Abstract
Local growth, invasion, and metastasis of malignancies of the head and neck involve extensive degradation and remodeling of the underlying, collagen-rich connective tissue. Urokinase plasminogen activator receptor-associated protein (uPARAP)/Endo180 is an endocytic receptor recently shown to play a critical role in the uptake and intracellular degradation of collagen by mesenchymal cells. As a step toward determining the putative function of uPARAP/Endo180 in head and neck cancer progression, we used immunohistochemistry to determine the expression of this collagen internalization receptor in 112 human squamous cell carcinomas and 19 normal or tumor-adjacent head and neck tissue samples from the tongue, gingiva, cheek, tonsils, palate, floor of mouth, larynx, maxillary sinus, upper jaw, nasopharynx/nasal cavity, and lymph nodes. Specificity of detection was verified by staining of serial sections with two different monoclonal antibodies against two non-overlapping epitopes on uPARAP/Endo180 and by the use of isotype-matched non-immune antibodies. uPARAP/Endo180 expression was observed in stromal fibroblast-like, vimentin-positive cells. Furthermore, expression of the collagen internalization receptor was increased in tumor stroma compared with tumor-adjacent connective tissue or normal submucosal connective tissue and was most prominent in poorly differentiated tumors. These data suggest that uPARAP/Endo180 participates in the connective tissue destruction during head and neck squamous cell carcinoma progression by mediating cellular uptake and lysosomal degradation of collagen.
Keywords
D
Urokinase plasminogen activator receptor-associated protein (uPARAP)/Endo180 is a recently identified member of the macrophage mannose receptor family of endocytic transmembrane glycoproteins (Behrendt et al. 2000; Sheikh et al. 2000). Little is known about the expression of uPARAP/Endo180 in vivo, but the receptor has been reported to be present on a subset of mesenchymal cells at sites of active tissue remodeling. These include bone-forming cells, activated hepatic stellate cells, and mammary tumor myoepithelial cells and fibroblasts (Engelholm et al. 2001; Schnack Nielsen et al. 2002; Mousavi et al. 2005). Gene-targeting studies in mice unexpectedly uncovered a near-complete requirement of uPARAP/Endo180 for cellular uptake and subsequent lysosomal degradation of a wide range of collagen species (East et al. 2003; Engelholm et al. 2003; Wienke et al. 2003; Kjoller et al. 2004). Furthermore, our recent study showed that uPARAP/Endo180 impaired collagen turnover critical to tumor expansion in a murine model of mammary adenocarcinoma, as evidenced by the abrogation of cellular collagen uptake, tumor fibrosis, and blunted tumor growth (Curino et al. 2005). Together these studies suggest that uPARAP/Endo180 could have a key function in collagen turnover during tumor dissemination.
In this study we investigated the expression of uPARAP/Endo180 in a panel of human head and neck squamous cell carcinomas presenting at a wide spectrum of anatomic locations and representing different histological grades. We report that stromal expression of uPARAP/Endo180 is a consistent feature of head and neck squamous cell carcinoma, suggesting that intracellular collagen degradation contributes to the devastating connective tissue destruction associated with this disease.
Materials and Methods
Tissue Samples
The “OR601, Oral squamous cancer tissue array with normal oral control tissue” (http://www.biomax.us/tissue-arrays/Oral/OR601) was obtained from US Biomax, Inc. (Rockville, MD). The “CC34-01-001, Head and neck squamous carcinoma (multisites, grade I- III) tissue array (http://www.cybrdi.com/viewproduct.php) was obtained from Cybrdi Inc. (Frederick, MD). Additionally, 49 oral squamous cell carcinomas were obtained from the Oral & Pharyngeal Cancer Branch Tissue Repository, National Institute of Dental and Craniofacial Research, Bethesda, MD.
Generation of Monoclonal Antibodies
A uPARAP/Endo180 fusion protein, including the third domain of the urokinase receptor as a purification tag, was produced and isolated as described previously for the C4.4A antigen (Hansen et al. 2004). uPARAP/Endo180-deficient mice (backcrossed >10 times in the mouse FVB strain) were immunized with the soluble human uPARAP/Endo180 fusion protein after which cell fusion, cloning of hybridomas, and generation of monoclonal antibodies (MAbs) were performed using routine techniques (Ronne et al. 1991).
