Abstract
For subsets of colorectal adenocarcinoma (CRC) patients, nuclear accumulation of p53 (p53nac) and Bcl-2 expression are prognostic indicators. To understand their role in the progression of CRC we evaluated 90 CRCs and their contiguous adenomatous components (CAdCs) for immunohistochemical expression of these markers. In general, p53nac and Bcl-2 expression was significantly increased when comparing normal colonic epithelia to CAdCs and CRCs. Thirteen (14%) CAdCs that demonstrated p53nac continued to express p53nac in their contiguous CRCs. A similar trend was observed in Bcl-2 expression in that the majority of CAdCs expressing Bcl-2 continued to express it in their matching CRCs (39/44). Patients whose CAdCs and their contiguous CRCs demonstrate p53nac had shorter median survival (35.9 months) than those patients whose CAdCs and CRCs did not (80.56 months). However, patients whose CAdCs had p53nac and lacked Bcl-2 expression had the lowest median survival (15.74 months) when compared with patients whose CAdCs did not demonstrate p53nac but had increased expression of Bcl-2 (71.77 months). These findings suggest that in those adenomas that demonstrate p53nac but lack Bcl-2 expression, their contiguous CRCs are more likely to be aggressive as they progress.
D
Some of the genes known to be involved in CRC progression from an adenoma to CRC are APC, β-catenin, K-ras, c-myc, SMAD4, and p53. Of these, p53 mutations (alterations) have been proposed to occur as late carcinogenic events. The wild-type p53 induces apoptosis in response to irreversible DNA damage and to an oncogenic stimulus (Efeyan et al. 2006), whereas mutant p53 inhibits apoptosis and confers an advantage for cell survival (Shaw et al. 1992; Lotem and Sachs 1993). Bcl-2 protooncogene acts as an inhibitor of apoptosis (Flohil et al. 1996). Transfection of p53 into lymphoid cells was shown to downregulate endogenous Bcl-2 expression, whereas simultaneous increase in the expression of Bax, a proapoptotic protooncogene, was observed (Miyashita et al. 1994; Selvakumaran et al. 1994). On the other hand, p53-induced cell death can be prevented by Bcl-2 expression (Hao et al. 1998); specifically the expression of Bcl-2 in p53-deficient mice is enhanced, with accompanying downregulation of Bax expression (Miyashita et al. 1994). These findings suggest that p53 and Bcl-2 may interact and have different roles in carcinogenesis.
Alterations in p53 and Bcl-2 were reported in early stages of CRC development (van den Berg et al. 1989; Purdie et al. 1991; Hague et al. 1994; Bosari et al. 1995; Mosnier et al. 1996; Yao et al. 1999; Krajewska et al. 2005). Furthermore, other studies as well as our own have demonstrated that nuclear accumulation of p53 (p53nac) and Bcl-2 are important prognostic molecular markers specifically for a subset of patients with CRC (van den Berg et al. 1989; Purdie et al. 1991; Ofner et al. 1995; Mosnier et al. 1996; Manne et al. 1997, 1998, 2000; Kaklamanis et al. 1998; Yao et al. 1999). Although multiple genetic alterations have been reported during the development of colorectal neoplasia, known molecular events that contribute to the aggressive behavior of adenomas are limited. In the present study we attempted to determine the roles of phenotypic expression of p53nac and Bcl-2 in contiguous adenomatous components (CAdCs) of CRCs to assess the aggressive behavior of their contiguous invasive carcinomas of the colorectum.
Materials and Methods
Patients and Tissues
The institutional review boards of the University of Alabama at Birmingham (UAB) Hospital approved this study. We identified a total of 620 CRC patients from the UAB Hospital who had undergone surgical resection for first primary CRC from 1981 through 1993. We obtained the medical records including surgical pathology reports of these patients, which were reviewed by two of the gastrointestinal (GI) pathologists (CS, NCJ) to ascertain key information. During our initial selection process, those patients who died within a week of their surgery; those who had inflammatory bowel disease; those patients whose archival tissues were not available; those patients with surgical margin involvement, unspecified tumor location, multiple primaries within the colorectum, or multiple malignancies; or those patients with family or personal history of CRC were all excluded from the study population. However, based on the information in patients’ charts, it may have been difficult to identify the familial vs. sporadic nature of the tumors; therefore, our patient populations can be described as ‘consecutive’ populations of CRC patients. To control for treatment bias, we included only those patients who underwent surgery as a therapeutic intervention and excluded patients who received any pre- or postsurgical therapies. Because the use of adjuvant chemotherapy was not widespread during the time frame of this study (1981–1993), we were able to obtain a large number of CRC patients who had not received adjuvant therapy. Only 90/620 cases had CRCs with contiguous adenomatous components. We obtained formalin-fixed paraffin-embedded archival tissue blocks of these cases from the files of The UAB–Surgical Pathology Department.
