Abstract
β-catenin plays a role in cell adhesion and as a transcriptional coactivator. Its levels are regulated in cells by controlling its degradation through ubiquitination by two different E3 ligase complexes. One complex contains β-transducing repeat containing (BTRC) protein, which binds to β-catenin when phosphorylated on specific (S33 and S37) residues, whereas the other involves calcyclin-binding protein (CACYBP). The aim of this study was to determine the localization and levels of total and active (S33/S37-dephosphorylated) β-catenin in the pregnant mouse uteri and those undergoing artificially stimulated decidualization. These two forms of β-catenin were localized almost exclusively to the endometrial epithelia just prior to the onset of implantation. Although this localization continued after the onset of implantation, there were less epithelial cells present in areas of the uterus undergoing decidualization. Rather, there was a progressive increase in β-catenin localization in endometrial stromal cells undergoing decidualization in the anti-mesometrial and, to a lesser extent, in the mesometrial regions. The presence of a conceptus was not required for the changes in localization seen in the pregnant uterus because similar findings were also seen in uteri undergoing artificially stimulated decidualization. Finally, overall levels of total, active (S33 and S37 dephosphorylated), and phosphorylated (S33/S37/T42) β-catenin protein and the steady-state levels of calcyclin-binding protein mRNA changed in the uterus during decidualization. The result of this study shows the changing localization and levels of β-catenin in the mouse uterus during decidualization. Further, the results suggest potential roles for both the BTRC and CACYBP E3 ligase mechanisms of β-catenin ubiquitination in the uterus during decidualization.
β-catenin protein (CTNNB1) is a multifunctional intra-cellular protein. One of the well-known roles of CTNNB1 is to help provide an anchor between the cytoskeleton and a family of transmembrane proteins called cadherins (Kemler 1993). The extracellular domains of these cadherins on adjacent cells can bind to each other, forming adhesive contacts called adherens junctions (Yap et al. 1997). Therefore, CTNNB1 is thought to play an important role in maintaining tissue architecture and morphogenesis. Notably, the regulation of CTNNB1 participation in the adherens junctions is controlled by its phosphorylation status of specific amino acids (Nelson and Nusse 2004; Bienz 2005; Harris and Peifer 2005; Lilien and Balsamo 2005) and by catenin β interacting protein 1 (CTNNBIP1, also called ICAT) (Gottardi and Gumbiner 2004b; Stow 2004). A second well-known role of CTNNB1 is to enter the nucleus and act as a cotranscriptional modulator of the expression of a growing number of target genes (Cadigan and Nusse 1997). Although the canonical Wnt signaling pathway has been well implicated in CTNNB1-mediated transcriptional regulation (Gordon and Nusse 2006), it is beginning to appear that additional factors play an important role in the regulation of the CTNNB1 function in the nucleus (Stow 2004; Krieghoff et al. 2006; Zeng et al. 2006). Regulation of CTNNB1 participation in adhesion or transcriptional activation is the topic of recent reviews to which the reader is referred (Bienz 2005; Harris and Peifer 2005).
One of the major factors regulating CTNNB1 functions in cells is modulating its ubiquitination and thus subsequent destruction by the proteasome. The best-described pathway regulating CTNNB1 degradation is the canonical Wnt pathway that acts to prevent its degradation (Logan and Nusse 2004). This pathway involves a group of secreted Wnt proteins that act as ligands for a group of membrane Frizzled receptors causing the activation of a well-characterized intracellular signaling cascade inhibiting glycogen synthase kinase 3-β (GSK3β) protein activity. GSK3β is a major regulator of CTNNB1 ubiquitination and thus degradation (Kitagawa et al. 1999; Latres et al. 1999; Liu et al. 1999; Winston et al. 1999). The phosphorylation status of a group of N-terminal residues of CTNNB1 plays a key role in regulating this GSK3β-dependent CTNNB1 degradation. Phosphorylation of amino acid residues S33 plus S37 of CTNNB1 by GSK3β leads to its rapid ubiquitination via an E3 ligase that contains the F-box protein called β-transducing repeat containing (BTRC) protein (Kitagawa et al. 1999; Winston et al. 1999). When these amino acids are not phosphorylated, such as when the canonical Wnt pathway is activated, BTRC is unable to bind to the N terminus and thus CTNNB1 is not ubiquitinated. Therefore, CTNNB1 dephosphorylated at S33 plus S37 is commonly referred to as “active CTNNB1” because it can function within the cell as opposed to being degraded. Notably, recent evidence indicates that this form of the protein can take part in both adherens junctions and cotranscriptional activation (Gottardi and Gumbiner 2004a).
Recently, a second E3 ligase complex has been identified and is believed to be another regulator of CTNNB1 ubiquitination and subsequent breakdown by the proteasome (Filipek 2006). This complex in mouse cells includes a protein called mouse calcyclin-binding protein (CACYBP; also commonly called SIAH-interacting protein) along with transducin (beta)-like 1 X-linked, S-phase kinase-associated protein 1A and seven in absentia 1A proteins (Matsuzawa and Reed 2001; Matsuzawa et al. 2003). Unlike the complex above, the actions of this ligase complex do not depend on the phosphorylation status of CTNNB1 (Filipek 2006). CACYBP does, however, show changes in cellular localization under different conditions, which might depend on its own serine phosphorylation status (Filipek et al. 2002; Wu et al. 2003). Two recent reviews (Filipek 2006; Fukushima et al. 2006) summarize what is presently known about the role of CACYBP in an E3 ligase complex, and it is clear that more work is needed before we completely understand its role in CTNNB1 degradation and how it is controlled.
