Abstract
The ontogeny of the innervation of human lymphoid organs has not been studied in detail. Our aim was to assess the nature and distribution of parenchymal nerves in human fetal thymus and spleen. We used the peroxidase immunohistochemical technique with antibodies specific to neuron-specific enolase (NSE), neurofilaments (NF), PGP9.5, S100 protein, and tyrosine hydroxylase (TH) and evaluated our results with image analysis. In human fetal thymus, NSE-, NF-, S100-, PGP9.5-, and TH-positive nerves were identified associated with large blood vessels from 18 gestational weeks (gw) onwards, increasing in density during development. Their branches penetrated the septal areas at 20 gw, reaching the cortex and the corticomedullary junction between 20 and 23 gw. Few nerve fibers were seen in the medulla in close association with Hassall's corpuscles. In human fetal spleen, NSE-, NF-, S100-, PGP9.5-, and TH-positive nerve fibers were localized in the connective tissue surrounding the splenic artery at 18 gw. Perivascular NSE-, NF-, S100-, PGP9.5-, and TH-positive nerve fibers were seen extending into the white pulp, mainly in association with the central artery and its branches, increasing in density during gestation. Scattered NSE-, NF-, S100-, PGP9.5-, and TH-positive nerve fibers and endings were localized in the red pulp from 18 gw onward. The predominant perivascular distribution of most parenchymal nerves implies that thymic and splenic innervation may play an important functional role during intrauterine life.
T
Evidence that lymphoid organs are directly innervated was first provided by Tonkoff as early as 1899 (Tonkoff 1899), when nerve fibers were observed extending into lymph nodes. The adult lymphoid organs are directly innervated mainly by the sympathetic nervous system, which plays a functional role in the immune system regulation in all species studied (Zetterstrom et al. 1973; Reilly et al. 1976; Giron et al. 1980; Williams and Felten 1981; Bulloch and Pomerantz 1984; Kendall et al. 1988; Felten et al. 1992; Felten 1993; Friedman and Irwin 1997).
In a variety of adult mammalian species, sympathetic noradrenergic innervation of lymphoid organs, including the thymus and spleen, is associated with the vasculature and is generally directed into zones of lymphocytes, mainly T cells (Felten et al. 1985; Kendall and al-Shawaf 1991; Kranz et al. 1997; Cavallotti et al. 1999).
In the adult human and rat thymus, postganglionic sympathetic nerve fibers extend from nerve bundles and plexuses around large blood vessels, travel into the thymic capsule and septa, and further branch into the parenchyma (Felten et al. 1985; Kendall 1989; de Leeuw et al. 1992; Vizi et al. 1995; Kranz et al. 1997; Cavallotti et al. 1999). Unaccompanied adrenergic nerve fibers have been observed in the cortical thymic parenchyma (Vizi et al. 1995) but are extremely sparse in the medulla (Felten et al. 1985; Kranz et al. 1997). Immunohistochemical studies have revealed the immunoreactivity of tyrosine hydroxylase (TH), an adrenergic marker, in association with T-lymphocytes, mast cells, and thymic epithelial cells (Vizi et al. 1995; Kurz et al. 1997).
In the spleen, intrinsic nerves are in their great majority part of the sympathetic nervous system (Heusermann and Stutte 1977; Kudoh et al. 1979; Klein et al. 1982; Heusermann 1988). In the adult human and rat spleen, noradrenergic postganglionic nerves enter the organ accompanying the splenic artery and run along the trabeculae, forming plexuses (Heusermann and Stutte 1977; Kudoh et al. 1979; Williams and Felten 1981; Heusermann 1988). They are further distributed mainly in the white pulp along the central artery, where they reach their greatest density and end up in the periarterial lymphatic sheath (Heusermann and Stutte 1977; Kudoh et al. 1979; Williams and Felten 1981; Felten et al. 1985; Heusermann 1988).
Although anatomical studies have partly revealed the origin, pattern of distribution, and targets of nerve fibers in the human adult lymphoid organs, the ontogeny of the innervation of human lymphoid organs has not been studied in detail. The aim of this study was to assess the nature and distribution of parenchymal nerves in human thymus and spleen during fetal life.
Materials and Methods
Clinical Data
Tissue samples from 23 human fetal thymuses (14 of 18–24 gestational weeks (gw) and 9 of 25–39 gw), 10 human fetal spleens (6 of 18–24 gw and 4 of 25–39 gw), and a human adult spleen were included in the study. Fetal thymus and spleen tissue samples were retrieved from the files of the Pathology Department of Athens Medical School, from October 1999 to November 2003, with the approval of the Ethics Committee of the National and Kapodistrian University of Athens and in accordance with the ethics standards described by the Helsinki Declaration of the World Medical Association (2000). All fetuses were aborted spontaneously. On autopsy, no fetal malformations or other pathology were detected. The parents' informed consent for the use of fetal tissue material for research was obtained for each case prior to inclusion in the study. Adult patients approved the use of nondiagnostic tissue from surgical specimens for research purposes.