Western Blotting
Specificity of the isolated antibodies was tested by Western blotting analysis using the Western Breeze kit (Invitrogen; Carlsbad, CA) according to the manufacturer's protocol. MAbs were diluted to 2 μg/ml. Procedures for the generation of uPARAP/Endo180-sufficient and -deficient dermal fibroblasts from newborn mice have been described previously (Engelholm et al. 2003). Detergent lysates of U937 cells and fibroblasts were prepared as described (Behrendt et al. 2000).
Immunohistochemistry
Tissue sections (5 μm) were deparaffinized in xylene (Mallinckrodt Baker; Phillipsburg, NJ), and the tissue was rehydrated in graded ethanol (100%, 95%, 70%, and H2O). Antigen retrieval was performed using proteinase-K (Fisher Scientific; Pittsburgh, PA) treatment (5 μg/ml in 50 mM Tris-HCl, pH 8.0; Quality Biologicals, Gaithersburg, MD), 5 mM EDTA (Quality Biologicals) at 37C for 10 min for detection of uPARAP/Endo180, or using 10 mM sodium citrate, pH 6.0, for 15 min for detection of vimentin. Endogenous peroxidase activity was quenched by treatment with 1% hydrogen peroxide (Fisher Scientific) in H2O for 15 min, after which the sections were washed in tap water and transferred to 1X TBS buffer (Quality Biologicals) with 0.5% Triton X-100 (Sigma-Aldrich; St Louis, MO). Mouse MAbs against uPARAP/Endo180 described above (2.h.9:F12 at 1.8 μg/ml and 5.f.4:D7 at 1.4 μg/ml, both IgG1κ), vimentin (1:500; DakoCytomation, Carpinteria, CA), or a non-immune mouse IgG1κ control antibody (1.5 μg/ml; eBiosciences, San Diego, CA) in 1X TBS with 0.25% BSA (Sigma-Aldrich) were incubated overnight at 4C. Slides were removed the following day from the cold and incubated for an additional 30 min at room temperature. Slides were then washed twice with 0.05% Tween-20 (Bio-Rad Laboratories; Hercules, CA) in 1X TBS. EnVision+ peroxidase anti-mouse antibody (DakoCytomation) was applied to the slides for 45 min at room temperature, followed by two washes with TBS-Tween-20 buffer. Slides were developed for 22 min for uPARAP/Endo180 and 4 min for vimentin using SIGMA FAST 3,3′-diaminobenzidine tablet sets (Sigma-Aldrich) according to the manufacturer's instructions. The staining reaction was stopped with tap water and the tissue was counterstained in Mayer's hematoxylin (Sigma-Aldrich) for 30 sec, blued using 1X TBS, and rinsed in tap water. Slides were dehydrated through graded ethanol and xylene. Coverslips were mounted with permount (Fisher Scientific) and the slides examined for uPARAP/Endo180 expression as described below.
Evaluation of Staining Intensities and Statistical Analysis
Assessment of staining intensities of tissue samples was performed by an investigator unaware of sample identity using a modification of procedures used previously to quantitatively assess expression of stromal antigens in oral squamous cell carcinomas (de Vicente et al. 2005; Mathe et al. 2006; Piattelli et al. 2006). Scores were assigned on the basis of the intensity and extent of stromal staining in random X20 microscopic fields using an arbitrary scale from zero to four where 0 = three or fewer stromal cells stain weakly, 1 = four or more stromal cells stain weakly, 2 = many stromal cells stain moderately or the majority of stromal cells stain weakly, 3 = the majority of stromal cells stain strongly, and 4 = almost all stromal cells stain at great intensity.
Two-tailed χ2 analysis was used to determine statistical significance of differences in the frequency of uPARAP/Endo180 staining between groups. Statistical significance of the intensity of uPARAP/Endo180 staining between groups was determined by the two-tailed Mann-Whitney U test.