In response to an implanting conceptus, endometrial fibroblast-like cells in the rodent uterus differentiate into large decidual cells in a process called the decidual cell reaction or decidualization (reviewed in (Abrahamsohn and Zorn 1993). This re-differentiation of cells occurs in a temporal and regional fashion. It first begins just after the onset of implantation in a small number of cells adjacent to the implanting conceptus in the anti-mesometrial region of the endometrium, forming what is referred to as the primary decidual zone (PDZ). This is followed by the progression of decidualization toward the myometrium in the anti-mesometrial region of the uterus, which forms the secondary decidual zone (SDZ). Together the PDZ and SDZ make up the anti-mesometrial decidual cells that either undergo apoptosis or make up the decidua capsularis later in pregnancy. Cells in the mesometrial region are the last to begin to undergo decidualization forming the mesometrial decidua, which is the area in which the placenta and its vascular supply develop. Although the precise molecular mechanisms involved are still being studied, molecular signals from the conceptus do not appear to be required for decidualization to occur. This is due to a large body of work, beginning first about a century ago on guinea pigs (Loeb 1908) and then on several other species (reviewed by Krehbiel 1937) that shows decidualization can occur in the absence of the conceptus. This allows one to study the details of decidualization without the confounding effects of an implanting conceptus and developing placenta. The aim of the present study was to determine the localization and levels of total and active CTNNB1 of the pregnant mouse uteri and those undergoing artificially stimulated decidualization. The levels of Cacybp gene expression in the uterus were also examined to see if its levels change during progression of decidualization.
Materials and Methods
Animals
For each experiment carried out in this study, samples were collected from three to four independent animals per tissue type and time of sampling. All procedures involving mice were approved by the Southern Illinois University Institutional Animal Care and Use Committee. Adult CD1 mice purchased from Charles River Breeding Laboratories (Wilmington, MA) were kept under controlled light conditions (lights on from 07:00 to 19:00 hr) with free access to food and water. Females were placed with fertile males and the morning a vaginal plug was detected was considered to be day 0.5 of pregnancy. Mice were killed at 09:00 hr on days 3.5 to 7.5 of pregnancy, which approximately corresponds to −1 to 3 days after the onset of decidualization, respectively. Segments of the uterus containing implanting embryos (implantation site) and not containing implanting embryos (non-implantation site) were dissected. To identify implantation sites of the uterus on day 5 of pregnancy just after the onset of implantation (Finn and McLaren 1967), 0.1 ml of Evan's blue dye (Sigma; St Louis, MO) solution (2% w/v in saline) was injected IV (tail vein) at 10 min prior to killing.
The artificial model of decidualization used in this study has been previously described in detail (Bany and Cross 2006). Briefly, animals were ovariectomized, allowed a 1-week recovery, and then injected SC with a regimen of estradiol-17β and/or progesterone (Sigma) to adequately sensitize the uterus for an artificial deciduogenic stimulus. Once sensitized for the deciduogenic stimulus, an intraluminal injection of 10-15 μl of sesame oil was used to stimulate decidualization in one uterine horn (stimulated), whereas the contralateral horn served as a control (non-stimulated). Mice were killed at exactly 0, 24, 48, or 72 hr after artificially inducing decidualization, corresponding to days 0-3 after the onset of decidualization. Non-stimulated and stimulated uterine horns were dissected.
Immunohistochemical Staining for Total and Active CTNNB1
Samples collected for immunohistochemical analyses were immersed in 4% paraformaldehyde (Fisher Scientific; Pittsburgh, PA) (w/v) in PBS (PFA-PBS) for 24 hr followed by immersion in 70% ethanol (MIDSCI; St Louis, MO) for 24 hr at 4C. Tissues were then dehydrated, cleared in xylene (Fisher Scientific), dehydrated in decreasing concentrations of ethanol, and then embedded in paraffin (Electron Microscopy Sciences; Hatfield, PA) using routine histological procedures. Cross-sections (5 μm) of the uteri were prepared and mounted onto silanized glass slides. Sections were placed in an 80C oven for 5 min, deparaffinized in xylene, rehydrated in decreasing concentrations of ethanol, and placed in water. For antigen recovery, sections were heated (95C) in citrate buffer (13 mM citric acid and 87 mM sodium citrate, pH 6.0) and then allowed to sit at room temperature for 20 min. After washing with PBS, sections were placed in PBS containing 0.05% (v/v) Tween-20 (PBST) and then were used immediately for total or active CTNNB1 immunostaining.
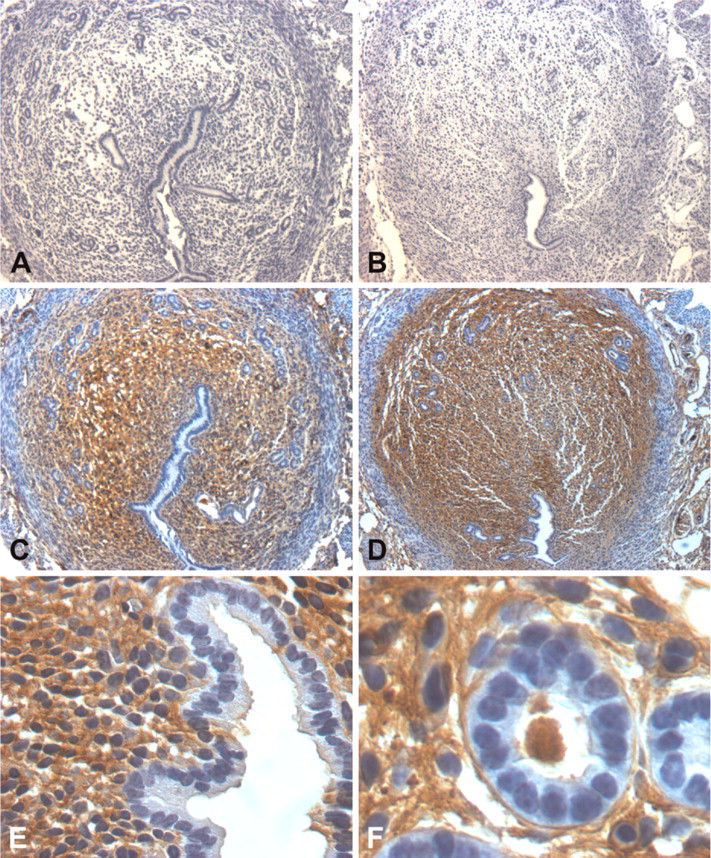
Control sections stained using the methods of Li et al. (2005) for active CTNNB1. Negative control uterine sections from non-stimulated (
Immunostaining for total CTNNB1 was carried out at room temperature and began by blocking nonspecific binding by incubating the slides for 1 hr in blocking buffer (2% goat serum in PBST). Next, the sections were incubated for 1 hr in blocking buffer containing 15 μg/ml of anti-CTNNB1 IgG (cat. #ab2982; Abcam, Cambridge, MA). After washing with PBST, sections were incubated for 1 hr in blocking buffer containing 4.5 μg/ml biotin-conjugated secondary goat anti-rabbit IgG (Vector Laboratories; Burlingame, CA). Next, sections were washed in PBST followed by incubation for 30 min in blocking buffer containing 2.5 μg/ml of streptavidin-alkaline phosphatase conjugate (Vector Laboratories). Next, to inhibit endogenous alkaline phosphatase activity, sections were washed for 5 min in a buffer containing 10 mM Tris, 150 mM NaCl, pH 7.8 (TBS) containing 0.6 mg/ml levamisole (Sigma). Vector blue substrate (Vector Laboratories) containing 1 mg/ml levamisole was then applied on the surface of each section for 5-20 min until a blue color developed. After washing with water, sections were counterstained in nuclear fast red (Vector Laboratories) and coverslipped using Fluoromount-G (Southern Biotech; Birmingham, AL) mounting medium. For negative control sections, primary antibody was replaced with the 15 μg/ml rabbit IgG (Sigma), and no staining was observed (data not shown).