Immunohistochemistry
The streptavidin-biotin immunohistochemical technique was performed on samples fixed in 10% neutral formalin and embedded in paraffin. The specific antibodies for pan-neural markers and the adrenergic marker TH that were used are shown in Table 1.
Five-μm-thick sections were dewaxed and rehydrated. Endogenous peroxidase was quenched with 0.9% H2O2 in methanol for 30 min. Antigen unmasking was performed in 0.01 M citrate buffer (pH 6.0) for three 4-min cycles in a household microwave oven (800 W). Sections were incubated overnight with mouse monoclonal anti-human antibodies to NF, NSE, PGP9.5, TH, and the rabbit polyclonal anti-human S100 antibody (Table 1), followed by the streptavidin-biotin peroxidase complex (Biogenex; San Ramon, CA). DAB was used as chromogen, and counterstaining was done with hematoxylin. Negative control sections, in which the primary antibody was omitted, were used for each case in every immunostaining run. Normal adrenal gland tissue sections were used as positive controls.
Computer-assisted Morphometric Analysis of Intrinsic Immunoreactive Nerve Fibers and Cells
Morphometric variables were determined by image analysis, using an IBM computer (IBM Computer, Inc.; Armonk, NY). Images were captured with a Leica DM LB microscope coupled to a Sony digital camera (DFW-X700) and digitalized with Path-Sight version 4.3 (Medical Solutions plc.; Nottingham, UK). Three hot spot areas per section were analyzed using Image Pro Plus (Media Cybernetics, Inc.; Silver Spring, MD). The computerized system measured the number of nerve fibers and immunoreactive cells per square millimeter in each of three fields of 10 × objective/section/region.
Statistical Analysis
All statistical analyses were done using the SPSS software program (version 14.0 for Windows; SPSS, Inc., Chicago, IL). Sample distribution was considered normal, and an independent t-test was used to evaluate the differences in number of nerve fibers between the second and third trimesters of gestation. The number of immunoreactive cells in thymic cortex and medulla were compared with a paired t-test. All p values were based on two-sided testing, and differences were considered significant at p<0.05.
Results
We investigated the immunohistochemical expression of the pan-neural markers NSE, NF, PGP9.5, and protein S100, and the adrenergic marker TH in human fetal thymus and spleen and in human adult spleen. In all cases of immunopositive nerves, the immunohistochemistry reaction was localized within the axoplasm.
Panel of antibodies used
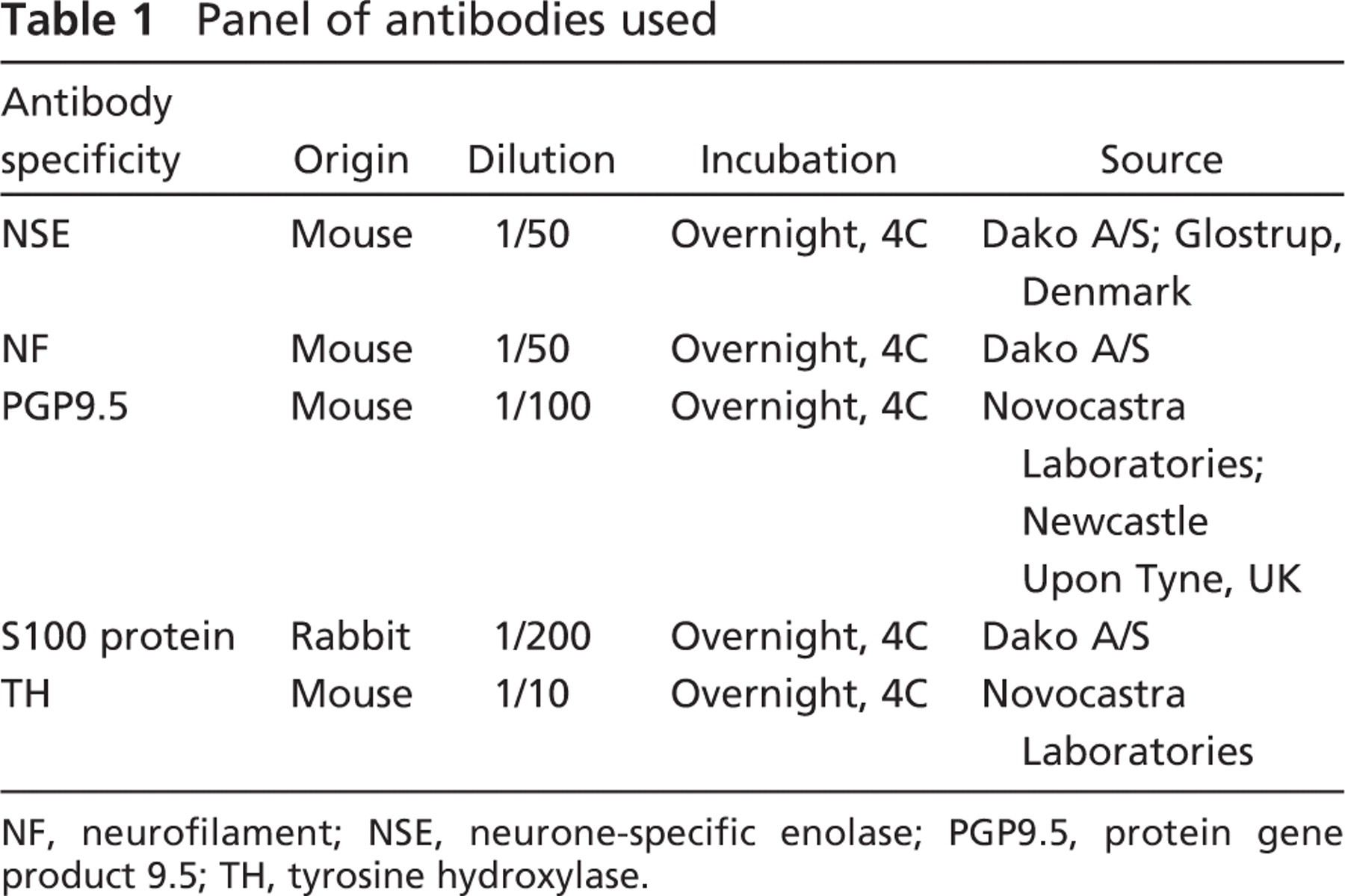
NF, neurofilament; NSE, neurone-specific enolase; PGP9.5, protein gene product 9.5; TH, tyrosine hydroxylase.
