Abstract
The 80-kDa human sperm antigen (HSA) has demonstrated to be a promising candidate for development of an antifertility vaccine because it is a sperm-specific, conserved, and immunogenic protein. The present study demonstrates the androgen-regulated expression of 80-kDa HSA in testis and epididymis of rat by immunohistochemistry (IHC), using its specific antibodies. Developmental expression of 80-kDa HSA was investigated on days 10, 20, 40, 60, and 90 of age in the testis and epididymis by IHC, and relative staining intensity was estimated by image analysis using BIOVIS software. On days 10 and 20, no significant staining was observed in the testis and epididymis, whereas it gradually increased from day 40 onwards. The highest staining was seen on day 90 in both testis and epididymis. Gradual increase in expression of 80-kDa HSA after day 40 suggests that it is possibly regulated by androgen. To study the androgen-regulated expression of 80-kDa, adult male rats were treated with 75 mg/kg body weight of ethylene dimethane sulfonate (EDS), which selectively destroys Leydig cells and thus induces complete androgen withdrawal. It was observed that the staining intensity decreased following EDS treatment in rat testis as well as epididymis, and it was regained after supplementation with dihydrotestosterone. Increased expression during sexual maturation at the time of testosterone surge and its regulation by anti-androgen/androgen treatment suggest androgen-dependent expression of 80-kDa HSA in rat testis and epididymis.
Keywords
S
The 80-kDa human sperm antigen (HSA) has been proven to be one of the promising candidate antigens for development of an antifertility vaccine because it is sperm specific, conserved, highly immunogenic, and induces immunological infertility following active/passive immunization. The 80-kDa HSA is a glycoprotein initially identified from human sperm extract by Western blot technique using serum of an immunoinfertile woman (Bandivdekar et al. 1991, 1992). Active immunization of male and female rats with purified 80-kDa HSA resulted in infertility (Bandivdekar et al. 1991, 1992, 2001a). Moreover, the partial N-terminal amino acid sequence of 80-kDa HSA (peptide NT) and the amino acid sequence corresponding to its peptides obtained by endoproteinase Lys-C digestion (peptides 1, 2, 3, and 4) and by endoproteinase Glu-C digestion was determined. All peptides did not show homology with any of the specific known proteins in the protein database. Peptides NT 1, 2, 3, and 4 were synthesized, conjugated to KLH, and used as an immunogen. These peptides, with the exception of peptide 3, elicited significant antibody titer in rabbits and mimicked immunobiological properties of native protein (Bandivdekar et al. 2001b). Passive administration of antibodies to peptides NT 1, 2, and 4 rendered male and female rats infertile (Vernekar et al. 2004; Bandivdekar et al. 2005). Immunofluorescent studies demonstrated its specific localization on rat and human spermatozoa (Bandivdekar et al. 1992, 2001a). By means of immunohistochemistry (IHC), the antigen was found to be specifically localized in the human testis and epididymis but absent in other somatic tissues such as brain, kidney, liver, heart, lung, spleen, intestine, appendix, and nerve (Bandivdekar et al. 2001a).
The present study describes the developmental expression of the 80-kDa HSA and its androgen-dependent expression in the rat testis and epididymis.
Materials and Methods
Animals
Belgium albino rabbits and Holtzman rats used for the study were bred at the Institute's animal house. Rats were housed in-group of four per cage, whereas rabbits were caged individually under standard laboratory conditions in an illumination-controlled room at 25C. Animals were provided with standard chow twice daily and water was supplied ad libitum.
Ethical clearance for use of animals for experimentation was obtained from the Institutional Animal Ethics Committee prior to the initiation of the study. Experiments were performed in accordance with the Guiding Principles for the Care and Use of Laboratory Animals.
Purification of 80-kDa HSA and Generation of Antibodies in Rabbits
The 80-kDa HSA was purified from human sperm extract as described earlier (Bandivdekar et al. 1991, 2001a). In brief, the sperm pellet obtained by centrifugation of semen sample was washed three times with 0.01 M PBS, pH 7.4, and sperm proteins were solubilized with 0.05% (w/v) sodium deoxycholate (Sigma; St Louis, MO) in 0.01 M Tris-HCl buffer, pH 8. Solubilized sperm proteins were then fractionated with 40% (w/v) ammonium sulfate (Qualigens; Mumbai, India). The supernatant was then further fractionated by sequential gel permeation chromatography followed by ion-exchange chromatography using fast protein liquid chromatography. Antigen purity was ascertained by SDS-PAGE.
Homogeneous 80-kDa HSA was used as an immunogen to generate polyclonal antibodies in rabbits. Belgium albino white rabbits were actively immunized with 100 μg of purified 80-kDa HSA emulsified with Freund's complete adjuvant (Sigma). Booster injections of 100 μg each of purified 80-kDa HSA emulsified with Freund's incomplete adjuvant (Sigma) were administered at 4-week intervals. Animals were bled at 2-week intervals and sera harvested. Antibody titer was determined by ELISA using the purified 80-kDa HSA as well as sperm extract-coated wells. Antibody specificity was checked by Western blot analysis of sperm extract.
Western Blot Analysis
Sperm proteins were resolved on discontinuous polyacrylamide gel (5% stacking and 12% resolving gel) at constant voltage (100 V) on a Bio-Rad minigel apparatus (Mini-Protean II; Bio-Rad, Richmond, CA) using the method of Laemmli (1970). Proteins were transferred to a nitrocellulose membrane at 100 V for 1 hr with cooling (Towbin et al. 1979). The unoccupied sites on the membrane were blocked by incubating with PBS containing 10% (w/v) non-fat dry milk powder for 2 hr at room temperature on a rocking platform. The membrane was then incubated for 11/2 hr with antibodies to 80-kDa HSA raised in rabbits diluted 1:200 with PBS. After washing twice with PBS containing 0.1% Tween-20 and once with PBS, the membrane was further incubated with alkaline phosphatase-conjugated goat anti-rabbit gamma globulin (Banglore Genei; Banglore, India) diluted to 1:1000 in PBS. The membrane was washed similarly as mentioned above, and the immunoreactive proteins were visualized using nitroblue tetrazolium and 5-bromo-4-chloro-3-indolyl phosphate (NBT-BCIP; Banglore Genei) as a substrate.