Antibodies used for Western blot analyses
Immunostaining for active CTNNB1 involved the use of the Animal Research Kit exactly as directed by the manufacturer (ARK; Dako North America, Carpinteria, CA). This kit is designed to minimize or prevent reactivity of the anti-mouse secondary antibody to endogenous mouse IgG in the sections. Primary antibody used was a mouse anti-dephosphorylated (S33, S37) CTNNB1 (cat. #05-601; Upstate, Lake Placid, NY), and the chromagen utilized in the kit is 3,3′-diaminobenzidine. Sections were counterstained in Harris hematoxylin (Fisher Scientific) before coverslipping using Clarion Mounting Medium (Sigma). For immunostaining some sections, primary antibody was replaced with 1 μg/ml mouse IgG (Sigma) as a negative control. Notably, every attempt to carry out immunostaining using routine methods, which do not take into account nonspecific binding of anti-mouse secondary antibody to endogenous mouse tissue IgG, led to strong staining in the endometrial stroma in areas not undergoing decidualization with no luminal and glandular epithelial staining (Figure 1). We believe this staining represents nonspecific binding of the secondary antibody for two reasons. First, when the staining was carried out with the omission of the secondary anti-mouse IgG and preimmune IgG in its place, there was no staining (Figures 1A and 1B). Second, staining was observed when immunohistochemical staining was carried out with the omission of the mouse anti-mouse IgG and preimmune mouse IgG in its place (Figures 1C-1F).
Western Blot Analyses
Uterine tissues were homogenized in Tissue Protein Extraction Reagent (TPER; Pierce Biotechnology, Rockford, IL). The homogenates were centrifuged (12,000 × g for 20 min at 4C) and the supernatants were then collected. Concentrations of proteins in the supernatants were then determined using a BCA Protein Assay Kit (Pierce Biotechnology) according to the manufacturer's instructions. Next, protein samples (10 mg per lane) were subjected to reducing SDS-PAGE as described elsewhere (Laemmli 1970) using 12% Tris-glycine PAGEr Gold Precast gels (Pierce Biotechnology). Proteins were then transferred onto Immobilon-P membrane (Millipore; Billerica, MA) using methods described elsewhere (Towbin et al. 1979). Finally, the membrane was rinsed in TBS and used for Western blot analyses.
Except where noted, Western blot analyses were carried out at room temperature and started with the immersion of the membranes in blocking buffer containing TBST along with either 5% powdered skim milk (Kroger; Carbondale, IL) or BSA (Fisher Scientific) (see Table 1) for 3 hr with gentle shaking. The membranes were then immersed overnight in blocking buffer containing the desired primary antibody (see Table 1) or negative control IgG (Sigma) at 4C. After washing with TBST, membranes were incubated for 40 min in blocking buffer containing the appropriate peroxidase-conjugated secondary antibody (see Table 1). After washing with TBST followed by TBS, SuperSignal West Femto Maximum Sensitivity Substrate kit (Pierce Biotechnology) was used to detect the peroxidize activity in the membranes as recommended by the manufacturer. Chemiluminescent signals produced by the peroxidase activity were documented and quantified using Kodak Image Station 2000R and Kodak 1D software (Eastman Kodak Company; New Haven, CT). Signals were normalized to the mean signal level in the 0-hr samples. In the case of active and phosphorylated β-catenin, results were further normalized to the levels of total β-catenin. The resulting measures of relative levels of protein were statistically analyzed using two-way analyses of variance followed by posthoc Duncan Multiple Range tests to compare differences between means.