Human Fetal Thymus
Immunoreactivity for Pan-neural Markers. The pattern of distribution and density of intraparenchymal thymic nerve fibers as evaluated by image analysis are presented in detail in Table 2. Nerve bundles showing immunopositivity for NSE, NF, protein S100, and PGP9.5 were identified associated with large blood vessels from 18 gw onwards (Figure 1A). At 20 gw, their branches traveled in the capsule [mean density (MD) 0.29, 0.32, 0.36, and 0.13 nerve fibers/mm2, respectively], penetrated the septal areas (MD 0.55, 0.31, 0.46, and 0.23, respectively), and ran close to intralobular arterioles and venules, reaching the cortex (MD 0.41, 0.18, 0.20, and 0.16, respectively) and the corticomedullary junction (MD 0.39, 0.15, 0.12, and 0.00, respectively) between 20 and 23 gw (Figure 1B). Nerve fibers were also seen in the medulla (MD 0.45, 0.24, 0.17, and 0.00, respectively), in close association with Hassall's corpuscles. An increase in density of intraparenchymal nerve fibers was observed with all markers, as evidenced by the p values shown in Table 2.
NSE-, PGP9.5-, and S100-immunoreactive thymic epithelial cells were detected in the cortex [mean cortical density (MCD) 0.09, 0.08, and 0.38, respectively], and immunoreactivity for NSE and S100 was also present in medullary epithelial thymic cells [mean medullary density (MMD) 0.41 and 1.08 cells/mm2, respectively] as early as 20 gw (Table 3). The majority of the latter were strongly immunostained with an intensity similar to that displayed by the immunoreactive nerve fibers, and increased in density with gestation (Figure 1C), as shown in Table 3. Interestingly, most of the S100- and NSE-positive cells were localized in the medulla in both the second (MMD 1.08 vs MCD 0.38, p = 0.07 for S100 and MMD 0.41 vs MCD 0.09, p = 10−6 for NSE) and the third trimester (MMD 0.73 vs MCD 0.32, p = 4 × 10−5 for S100, and MMD 1.03 vs MCD 0.06, p = 9 × 10−6 for NSE), whereas PGP9.5-positive cells were preferably distributed in the cortex in both trimesters (MCD 0.08 vs MMD 0.00, p = 3 × 10−4 for the second and MCD 0.14 vs MMD 0.00, p = 2 × 10−5 for the third trimester) (Table 3).
Immunoreactivity for TH. Most of the human fetal thymic nerves were positive for the adrenergic marker TH. The pattern of distribution, as well as the density of the adrenergic innervation, was similar to those highlighted by the pan-neural markers (Table 4). Specifically, TH-positive nerve bundles and fibers were immunolocalized close to thymic large blood vessels from 18 gw onwards (Figure 1D), and their density increased during gestation (Table 4). TH-immunoreactive nerve fibers were seen in the thymic capsule (MD 0.69) and septa (MD 0.43), as well as extending further into the cortex (MD 0.12) in association with the vasculature as early as 20 gw. At 20 gw, TH-specific immunoreactive nerve fibers were also present in the medulla (MD 0.10); however, they appeared less abundant than the cortical nerve fibers. Additionally, TH immunoreactivity was observed in cortical and, to a lesser extent, in medullary epithelial thymic cells (MCD 0.46 vs MMD 0.13, p = 6 × 10−7 in the third trimester) (Table 3).