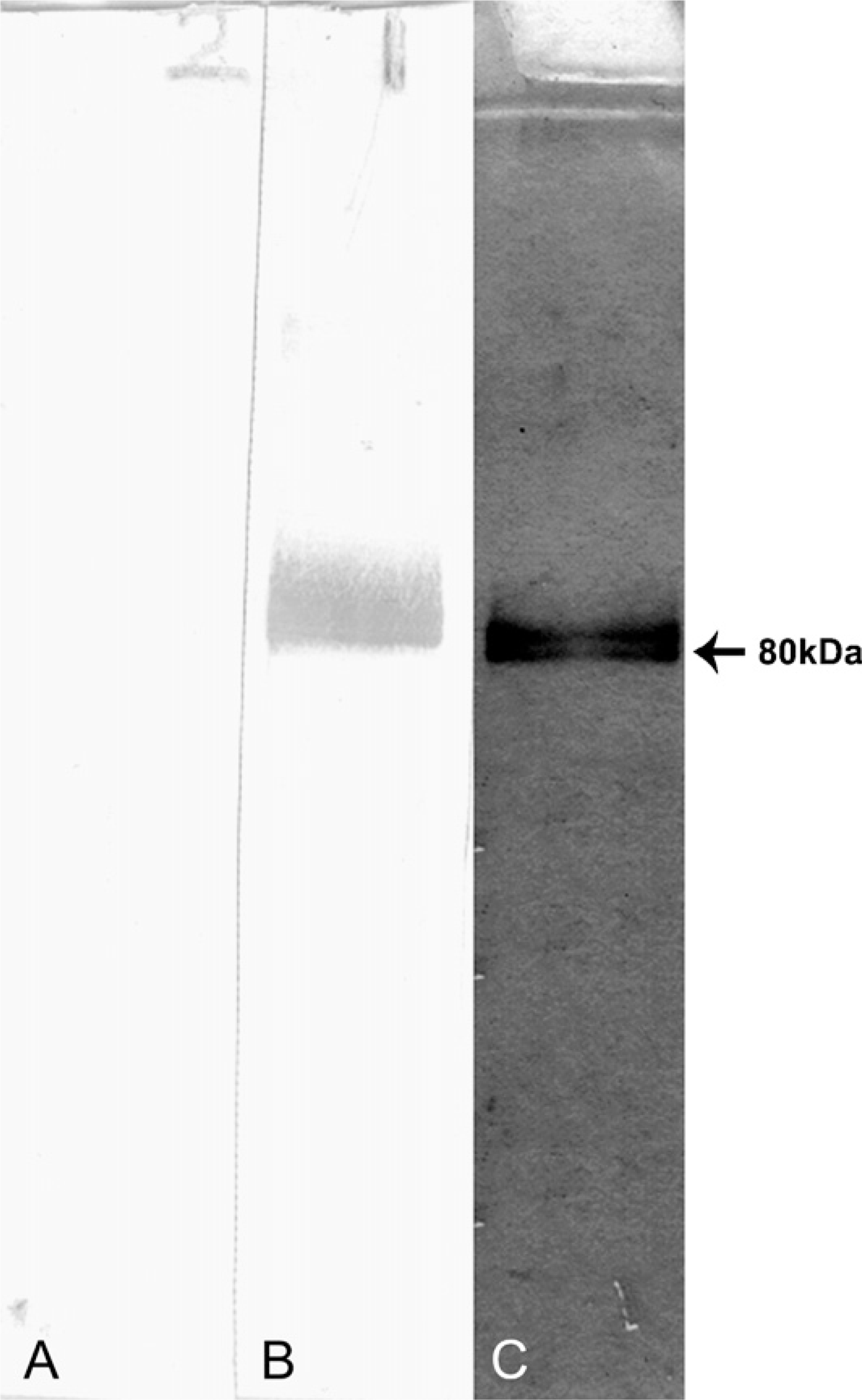
Homogeneity of purified 80-kDa human sperm antingen (HSA) was checked by SDS-PAGE. The protein resolved on 12% SDS-PAGE and was silver stained showing a single band of ~80 kDa (
Indirect Immunofluorescence Staining of Marmoset Spermatozoa
Indirect immunofluorescent localization of 80-kDa HSA was evaluated on live as well as fixed rat spermatozoa from cauda epididymis. For staining of live spermatozoa, sperm were washed and suspended to the concentration of 10 × 106 sperm/ml in Ham's F-10 nutrient medium (HiMedia; Mumbai, India) containing 0.2% BSA (sperm-washing medium); 0.2 ml of sperm suspension was incubated for 30 min in the presence of Ham's F-10 medium containing 5% BSA (wt/v) to block nonspecific sites at 37C. Following centrifugation, the sperm pellet was incubated for 1 hr at 37C with a 1:100 dilution of either polyclonal antibodies to 80-kDa HSA raised in rabbit or with preimmune rabbit sera (negative control). The sperm suspension was washed four times with sperm-washing medium and then incubated with 1:50 diluted fluorescein isothiocynate (FITC)-conjugated goat anti-rabbit gamma globulin for 1 hr at 37C in the dark. The sperm suspension was washed four times with sperm-washing medium as described previously and mounted on slides with coverslips using 90% glycerol solution as mountant containing
For staining of fixed spermatozoa, sperm were collected and washed three times with PBS by centrifugation at 700 × g for 10 min and then resuspended in PBS at a concentration of 5 × 106 sperm/ml. This sperm suspension was smeared on glass slides, air dried, fixed in 4% (wt/v) paraformaldehyde in PBS for 10 min at 4C, and then washed three times in PBS for 10 min each. Nonspecific sites were blocked for 2 hr using PBS containing 5% (wt/v) BSA, and preparations were placed in a humidified air chamber at 37C. Excess of blocking buffer was removed before incubation with primary antibody. Slides were incubated overnight at 4C with 1:100 dilution of either polyclonal antibodies to 80-kDa HSA raised in rabbit or with preimmune rabbit sera (negative control). Slides were washed four times with plain PBS for 10 min each and then incubated with 1:50 diluted FITC-conjugated goat anti-rabbit gamma globulin for 2 hr at 37C in the dark. Slides were washed with PBS as described previously, mounted, and examined as stated above.
