Abstract
Vascular permeability factor/vascular endothelial growth factor (VPF/VEGF) interacts with two high-affinity tyrosine kinase receptors, VEGFR-1 and VEGFR-2, to increase microvascular permeability and induce angiogenesis. Both receptors are selectively expressed by vascular endothelial cells and are strikingly increased in tumor vessels. We used a specific antibody to localize VEGFR-2 (FLK-1, KDR) in microvascular endothelium of normal mouse kidneys and in the microvessels induced by the TA3/St mammary tumor or by infection with an adenoviral vector engineered to express VPF/VEGF. A pre-embedding method was employed at the light and electron microscopic levels using either nanogold or peroxidase as reporters. Equivalent staining was observed on both the luminal and abluminal surfaces of tumor- and adenovirus-induced vascular endothelium, but plasma membranes at interendothelial junctions were spared except at sites connected to vesiculovacuolar organelles (VVOs). VEGFR-2 was also localized to the membranes and stomatal diaphragms of some VVOs. This staining distribution is consistent with a model in which VPF/VEGF increases microvascular permeability by opening VVOs to allow the transendothelial cell passage of plasma and plasma proteins.
Keywords
Vascular permeability factor/vascular endothelial growth factor (VPF/VEGF) is a potent multifunctional cytokine that permeabilizes vascular endothelium to plasma proteins and reprograms endothelial cell gene expression so as to induce angiogenesis (Clark et al. 1996; Brown et al. 1997; Ferrara and Davis-Smyth 1997; Dvorak et al. 1999; Feng et al. 1999a; Ferrara 1999). VPF/VEGF is secreted by many tumors and by activated macrophages, keratinocytes, synovial cells, various embryonic cells, and cultured epithelial and mesenchymal cell lines. VPF/VEGF induces its biological effects by binding to two tyrosine kinase receptors, VEGFR-1 (fms-like tyrosine kinase receptor, or Flt) and VEGFR-2 (fetal liver kinase 1, or Flk-1, in rodents; kinase insert domain-containing receptor, or KDR, in humans) which are selectively expressed in vascular endothelium (Matthews et al. 1991; Terman et al. 1991, 1992; de Vries et al. 1992; Quinn et al. 1993; Mustonen and Alitalo 1995). Both receptors are strikingly upregulated in tumors, wounds, and in certain types of inflammation (e.g., rheumatoid arthritis, psoriasis) in which VPF/VEGF is overexpressed (Brown et al. 1997). The complex that forms between tumor-secreted VPF/VEGF and its receptors has been recognized as an attractive potential target for antiangiogenesis therapy (Dvorak et al. 1991; Brekken et al. 1998). Recently, a third non-tyrosine kinase receptor for VPF/VEGF, neuropilin, has also been described (Soker et al. 1998).
Although both VEGFR-1 and VEGFR-2 undergo phosphorylation after interaction with VPF/VEGF, VEGFR-2 is believed to be the more important receptor with regard to induction of increased microvascular permeability and angiogenesis (Millauer et al. 1993; Waltenberger et al. 1994; Kroll and Waltenberger 1997; Gerber et al. 1998; Feng et al. 1999b). VEGFR-2 is also a receptor for other members of the VEGF family, including VEGF-C, VEGF-D, and VEGF-E (Jeltsch et al. 1997; Achen et al. 1998; Ogawa et al. 1998).
Expression of both VEGFR-1 and VEGFR-2 has been localized by in situ hybridization to microvascular endothelium of normal kidneys and to tumors, healing wounds, and inflammatory sites (Brown et al. 1997). VEGFR-2 has also been identified in the blood vessels of human placentas, breast cancers, and gastric carcinomas by light microscopic immunohistochemistry (Vuckovic et al. 1996; Tanigawa et al. 1997; de Jong et al. 1998a,b). However, there have been no reports describing the ultrastructural localization of either receptor in vascular endothelium.
We report here the ultrastructural localization of VEGFR-2 (flk-1/KDR) in vascular endothelium in three model systems in which VPF/VEGF is highly expressed: (a) glomerular and peritubular capillaries of normal mouse kidney; (b) microvessels supplying a well-characterized mouse mammary carcinoma; and (c) new vessels induced by an adenoviral vector engineered to overexpress VPF/VEGF (adeno-vpf/vegf) (Pettersson et al. in press). Similar results were obtained using preembedding methods that used either silver-enhanced nanogold or horseradish peroxidase as reporters. Microvascular endothelial cells were positive for VEGFR-2 in all three models and, in the latter two, could be localized to the luminal and abluminal surfaces and to the membranes of cytoplasmic vesiculovacuolar organelles (VVOs). These findings are consistent with other evidence that VVOs provide an important pathway for extravasation of plasma and plasma proteins in response to VPF/VEGF (Feng et al. 1996, 1997, 1999a). VEGFR-2 was also localized to lymphatic endothelium at adeno-vpf/vegf-injected sites but was not detected in multivesicular bodies, Weibel-Palade bodies, coated pits, coated vesicles, or in the basal lamina underlying VEGFR-2-positive endothelium.
Materials and Methods
Animals, Tumors, and Vectors
TA3/St tumors, originally derived from a mammary adenocarcinoma that developed spontaneously in an A/HeHa strain mouse, were passaged weekly in ascites form in the peritoneal cavities of female A/Jax mice. To induce solid tumors, 1 × 106 tumor cells were injected into the subcutaneous space of ∼20-g female A/Jax mice. Adenoviral vectors were prepared as previously described (Ory et al. 1996; Hardy et al. 1997). Female BALB/c ByJ Hfh11nu immunodeficient nude mice, 4–6 weeks of age, were obtained from the Jackson Laboratory (Bar Harbor, ME). An adenoviral vector was engineered to express murine VPF/VEGF164 (Guzman et al. 1993) and 2.5 × 108 pfu was injected into flank skin or leg muscle of nude (immunodeficient) mice (Pettersson et al. in press). In brief, adeno-vpf/vegf-injected animals develop a strong angiogenic response accompanied by vascular hyperpermeability and edema but without inflammatory cells.