In our study, two GI pathologists (CS, NCJ) reviewed hematoxylin- and eosin-stained slides of all cases to determine the histomorphological type (tubular, tubulovillous, and villous) and the degree of dysplasia of the CAdCs (Hamilton and Aaltonen 2000). Degree of histological differentiation of CRCs was graded as well, moderate, poor or undifferentiated, as suggested by WHO (Hamilton and Aaltonen 2000). Both pathologists were blinded to the pathological evaluations of the other; however, if there were any discrepancies they were resolved by analyzing cases together to reach a final consensus. Subsequently, we pooled well- and moderately differentiated CRCs into a low-grade group and poor and undifferentiated tumors into a high-grade group (Compton et al. 2000). Pathological staging was performed according to the criteria of the American Joint Commission on Cancer (stages I, II, III, and IV) (Green et al. 2002). The International Classification of Diseases for Oncology (ICD-O) codes were used to specify anatomic location of the tumor (World Health Organization 1990). Anatomic sub-sites were grouped into the proximal colon (cecum, ascending colon, and proximal two thirds of the transverse colon), the distal colon (distal one third of the transverse colon, descending colon, and sigmoid colon), and the rectum.
Patients were followed by The UAB tumor registry until their death or the date of the last documented contact within the study time frame. The tumor registries ascertain outcome (mortality) information directly from patients (or living relatives) and from the physicians of the patients through telephone and mail contacts. This information is further validated against State Death Lists. Tumor registries update follow-up information every 6 months, and follow-up of our cohort ended in April 2007.
Immunohistochemical Analysis
Five-μm tissue sections were cut from paraffin blocks representative of normal and tumor with contiguous CAdC of each case and were mounted on Superfrost/ Plus slides (Fisher Scientific; Pittsburgh, PA). We cut the tissue sections 1–2 days prior to immunostaining to avoid potential problems in antigen recognition due to storage degradation of cut tissue sections on glass slides (Prioleau and Schnitt 1995; Jacobs et al. 1996). Immunostaining was performed as described in our earlier studies of antigen expression in different tissues (Manne et al. 1997, 1998, 2000). In brief, sections were melted and incubated overnight at 37C prior to staining. Tissue sections were then deparaffinized in xylene and subsequently rehydrated in graded alcohols. Sections were then transferred to a Tris-buffer bath (0.05 M Tris base, 0.15 M NaCl, and 0.01% Triton X-100, pH 7.6). Microwave antigen retrieval was performed on the sections for 7 min using citrate buffer 0.01 M, pH 6 (citric acid monohydrate 2.1 g, H2O 1000 ml, adjusting pH with NaOH) for Bcl-2; however, no antigen retrieval was performed for tissue sections stained for p53nac (Baas et al. 1996; Manne et al. 1997). Each section was treated with an aqueous solution of 3% H2O2 for 5 min to quench endogenous peroxidase activity. Each section was then incubated with 3% goat serum at room temperature for 1 hr to reduce nonspecific immunostaining. Tissue sections were then incubated with anti-human p53 (clone BP53-12.1, dilution 1:80; BioGenex, San Ramon, CA) and anti-human Bcl-2 (clone 124, dilution 1:60; Roche Diagnostics, Indianapolis, IN) primary monoclonal antibodies. Sections on which the primary antibody was not applied were utilized as negative controls.
Secondary detection was accomplished using a multispecies detection system (Signet Laboratories; Dedham, MA). Sections were exposed to biotinylated multispecies antibodies including anti-mouse antibodies for 20 min and then incubated with peroxidase-labeled streptavidin for 20 min. A diaminobenzidine tetrachloride supersensitive substrate kit (BioGenex) was used to visualize the antibody–antigen complex. Each section was then counterstained using hematoxylin, dehydrated using graded alcohols, and soaked in xylene before coverslipping.