IHC Localization of 80-kDa HSA in Testis and Epididymis of Rats at Different Ages
Testis and epididymis of rats (
Quantitative Image Analysis
Staining intensity was analyzed using BIOVIS Image Plus Software (Expert Vision Labs Pvt Ltd; Mumbai, India) to assess the relative levels of expression of 80-kDa HSA in testicular and epididymal sections of rats from different groups. Five different areas were digitally captured at random within the testicular and epididymal sections of each rat, providing a systematic survey throughout each region for each case using a live color video camera mounted onto a Zeiss Axioplan microscope (Carl Zeiss) with a neutralizing blue filter. All parameters including the lamp intensity video camera setup and microscope calibration were held constant. Immunoreactivity of each tissue section was measured by subtracting integrated optical density (IOD) of the staining of the sections treated with preimmune sera (negative control) from that of the IOD of the staining of the sections treated with immune sera (test). Data were plotted as the immunoreactivity per field ± SD for each group.
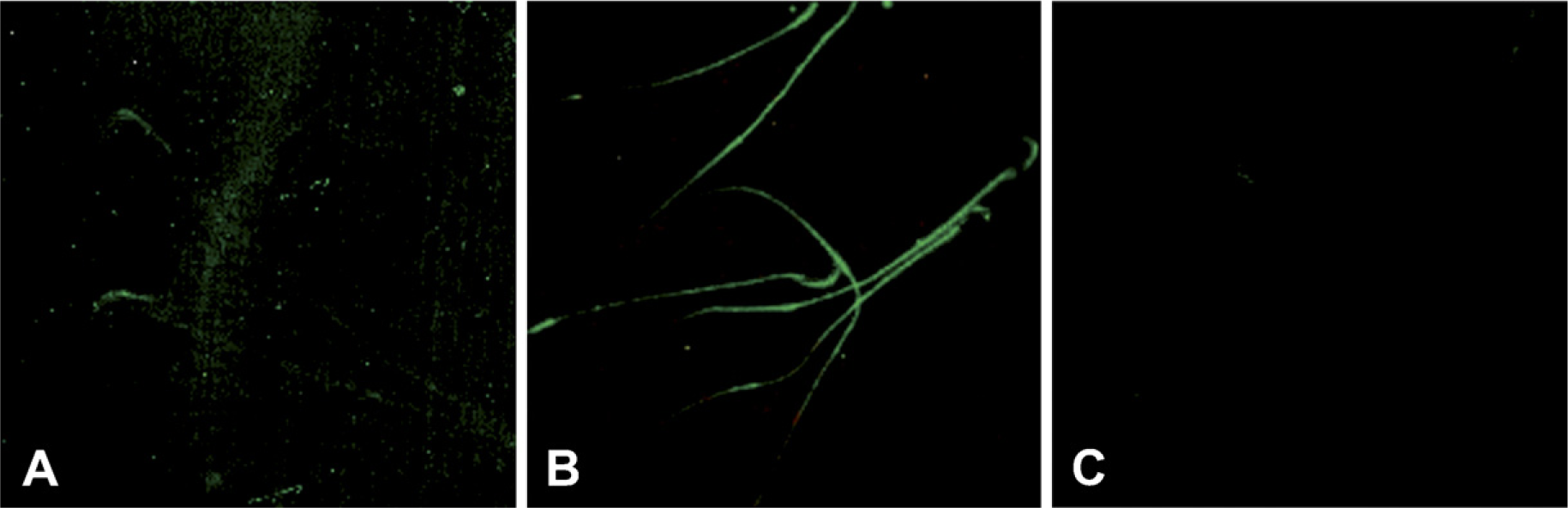
Immunofluorescent localization of 80-kDa HSA in live and 4% paraformaldehyde-fixed rat spermatozoa. Localization of 80-kDa HSA was observed on the acrosomal region of live spermatozoa (