Antibodies to VEGFR-2 (Flk-1)
Mouse Flk-1/SEAP or the extracellular soluble domain of Flk-1 (sFlk-1) was used as immunogen with equivalent results. Flk-1/SEAP was isolated and purified as previously described (Tessler et al. 1994; Brekken et al. 1998). After purification, the receptor (in PBS) was sterile-filtered and frozen in aliquots until use. Antibodies were generated by immunizing three female rabbits with 60–70 μg of either Flk-1/SEAP or the soluble extracellular domain of Flk-1 (sFlk-1). Injections (50 μl each) were split over three subcutaneous sites on each rabbit. Subsequently, SC booster injections of 25–40 μg of sFlk-1 were administered in TiterMax every 5 weeks for a period of 6 months to 1 year. Animals were bled from the ear and sera collected and pooled 2–3 weeks after each booster injection. Titers were measured with an indirect ELISA assay using sFlk-1 as antigen (Huang et al. 1998) and also by FACS analysis on cells that were either VEGFR-2 (Flk-1)-positive or -negative [kindly supplied by Dr. J. Waltenberger, Ulm, Germany (Waltenberger et al. 1994)]. Antibodies were then affinity-purified from high-titer (≥ 1:50,000) sera. An affinity column was prepared by linking 1–2 mg of purified sFlk-1 to a 1.0-ml HiTrap NHS-activated Sepharose column (Pharmacia; Piscataway, NJ) according to the manufacturer's instructions. Sera were diluted 1:2 in PBS with NaN3 and recirculated on the column for 24–48 hr at 4C. The column was then washed with 50–100 column volumes of PBS and eluted at low and high pH with 0.1 M citric acid, pH 2.8, and 0.1 M triethylamine, pH 11.5, respectively. Typical yields were ∼200 μg of specific IgG/ml serum. The antibody was shown to be specific for VEGFR-2 by ELISA and Western blot analysis (Huang et al. 1998).
Tissue Fixation and Preparation for Immunohisto/cytochemistry
Nine days after receiving TA3/St tumor, six A/Jax mice bearing solid TA3/St tumors and six control mice of the same strain were sacrificed by CO2 narcosis and tumors, along with skin samples and kidneys of control A/Jax mice, were harvested. Portions of ear skin and leg muscles were similarly harvested from nude mice at 1, 3, 5, or 8 days after injection of adeno-vpf/vegf or adenovirus bearing a control insert (lacZ or green fluorescent protein).
Attempts to demonstrate VEGFR-2 at the electron microscopic level by postembedding procedures were unsuccessful. Therefore, we adopted a pre-embedding protocol using either nanogold or peroxidase as a reporter. Tissues were immersed immediately in freshly prepared 4% paraformaldehyde in 0.02 M phosphate buffer, pH 7.4. After 4 hr of fixation at room temperature (RT), tissues were transferred to 30% sucrose in the same PBS buffer overnight at 4C. Finally, tissues were embedded in OCT compound (Miles; Elkhart, IN) and stored in liquid nitrogen at −176C for subsequent use. Frozen sections, 5 or 10 μm thick, were cut on a standard cryostat (Tissue-Tek II) and were collected on pre-cleaned microscopic slides (Fisherbrand, Colorfrost/Plus; Fisher Scientific, Pittsburgh, PA). Slides were air-dried for 20 min before staining for light or electron microscopy.
Immunoperoxidase Protocol
Five-μm cryostat sections were cut and mounted on glass slides for light microscopic immunohistochemistry. Ten-μm cryostat sections were cut, mounted, and processed according to the same immunostaining protocol for subsequent embedment in Epon for 1μm light microscopic sections and also for electron microscopy. All of the following immunostaining steps were performed at RT with cryostat sections mounted on glass slides: (a) wash in 0.02 M PBS, pH 7.6, 5 min; (b) immerse in 0.3% hydrogen peroxide (Fisher Scientific; Fair Lawn, NJ) in methanol, 30 min; (c) wash in 0.02 M PBS, pH 7.4, 5 min; (d) immerse in 5% normal goat serum (NGS) (Vector Laboratories; Burlingame, CA), 20 min; (e) primary affinity-purified polyclonal rabbit antibody to VEGFR-2 (T014) prepared by two of the authors (RAB, PET) (Huang et al. 1998) was used at a dilution of 1:40 (= 1 μg protein/μl) in 0.02 M PBS, 60 min; (f) three washes in 0.02 M PBS, pH 7.4, 5 min each; (g) secondary antibody (affinity-purified, biotinylated goat anti-rabbit IgG antibody; (Vector), 1:100 in 0.02 M PBS, pH 7.4, 60 min; (h) three washes in 0.02 M PBS, pH 7.4, 5 min each; (i) avidin-biotin-horseradish peroxidase complex (ABC) (Vector) in 0.02 M PBS, pH 7.4, 30 min; (j) three washes in 0.02 M PBS, pH 7.4, 5 min each; (k) 0.05% 3,3′-diaminobenzidine tetrahydrochloride (DAB) in 0.02 M PBS, pH 7.4, containing 0.003% hydrogen peroxide, 5 min; (l) wash in 0.02 M PBS, pH 7.4, 5 min; (m) postfix in 1% glutaraldehyde in 0.02 M PBS, pH 7.4, for 5 min; (n) after rinsing with water, sections were counterstained with modified Mayer's hematoxylin (American Histology Reagents; Stockton, CA) for 5 min; (o) wash in water for 30 min followed by dehydration through a series of ethanols and mount the sections under coverslips.