Distribution and density (nerve fibers/mm2) of pan-neural marker–positive nerve fibers in human fetal thymus using image analysis
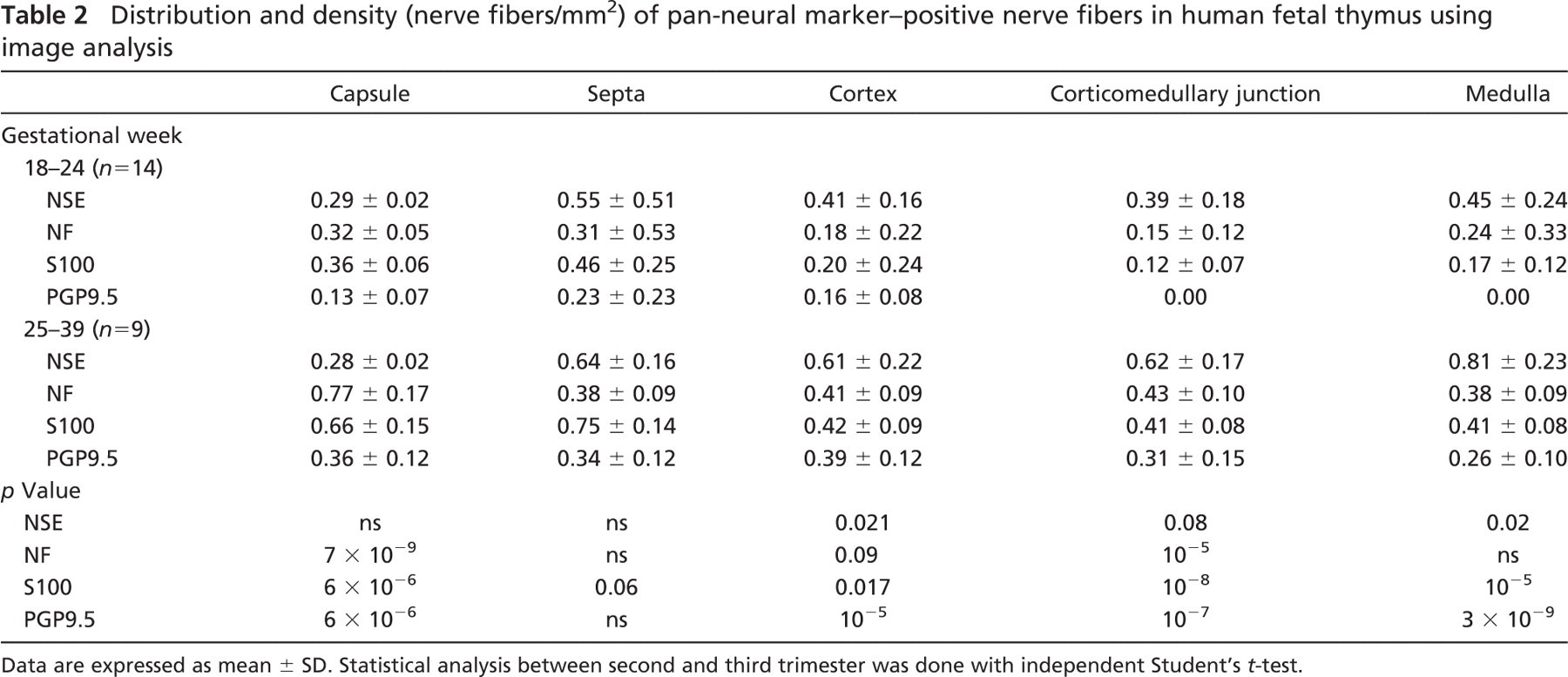
Data are expressed as mean ± SD. Statistical analysis between second and third trimester was done with independent Student's t-test.
Human Fetal Spleen
Immunoreactivity for Pan-neural Markers. In human fetal spleen, immunoreactive intraparenchymal nerves showed a predominantly perivascular distribution, and increased in density during gestation (Table 5). NSE-, NF-, S100-, and PGP9.5-positive nerve bundles entered the spleen in association with the splenic artery and its branches at 18 gw (Figure 1E) and extended into the capsule (MD 0.36, 0.05, 0.09, and 0.08, respectively) and the trabeculae (MD 0.59, 0.05, 0.56, and 0.00, respectively) (Figure 1F). Later in the second trimester, perivascular NSE-, and S100-positive nerve fibers were seen extending further into the white pulp, mainly in association with the central artery (MD 0.60 and 0.56, respectively) and its branches, to innervate the periarterial lymphatic sheath and the marginal zone. Scattered NSE-, NF-, S100-, and PGP9.5-positive nerve fibers and endings were localized in the red pulp in the vicinity of the arterial capillaries and the splenic sinuses from 18 gw onward (Table 5). S100 immunoreactivity was also observed in macrophage-like cells in the white pulp at 20 gw. The density of intraparenchymal splenic nerves seemed to increase with gestation; however, no statistical analysis was performed, because of the small number of cases studied.