Results
uPARAP/Endo180 expression was studied by immunohistochemical (IHC) analysis of a panel of human squamous cell carcinomas of the head and neck area, as well as a number of tumor-adjacent head and neck and normal head and neck tissues. The combined material successfully analyzed included 112 squamous cell carcinomas derived from the tongue (n=39), gingiva (n=15), larynx (n=8), cheek (n=9), tonsils (n=4), nasopharynx/nasal cavity (n=4), palatal surface of maxilla and mandible (n=7), lymph node metastases (n=2), maxillary sinus (n=2), ethmoid sinus (n=1), or from an unknown location in the oral cavity (n= 21). The normal or tumor-adjacent tissues analyzed included tongue (n=11), gingiva (n=5), palate (n=1), cheek (n=1), or from an unknown location in the oral cavity (n=1). The material was derived from the Biomax array (43 tumor samples and 13 normal or tumoradjacent samples) and the Cybrdi array (20 tumor samples in triplicate and six normal or tumor-adjacent samples in duplicate), and 49 oral squamous cell carcinomas from the Oral & Pharyngeal Cancer Branch Tissue Repository. For IHC analysis, we used mouse MAbs (2.h.9:F12 and 5.f.4:D7) that recognize speciesconserved, non-overlapping epitopes on uPARAP/Endo180 (unpublished data). Both MAbs were specific for uPARAP/Endo180, as demonstrated by Western blotting (Figure 1). Thus, in protein extracts of human U937 cells, which are known to express uPARAP/Endo180 (Behrendt et al. 2000), both MAbs recognized exclusively one band representing a protein with the molecular mass of uPARAP/Endo180 (180,000) (Sheikh et al. 2000) (Figure 1A, Lanes 1 and 2). No protein was recognized by an irrelevant