells in comparison with control cells.
The cytotoxicity results were submitted to a Student t test in a 95% confidence interval, using the computer software, IBM SPSS Statistics for Windows (version 19.0). p-values less than 0.05 were considered statistically significant.
Cotton Surface Activation
First, the cotton fabrics were washed with 1 g/L of a solution of AATCC 1993 Standard Reference Detergent WOB to remove any surface residues that could provide false antimicrobial activity results.
CDI Activation
All steps in this procedure, including preparation of solutions, were performed in a glove-box chamber with an inert atmosphere of liquid nitrogen to avoid reaction with oxygen and water in the normal atmosphere. The 4% (w/v) activation solution was prepared by dissolving CDI in anhydrous tetrahydrofuran (THF)—this concentration was based on unpublished results. The cotton fiber samples were submerged in the surface-activator solution for 2 h under moderate stirring. Afterwards, the samples were rinsed 3 times in THF and then deionized water. The proposed mechanism is represented in Fig. 1A.
TEMPO Radical Activation
The fabric surface was oxidized by reaction with TEMPO, which converts hydroxyl groups of carbon-6 into carboxylate groups. A solution of 0.0125% (w/v) of TEMPO, 0.125% (w/v) of sodium bromide, and 3.2% (v/v) of sodium hydro-chloride was prepared and its pH value was adjusted at 10.5. Ten, 2 g of fabric samples were submersed in 50 mL of the previous solution and stirred for 60 min. Afterwards, the pH of the solution was adjusted to 7 and the samples were washed in deionized water. The proposed mechanism is represented in Fig. 1B.
AMP Functionalization
For the exhaustion functionalization process following cotton fiber surface activation, solutions of Cys-LC-LL-37 were diluted to 1.91 μg/mL in deionized water. This concentration corresponded to about 40% of the antimicrobial peptide MIC value. This concentration is expected to be enough to provide antibacterial activity in cotton, since immobilization often promotes the activity of AMPs. 6 Also, antimicrobial peptides are very expensive and have a powerful antimicrobial activity, so it seemed promising to use 40% of MIC to functionalize the samples.
Activated samples (total of 4 g) were immersed in 100 mL of the bioactive solution, placed in the containers of the Datacolor Ahiba dying machine, and functionalized at 45 °C for 60 min at 15 rpm.
Finally, all samples were washed in a 1 g/L of a solution of AATCC 1993 Standard Reference Detergent WOB for 5 wash cycles performed at 40 °C over 60 min, a method adapted from the international standard EN ISO 105-C06:2010, 11 followed by drying at 37 °C for ∼4 h. Soaping was performed over the samples treated with the AMPs prior to further investigation. Assessment of antibacterial activity was performed to give evidence of durable functionalization.
Color Strength
A color strength test was performed using Coomassie Brilliant Blue reagent to assess the presence of Cys-LC-LL-37 in the modified-cotton samples, and K/S values were determined. Textile samples were immersed in 10 mL of Coomassie Brilliant Blue G-250 reagent solution at room temperature (RT) and under constant stirring for 15 min. The samples were then washed with distilled water to remove unbound peptides from the fibers.
Sample color measurement was performed using a Spectraflash 110 spectrophotometer from Datacolor at 595 nm. The ratio of light absorption to light scattering at a given wavelength is proportional to the concentration of a dye in the sample. The relationship is derived from the Kubelka-Munk equation (Eq. 1). 12
R is the reflectance, K is the light absorbed, and S is the light scattered. Also the relationship between K/S and concentration of the dye is given by Eq. 2. 12
k is the proportionality constant and C is the colorant concentration. 12 An elevated K/S value indicates that more color reagent reacted with the peptides adsorbed in the cotton. Consequently, a high K/S indicates that a higher concentration of AMP, which reacts with the color reagent, is present on the cotton surface.
Antibacterial Efficacy
The antibacterial efficacy of functionalized textile samples was tested according to JIS L Standard 1902:2002. 13 Antimicrobial activity against S. aureus and K. pneumoniae of the AMP-functionalized samples was performed by a suspension quantitative test, in which there was direct contact of the biomaterial with a bacterial cell suspension. Control samples (without AMP) and AMP-functionalized samples were tested for antimicrobial activity. The bacterial growth reduction rate was calculated based on the difference between the number of colony forming units (CFU) on the control fabric and the functionalized fabric, using Eq. 3.
[Control] is the CFU/mL of the control fabric (without AMP) and [Functionalized] is the CFU/mL of the AMP-functionalized fabric. Growth reduction was calculated after 18 h of incubation between textiles and bacterial suspension.