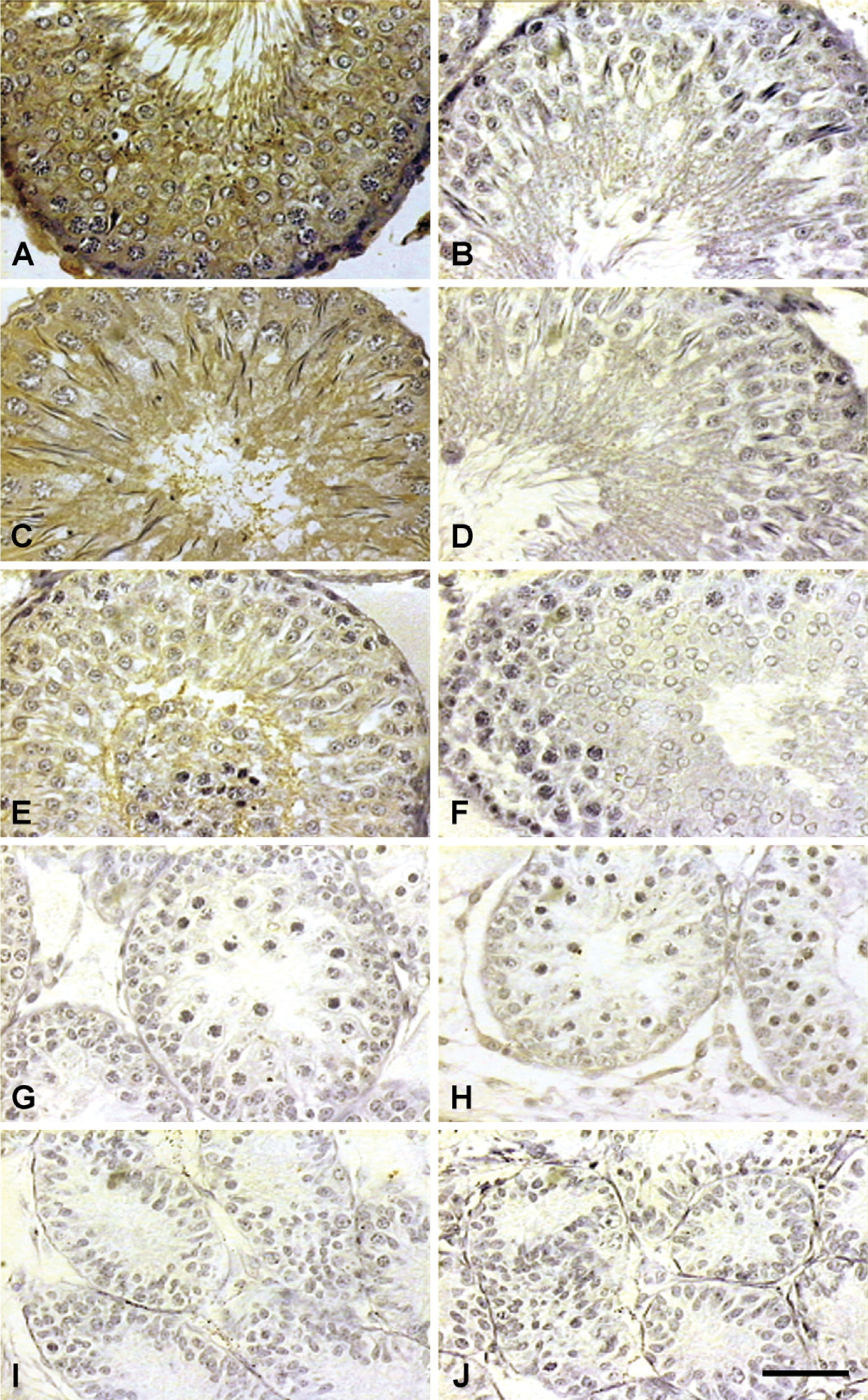
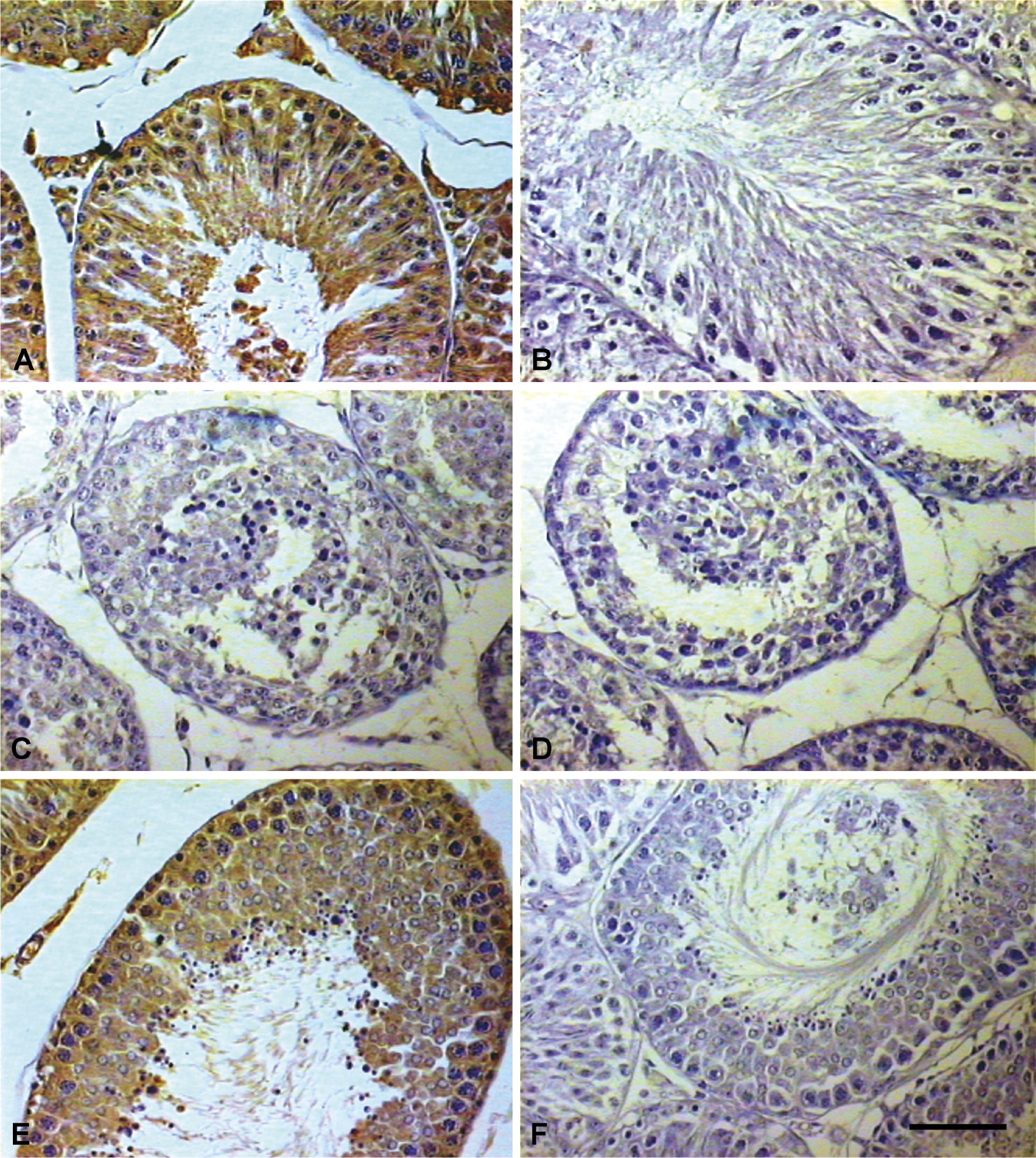
Immunohistochemical (IHC) localization of 80-kDa HSA in testis of rat at different ages. Expression of 80-kDa HSA gradually increased from days 40 (
IHC localization of 80-kDa HSA in epididymis of rats at different age. Expression of 80-kDa HSA in caput on day 90 (
Localization of 80-kDa HSA in Testis and Epididymis of Ethylene Dimethane Sulfonate (EDS)-treated Rats
Androgen-regulated expression of 80-kDa HSA was studied in 90-day-old adult male rats weighing ~250 ± 25 g.
To induce androgen withdrawal, rats received a single IP injection of 75 mg/kg EDS (Sigma) in DMSO and water (1:3 v/v). In another group of EDS-treated rats, 250 μg dihydrotestosterone (DHT; Sigma) was administered IP in 100 μl olive oil daily from day 5 of EDS treatment. Control animals were also administered an equal volume of DMSO: water (1:3 v/v) to parallel the EDS treatment.
Animals were sacrificed on the 10th day after the injection of DMSO or EDS, and the testis and epididymis were fixed in Bouin's solution and processed for IHC using antibodies to 80-kDa HSA as described above. Animals were bled on day of EDS treatment (day 1) and then on days 5 and 10 and sera harvested. Serum androgen levels of all animals were estimated by radioimmunoassay (RIA).