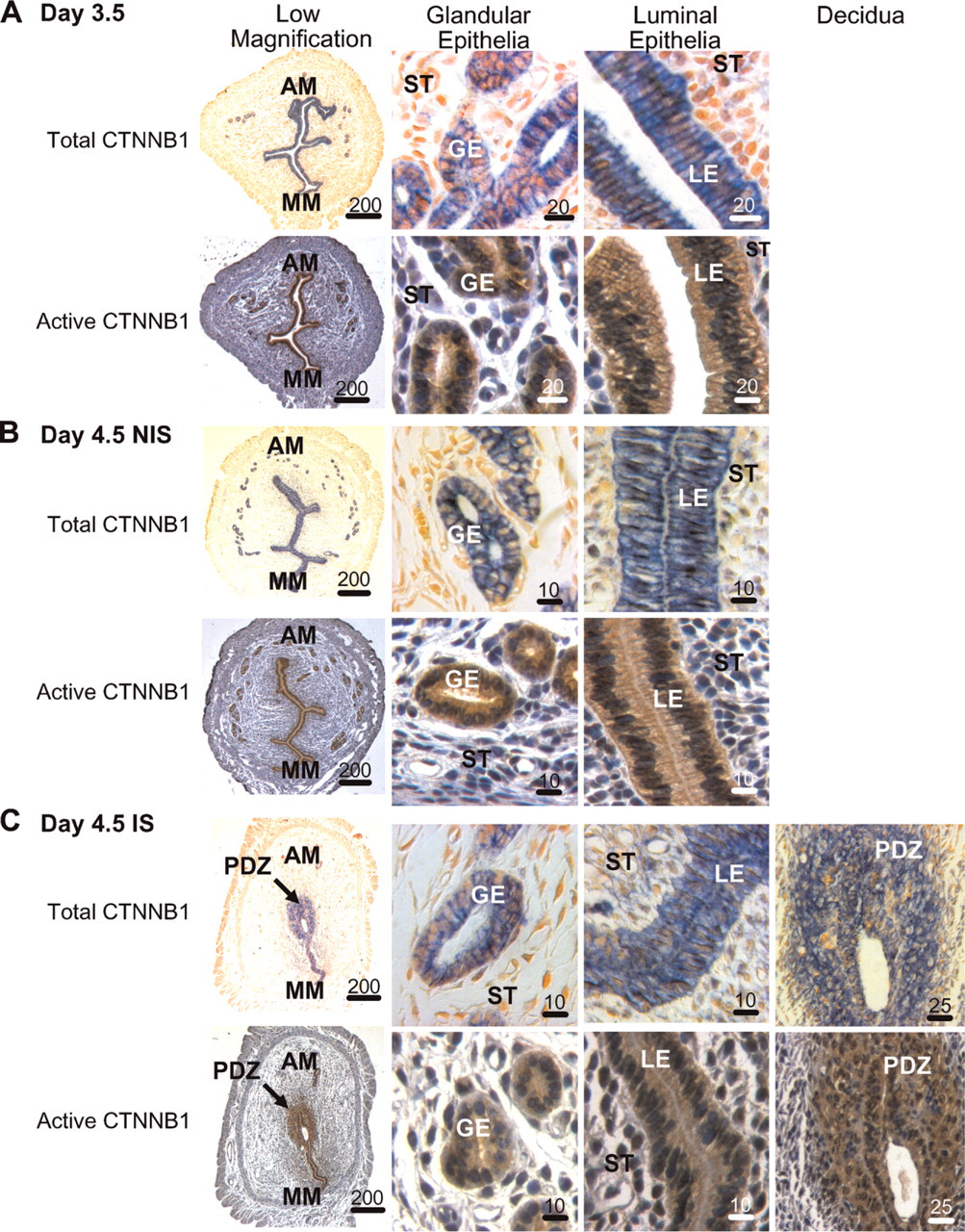
Immunohistochemical localization of total (blue) and active (brown) CTNNB1 in the mouse uterus around the time implantation begins. (
Northern Blot Analyses
Tissues were homogenized in Trizol (Invitrogen; Carlsbad, CA), and total RNA was isolated as recommended by the manufacturer. Random-primer 32P-labeling of probes and Northern blot preparation and hybridization plus washing were carried out exactly as previously described (Bany and Schultz 2001). Hybridization signals for Cacybp mRNA and 18S rRNA were evaluated using densitometry. Cacybp cDNA used was generated using routine RT-PCR (upstream primer 5′-TGATGTGGAATGCTCGTGTT-3′; downstream primer 5′-CTCCTGCATGCA-ATAAGTTCA-3′; Integrated DNA Technologies, Coralville, IA), which resulted in an 893-base pair amplicon. This amplicon was then cloned into pGEM-Teasy vector using methods recommended by the manufacturer (Promega; Madison, WI). Clones were grown in bacteria using routine methods and then isolated and purified using a Qiagen Maxiprep kit (Qiagen; Valencia, CA). Purified clones were then sequenced for verification (UIUC Keck Center for Comparative and Functional Genomics; Urbana-Champaign, IL).
Results
CTNNB1 Localization in Pregnant Uteri
Day 3.5. At a time just prior to the onset of implantation (Finn and McLaren 1967), total and active (Figure 2A) CTNNB1 were localized almost exclusively to the glandular and luminal epithelia of the endometrium. For all sample times and types, no immunostaining was seen when rabbit and mouse IgG were used in place of the primary antibodies against total and active CTNNB1, respectively (data not shown).
Day 4.5. Just after the onset of implantation (Finn and McLaren 1967), a high level of staining for total and active CTNNB1 remained in the glandular and luminal epithelia in non-implantation (Figure 2B) and implantation (Figure 2C) sites. In only the sections from implantation sites, there was an additional high level of localization for both proteins throughout the PDZ around the anti-mesometrial pole of the uterine lumen where the blastocyst stage embryo has started to implant. Total and active CTNNB1 were also localized to some cells in the endometrial stroma outside of the PDZ, especially adjacent to the luminal epithelia around the mesometrial part of the lumen.
Day 5.5. Sections from non-implantation sites of pregnant uteri from days 5.5 (Figure 3A) to 7.5 (Figure 3D) showed identical localization of total and active CTNNB1 to the glandular and luminal epithelia as seen in day 3.5 sections as well as those from day 4.5 non-implantation sites. However, their localization in implantation sites continued to change compared with day 4.5. Total and active CTNNB1 were strongly localized to the glandular and mesometrially located luminal epithelia in sections from implantation sites on day 5.5 (Figure 3B). Staining of the luminal epithelial cells adjacent to the implanting embryo either disappeared or was lighter. Both total and active CTNNB1 were also localized to the mesometrial decidua in cells adjacent to the luminal epithelia as well as in a larger region of the anti-mesometrial decidua compared with day 4.5.
Days 6.5 and 7.5. For implantation sites at day 6.5 (Figure 3C) and 7.5 (Figure 3E) of pregnancy, total and active CTNNB1 were localized to the glandular and luminal epithelia, embryo, anti-mesometrial decidua, and in parts of the mesometrial decidua. In the anti-mesometrial and mesometrial decidua, strong staining was seen especially in the decidual cells adjacent to the remaining endometrial cells not undergoing decidualization and are adjacent to the myometrium. Finally, endometrial glands and luminal epithelia were almost absent in the implantation sites at these times. However, when present, glands were localized in the undifferentiated anti-mesometrial stroma adjacent to the myometrium. In this region the epithelial, but not stromal, cells stained positive for total and active CTNNB1.
CTNNB1 Localization in Uteri Undergoing Artificially Stimulated Decidualization
Twenty-four Hr Poststimulation. Total and active CTNBB1 were localized almost exclusively to the glandular and luminal epithelia within the endometrium in sections from the non-stimulated uterine horns at 24 hr (Figure 4A). This staining was similar to that seen in the sections from non-implantation sites of the pregnant uteri and the preimplantation uterus (Figure 2 and Figure 3). In the uterine horns undergoing artificially stimulated decidualization at 24 hr, total and active CTNNB1 were strongly localized to the glandular epithelia, luminal epithelia in the mesometrial region, and in the anti-mesometrial decidual cells adjacent to lumen (Figure 4B). Unlike in the implantation sites after the onset of decidualization, luminal epithelial cells in the anti-mesometrial region remain, but instead of being columnar as the ones located mesometrially, they appear cuboidal in shape and stain only weakly for total CTNNB1.