Results and Discussion
MICs of AMPs
The MIC of Cys-LC-LL-37 against S. aureus and K. pneumoniae was 5.00 μg/mL. According to Dürr et al., LL-37 gave an MIC against K. pneumoniae of 4.20 μg/mL, 14 which was very similar to the value for Cys-LC-LL-37 obtained in this study (5.00 μg/mL); and an MIC of 144 μg/mL against S. aureus, 14 which was a much higher value than the one for Cys-LC-LL-37 (5.00 μg/mL).
Cytotoxicity Assay
The MTT viability assay was used to determine the Cys-LC-LL-37 cytotoxicity on normal human dermal fibroblasts (NHDF). The viability percentage of fibroblasts treated with two concentrations of AMP were compared to the controls that had no contact with the tested agents (Fig. 2). When fibroblasts were contacted with Cys-LC-LL-37 at concentrations of 2.00 and 5.00 μg/mL, there was an increase in fibroblast viability of 10% and a decrease of 0.2% compared to the control, respectively.
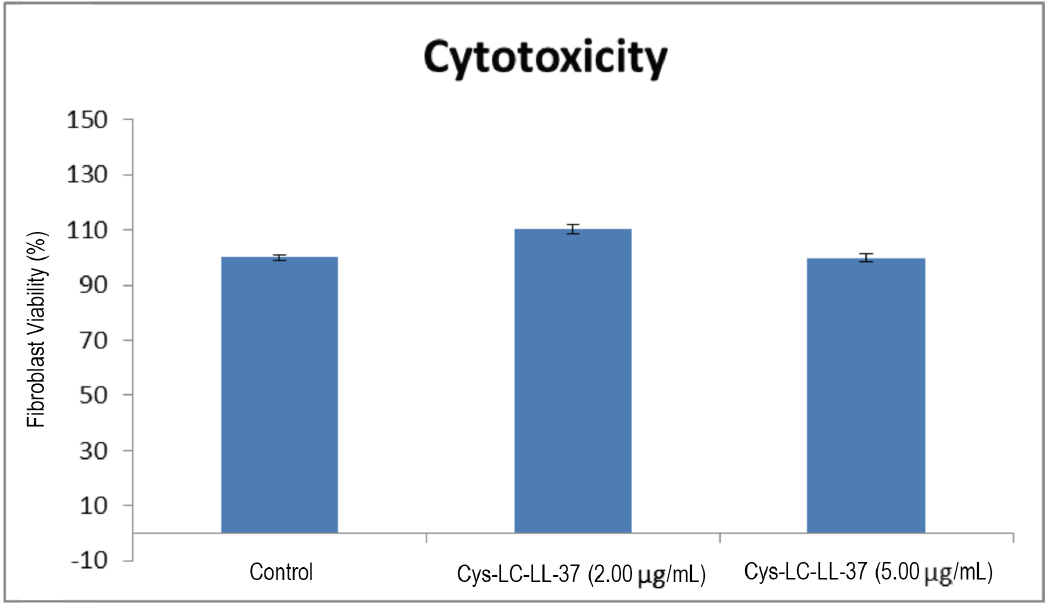
Fibroblast viability percentage (mean ± SD) when in contact with two concentrations of Cys-LC-LL-37 and the untreated control.
These results indicate that the Cys-LC-LL-37 concentrations tested were not cytotoxic to the NHDFs. According to Gouveia et al., only a difference greater than 30% as compared with the control is considered toxic to NHDF cells. 15 Consequently, these AMPs were considered safe to be applied as antimicrobial agents in contact with the human skin without causing any adverse cutaneous reaction in the concentrations tested. The results were statistically significant for a p-value < 0.05, according to a Student t test with a 95% confidence interval.
Lehrer and Ganz stated that, at 50–100 μg/mL, LL-37 was cytotoxic. 16 Cys-LC-LL-37 tested concentrations were much less than 50 μg/mL and, as expected, no cytotoxic effect was observed.
Color Strength
The greater the percent Coomassie Brilliant Blue dye strength, the greater the peptide content on the sample. The covalent surface modification with CDI allowed a greater retention of peptide on the cotton (∼49%). TEMPO-activated cotton retained Cys-LC-LL-37 at ∼26%. Covalent surface activation improved peptide retention on the cotton fabric.