Testosterone Assay
Serum testosterone levels were estimated in the samples (
Statistical Analysis
Data were analyzed by ANOVA with Dunnet's multiple range tests using SPSS for Windows (v. 11.0; SPSS Inc., Chicago, IL) to ascertain the significance of variance of 80-kDa HSA expression among different groups of rats. Data were expressed as mean ± SD;
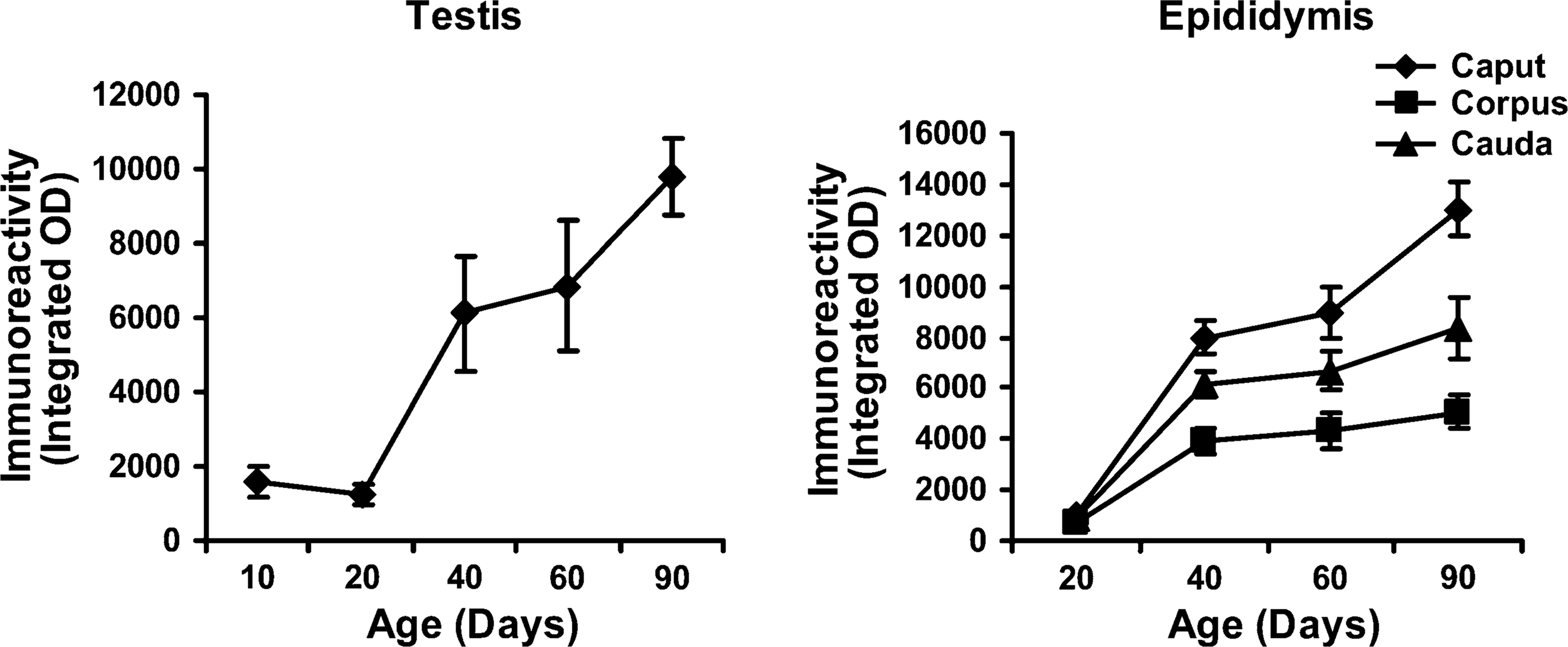
Graphic representation of quantitative analysis of the staining intensity showing developmental expression of 80-kDa HSA in rat testis and epididymis. Data are expressed as mean of the immunoreactivity ± SD (
Results
Purification of 80-kDa HSA and Characterization of Antibodies to 80-kDa HSA
Homogeneous 80-kDa HSA was obtained following sequential gel permeation and ion-exchange chromatography of human sperm extract. Purified protein showed the single protein band of ~80 kDa following SDS-PAGE (Figure 1). Immunization of rabbits with purified 80-kDa HSA elicited a gradual increase in antibody titer. Antibodies specifically reacted with purified 80-kDa HSA as well as sperm extract coated into the wells of microtiter ELISA plates. Western blot analysis of sperm extract using antibodies to 80-kDa HSA showed the specific reactivity with a single 80-kDa protein band (Figure 1). Preimmune serum did not show any reactivity.