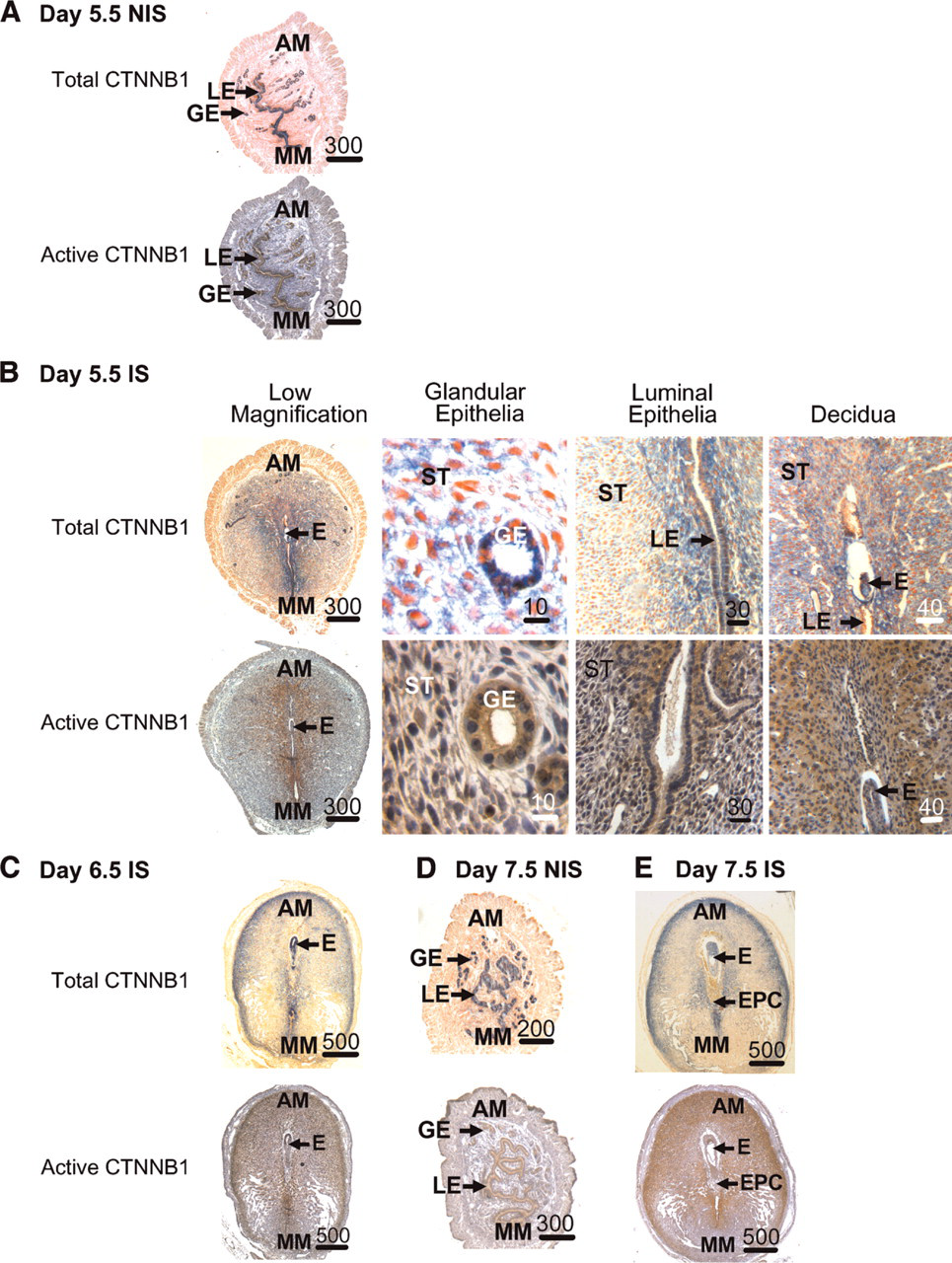
Immunohistochemical localization of total (blue) and active (brown) CTNNB1 in mouse uteri during the progression of decidualization. Localization of total and active CTNNB1 in non-implantation sites (NIS) on days 5.5 (
Forty-eight and 72 Hr Poststimulation. Total and active CTNBB1 were localized almost exclusively to the glandular and luminal epithelia within the endometrium in sections from the non-stimulated uterine horns (Figure 4D). By 48 hr (Figure 4C) and 72 hr (Figure 4E), staining for total and active CTNNB1 in stimulated horns continued in the glandular and luminal epithelia. However, as in the implantation sites, the numbers of endometrial glands were almost absent in the stimulated uterine horns at these times but, if present, were found in the undifferentiated anti-mesometrial stroma adjacent to the myometrium. Similar, but not identical, localization patterns of total and active CTNNB1 were seen between the artificially stimulated deciduomas at 48 hr and 72 hr compared with that at 24 hr after the onset of decidualization. More decidual cells stained for total and active CTNNB1 when compared with that at 24 hr. Similar to the deciduas on days 6.5 and 7.5 of pregnancy, glandular epithelia were found in the deciduomas at 48 hr and 72 hr in the areas of the endometrium just adjacent to the myometrium where the stromal cells do not undergo decidualization. In this region the epithelial cells, but not stromal cells, stained positive for total and active CTNNB1.
Active and Phosphorylated Forms of CTNNB1 Levels Change in the Uterus During Decidualization
As shown in Figure 5, all forms (total, active, and phosphorylated) of CTNNB1 could be detected in protein extracts from non-stimulated and stimulated uterine horns at 0, 24, 48, and 72 hr after artificially stimulating decidualization. The level of total CTNNB1 was not significantly (p>0.05) different between non-stimulated and stimulated uterine horns on each day examined. However, levels of active CTNNB1 were significantly (p<0.05) greater in the non-stimulated compared with stimulated uterine horns at 24 and 72 hr. Finally, the level of phosphorylated CTNNB1 was significantly (p<0.05) greater in the stimulated compared with non-stimulated uterine horns at 24, 48, and 72 hr. No bands were seen when Western blot analyses were carried out using IgG or serum in place of the CTNNB1 or ACTB antibodies, respectively (data not shown).