Five controls were performed to ensure the specificity of peroxidase immunostaining: (a) replacement of primary antibody by an irrelevant rabbit IgG; (b) omission of specific primary antibody; (c) omission of the secondary antibody; (d) omission of the ABC complex; (e) omission of DAB.
Immuno-Nanogold-Silver Protocol
Sections were prepared as for the immunoperoxidase protocol. The following steps were performed at RT with cryostat sections mounted on glass slides: (a) wash in 0.02 M PBS, pH 7.6, 5 min; (b) immerse in 50 mM glycine in 0.02 M PBS, pH 7.4, 10 min; (c) wash in 0.02 M PBS, pH 7.4, 5 min; (d) immerse in 5% NGS (Vector), 20 min; (e) incubate in the primary antibody, T014, as for immunoperoxidase staining; (f) three washes in 0.02 M PBS, pH 7.4, 5 min each; (g) incubate in the secondary antibody (affinity-purified nanogold, 1.4 nm in diameter-conjugated goat anti-rabbit IgG antibody or affinity-purified nanogold with 1.4 nm in diameter-conjugated goat anti-rabbit Fab′ (Nanoprobes; Stony Brook, NY), 1:100 in 0.02 M PBS, pH 7.4, 60 min; (h) three washes in 0.02 M PBS, pH 7.4, 5 min each; (i) postfix in 1% glutaraldehyde in 0.02 M PBS, pH 7.4, 5 min; (j) three washes in distilled water, 5 min each; (k) develop with HQ silver enhancement solution (Nanoprobes) for 6–11 min in the darkroom; (l) two washes in distilled water, 2 min each; (m) fix in 5% sodium thiosulfate, 1 min; (n) rinse with water and counterstain with modified Mayer's hematoxylin for 5 min; (o) wash in water for 30 min, followed by dehydration through a series of ethanols, and mount the sections under a coverslip.
Four controls were performed to ensure the specificity of nanogold immunostaining: (a) replacement of primary antibody by an irrelevant rabbit IgG; (b) omission of specific primary antibody; (c) omission of the secondary antibody; (d) omission of the HQ silver enhancement solution.
Tissue Processing for 1-μm Epon Sections and for Electron Microscopy
After immunostaining with either of the two protocols, 10-μm sections attached to glass slides were postfixed in 1% osmium tetroxide in Sym-Collidine buffer, pH 7.4, for 10 min at RT, washed with 0.05 M sodium maleate buffer, pH 5.2, and stained with 2% uranyl acetate in 0.05 M sodium maleate buffer, pH 6.0, for 5 min at RT. Sections were washed with distilled water and then dehydrated in graded ethanols and infiltrated with a propylene oxide-Epon 812 sequence. Embedment was performed by inverting Eponfilled plastic capsules over the slide-attached tissue sections. After polymerization for 16 hr at 60C, Epon blocks were separated from glass slides by brief immersion in liquid nitrogen.