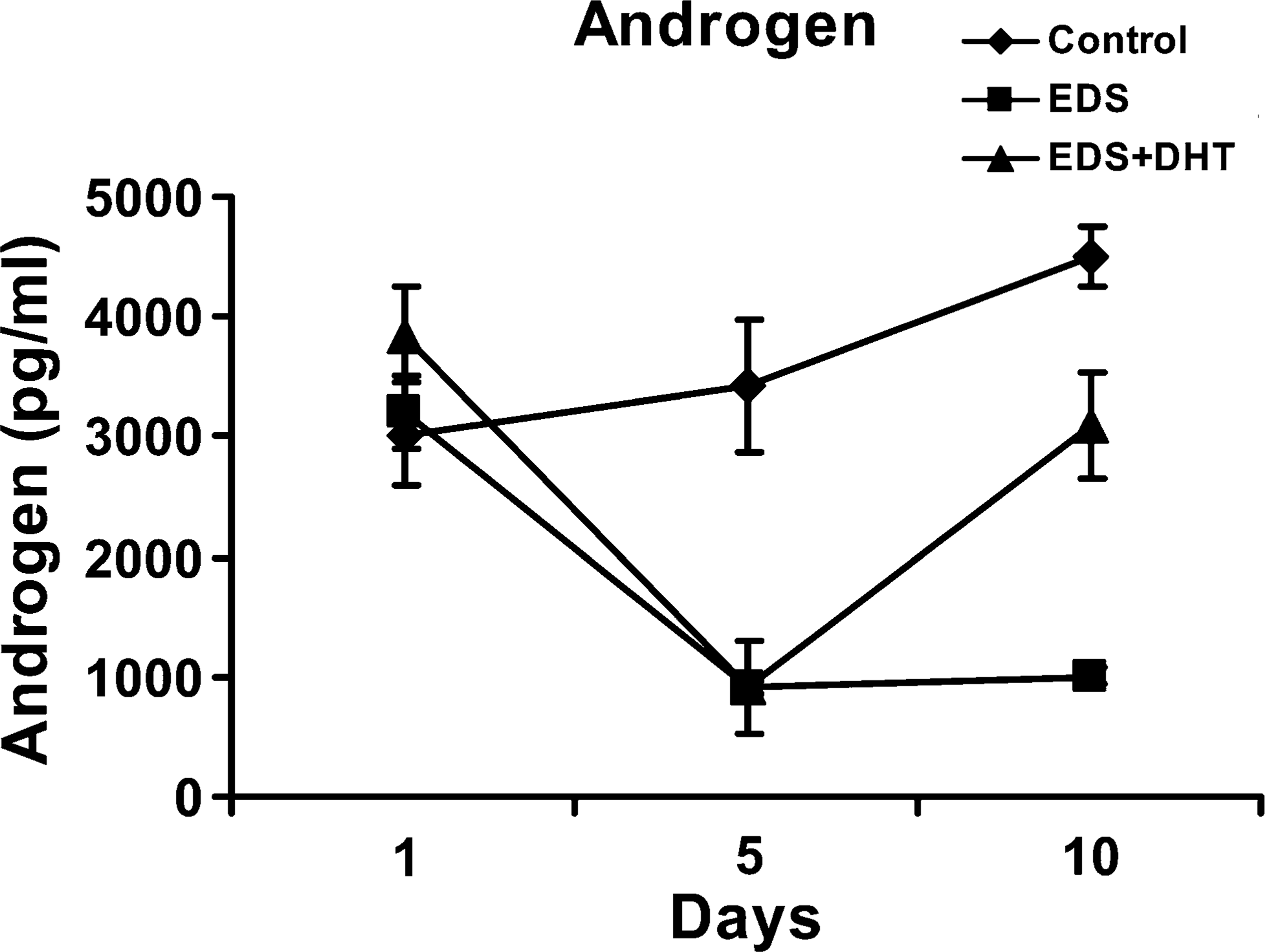
Graphic representation showing the effect of ethylene dimethane sulfonate (EDS) and supplementation of dihydrotestosterone (DHT) on serum androgen levels. Data are expressed as mean of the androgen level (pg/ml) ± SD (
Localization of 80-kDa HSA in Rat Sperm
Immunofluorescent localization of 80-kDa HSA using its specific antibody raised in rabbits was observed predominantly on the acrosomal region of live and fixed rat spermatozoa. In the case of immunofluorescent localization of fixed spermatozoa, the protein was also localized on the tail region (Figure 2). Sperm incubated with preimmune rabbit serum as the negative control did not show any staining.
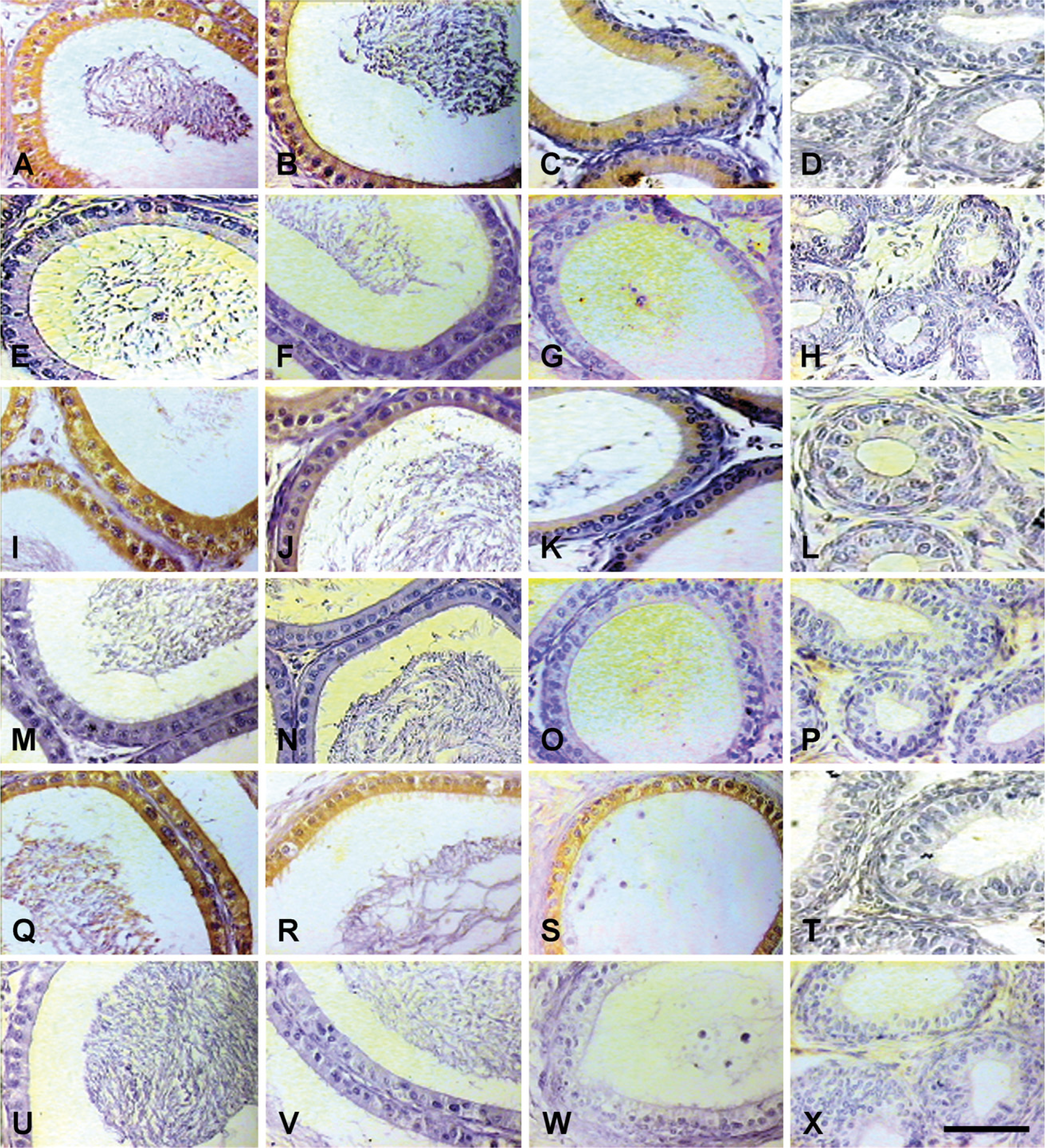
IHC localization of 80-kDa HSA in testis of rats treated with EDS only and EDS supplemented with DHT. Expression of 80-kDa HSA was suppressed significantly in rats treated with EDS (
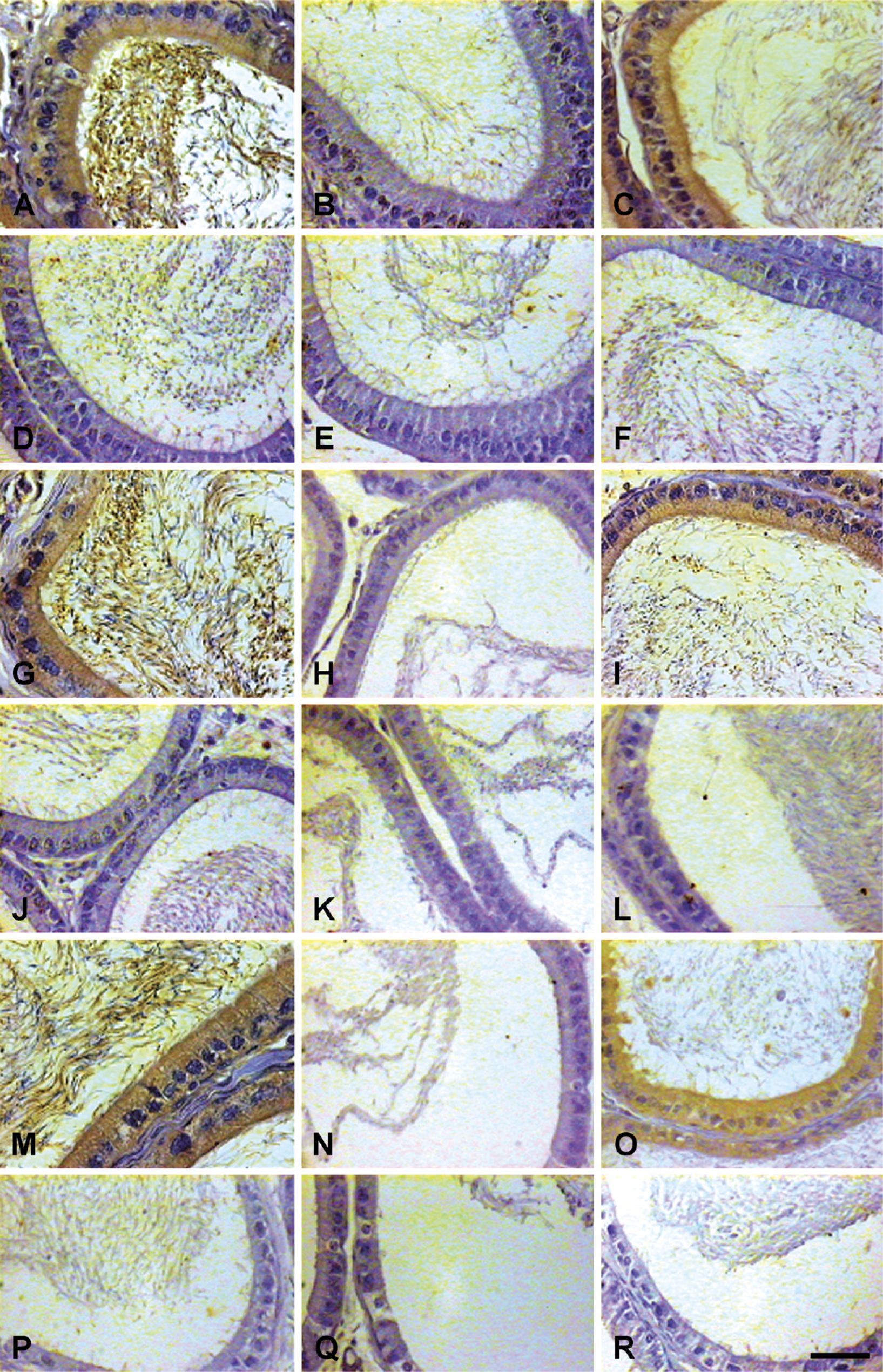
IHC localization of 80-kDa HSA in epididymis of EDS-treated and DHT-supplemented rats. Expression of 80-kDa HSA was suppressed significantly in rats treated with EDS (
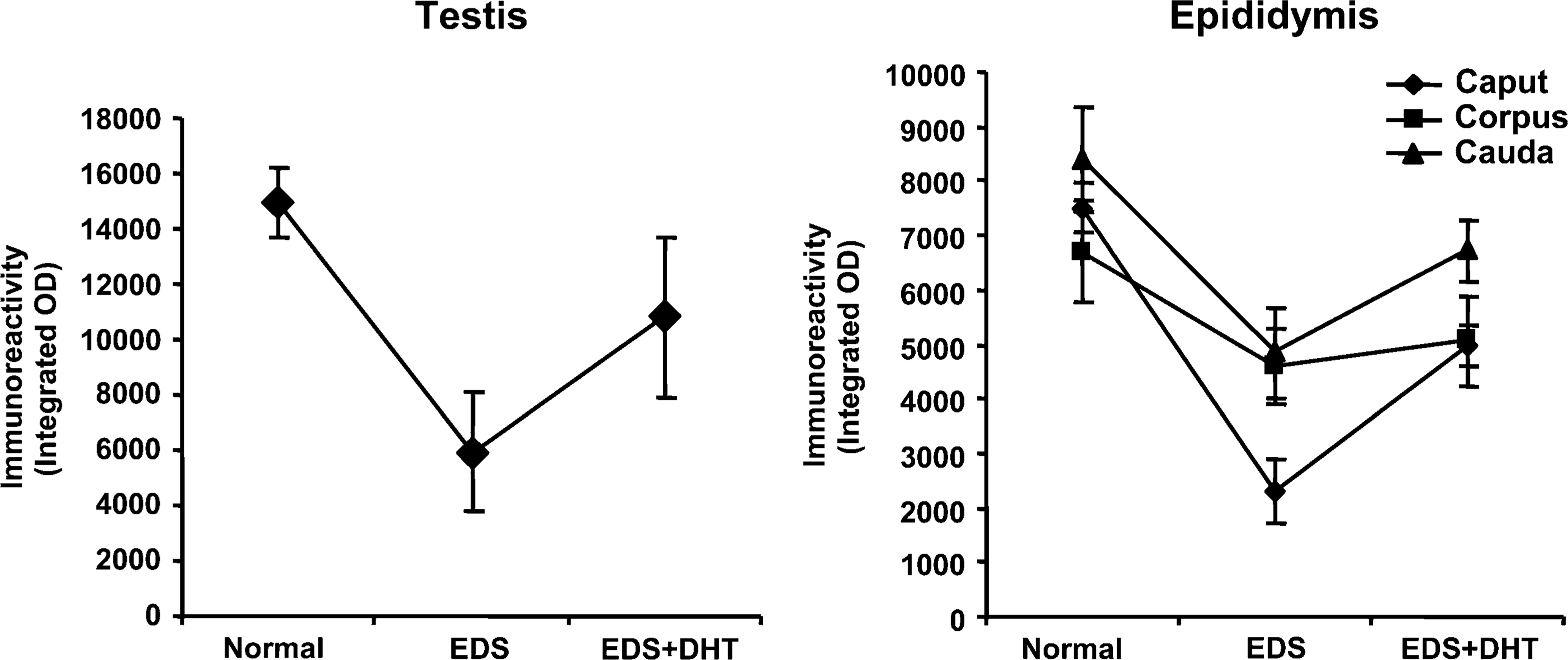
Graphic representation of quantitative analysis of the staining intensity showing androgen-regulated expression of 80-kDa HSA in rat testis and epididymis. Data are expressed as mean of the immunoreactivity ± SD (
Developmental Expression of 80-kDa HAS
Figure 3 and Figure 4 show the IHC localization and Figure 5 shows the estimate of immunoreactivity of 80-kDa HSA in testis and epididymis of 10-, 20-, 40-, 60-, and 90-day-old rats. Staining intensity or the immunoreactive expression of 80-kDa HSA gradually increased from day 40 onwards, whereas on days 10 and 20 no significant staining was observed in the testis and epididymis of the rats. The significantly increased expression of antigen from day 40 is suggestive of androgen-dependent expression of 80-kDa HSA in the testis and epididymis.
Androgen-regulated Expression of 80-kDa HAS