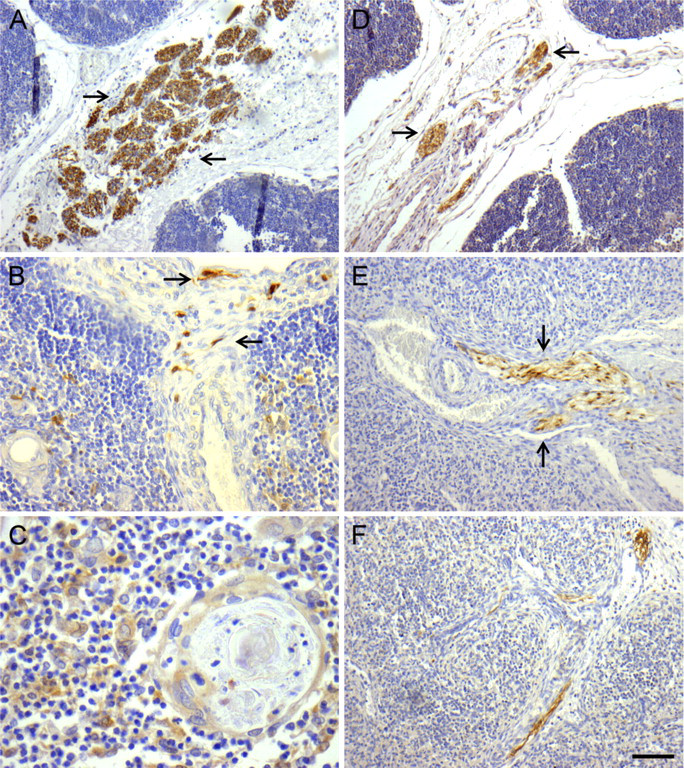
Localization of neural markers-specific immunoreactivity in human fetal thymus and spleen. (
Pattern of distribution and density (cells/mm2) of immunoreactive epithelial thymic cells in the human thymic fetal cortex and medulla using image analysis
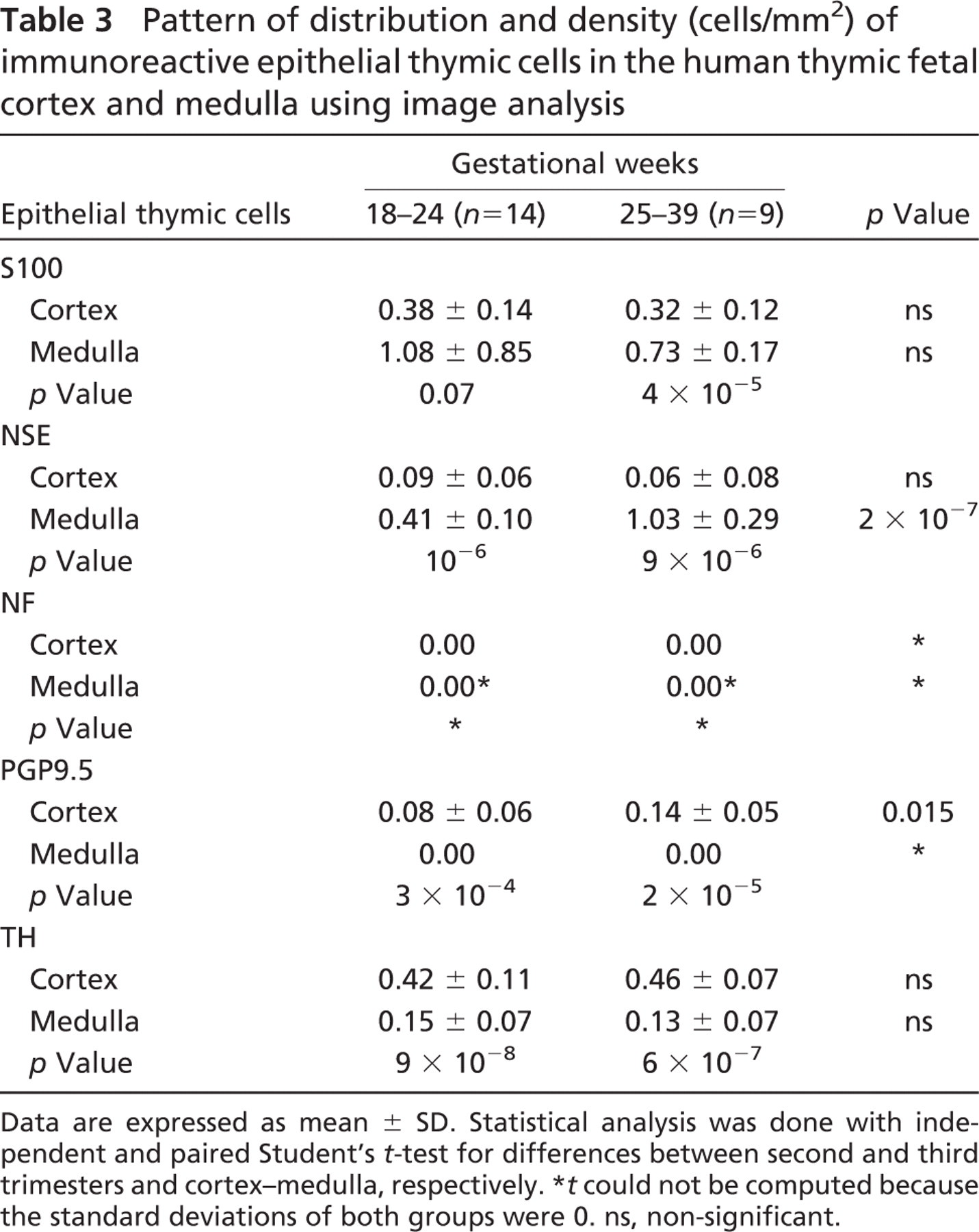
Data are expressed as mean ± SD. Statistical analysis was done with independent and paired Student's t-test for differences between second and third trimesters and cortex–medulla, respectively.
t could not be computed because the standard deviations of both groups were 0. ns, non-significant.