The staining evaluation strategy of p53nac and Bcl-2 expression was described in our earlier studies (Manne et al. 1997, 1998). Slides were independently examined by three pathologists (CS, NCJ, and WEG); however, if there was a discrepancy in individual scores all three pathologists reevaluated together by reaching a consensus agreement before combining the individual scores. As described in our earlier studies (Manne et al. 1997, 1998, 2000), we considered only tumor cells with distinct nuclear immunostaining for p53nac as positive and considered the tumor positive only if nuclear accumulation was identified in ≥10% of all malignant cells in a tissue section. We chose this cut-off value of 10% positivity because this value showed the highest concordance between immunohistochemical detection of p53nac and point mutations of the p53 gene as detected by single-strand conformational polymorphism analysis (95% of point mutations) (Grizzle et al. 1998).
A semiquantitative immunostaining score (ISS) for Bcl-2 was obtained as described previously (Manne et al. 1997, 2000; Chatla et al. 2005). In brief, each of the three IHC reviewing pathologists estimated the proportion of cells stained and the intensity of staining in the whole tissue section. Intensity of immunostaining of individual cells was scored onascale from 0(no staining) to 4 (strongest intensity). In addition, each pathologist estimated the proportion of cells stained at each intensity. The percent of cells at each intensity was multiplied by the corresponding intensity value to obtain an ISS that ranged from 0 to 4. Scores of the three investigators were combined to obtain an overall mean ISS. An ISS of ≥0.5 was chosen as a cut-off value for Bcl-2 expression as described earlier (Manne et al. 1997, 2000).
Results
Mean age of patients at the time of surgical resection was 66.5 years (range, 35–86 years). Based on the histopathological features, there were 53 tubular, 14 tubulovillous, and 23 villous types of CAdCs in this study. Further analysis of these histological types based on the degree of dysplasia revealed that 13/53 tubular, 2/14 tubulovillous, and 2/23 villous types were classified as CAdCs with high-grade dysplasia.
Phenotypic expression patterns of p53nac and Bcl-2 are shown in Figure 1. None of the adjacent normal epithelial mucosa was positive for p53nac, but Bcl-2 expression was observed in the basal crypts. In the CAdCs and CRCs, p53nac was localized to the nucleus and Bcl-2 to the cytoplasm. Tubular CAdCs (6/53, 11%) were less positive for p53nac than tubulovillous (3/14, 21%) and villous CAdCs (4/23, 17%), regardless of degree of dysplasia.
Expression levels of these two makers significantly increased as one moved from adjacent normal-appearing epithelium to CAdC to CRC (Figure 2). Clinicopathological features and expression profiles of p53nac and Bcl-2 of 90 CRCs and their CAdCs are given in Table 1.p53nac was observed in 13/90 (14%) CAdCs as compared with 35/90 (39%) of CRCs (Table 1). It is interesting to note that all CAdCs with p53nac also exhibited p53nac in their corresponding CRCs.
Increased cytoplasmic expression of Bcl-2 was observed in 44/90 (49%) CAdCs and in 62/90 (69%) CRCs (Table 1). Of 44 Bcl-2-expressing CAdCs, 39 (89%) showed Bcl-2 positivity in their corresponding CRCs. Only 5/90 (5.5%) cases showed loss of Bcl-2 expression in the CRCs with respect to their corresponding contiguous CAdCs, and all these cases were associated with increased expression of p53nac (data not shown). Individual and/or concomitant expression of Bcl-2 and p53nac did not show significant correlations with any of the prognostic features of CAdCs like grade of dysplasia, histological subtype, and extent of adenomatous component as well as the stage of their corresponding CRCs, suggesting that expression of both these markers are not dependent on these aggressive tumor features (data not shown).
Table 2 shows patient characteristics and their correlation with possible combinations of p53nac and Bcl-2 expression in CAdCs and in their corresponding CRCs. These correlation analyses suggested that the poor prognostic combination, p53nac positive plus Bcl-2 negative, was predominantly observed in patients with stage III tumor; however, this association was not statistically significant. However, there was a significant association between the proximal colon site and this poor prognostic combination of p53nac and Bcl-2 expression (χ2 p value, 0.028) (Table 2). Also, the phenotypic expression of p53nac and lack of Bcl-2 expression in CRCs (11/17, 65%) with adenomatous components were associated with regional lymph node involvement (data not shown).