IP administration of a single dose of EDS (75 mg/kg body weight) resulted in selective elimination of Leydig cells with endogenous suppression of androgen levels. In the animal control group, seminiferous tubules and interstitium were found tightly packed in the testis, whereas no histologically recognizable Leydig cells were detected on day 10 in the testicular sections of rats treated with EDS. Testosterone levels were suppressed in the EDS-treated animals. Circulating androgen levels were elevated following supplementation of EDS-treated animals with DHT to replace endogenous androgen levels (Figure 6). Immunohistochemically, expression of 80-kDa HSA (Figure 7 and Figure 8) and the estimated immunoreactivity (Figure 9) in the testicular and epididymal sections decreased significantly (
Discussion
The 80-kDa HSA is a sperm-specific, immunogenic, and conserved protein responsible for inducing immunological infertility in both male and female rats, suggesting its suitability as a candidate antigen for the development of a contraceptive vaccine (Bandivdekar et al. 1991, 1992, 2001a). The partial N-terminal amino acid sequence of 80-kDa HSA (peptide NT) and the amino acid sequence of its peptides obtained by enzymatic digestion (peptides 1–6) revealed no significant sequence homology with any of the known proteins in the gene bank (Bandivdekar et al. 2001a, b). Subsequently, peptides NT 1, 2, and 4 were synthesized, and these peptides were demonstrated to immunobiologically mimic the native protein. Passive administration of antibodies to these peptides rendered male and female rats infertile (Vernekar et al. 2004; Bandivdekar et al. 2005).
Western blot analysis of sperm extract reacted specifically to a single protein band of 80 kDa with rabbit anti-80 kDa HSA antibody. This suggests that the antibody used for the study was highly specific to 80-kDa HSA. Immunohistochemically, it was observed that 80-kDa HSA is specifically expressed in human testis and epididymis and not in other somatic tissues (Bandivdekar et al. 2001a). The present study evaluates the regulation of 80-kDa HSA expression in rat testis and epididymis. It was observed that expression of 80-kDa HSA gradually increased in the testis and epididymis of rats after day 40. No significant expression of 80-kDa HSA was observed in the testis and epididymis of rat on days 10 and 20, suggesting the onset of its expression with initiation of puberty.