control MAb of the same isotype (anti-TNP; Figure 1A, Lane 3). Utilization of uPARAP/Endo180-deficient mice for immunization led to the generation of MAbs (including the two MAbs of this study) that are reactive against both human and murine uPARAP/Endo180. As evident in Figure 1B, both antibodies reacted with a single protein (molecular mass 180,000) in lysates of wildtype mouse fibroblasts known to express uPARAP/Endo180 (Engelholm et al. 2003), whereas no protein was recognized in lysates of fibroblasts from littermate uPARAP/Endo180-deficient mice.
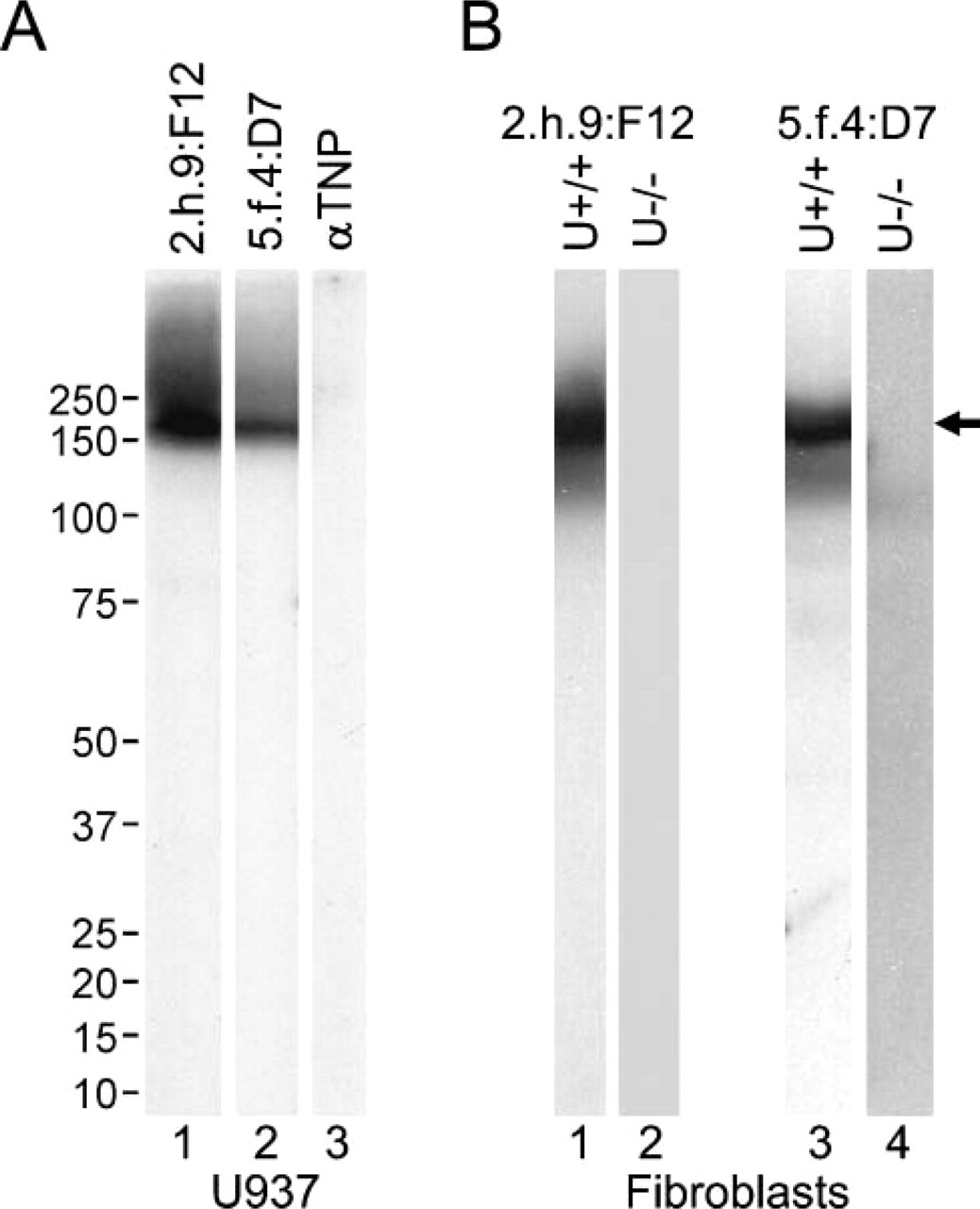
Monoclonal antibodies (MAbs) 2.h.9:F12 and 5.f.4:D7 specifically recognize uPARAP/Endo180. (
To validate the use of the MAbs for detection of uPARAP/Endo180 in tissue sections, we first performed IHC of serial sections from 17 randomly selected tumors. This analysis, which included well-differentiated, moderately differentially, and poorly differentiated tumors, showed that the two MAbs displayed an identical staining pattern, presenting a predominantly granular polarized intracellular and cell surface staining of a subset of stromal cells with fibroblast-like morphology, compatible with the distribution of an endocytic receptor (compare representative examples in Figures 2B and 2F, Figures 2C and 2G, and Figures 2D and 2H). Furthermore, no stromal staining was observed when substituting the uPARAP/Endo180 MAbs with a non-immune, isotype-matched IgG (Figures 2J-2L). No, or very little, staining was observed in normal or tumor-adjacent submucosal connective tissue, suggesting a specific upregulation of uPARAP/Endo180 in oral cancer stroma (Figures 2A and 2E).
