Abstract
Lung development takes place in a relatively low-oxygen environment, which is beneficial for lung organogenesis, including vascular development. Hypoxia-inducible factor (HIF)-1 plays an important role in mediating oxygen-regulated events. HIF-1 is stable and initiates gene transcription under hypoxia, whereas in normoxia, interaction with the von Hippel-Lindau (VHL) tumor suppressor protein leads to rapid degradation of the HIF-1α subunit. Interaction with VHL requires hydroxylation of HIF-1α proline residues by prolyl hydroxylases (PHDs). We investigated the expression of the various components regulating HIF-1α stability in first trimester (8-14 weeks) human lungs. Spatial expression was assessed by immunohistochemistry and temporal expression by quantitative PCR. Immunoreactivity for PHD1, PHD3, and seven in absentia homolog (SIAH) 1 was noted in the pulmonary epithelium. PHD2 was not expressed in the airway epithelium, but in the lung parenchyma. HIF-1α and vascular endothelial growth factor (VEGF) immunoreactivity were primarily detected in the branching epithelium. HIF-2α and ARNT proteins localized to the developing epithelium as well as mesenchymal, most likely vascular, structures in the parenchyma. VEGF receptor 2 (VEGFR2) was found in the subepithelium as well as in vascular structures of the mesenchyme. All components of the VEC complex (VHL, NEDD8, and Cullin2) were found in the epithelium. Quantitative PCR analysis demonstrated that VEGF, VEGFR1, HIF-1α, HIF-2α, ARNT, PHD1, PHD2, PHD3, and SIAH1 gene expression was constant during early pulmonary organogenesis. Cumulatively, the data suggest that the lung develops in a low-oxygen environment that allows for proper vascular development through HIF-regulated pathways.
L
Normal pulmonary development takes place in the relative hypoxic environment of the uterus (Lee et al. 2001), and this low fetal oxygen environment is beneficial for lung organogenesis, including vascular development (Semenza 2005; van Tuyl et al. 2005). In most mammalian systems, the cellular responses to oxygen alterations are mediated through the highly conserved hypoxia-inducible factor (HIF) family of transcriptional regulators. The HIF transcriptional complex is a heterodimer composed of one of three α-subunits (HIF-1α, HIF-2α, or HIF-3α) and a β-subunit (ARNT; also known as aryl hydrocarbon receptor nuclear translocator) (Paul et al. 2004). Regulation of HIF by oxygen occurs through modifications of the α-subunit, whereas ARNT is a constitutive nuclear protein and is not affected by oxygen. Under hypoxic conditions, the α-subunit is stable, allowing it to accumulate in the nucleus where, upon binding to ARNT, it recognizes HIF-response elements within the promoter regions of hypoxia-responsive target genes. Under normoxic conditions, the α-subunit is rapidly degraded by means of ubiquitination and proteasomal degradation (half-life <5 min in 21% O2) (Berra et al. 2006). Ubiquitination and proteasomal degradation of the α-subunit require von Hippel-Lindau (VHL) tumor suppressor protein (Maxwell et al. 1999). VHL is the HIF-α recognition component of a ubiquitin E3 ligase complex, VEC (Kim and Kaelin 2003), which consists of elongins B and C, Rbx1, and Cullin2 (Stickle et al. 2004). Recognition of HIF-α by VHL occurs only when two conserved proline residues (Pro 402 and Pro 564) in the oxygen-dependent degradation domain of HIF-α are hydroxylated (Ivan et al. 2001; Jaakkola et al. 2001). This hydroxylation is catalyzed by a family of prolyl hydroxylase domain enzymes termed PHD1, PHD2, and PHD3 (Berra et al. 2003; Appelhoff et al. 2004). PHDs are also degraded by E3-ubiquitin ligases, namely, SIAH (seven in absentia homolog) 1 and 2. The degradation is enhanced by hypoxia and SIAHs are transcriptionally upregulated during hypoxia (Berra et al. 2006). Thus, various proteolytic mechanisms are responsible for control of stability of HIF-1α.
We (van Tuyl et al. 2005) and others (Asikainen et al. 2005,2006) have reported that HIF-1α plays an important role in mediating oxygen-regulated events of pulmonary vascular development. However, we did not investigate the components regulating HIF-1α stability in the developing lung. We determined the spatial expression of various components of the HIF system in the human lung during the pseudoglandular stage of lung development (8-14 weeks) when the primitive pulmonary vascular system develops.