One-μm plastic sections for light microscopy were cut with an ultratome (Reichert; Vienna, Austria) and stained with alkaline Giemsa in a 60C oven for 60 min. For electron microscopy, thin sections were cut with a diamond knife on an ultratome (Reichert) and collected on uncoated 200-mesh copper grids (Ted Pella; Austin, TX). Grids were viewed unstained with a transmission electron microscope (CM 10; Philips, Eindhoven, The Netherlands).
Electron Microscopy and Statistical Analysis
Kidney, tumor, and adenovirus-injected skin and skeletal muscle were studied in three animals at the times indicated in the text. We studied six separate samples of each tissue and a total of 30 EM sections of kidney, 24 of tumor, and 36 of adenovirus-injected tissues. Statistical analysis was performed with the Mann-Whitney U-test.
Results
Localization of VEGFR-2 in Renal Microvessels of Normal Mice
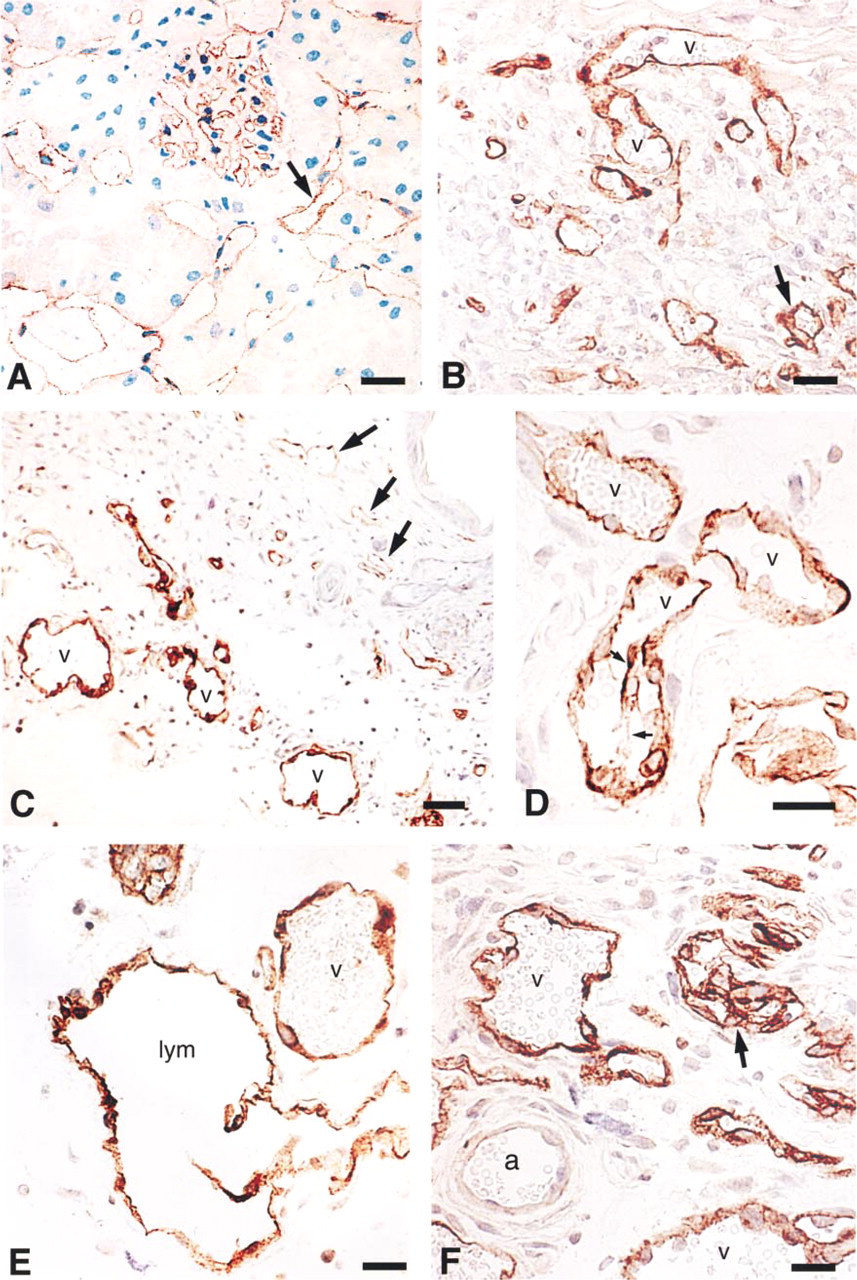
VPF/VEGF is strongly expressed by glomerular podocytes and tubular epithelium of normal kidney, and kidney is one of the few normal adult tissues in which VPF/VEGF receptor expression is readily detected by in situ hybridization (Berse et al. 1992; Brown et al. 1992a, 1997; Dvorak et al. 1999). By light microscopic immunohistochemistry, glomerular and peritubular microvascular endothelial cells stained intensely with the antibody to VEGFR-2 with either reporter (Figure 1A illustrates immuno-nanogold only). However, the endothelium lining arterioles and small arteries stained lightly or not at all. Podocytes and tubular epithelium were also not stained.
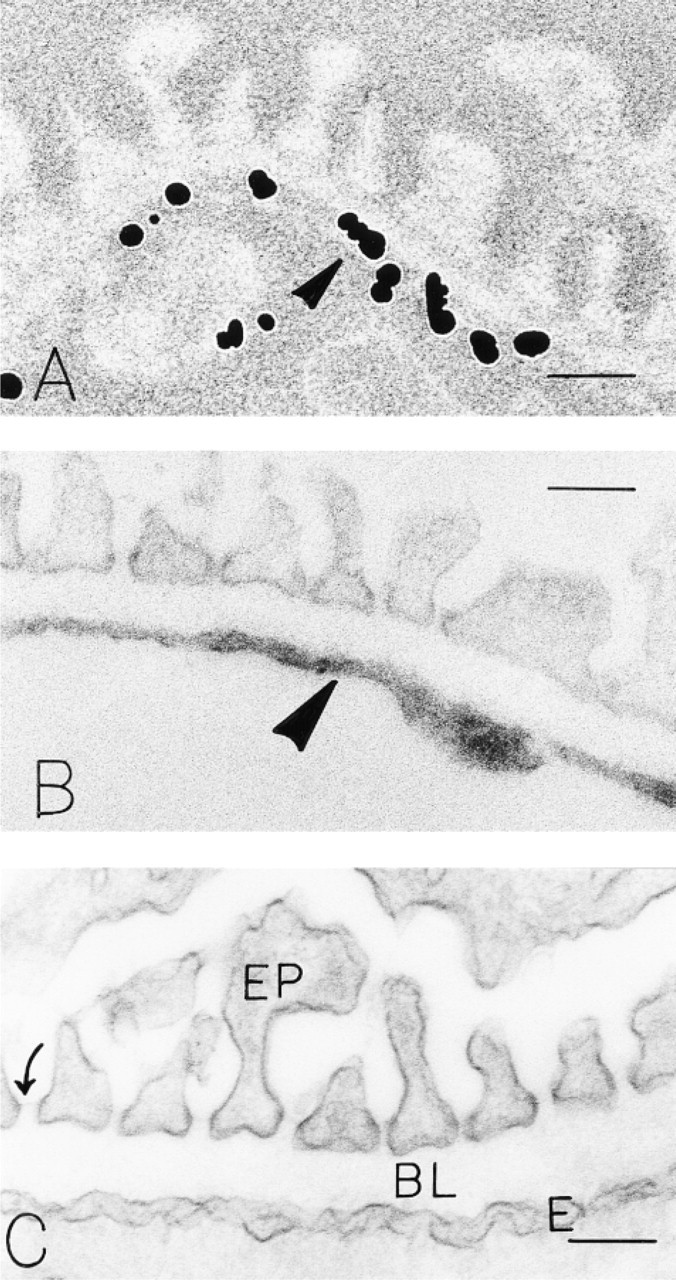
By electron microscopy, the particulate nanogold (Figure 2A) and peroxidase enzyme (Figure 2B) reporters precisely labeled glomerular endothelium, sparing the subjacent basal laminae as well as epithelial podocytes and their foot processes. Because the glomerular endothelium is extremely thin, we were not able to localize staining more precisely, i.e., to the luminal or abluminal surface. Controls for each technique were negative (Figure 2C).