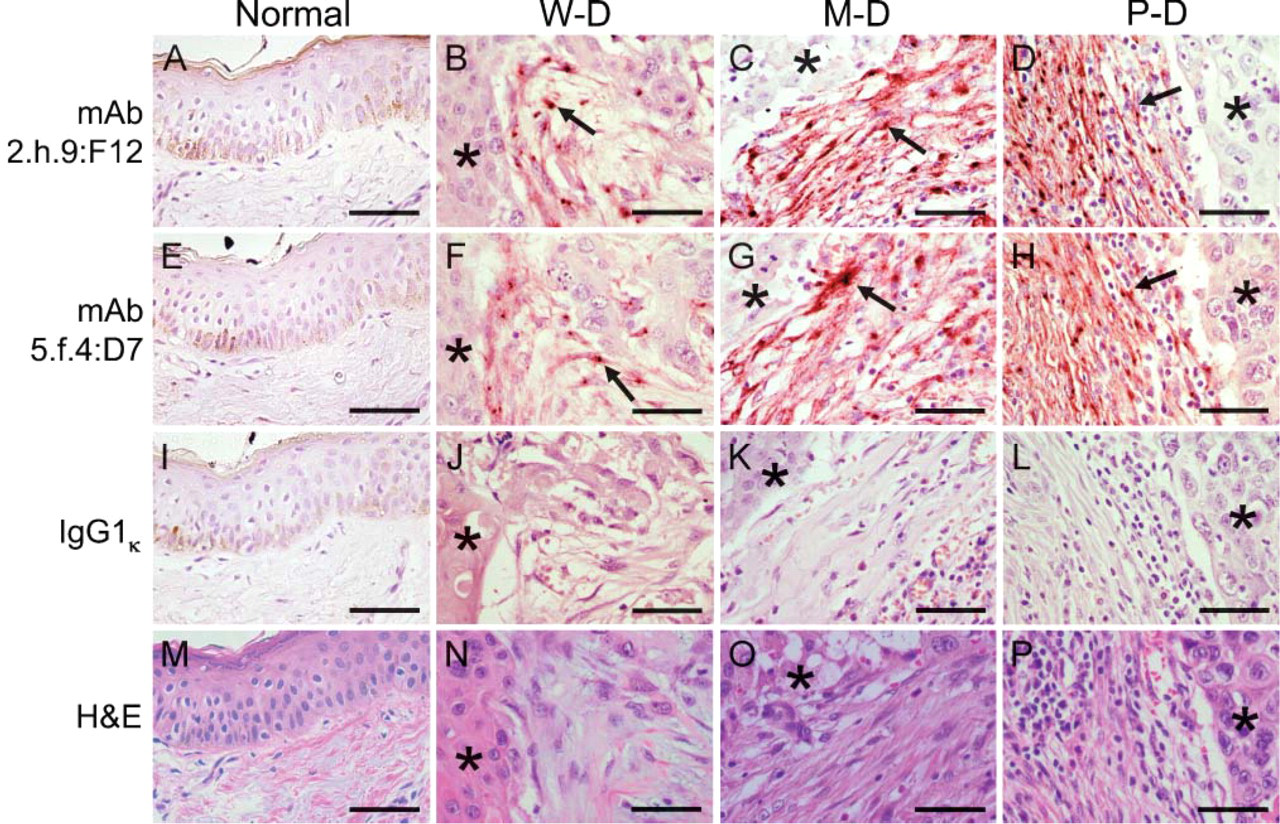
uPARAP/Endo180 can be detected in head and neck squamous cell carcinoma by immunohistochemistry (IHC). Representative examples of IHC staining of serial sections of normal (
Immunostaining of serial sections of the tumors using the uPARAP/Endo180 MAbs and an antibody against the mesenchymal cell marker, vimentin, was performed next to verify that uPARAP/Endo180 was specifically located in stromal cells of head and neck squamous cell carcinoma (Figure 3). This analysis showed that uPARAP/Endo180-positive cells also stained strongly positive for vimentin, providing positive identification of uPARAP/Endo-180-positive cells as stromal fibroblast-like cells (compare Figure 3A with Figure 3B and Figure 3D with Figure 3E). Together this analysis showed that uPARAP/Endo180 could be specifically detected by IHC of paraffin-fixed, archived tissues. The 2.h.9:F12 MAb was used for further studies of uPARAP/Endo180 expression, and staining of serial sections with isotype-matched, non-immune antibodies was included as a negative control in all sections analyzed. Infrequent ambiguous staining (2/112 tumors) was resolved by the staining of parallel sections by MAb 5.f.4:D7. The intensity of stromal immunostaining was estimated for all samples using an arbitrary scale from 0 to 4, as described in Materials and Methods.