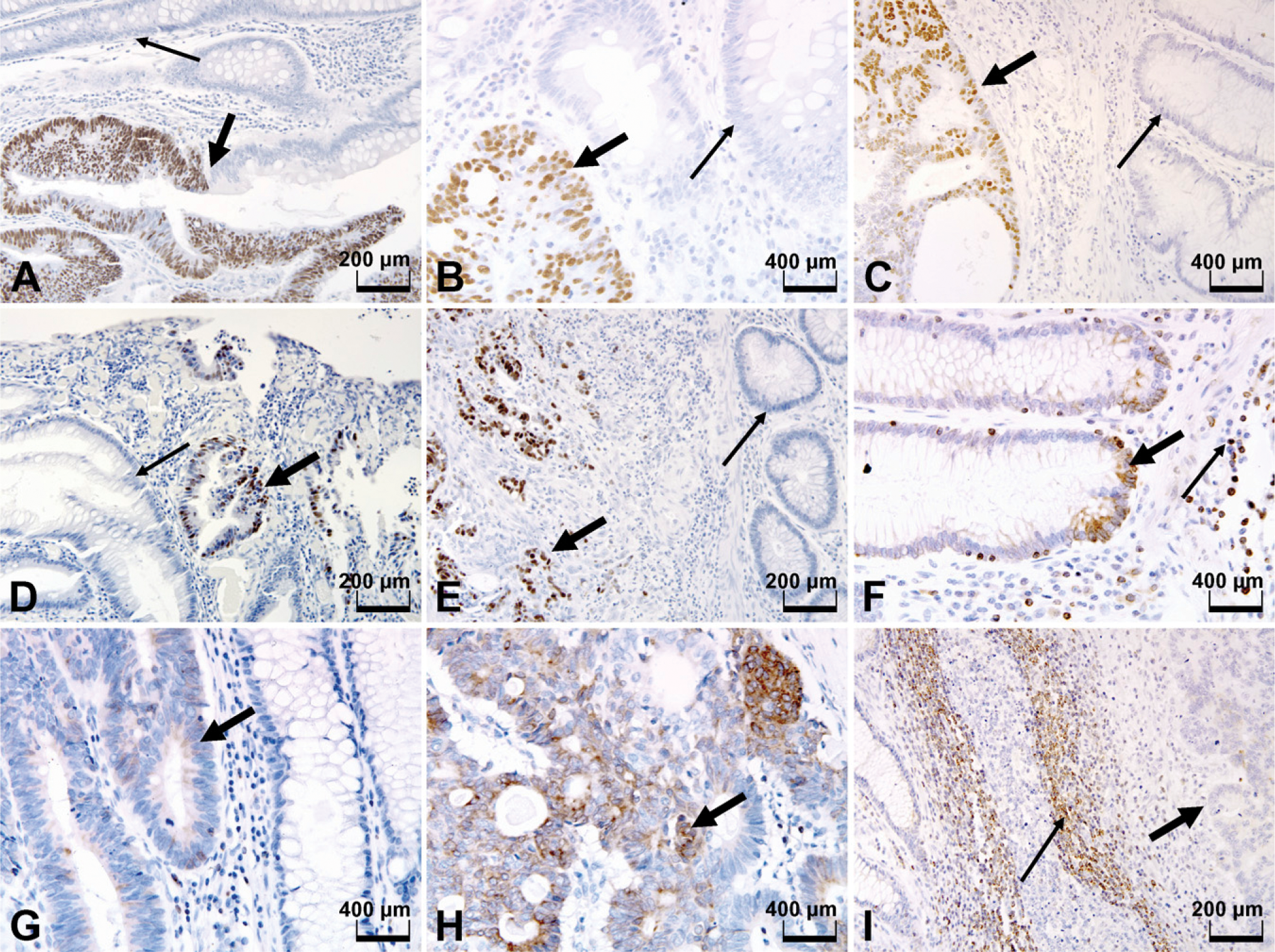
Immunohistochemical staining of p53nac and Bcl-2 expression in colorectal lesions. (
Median survival of patients with p53nac in both CAdCs and their corresponding CRCs was markedly lower (35.9 months) than those patients whose CAdCs were negative but their CRCs were positive for p53nac (74.51 months) or to those patients without p53nac in both CAdCs and CRCs (80.56 months) (Table 3). Bcl-2 expression in these lesions did not show such a significant difference in their median survivals (Table 3); however, when the concomitant expression of p53nac and Bcl-2 was considered, patients whose CAdCs were positive for p53nac and lacked Bcl-2 expression had the lowest median survival (15.74 months) when compared with patients whose CAdCs did not demonstrate p53nac but had increased expression of Bcl-2 (71.77 months) (Table 3). A similar trend in survival was observed in patients whose CRCs were categorized based on these two markers (50.89 vs 77.64 months) (Table 3).