Steady-state Cacybp mRNA Levels in the Uterus During Decidualization
As shown in Figure 6, Cacybp mRNA was detected in non-stimulated and stimulated uterine horns at 24, 48, and 72 hr after the onset of decidualization. The level of this mRNA was significantly (p<0.05) greater in the stimulated horn compared with non-simulated horns at 24 hr, but not at 48 and 72 hr.
Discussion
CTNNB1 likely plays a role in cell adhesion in the endometrium during decidualization. One of the roles of CTNNB1 is to be part of the anchor whereby the cytoskeleton is connected to cadherins, which are involved in cell-cell adherens junctions (Kemler 1993). The luminal and glandular epithelia of the mouse endometrium is a major site of E-cadherin gene (Cdh1) expression under various physiological states (Potter et al. 1996; Paria et al. 1999; Jha et al. 2006). During pregnancy, Cdh1 gene expression is found mainly in the luminal and glandular epithelia just prior to and after the onset of implantation (Paria et al. 1999). However, there is also additional expression in the primary decidual zone. Similarly, the present study shows that CTNNB1 is localized to the glandular and luminal epithelial just before and after the onset of implantation with the added expression in the primary decidual zone. Further, during the progression of decidualization on days 6.5 and 7.5 of pregnancy, Cdh1 expression is found in the secondary decidual zone, subepithelial stroma in the anti-mesometrial region, and also in the epithelial cells (Paria et al. 1999). Again, the localization of CTNNB1 in the present study followed the same localization. Therefore, during the peri-implantation period on days 3.5-7.5 of pregnancy, localization of level of expression of CTNNB1 correlates well with the localization of Cdh1 expression. This provides support to the idea that one of the key roles of CTNNB1 in the endometrium during implantation might be to be a part of the anchor between the cytoskeleton and membrane E-cadherin protein (CDH1) of endometrial cells. However, the exact importance of this remains uncertain in the context of implantation and decidualization. In addition to playing a role in homotypic adherens junctions between endometrial cells, it has been suggested that CDH1 might also interact with the implanting conceptus and guide trophoblast invasion into the uterus (Paria et al. 1999).
The N-terminal region of CTNNB1 contains three amino acids whose phosphorylation status determines the fate of the molecule. When CTNNB1 is phosphorylated at two of them (S33 and S37) by GSK3β, BTRC is able to bind to CTNNB1, which results in its ubiquitination and then degradation by the proteasome (Kitagawa et al. 1999; Winston et al. 1999). One of the antibodies used in the present study was raised against a peptide consisting of CTNBB1 residues 27-37 and recognizes the protein only when dephosphorylated at S33 and S37. This form