Antibacterial Properties
The antibacterial effect of 1.91 μg/mL Cys-LC-LL-37 functionalized textile samples is shown in Table I. Peptide surface concentration depends on the immobilization strategy used as the limited accessibility of peptide reactive groups and different coupling procedures can affect the efficiency of peptide immobilization. 6 Most studies indicate that peptide concentrations, although important, do not appear to be the most critical parameter for antimicrobial activity. 6 Additionally it was observed that the bactericidal activity of LL-37 immobilized onto titanium surfaces was independent of the peptide concentration. In most cases, the immobilized AMPs displayed an increase in their MIC value compared to the soluble peptide. 6 Therefore, in this work, 1.91 μg/ mL of Cys-LC-LL-37, about 40% of the 5.00 μg/mL MIC value, was used. This AMP is expensive unless very small concentrations are used, and since other studies revealed the immobilized AMPs presented a 50-fold higher MIC compared to soluble peptides, 6 an antibacterial effect with a 40% lower MIC value of peptide was targeted.
Percent Bacterial Reduction after 18 h Incubation between Fabric and Microorganism
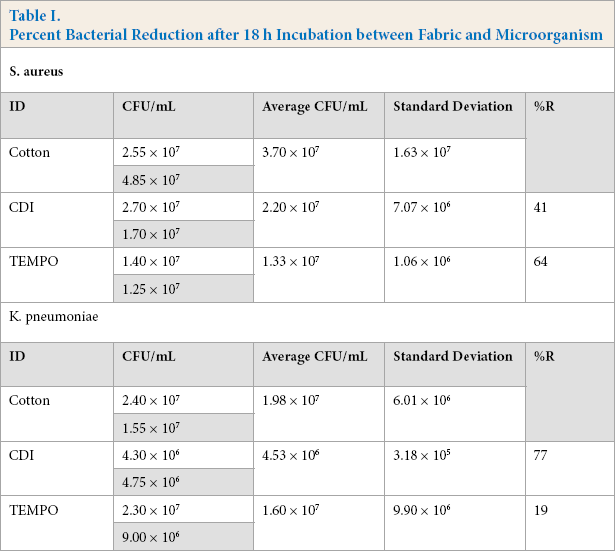
CDI-activated cotton functionalized with Cys-LC-LL-37 showed a bacterial reduction of 77% and 41% against K. pneumoniae and S. aureus, respectively, meaning that this bioactive fabric more effectively reduced the growth of the Gram-negative bacterium. On the other hand, TEMPO-activated cotton reduced the development of S. aureus by 64% and K. pneumoniae by 19%, thus being more effective against the Gram-positive microorganism. According to the work by Rajendran et al., cotton treated with bulk ZnO led to an S. aureus reduction of 19% and a reduction of Escherichia coli of 11% after five wash cycles. 17 These percentages are less than the ones obtained here, meaning that the treatment with Cys-LC-LL-37 of the covalent and ionic activated cotton fabrics had a significant antibacterial effect. Moreover, ZnO is toxic to mammalian cells 18 and Cys-LC-LL-37 can safely contact with human skin as determined by the cytotoxicity studies.
Conclusion
This research studied the development of two cotton-based textiles with an antibacterial finishing that could be used in the medical field to aid in the prevention of development of microorganisms in textiles. The bioactive agent, Cys-LC-LL-37, was tested for cytotoxic effects against NHDFs and no cell toxicity was observed, indicating that this agent was safe for skin contact. Cotton fibers were activated with either TEMPO or with CDI, which allowed the creation of ionic and covalent bonds, respectively, between the textile surface and the peptide. The two textile materials were compared in terms of functionalization efficacy and antibacterial activity. Results showed that the covalent surface modification with CDI allowed a greater retention of peptide in the cotton than the TEMPO-activated cotton.
The overall antibacterial activity of the TEMPO-activated cotton functionalized with the Cys-LC-LL-37 was more effective against S. aureus, while the effect of the Cys-LC-LL-37 covalently bonded to the textile was more effective against K. pneumoniae. To conclude, the covalently linked Cys-LC-LL-37 seemed to be more effective against the microorganisms in this study, especially against the Gram-negative bacterium. This bioactive textile also retained more Cys-LC-LL-37 which might contribute to the increased action. Comparatively, the TEMPO-modified cotton functionalized with the AMP presented a very low activity against the Gram-negative bacterium.
Footnotes
Acknowledgments
The authors thank Fundação para a Ciência e Tecnologia (FCT) for funding granted concerning the project-PTDC/EBB-BIO/113671/2009 (FCOMP-01-0124-FEDER-014752) Skin2Tex. We also thank Fundo Europeu de Desenvolvimento Regional (FEDER) through COMPETE–Programa Operacional Factores de Competitividade (POFC) for the co-funding.