Increased expression after day 40 relates to the distinct elevation of androgen levels in the rat during sexual maturation (Premchandran and Hegde 1991), suggesting that expression of 80-kDa HSA is possibly regulated by androgen. The effect of androgen on expression of 80-kDa HSA was investigated by treatment of adult male rats with Leydig cell toxin, EDS, followed by supplementation with DHT. EDS treatment has been reported to cause selective destruction of Leydig cells with suppression of testicular and serum testosterone level, and the effect was reversed after day 21 following treatment (Morris et al. 1986; O'Leary et al. 1987; Yang et al. 2006). The present study also demonstrates suppression of androgen levels following EDS treatment, which resulted in suppression of 80-kDa HSA expression in testis and epididymis. DHT supplementation following EDS treatment resulted in elevation of androgen levels with a significant increase in 80-kDa HSA expression.
In conclusion, the present study demonstrates that 80-kDa HSA is developmentally expressed. The gradual increase in the expression of 80-kDa HSA in the testis and epididymis of adult rat appears to be directly correlated with the increase in androgen levels. Data suggest the androgen-dependent expression of 80-kDa HSA.
Footnotes
Acknowledgements
The authors acknowledge the Lady Tata Memorial Trust, Mumbai, India for providing a Senior Research Fellowship (to BGK).
The authors also acknowledge Dr. C.P. Puri, Director, National Institute for Research in Reproductive Health for his constant support and encouragement. The technical assistance of Ravi Kadam, Shilpa Velhal, Jacintha Pereira, biochemistry staff, and animal house staff is gratefully acknowledged. This manuscript has been assigned the Institute publication number NIRRH/MS/16/2006.