Comparison of 43 squamous cell carcinomas and 13 normal head and neck tissues represented on the Biomax tissue array showed increased expression of uPARAP/Endo180 in tumor stroma compared with normal or tumor-adjacent submucosal connective tissue (Figure 4A). The frequency of uPARAP/Endo180-positive tumor samples (defined as having a score of 1 or higher) was 29/43 (67%), as opposed to 4/13 (31%) of normal or tumor-adjacent samples (p<0.025). Likewise, intensity of stromal uPARAP/Endo180 staining was significantly increased in tumor stroma compared with normal or tumor-adjacent submucosal connective tissue with a median intensity score of 1 (range 0-3) for tumors compared with a median intensity score of 0 (range 0-1) for normal tissue (p<0.0012). The identical trend was observed after analysis of tumors represented on the Cybrdi tissue array, although statistical significance was not obtained (Figure 3B). With this array, the frequency of uPARAP/Endo180-positive tumor samples was 15/20 (75%) as opposed to 2/6 (33%) of normal or tumor-adjacent samples (p<0.1). The median intensity score of the tumor stroma was 0.3 (range 0-0.7), compared with 0 (range 0-1) for normal or tumor-adjacent samples (p=0.29).
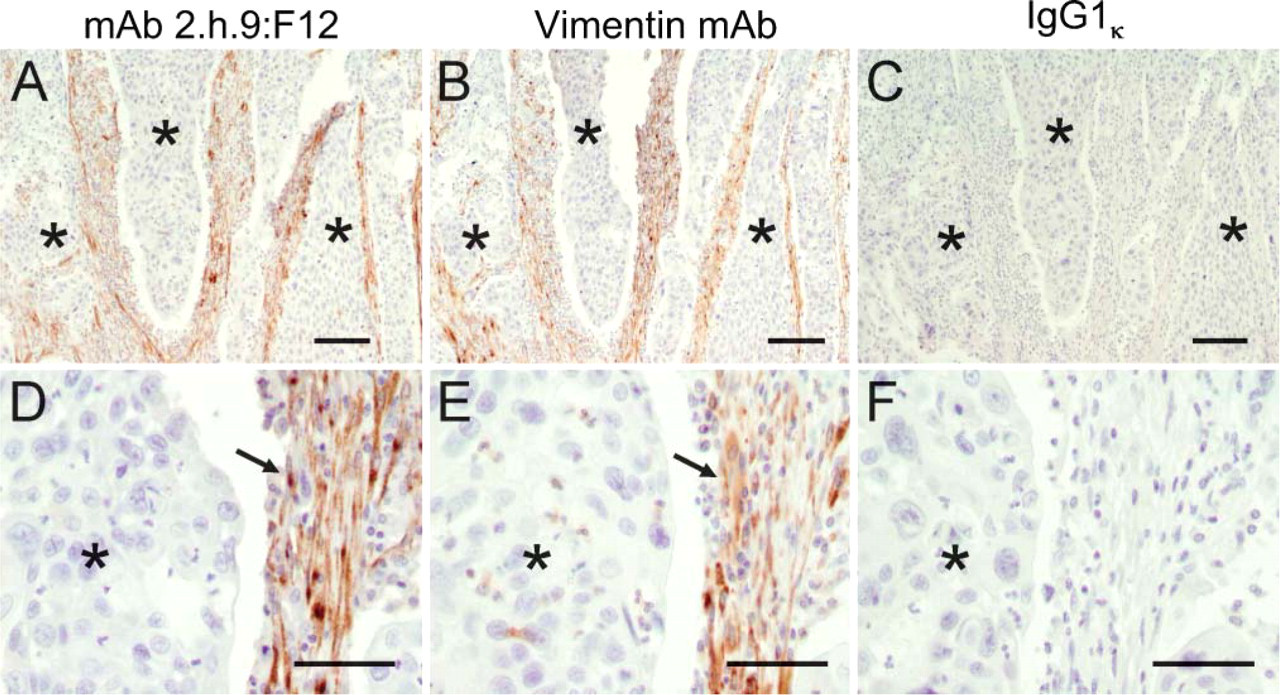
uPARAP/Endo180 is located in stromal cells of head and neck squamous cell carcinoma. Representative examples of low magnification (
Inspection of the staining intensity of tumor samples represented on the Biomax and Cybrdi tissue arrays did not show a clear association between the differentiation grade and extent of uPARAP/Endo180 staining. However, the limited number of samples included on these arrays and potential variability in histological classification criteria precluded a quantitative assessment. Therefore, to investigate the association between tumor differentiation grade and uPARAP/Endo180 expression, we analyzed the expression of an additional 48 tumors classified prior to immunostaining by one pathologist as well-differentiated (n=15), moderately differentiated (n=19), or poorly differentiated (n=15) head and neck squamous cell carcinoma (Figure 3C). This analysis showed no difference in the intensity of uPARAP/Endo180 expression between well-differentiated and moderately differentiated tumors (median 1, range 0-4 for both). However, a significant increase in uPARAP/Endo180 expression was observed in poorly differentiated tumors (median 2, range 0-4) when compared with well-differentiated and moderately differentiated tumors (p<0.04).