of CTNNB1, called active CTNNB1, is not targeted for degradation and can enter the nucleus and act as a cotranscriptional activator or take part in cell adhesion junctions (Gottardi and Gumbiner 2004a). In the present study, active CTNNB1 appears in the endometrial stromal cells undergoing decidualization in pregnant uteri and those undergoing artificially stimulated decidualization. It was not localized to stromal cells in areas of these uteri not undergoing decidualization. This suggests CTNNB1 localization and function dramatically change in areas of the mouse uterus undergoing decidualization. Further, because GSK3β activity is responsible for the phosphorylation status of CTNNB1, results of the present study imply that its overall activity also changes in the endometrial stroma during decidualization. Although its function in endometrium during decidualization remains to be completely elucidated, results of this study suggest that GSK3β is a regulator of CTNNB1 degradation in the uterus during decidualization in the mouse uterus.
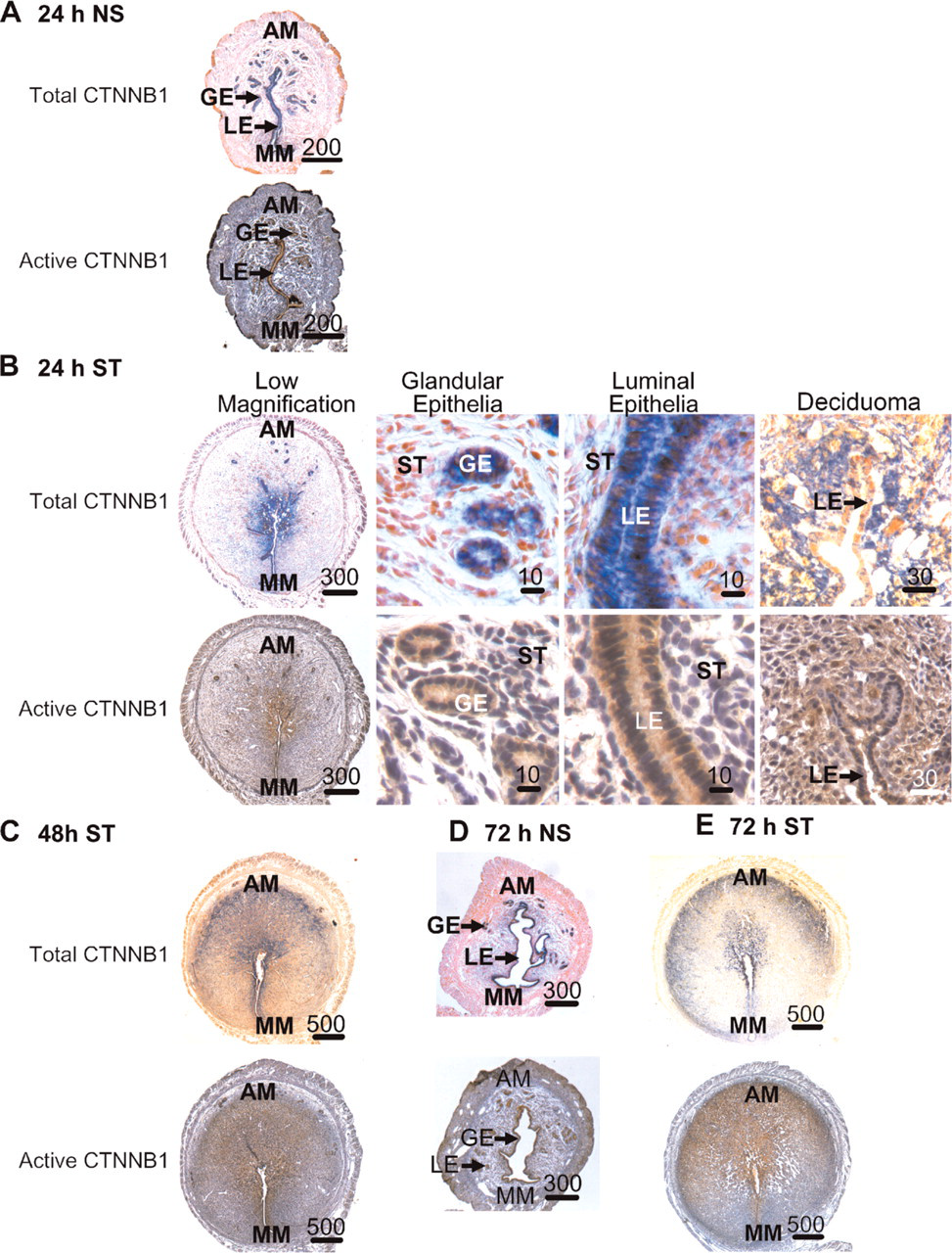
Immunohistochemical localization of total (blue) and active (brown) CTNNB1 in mouse uteri during artificially stimulated decidualization. Localization of total and active CTNNB1 in non-stimulated (NS) uterine horns 24 hr (
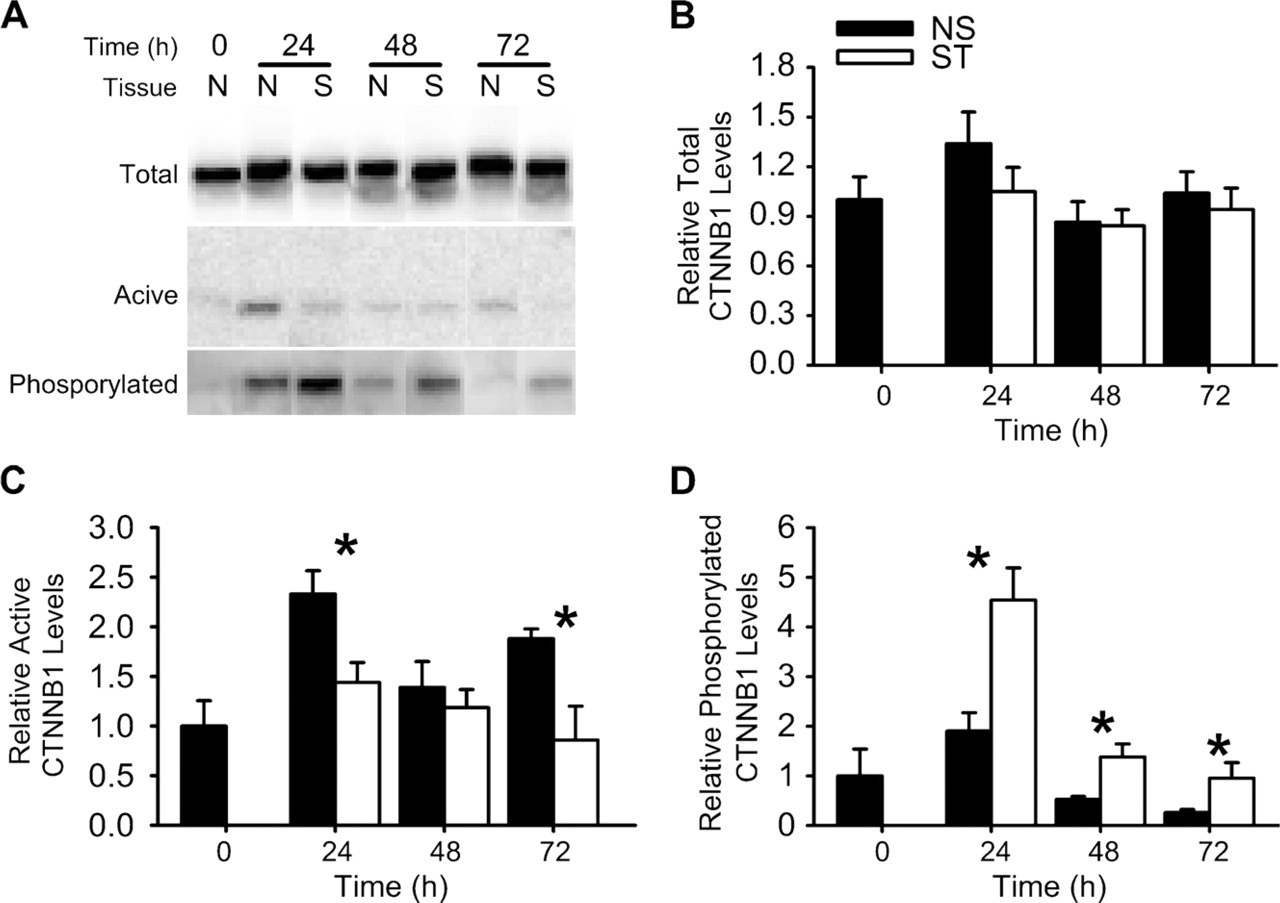
Western blot analyses of CTNNB1 in the mouse uterus during artificially stimulated decidualization. (
Recently, a new pathway was identified that targets CTNNB1 degradation and is independent of GSK3β. This new pathway involves a novel E3-ligase complex that contains CACYBP and is not regulated by the phosphorylation status of S33 and S37 of CTNNB1 (Matsuzawa and Reed 2001; Matsuzawa et al. 2003). Cacybp gene expression in the mouse uterus has recently been demonstrated in the luminal and glandular epithelia during the peri-implantation period with the appearance of expression in endometrial stromal cells undergoing decidualization (Yang et al. 2006). In the same study, Cacybp antisense oligodeoxynucleotides inhibited implantation in vivo (Yang et al. 2006) and anti-apoptotic effects on cultured endometrial stromal cells. In the present study we found that the level of Cacybp expression in uterine horns undergoing artificially stimulated decidualization was greater than contralateral control horns not undergoing decidualization soon after the onset of decidualization. Further, this gene was expressed in all uterine samples examined. Although its exact function in the endometrium during decidualization remains to be determined, our results and those of others (Yang et al. 2006) clearly indicate that CACYBP is in the uterus during the process of decidualization. It is tempting to speculate that this protein plays a role in CTNNB1 ubiquitination in the uterus at this time because it is already known to perform such a role in other cell types.