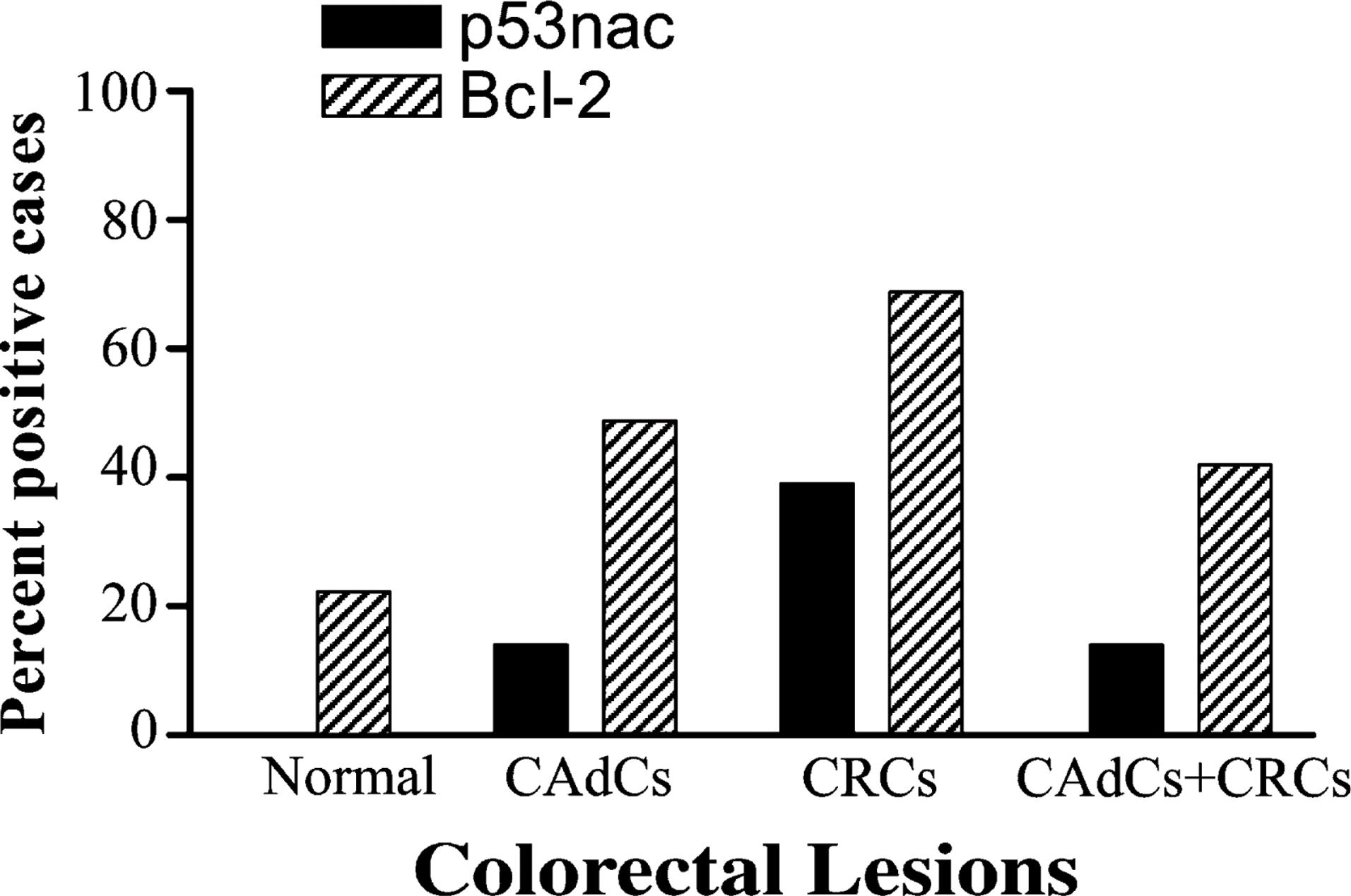
Expression patterns of p53nac and Bcl-2 in normal colonic epithelia, adenomatous components (CAdCs), adenocarcinomas (CRCs), and concomitantly expressing CAdCs and CRCs (CAdCs + CRCs). None of the adjacent normal colonic epithelium expressed p53nac.