Distribution and density (nerve fibers/mm2) of TH-positive nerves in the human fetal thymus using image analysis
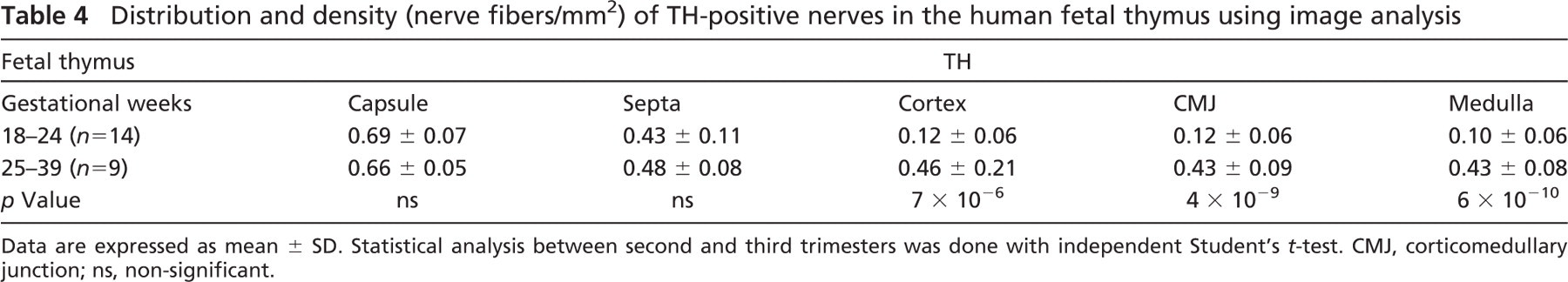
Data are expressed as mean ± SD. Statistical analysis between second and third trimesters was done with independent Student's t-test. CMJ, corticomedullary junction; ns, non-significant.
Density (nerve fibers/mm2) and distribution pattern of pan-neural markers and tyrosine hydroxylase–positive nerves in the human fetal spleen using image analysis
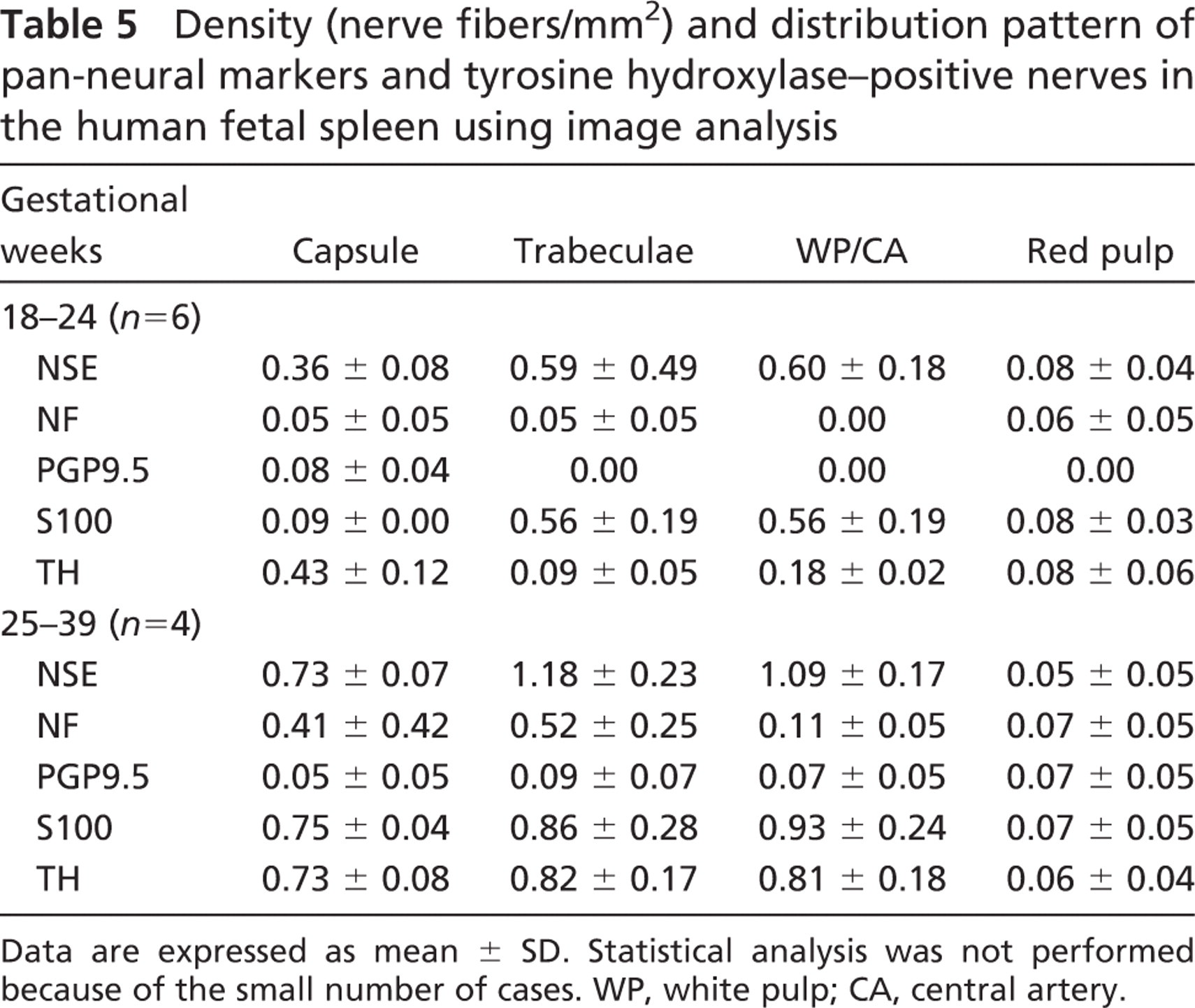
Data are expressed as mean ± SD. Statistical analysis was not performed because of the small number of cases. WP, white pulp; CA, central artery.