Localization of VEGFR-2 in Hyperpermeable Vascular Endothelium Supplying TA3/St Mouse Mammary Carcinomas
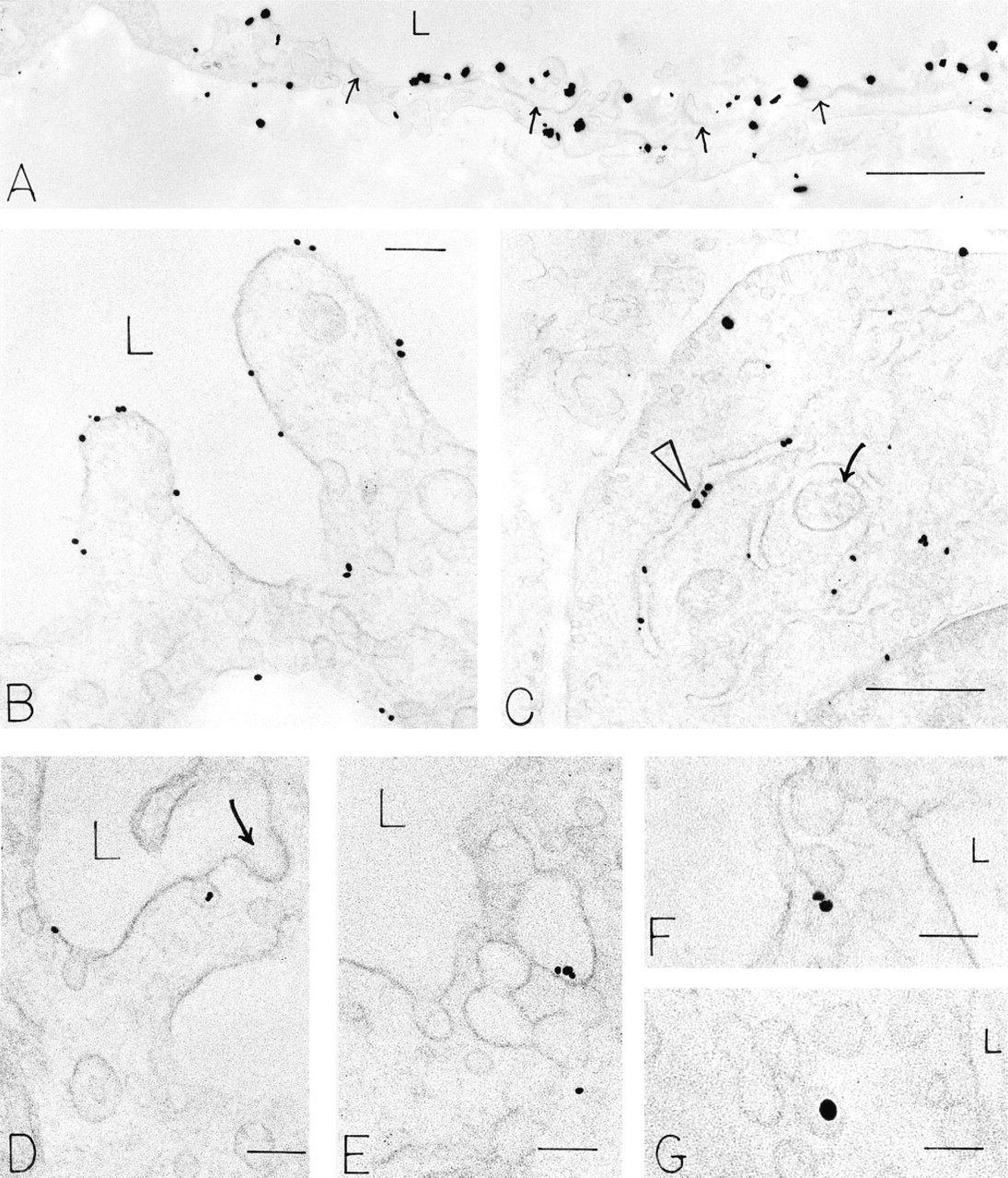
By light microscopy, both tumor-associated microvessels (Figure 1B) and lymphatic vessels (not shown) exhibited intense and specific linear staining for VEGFR-2, whereas endothelial cell nuclei and tumor cells did not stain. The first new blood vessels induced by TA3/St tumors are enlarged, pericyte-poor sinusoids termed “mother” vessels (Nagy et al. 1995; Pettersson et al. in press). Mother vessels subsequently split into smaller daughter vessels by a process of transluminal bridging of endothelial cells (Nagy et al. 1995; Feng et al. in press; Pettersson et al. in press). Bridging endothelium was also strongly VEGFR-2-positive (Figures 1B and 1D, arrows). Blood vessels and lymphatics in normal skin adjacent to tumors stained weakly or not at all for VEGFR-2 (Figure 1C, arrows). Both nano-gold and peroxidase reporters gave comparable results by light microscopy.