Discussion
Our study demonstrated that the phenotypic expression levels of p53nac and Bcl-2 significantly increased in the transition from adjacent normal-appearing epithelia to contiguous CAdCs to CRCs. CAdCs that demonstrated p53nac continued to express p53nac in their contiguous CRCs, and a similar pattern was observed for Bcl-2 in that reversal of phenotypes of p53nac or Bcl-2 expression was rare. Also, even though an abnormality in p53 is considered a late event, p53nac was present in 14% of CAdCs. A shorter survival period was observed in patients in whom their CAdCs and their contiguous CRCs exhibited p53nac as compared with those patients with CAdCs negative and CRCs positive for p53nac or with patients whose CAdCs and CRCs did not demonstrate p53nac. When CAdCs were immunoreactive for p53nac but lacked Bcl-2 expression, patients’ tumors were more aggressive than those CAdCs that lacked p53nac and increased Bcl-2 expression. These results suggest that once p53nac has been detected and Bcl-2 is downregulated in an adenoma, its contiguous CRC behaves aggressively.
Coexistence of CAdCs in juxtaposition to CRCs supports the now well-accepted hypothesis that a subgroup of adenocarcinomas do develop from adenomas. However, only a small proportion of all adenomas progress to become invasive carcinomas (Burgart 2002). Alterations present at the early stages are likely to play a crucial role in CRC progression. Understanding of the molecular alterations underlying this progression will aid in determining the aggressive behavior of precursor lesions as well as CRCs.
The majority of studies in CRC have demonstrated that abnormal p53, detected by IHC, is a marker of poor patient survival; however, some studies found that p53nac has limited value in predicting clinical outcome (as reviewed in Grewal et al. 1995; Manne et al. 1997). Specifically, others and our own prior studies have suggested that p53nac is a poor prognostic indicator for patients with proximal but not distal colon tumors, specifically for non-Hispanic Caucasian patients (Manne et al. 1998; Diez et al. 2000). In this study, 39% of CRC showed p53nac as compared with 14% of their contiguous CAdCs. A study by Yao et al. (1999) has also demonstrated a similar incidence of p53nac in independent villous adenomas. Low p53nac expression in CAdCs, as compared with the juxtaposed CRCs, indicates that p53nac is a relatively late event in colorectal carcinogenesis as demonstrated by several other studies (van den Berg et al. 1989; Purdie et al. 1991; Hague et al. 1994; Bosari et al. 1995; Mosnier et al. 1996; Yao et al. 1999; Krajewska et al. 2005). All cases with CAdCs demonstrating p53nac also exhibited p53nac in their corresponding CRCs. This finding suggests that an adenoma exhibiting p53nac is more likely to develop into an aggressive (invasive) carcinoma.
Clinicopathological characteristics of the study cohort
Expression of Bcl-2 in CRC has been shown to be a favorable prognostic marker in several studies (Ofner et al. 1995; Manne et al. 1997, 2000; Kaklamanis et al. 1998; Krajewska et al. 2005). Association of this anti-apoptotic protein with patients having a better survival may be due to the ability of Bcl-2 to be converted from a protector to a killer through interactions with other proteins (Lin et al. 2004) or being related to the as yet unexplained role of Bcl-2 in suppressing cell cycle entry (Linette et al. 1996; O'Reilly et al. 1996). As demonstrated in our earlier studies (Manne et al. 1997, 2000), the current study also showed a higher incidence of Bcl-2 expression in CRCs (69%) than in the corresponding CAdCs (49%); however, if Bcl-2 was expressed in the CAdCs it continued to be expressed in the majority of matching CRCs. Because Bcl-2 expression is linked to a favorable prognosis, this suggests that adenomas expressing Bcl-2 may similarly be tied to a favorable outcome even if CRC develops. These results suggest that Bcl-2 oncogenic protein may play a role in colorectal tumorigenesis, possibly in the early phases of the adenoma–carcinoma sequence.