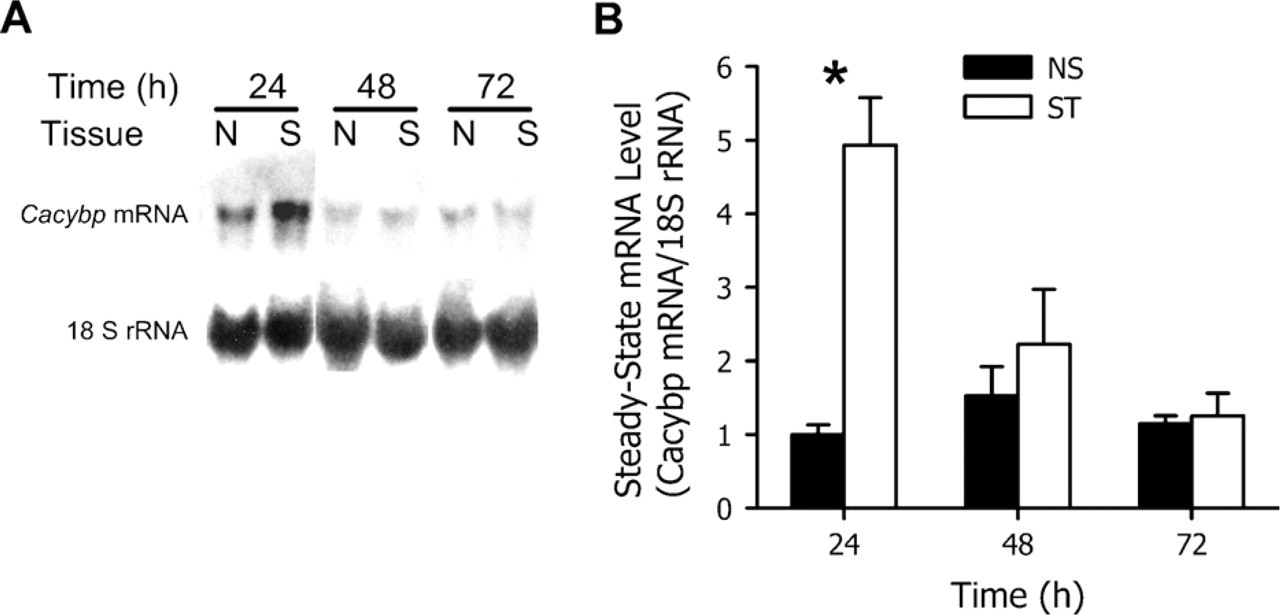
Northern blot analysis of the steady-state Cacybp mRNA levels in the mouse uterus during artificially stimulated decidualization in the non-stimulated (NS) and stimulated (ST) uterine horns at 24, 48, and 72 hr after the onset of decidualization. Representative autoradiograms (
Little is known about CTNNB1 function and regulation in the uterus during decidualization. Mohamed et al. (2005) recently determined that uterine Wnt/CTNNB1 signaling is required for implantation. Their experiments were carried out on transgenic LEF-reporter mice, and areas of reporter expression were interpreted as being sites of CTNNB1 nuclear signaling. It has recently been pointed out that one should be cautious about interpreting results of data using such transgenic reporters in general, including the one used in their study with regard to in vivo Wnt responsiveness (Barolo 2006). This may explain, in part, the discrepancies between the localization of reporter activity only to the luminal epithelium on day 4.5 of pregnancy in their study compared with the localization of active CTNNB1 to both the epithelia and primary decidual zone in the present study. Further, localization of active CTNNB1 in the present study in no way proves it enters the nuclei of cells to act as a cotranscriptional activator. Active CTNNB1 can also participate in cell adhesion junctions (Gottardi and Gumbiner 2004a). Indeed, recent evidence suggests that the regulation of the subcellular localization of CTNNB1 might be another level by which this signaling system is regulated (Krieghoff et al. 2006). More work is required to clearly determine the role and regulation of CTNNB1 in the mouse uterus during the progression of decidualization. However, given that the localization of both total and active CTNNB1 were similar in the pregnant uteri and those undergoing artificially stimulated decidualization, it is possible that signals from the conceptus might not regulate its function in the uterus during decidualization.
Immunohistochemical localization of active β-catenin within the mouse uterus during the peri-implantation period in this study contradicts recent findings (Li et al. 2005) where the identical antibody was utilized. As mentioned in Materials and Methods, the use of routine indirect immunostaining procedures on mouse tissues utilizing an antibody raised in mice requires special attention so that the staining seen is not background binding of the secondary antibody to endogenous IgG. Although Li et al. (2005) reported that they conducted preimmune IgG controls and got no staining, we did get staining. In fact, this staining was similar to what they found for active β-catenin. We cannot explain this discrepancy with regard to the IgG controls. However, when using methods to eliminate background binding of the secondary antibody to endogenous mouse IgG, control sections had no staining in the present work. Notably, the staining pattern of Li et al. (2005) looks similar to that found for IgG localization (Bernard et al. 1981; Rachman et al. 1986) in the mouse uterus to endometrial stromal and not luminal or glandular epithelial cells. Further, there is good evidence that endogenous IgG is absent from the primary decidual zone (Parr and Parr 1989), an area where Li et al. (2005) did not find localization. Because we used a special method of using mouse antibodies on mouse sections and found no staining using IgG controls and localized much of the active β-catenin to places where there is little to no endogenous IgG (luminal plus glandular epithelia and primary decidual zone), we are convinced that the staining seen in this study is specific for active β-catenin.
In conclusion, results of this study show the changing localization and levels of total and active β-catenin in the uterus during decidualization and provide evidence that both of the E3 ligase mechanisms known to regulate its degradation might play important roles in the endometrium during decidualization.
Footnotes
Acknowledgements
This work was supported by an Office of Research Development and Administration (ORDA) Faculty Seed Grant from Southern Illinois University-Carbondale.