Discussion
Intracellular collagen degradation mediated by uPARAP/Endo180 is emerging as a quantitatively important pathway for ECM degradation in the context of malignant tissue remodeling (East et al. 2003; Engelholm et al. 2003; Curino et al. 2005). Recent studies have established that uPARAP/Endo180 expression by tumor stromal cells is critical to tumor expansion by limiting the deposition of collagen within the tumor stroma (Curino et al. 2005). However, studies of the expression and localization of the collagen internalization receptor in human malignancies are still very limited (St Croix et al. 2000; Schnack Nielsen et al. 2002). Here we performed a detailed IHC study of the expression of uPARAP/Endo180 in human head and neck squamous cell carcinomas of diverse origin and variable histological grades. Use of independently derived MAbs defining non-overlapping epitopes on uPARAP/Endo180, as well as the use of isotype-matched control antibodies, allowed the unambiguous identification of uPARAP/Endo180-expressing cells in these head and neck malignancies. Our analysis revealed that the collagen internalization receptor is frequently expressed by fibroblast-like, vimentin-positive cells embedded in collagen-rich stroma adjacent to nests of tumor cells. Furthermore, uPARAP/Endo180 expression was increased in the tumor stroma as compared with tumor-adjacent connective tissue or normal submucosal connective tissue, and the receptor displayed the most prominent expression in the stroma of poorly differentiated tumors. These data strongly suggest that uPARAP/Endo180 contributes to the pathological destruction of connective tissue during head and neck squamous cell carcinoma progression by facilitating cellular uptake and intracellular degradation of collagen by tumor stromal cells. In addition to its role in collagen catabolism, uPARAP/Endo180 forms a trimolecular complex with urokinase plasminogen activator and the urokinase plasminogen activator receptor (Behrendt et al. 2000). Interestingly, expression of the urokinase plasminogen activator receptor is also strongly upregulated in stromal fibroblasts of oral squamous cell carcinoma (Lindberg et al. 2006), suggesting that uPARAP/Endo180 may also promote oral squamous cell carcinoma progression by modulating the activity of the plasminogen activation system.