Materials and Methods
Human Lung Samples
All tissues were collected after informed consent in accordance with the Ethics Guidelines of the Faculty of Medicine, University of Toronto. Human lung samples were obtained from elective terminations of pregnancies. Samples were processed for immunohistochemistry (IHC) and for quantitative RT-PCR. Gestational ages for the samples used for IHC were 8 weeks (
IHC
Lung samples were fixed overnight in 4% (v/v) paraformaldehyde in PBS, dehydrated, and embedded in paraplast. Immunostaining was performed using the avidin-biotin (ABC) immunoperoxidase method as described by Hsu et al. (1981). Seven-μm-thick sections were deparaffinized and rehydrated in a graded series of ethanol. Antigen retrieval was achieved with heating in 10 mM sodium citrate, pH 6.0. Sections were washed in PBS, and endogenous peroxidase was blocked in 3% (v/v) H2O2 in methanol. Blocking was done with 5% (w/v) normal goat serum (NGS) and 1% (w/v) BSA in PBS (for SIAH staining, NGS was substituted with donkey serum). Sections were then incubated overnight at 4C with antibodies. Primary antibodies were rabbit polyclonal anti-human Cullin2 antibody (1:200, RB-046-p1; Neomarkers, Fremont, CA), a rabbit polyclonal anti-human NEDD8 antibody (1:500, ALX-210-194-R200; Alexis Biochemicals, San Diego, CA), a rabbit polyclonal anti-human PHD1 antibody (1:400, AB100-310; Novus Biologicals, Littleton, CO), a rabbit polyclonal anti-human PHD2 antibody (1:400, AB4561; Abcam, Cambridge, MA), a rabbit polyclonal anti-human PHD3 antibody (1:200; AB4562, Abcam), a goat polyclonal anti-human SIAH1 antibody (1:50, AB2237; Abcam), a mouse monoclonal anti-human HIF-1α antibody (1:200; NB 100-105, Novus Biologicals), a rabbit polyclonal anti-human ARNT antibody (1:500, NB 100-110; Novus Biologicals), a rabbit polyclonal anti-mouse HIF-2α antibody (1:600, NB 100-122; Novus Biologicals), a mouse monoclonal anti-human VHL antibody (1:60, OP102; Oncogene, Mississauga, ON), a mouse monoclonal anti-mouse vascular endothelial growth factor receptor (VEGFR2) (KDR/Flk-1) antibody (1:400, MAB1667; Upstate USA, Charlottesville, VA), or a rabbit polyclonal anti-human VEGF antibody (1:200, SC152; Santa Cruz Biotechnology, Santa Cruz, CA). All were diluted in blocking solution (5% NGS and 1% BSA in PBS, whereas donkey serum was used for SIAH1). Sections were subsequently incubated with biotinylated secondary antibodies [1:300 rabbit anti-mouse for HIF-1α and VHL; 1:300 donkey anti-rabbit for Cullin2, ARNT, PHDs, VEGF, and VEGFR2, and 1:300 donkey anti-goat for SIAH1 (Santa Cruz Biotechnology)], and color detection was performed according to instructions in the Vectastain ABC and DAB kit (Vector Laboratories; Burlingame, CA). Sections were counterstained with Carazzi's hematoxylin and mounted in Permount (Fisher Scientific; Unionville, ON). We have previously confirmed the specificity of all antibodies using human placental tissues (Ietta et al. 2006).
RNA Isolation and Quantitative PCR
Human lungs were snap frozen in liquid nitrogen and stored at −70C. Total RNA was extracted using the TRIzol Reagent (Invitrogen; Burlington, ON), and total RNA was reverse transcribed (37C) using random hexamers (Applied Biosystems; Foster City, CA). Quantitative real-time PCR (qPCR) was used to validate differential gene expression. Primers and Taqman probes for VEGF, VEGFR1 (Flt-1), HIF-1α, HIF-2α, ARNT, PHD1, PHD2, PHD3, and SIAH1 were purchased from Applied Biosystems as Assays-on-Demand for human genes. For each probe, a dilution series determined the efficiency of amplification of each primer-probe set, allowing the relative quantification method to be employed (Livak and Schmittgen 2001). For the relative quantitation, PCR signals were compared among groups after normalization using 18S as an internal reference. Briefly, relative fold change was calculated as 2−(ΔCtexp-ΔCtcontrol), where ΔCt = average of (Ctgene of interest-Ct18S) of each sample within a group. Experimental groups (lungs of 10 and 13 weeks gestation) were compared with the control group (lungs of 16 weeks gestation).