Correlation between clinicopathologic features and the concomitant expression patterns of p53 and Bcl-2
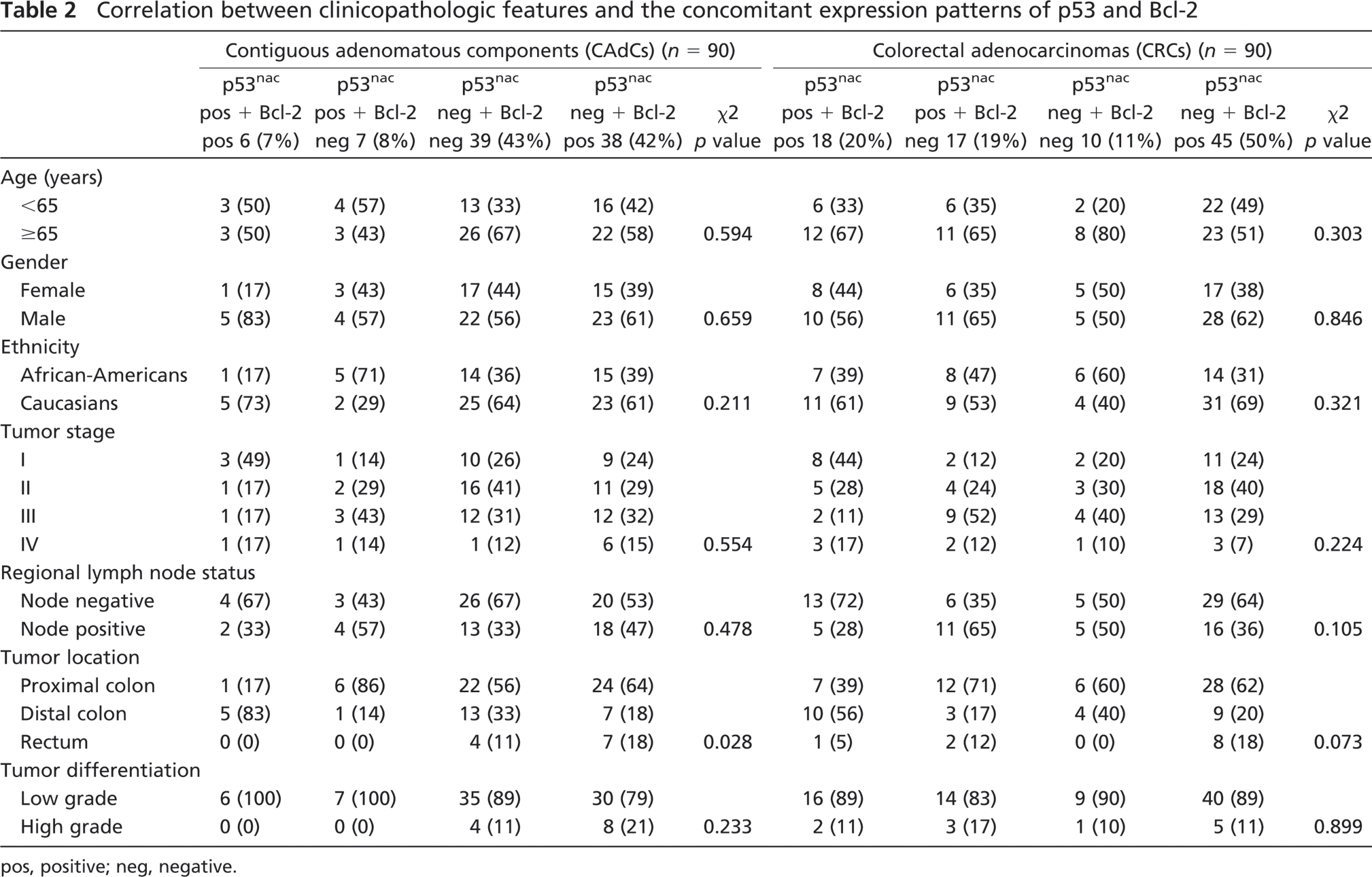
pos, positive; neg, negative.