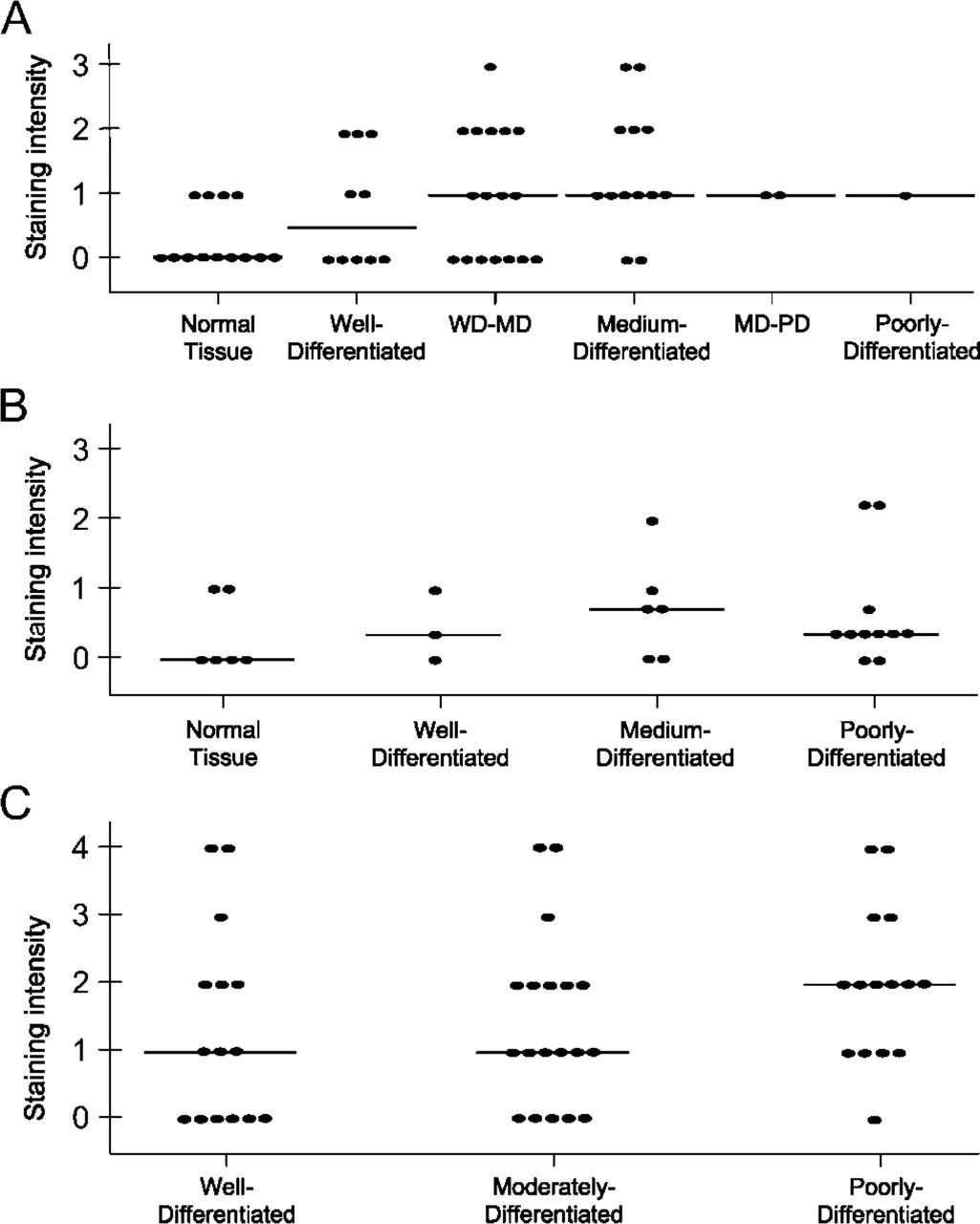
Increased expression of uPARAP/Endo180 in head and neck squamous cell carcinoma. Scatterplot of the intensity of IHC staining of uPARAP/Endo180 in tissue sections represented on the Biomax (
Many mechanistic aspects of intracellular collagen degradation remain poorly understood. However, extra-and intracellular collagen degradation pathways appear to operate at least partially independently of each other (Everts et al. 1989; Holmbeck et al. 1999). This property of the intracellular collagen degradation pathway, when combined with the findings in this paper, suggests that uPARAP/Endo180 could represent a new therapeutic target for the clinical management of head and neck malignancies.
Footnotes
Acknowledgements
The work was supported by the National Institute of Dental and Craniofacial Research Intramural Research Program and by a grant from the Department of Defense (DAMD-17-02-0693 to THB). J. Shireman was supported by the Howard Hughes Medical Research-NIH Research Scholars Program. L.H.E, N.B., and D.H.M were supported by EU contract LSHC-CT-2003-503297 and by grants from the Danish Cancer Society, the Danish Cancer Research Foundation, and the Danish Medical Research Council.
We thank Drs. Mary Jo Danton and J. Silvio Gutkind for carefully reading this manuscript.