By electron microscopy, abluminal and luminal vascular endothelium was labeled patchily, not continuously, with antibody to VEGFR-2 with both immunocytochemical methods (Figure 3; only peroxidase data shown). There was no difference in nanogold labeling density between the endothelial lumen and the ablumen (Table 1). The endothelial cells lining tumor-associated vessels, like normal venular endothelium, contain many VVOs, prominent clusters of uncoated caveolae-like vesicles, and vacuoles that span the endothelial cytoplasm from lumen to ablumen (Kohn et al. 1992; Dvorak et al. 1996; Feng et al. 1996, 1999a). In tumor vessels and in normal venules stimulated with VPF/VEGF, VVOs provide a pathway for transendothelial extravasation of plasma proteins and other circulating macromolecules. VVOs also stained specifically with the antibody to VEGFR-2; 52% of 102 VVOs counted were labeled (mean 1.5 gold particles/VVO). Lateral plasma membranes of interendothelial cell contacts were sometimes focally positive, but only in those uncommon instances in which a VEGFR-2-positive VVO vesicle happened to be attached (Figure 3A). The stomatal diaphragms of some VVOs and caveolae were VEGFR-2-positive, best seen with the peroxidase reporter when the entire vesicle was not stained (see arrows in Figures 3B and 3D). VVO vesicles and vacuoles deeper within the endothelial cytoplasm were less frequently labeled than those closer to the luminal or abluminal surfaces (Figure 3C).
Plasma membranes and components of VVOs of normal skin vessels remote from sc tumors were also labeled for VEGFR-2, but less frequently and with reduced intensity compared with vessels more closely associated with SC tumor nodules.
Immunostaining of microvessels with antibody to VEGFR-2. (
Sections of mouse kidney prepared with the immuno-Nanogold-silver (
Localization of VEGFR-2 to Hyperpermeable New Vessels Induced in Normal Mice by an Adenoviral Vector Expressing Murine VPF/VEGF164 (adeno-vpf/vegf)
Adeno-vpf/vegf was injected into the skin or leg muscle of normal immunodeficient nude mice. Transduced stromal cells expressed VPF/VEGF, as determined by in situ hybridization (Pettersson et al. in press), leading to a local angiogenic response. As in TA3/St tumors, angiogenesis was initially characterized by the formation of large mother vessels, followed by their subsequent splitting into smaller daughter vessels by a process of endothelial cell bridging (Nagy et al. 1995; Feng et al. in press). As shown in Figures 1C-1F, VEGFR-2 expression was strikingly upregulated in both mother and daughter vessels compared with nearby normal microvessels. Lymphatic endothelium also stained strongly for VEGFR-2 (Figure 1E), but the endothelium lining arterioles did not stain (Figure 1F).
By electron microscopy, specific VEGFR-2 labeling was also strikingly increased in adeno-vpf/vegf-induced microvessels compared with normal microvascular endothelium (Table 1). The luminal and abluminal plasma membranes exhibited equivalent labeling with both the peroxidase and the nanogold method (Table 1; Figures 4A and 4B). However, lateral endothelial cell plasma membranes at cell junctions were not labeled (Figure 4A). VVO vesicles, vacuoles, and stomata interconnecting adjacent vesicles and vacuoles were frequently labeled. Thus, 54% of 111 VVOs counted were labeled (mean 1.8 gold particles/VVO) (Figures 4E-4G). In addition, some endothelial cells exhibited expanded cisternae of rough endoplasmic reticulum and these, as well as perinuclear cisternae, were also labeled with antibody to VEGFR-2 (Figure 4C). Caveolae were also labeled rarely, but coated vesicles were not (Figure 4D).
Discussion
By making use of two different pre-embedding protocols for immunocytochemistry we have localized VEGFR-2 (flk-1/KDR), one of the two high-affinity tyrosine kinase VPF/VEGF receptors, to vascular endothelium at three sites of strong VPF/VEGF expression in the mouse: normal kidney, the TA3/St mammary carcinoma, and skin injected with an adenoviral vector engineered to overexpress murine VPF/VEGF164. In the latter two instances, both the luminal and abluminal membranes of vascular endothelium stained strongly and specifically, along with the membranes and diaphragms of some caveolae and VVOs. Because glomerular and peritubular capillaries are lined by unusually thin endothelium, we could not distinguish luminal from abluminal staining in these vessels. VEGFR-2 was also localized to the expanded RER of endothelial cells and connected to the perinuclear cisternae of growing endothelial cells at skin sites injected with adeno-vpf/vegf. VEGFR-2 staining was not observed in several other locations in vascular endothelium, including the lateral plasma membranes at interendothelial cell junctions, mitochondria, nuclei, multivesicular bodies, Weibel-Palade bodies, coated pits, coated vesicles, lysosomes, and basal laminae. Fenestral diaphragms of fenestrated portions of endothelium, glomerular epithelial cells, their foot processes, and their slit diaphragms were also not stained for VEGFR-2 in the kidney. Tubular epithelial cells adjacent to VEGFR-2-positive peritubular capillaries were also negative. TA3/St tumor cells were not stained for VEGFR-2, nor were nerves or interstitial collagen in any of the tissues studied. Controls for each immunocytochemical procedure were negative.
Both of the high-affinity VPF/VEGF tyrosine kinase receptors, VEGFR-1 and VEGFR-2, are expressed at relatively low copy numbers in several types of cultured endothelial cells; published estimates vary from 27,000 to 150,000 copies of VEGFR-2 per cell and the frequency of VEGFR-1 is ∼10-fold lower (Vaisman et al. 1990; Detmar et al. 1995; Thieme et al. 1995). However, little is known about the frequency of either receptor on vascular endothelium in vivo. As determined by in situ hybridization, both receptors are expressed at very low to undetectable levels in most normal tissues, with the exception of kidney (Brown et al. 1997; Dvorak et al. 1999). However, expression of both receptors is strikingly upregulated in microvascular endothelium adjacent to sites of VPF/VEGF overexpression, i.e., in tumor-associated blood vessels (Brown et al. 1997) and in the new vessels that form in response to local injection of adeno-vpf/vegf (Pettersson et al. in press). The data presented here are consistent with earlier observations in that VEGFR-2 was undetectable by immunohistochemistry in the microvasculature of most normal tissues, except for kidney, but was strongly expressed in two sites of VPF/VEGF overexpression (TA3/St mammary carcinoma and skin sites injected with adeno-vpf/vegf).