Adrenergic Innervation. Most of the intraparenchymal splenic nerves were adrenergic and showed a predominantly perivascular distribution. At 20 gw, TH-immunoreactive nerve fibers were seen extending from the splenic capsule (MD 0.43) to the red pulp (MD 0.08) and the white pulp (MD 0.18), mainly associated with the central artery. Their density increased during development, as shown in Table 5. TH-positive cells were also observed in the white pulp during the third trimester.
Human Adult Spleen
In human adult spleen, scarce small nerve fibers showing weak immunoreactivity with both pan-neural and andrenergic markers were noted in association with the central artery, whereas no immunostaining was detected in the red pulp.
Discussion
Anatomical studies have revealed an extensive intraparenchymal innervation of the human adult primary and secondary lymphoid organs (Heusermann and Stutte 1977; Kudoh et al. 1979; Heusermann 1988; Kendall 1989; de Leeuw et al. 1992). Data on the innervation of human fetal lymphoid organs are, however, limited, with only two reports in the literature, to the best of our knowledge.
Hammar (1935) first detected the presence of silver-stained neural profiles in the developing human thymus, branching into the parenchyma and forming a plexus in the medulla by 16–20 gw. Ghali et al. (1980) subsequently showed that nerves are mainly associated with the vasculature and further distribute into the cortex and medulla of the pre-and postnatal human thymus. These data have not been confirmed to date, and most of our knowledge is based on animal models (Ackerman et al. 1987a; Felten et al. 1992; Vizi et al. 1995).
We report a predominantly perivascular intraparenchymal innervation of the human fetal thymus, with nerve fibers extending from large hilar plexuses, traveling into the capsule and septa and further penetrating the cortex and, to a lesser extent, the medulla. Our findings are in agreement with the perivascular distribution pattern and the early presence of adrenergic innervation in specific compartments of the thymus and spleen during critical periods of development, reported in other species (Felten et al. 1992; Blennerhassett and Bienenstock 1998). The latter may suggest a possible role of the sympathetic nervous system in blood flow control and lymphocyte traffic as well as in maturation of the immune system.
In the human fetal spleen, we observed nerve fibers in the proximity of hilar splenic vessels irradiating into the parenchyma, associated mainly with the vasculature and ending into the periarteriolar lymphatic sheath. In the rat spleen, the adult pattern of sympathetic innervation persists through 12 months of age (Bellinger et al. 1987). With normal aging, a decline in the density of the noradrenergic innervation is observed, following the loss of T-lymphocytes in the periarteriolar lymphatic sheaths and that of macrophages of the marginal zone (Bellinger et al. 1992). Interestingly, a decline in T cell–mediated immunity is observed at the same time. This phenomenon has not been documented in the human adult spleen. Our results show that the density of intraparenchymal nerves is greater in the fetal spleen compared with the adult. It is clear that further studies using material from different periods of development are needed to assess the full spectrum of the evolution of human spleen innervation.
Only a few data are available on the presence of neural marker–positive cells in human fetal lymphoid organs. In our study, cells displaying specific immunoreactivity for nerve markers were localized in distinct areas of the fetal thymus and spleen, which differ from each other in their cellular composition and physiological role.