Patient survival based on the molecular marker expression status of colorectal lesions
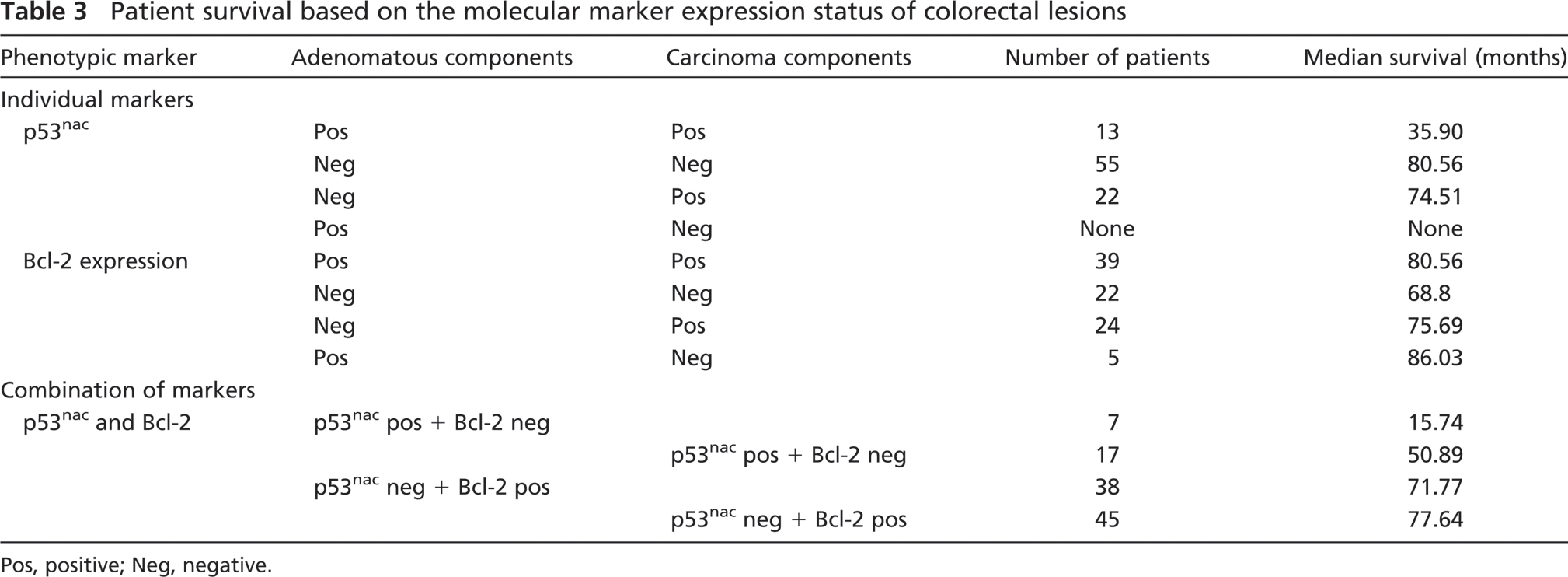
Pos, positive; Neg, negative.
We conclude that the phenotypic expression of p53 and Bcl-2 progressively increased from adjacent normal-appearing epithelium to CAdC to invasive CRC. The presence of p53nac in the CAdC is an indicator of aggressive behavior of colonic lesions, and these patients are more likely to develop aggressive invasive cancer. Although the additional prognostic pathological features including tumor budding, host responses (e.g., Crohn-like reaction), extramural invasion, vascular invasion, etc. will significantly influence the clinical outcome, evaluation of a combination of p53 and Bcl-2 expression in CAdCs correlates with patient survival. Therefore, it is of great value to assess the adenomas for the phenotypic expression of these markers, which may aid in patient follow-up and surveillance. Furthermore, these findings may lead to the development of new tools for cancer prevention.
Footnotes
Acknowledgements
This work is supported by the National Institutes of Health/National Cancer Institute (Grant RO1-CA-98932-01) and the Early Detection Research Network (Grant U24-CA-086359).
We thank Harpreet Singh, MD, for assistance in database creation and data processing. We thank the Tissue Procurement Facility of The UAB–Comprehensive Cancer Center for histological services.