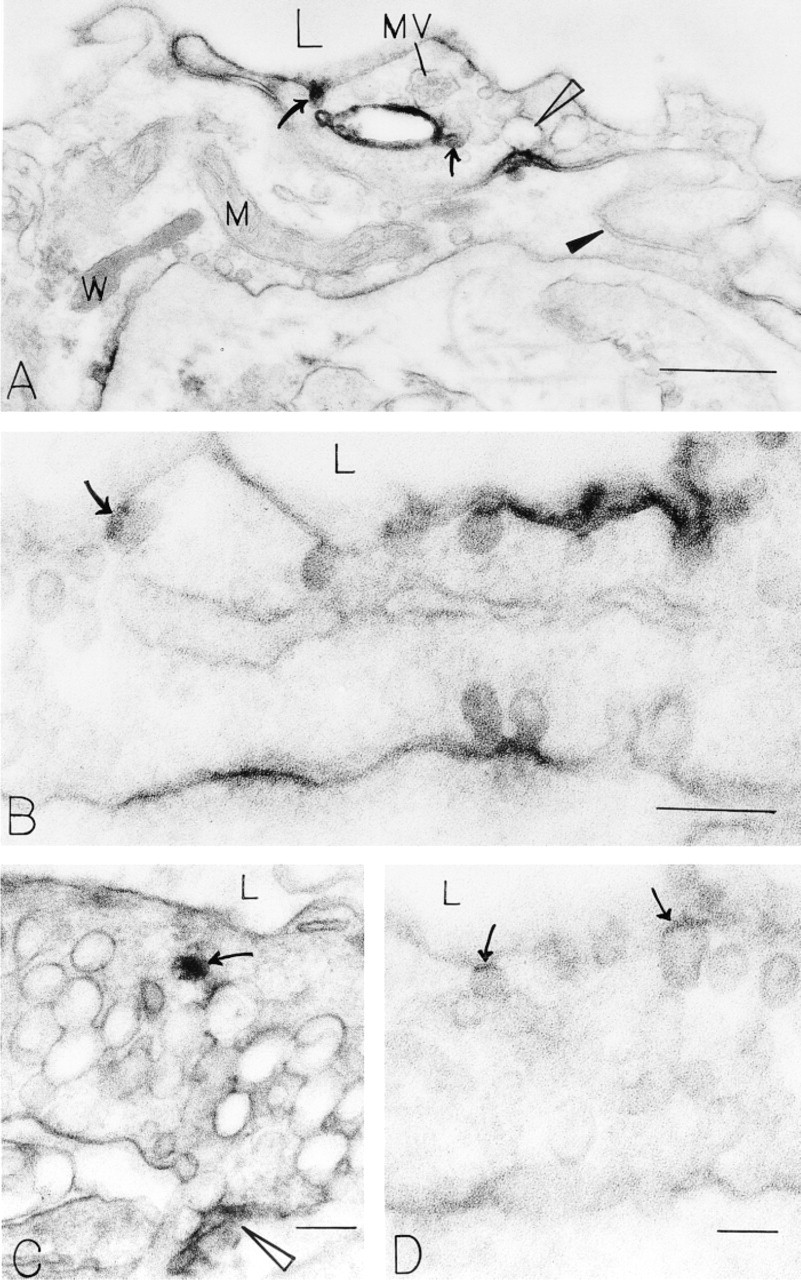
Unstained immunoperoxidase preparations localize VEGFR-2 to endothelial structures in TA3/St mouse mammary adenocarcinoma 9 days after injection of 1 × 106 tumor cells into the SC space of mice. VEGFR-2 is focally present (as determined by electron-dense peroxidase reaction product) on luminal and abluminal plasma membranes (
Surface labeling density of microvascular endothelium from 9 day TA3/St tumors, 4-day adeno-vpf/vegf-induced skin mother vessels, and normal control skin with antibody to VEGFR-2; silver-enhanced nanogold served as the reporter; luminal and abluminal perimeters were measured separately and compared statistically with the Mann-Whitney U-test
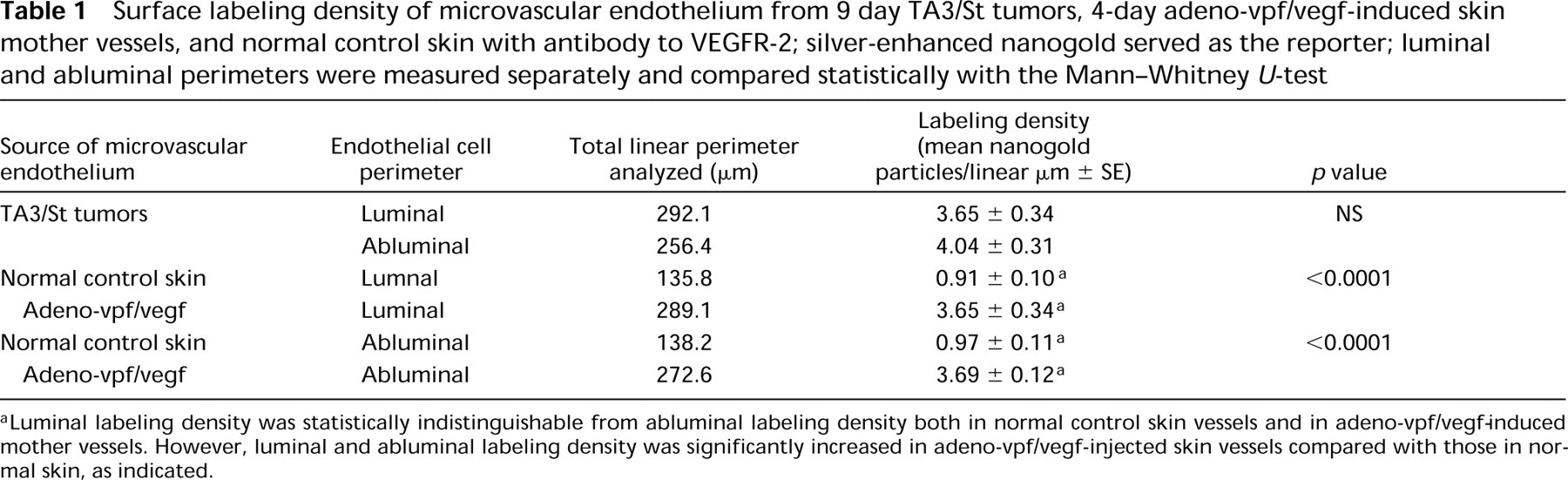
Luminal labeling density was statistically indistinguishable from abluminal labeling density both in normal control skin vessels and in adeno-vpf/vegf-induced mother vessels. However, luminal and abluminal labeling density was significantly increased in adeno-vpf/vegf-injected skin vessels compared with those in normal skin, as indicated.