We report a predominantly medullary localization of S100-positive cells within the human fetal thymus in both the second and third trimesters of gestation. The majority of them, with their large size and rounded morphology, are indicative of an epithelial origin. This is the first time that S100 immunoreactivity in the thymic medulla in both isolated and epithelial cells of Hassall's corpuscles has been reported. However, it is possible that a subset of these cells with a more dendritic morphology are similar to the S100-positive dendritic cells described by Uccini et al. (1986) and Savchenko et al. (2006). Savschenko et al. showed that S100-positive dendritic cells are present in the corticomedullary region and the medulla of the human thymus at an early stage of gestation, suggesting that S100-positive cells are candidates for the thymic dendritic cell precursor. In the human fetal lymphoreticular system, S100 protein has also been detected in monocytemacrophages, nonphagocytic histiocytes, and in some CD8+ T cells (Akagi et al. 1989). Akagi et al. (1989) investigated the ontogeny of these S100-expressing cells in humans and noted that they were first detected in the thymus at 12 gw and were then subsequently observed in the other lymphoid organs.
We demonstrated a higher density of NSE-positive cells in the thymic medulla in the second and third trimesters of gestation. In the adult rat thymus, NSE-positive cells reside preferably in the medulla and represent subtypes of thymic epithelial cells (Brelinska et al. 2000). An ultrastructural study of rat medullary epithelial cells has shown that these distinct subtypes of NSE-positive thymic epithelial cells represent sequential stages of medullary cell proliferation (De Waal and Rademakers 1997).
The distribution of PGP9.5-positive nerves differs between the cortex and medulla of adult rat (Brelinska et al. 2000) and human thymus (de Leeuw et al. 1992). PGP9.5-specific immunostaining is prominent in the inner cortex, with a dendritic labeling pattern compatible with the epithelial network, whereas in the medulla, it is sparse and detected mainly in the vicinity of Hassall's corpuscles. Interestingly, NF 160 kDa immunoreactivity has been detected mainly in the medulla of the adult thymus (de Leeuw et al. 1992). We did not observe any differences in the distribution pattern of PGP9.5- and NF-immunoreactive cells in the different compartments of fetal thymus.
Recent data suggest that lymphocytes may express TH and can thus synthesize catecholamines, especially when activated (Qiu et al. 2004). According to Kranz et al. (1997), TH-specific immunoreactivity has been identified in thymic cortical epithelial cells of children, whereas only a few TH-positive cells have been found in the medulla. The participation of these immunopositive cells in immune responses remains to be defined. They may be part of a specialized environment, involved in thymocyte phenotypic differentiation and in the procedures of both negative and positive selection (Brelinska et al. 1991, Hiramine et al. 1995). Our findings in human fetal thymus are consistent with the data above, inasmuch as most of the TH-positive cells were localized in the cortex.
In the human adult spleen, immunoreactivity for S100 has been demonstrated in dendritic cells and lymphocyte-like cells in the splenic periarterial lymphatic sheath (Uccini et al. 1986). In addition, Buckley (1991) has shown that in humans, there are several NSE-positive subpopulations of macrophages and dendritic cells forming clusters of variable sizes within the red pulp. We observed S100-specific immunoreactivity in splenic macrophages and dendritic cells as early as 20 gw, whereas TH-specific immunoreactivity was present in cells of the white pulp during the third trimester of gestation.
Experimental studies have shown that the neuronal pathway is involved in the control of immune responsiveness at several levels, from cellular functions and interactions to overall immunological responses (Rogausch et al. 2004, Wrona 2006). It seems that stimulation of the sympathetic nervous system results in localization of the inflammatory response, suppression of T1 helper–mediated responses (Vizi et al. 1995; Friedman and Irwin 1997; Kurz et al. 1997), and protection from the detrimental effects of pro-inflammatory cytokines (Hasko et al. 1998; Elenkov et al. 2000; Pavlov et al. 2003). Parasympathetic innervation also influences the immune function (Rinner et al. 1995; Tracey 2002; Saeed et al. 2005; Zimring et al. 2005). The development and differentiation of the lymphoid organs appears to depend on the presence of neural factors and especially on sympathetic innervation (Ackerman et al. 1987b; Bulloch et al. 1987). Stimulation of the sympathetic nervous system affects lymphocyte proliferation and circulation, cytokine production, and activation of several lymphoid cells expressing adrenoreceptors (Elenkov et al. 2000). In addition, splenic chemical sympathectomy in neonatal rats is implicated in deranged natural killer (NK) cell activity, T- and B-lymphocyte proliferation, immunoglobulin M production, and antibody responses (Ackerman et al. 1991; Madden et al. 1993). Our results on human fetal thymus and spleen concur with these data and suggest that even during fetal life, there is a close link between the thymus, the spleen, and the nervous/neuroendocrine system.
In conclusion, human fetal thymus appears richly innervated during the second and third trimesters, in contrast to fetal spleen. The density of intraparenchymal nerve fibers in both fetal thymus and spleen increases during development. Most of the intraparenchymal nerve fibers in the human fetal thymus and spleen were observed following the vasculature, and this predominantly perivascular distribution implies that the intrinsic innervation of lymphoid organs plays an important functional role during intrauterine life.
Footnotes
Acknowledgements
This study was supported by the Kapodistrias Research Program, Medical School, National and Kapodistrian University of Athens, grant 70/4/6549. We thank Drs. Alekos Kateris and Serafim Tsoukos for technical assistance.