In earlier studies we localized VPF/VEGF to the vascular endothelium of tumor-associated vessels by immunohistochemistry (Dvorak et al. 1991; Brown et al. 1997). Because these endothelial cells were not themselves expressing VPF/VEGF, we concluded that the staining observed represented tumor cell-secreted VPF/VEGF that was bound to its receptors on vascular endothelium. Extending this approach to the electron microscopic level, we found that antibodies to VPF/VEGF strongly stained the abluminal surfaces of tumor vascular endothelium, along with VVO vesicles and vacuoles. The luminal surfaces of the same endothelial cells were also stained, but to a significantly lesser extent (Qu-Hong et al. 1995). We offered two explanations for these findings. One possibility was that VPF/VEGF receptors were asymmetrically distributed, with more being present on the abluminal than on the luminal endothelial cell surface. This might make sense in that in most situations, e.g., tumors, healing wounds, and inflammation, VPF/VEGF is secreted by tissue cells and therefore encounters the abluminal surface of microvascular endothelium before the luminal surface (Brown et al. 1992b, 1997; Detmar et al. 1994; Fava et al. 1994; Koch et al. 1994). On the other hand, exposure of the abluminal surfaces of vascular endothelium to higher concentrations of VPF/VEGF that was secreted locally by tumor or other tissue cells could equally well account for our observations of asymmetric VPF/VEGF distribution, even if equal numbers of VEGF receptors were to be found on luminal and abluminal surfaces. The data presented here indicate that when VPF/VEGF was overexpressed locally in skin by local administration of a VPF/VEGF-engineered adenoviral vector, equivalent numbers of VEGFR-2 were found on the luminal and abluminal surfaces of vascular endothelium. Of course, these data say nothing about the signaling efficiency of VPF/VEGF when it interacts with receptors that are disposed on the luminal compared with the abluminal endothelial surface.
Localization of VEGFR-2 to VVO membranes and to the luminal and abluminal plasma membranes of vascular endothelium, but not to the lateral plasma membranes at interendothelial cell junctions, is consistent with the mechanisms we have proposed for the increased microvascular permeability that is induced by VPF/VEGF and other vasoactive mediators (Feng et al. 1996, 1997). In brief, we have presented evidence that VVOs, structures that are often strategically concentrated in parajunctional zones of normal venule and tumor endothelium, provide a primary transcellular pathway for extravasation of macromolecules across continuous vascular endothelium. In response to vasoactive mediators, the diaphragms separating individual VVO vesicles from each other and from the vascular lumen and ablumen open, allowing the transendothelial passage of tracers from one vesicle or vacuole to the next until they finally cross the endothelial barrier. This view stands in contrast to the traditional view, originally proposed by Majno and associates (1969) and more recently advocated by the McDonald laboratory (Baluk et al. 1997), that vasoactive mediators promote endothelial cell contractions that pull apart adjacent endothelial cells from each other to generate interendothelial cell gaps through which plasma can leak. For a detailed discussion of the controversy surrounding the pathways taken by circulating macro-molecules in crossing vascular endothelium in response to vasoactive mediators, readers are referred to several recent review articles (Dvorak et al. 1999; Feng et al. 1999a; McDonald et al. 1999; Michel and Neal 1999).
Immuno-Nanogold-silver preparations localize VEGFR-2 to endothelial structures in angiogenic sites induced 4 days earlier in the skin of nude mice by adeno-vpf/vegf (
Finally, it is of interest that lymphatic vessels at sites of adeno-vpf/vegf injection also stained strongly with the antibody to VEGFR-2. Lymphatic vessels have not been reported to overexpress VEGFR-2 at tumor sites, but this is not surprising because tumors have generally not been believed to induce the formation of new lymphatics. In addition, lymphatic channels may be difficult to distinguish from new blood vessels by in situ hybridization. On the other hand, lymphatics of the chick chorioallantoic membrane are reported to express VEGFR-2 and VEGFR-3 (Oh et al. 1997), and VEGF-C, which reacts with both of these receptors, is reported to induce the formation of both lymphatics and blood vessels (Cao et al. 1998).
Footnotes
Acknowledgements
Supported by US Public Health Service National Institutes of Health grants AI-33372 (AMD), CA-50453 (HFD), CA74951 and CA54168 (PET), T32 GM07062 (RAB), and HL-59316, and by funding from the Howard Hughes Medical Institute (RCM). AP received fellowship support from the Wenner-Gren Foundation, Stockholm, Sweden.