Results
HIF and VEGF
IHC staining for HIF-1α, HIF-2α, and ARNT revealed that all three HIF subunits were expressed in the pseudoglandular human lung of 8- to 13-weeks gestation (Figure 1 shows 10 weeks). HIF-1α immunoreactivity was primarily detected in the branching epithelium of the first trimester lung (Figures 1A and 1A-I). Some mesenchymal cells stained positive for HIF-1α (arrowheads in Figure 1A-I). Although HIF-2α protein was found in the airway epithelium (Figure 1B), it was also detected in mesenchymal, most likely vascular, structures (arrowheads in Figure 1B-I). ARNT was predominantly found in the epithelium (Figure 1C), although some positive staining was noted in the lung parenchyma (arrow in Figure 1C-I). Because VEGF is a downstream target of HIF-1 (Maynard and Ohh 2005), we determined the spatial expression of VEGF in the first trimester human lungs and observed that VEGF was strongly expressed in the epithelium (Figures 2B, 2B-I, and 2B-II), in agreement with previous studies in humans (Acarregui et al. 1999) and mice (Greenberg et al. 2002). As reported for mice (Greenberg et al. 2002), positive VEGF staining was also noted in mesenchymal, most likely vascular, structures of the first trimester human lung (Figure 2B-I). Positive immunoreactivity for VEGFR2, an endothelial-specific tyrosine kinase receptor for VEGF (Carmeliet and Collen 1999; Shibuya 2006), was found in mesenchymal cells immediately adjacent to the epithelium as well as in vascular (arrowheads) structures of the mesenchyme (Figures 2C, 2C-I, and 2C-II). The VEGFR2 antibody was also reactive with the luminal airway epithelium. No immunoreactivity was observed in control sections in which primary HIF-1α monoclonal antibodies (Figure 3A) or HIF-2α, ARNT, VEGF, and VEGFR2 polyclonal antibodies (Figure 2A) were omitted.
VEC Complex
At 10 weeks gestation, strong positive immunoreactivity for VHL was noted in the lung epithelium of developing airways (Figures 3B-I and 3B-II). No mesenchymal staining was observed. VHL-associated ubiquitination activity requires Cullin2, which was primarily detected in the branching epithelium of the first trimester human lung (Figure 3C). However, some mesenchymal staining was apparent. Because ligation of NEDD8 (neural precursor cell expressed, developmentally downregulated) to Cullin2 may be important for VHL-mediated HIF-1α degradation (Kamitani et al. 1997; Ohh et al. 2002), we also analyzed the first trimester human lung for NEDD8 protein expression. NEDD8 exhibited a similar cellular distribution as Cullin2 (Figure 3D). Interestingly, both Cullin2 and NEDD8 were absent from mesenchymal cells immediately adjacent to the epithelium (see inserts in Figures 3C and 3D). The staining patterns for all three VEC proteins were similar for all age groups, i.e., 8 weeks, 10-12 weeks, and 13 weeks (not shown). No immunoreactivity was observed in control sections in which primary VHL (Figure 3A), Cullin2, and NEDD8 (Figure 2A) antibodies were omitted.
PHDs and SIAH1
Strong positive immunoreactivity for PHD1 and PHD3 was noted in the branching epithelium of the first trimester lung (Figures 4B and 4D). In contrast, PHD2 was not expressed in the airway epithelium (Figure 4C). Positive staining for PHD2 was detected in mesenchymal cells subjacent to the newly forming airways as well as in mesenchymal, most likely vascular, structures in the parenchyma. SIAH1 immunoreactivity was found only in the airway epithelium (Figure 4F). No differences between age groups were observed. Again, no immunoreactivity was observed in control sections in which primary PHDs (Figure 4A) and SIAH1 (Figure 4E) antibodies were omitted.
Quantitative RT-PCR
To quantify mRNA expression of HIF pathway-related molecules in the different age groups, we performed real-time PCR. PCR analysis showed that VEGF, VEGFR1 (Flt-1), HIF-1α, HIF-2α, ARNT, PHD1, PHD2, PHD3, and SIAH1 were all expressed during early pulmonary organogenesis (Figure 5). No changes in expression were noted between the early age groups (10 weeks vs. 13 weeks), corroborating the IHC findings. Most HIF-related genes decreased with advancing gestation (16 weeks).
Discussion
During the early phase of pregnancy, oxygen supply to the fetus changes dramatically. The intervillous space in the placenta opens to maternal blood after 10-12 weeks of gestation, and oxygen saturation of blood supplied to the fetus rises from 2-3% O2 to 8-10% O2 (Rodesch et al. 1992; Burton et al. 1999). It has been shown that placental degradation of HIF-1α takes place after 10 weeks of gestation at the time when placental oxygenation increases (Ietta et al. 2006). This also implies a changed oxygen concentration in the blood going to the fetus. Fetal blood oxygen rises and subsequently more oxygen is delivered to the organs. Angiogenesis and branching morphogenesis are very important during this phase of development in the fetal lung. Therefore, we investigated the spatial and temporal expression of HIF/VEGF pathway-related molecules in the lung. All molecules examined (HIF-1α, HIF-2α, ARNT, VHL, Cullin2, NEDD8, VEGF, VEGFR1-2, PHD1, PHD2, PHD3, and SIAH1) were expressed simultaneously, suggesting that HIF regulatory mechanisms are already in effect from week 8 of gestation.
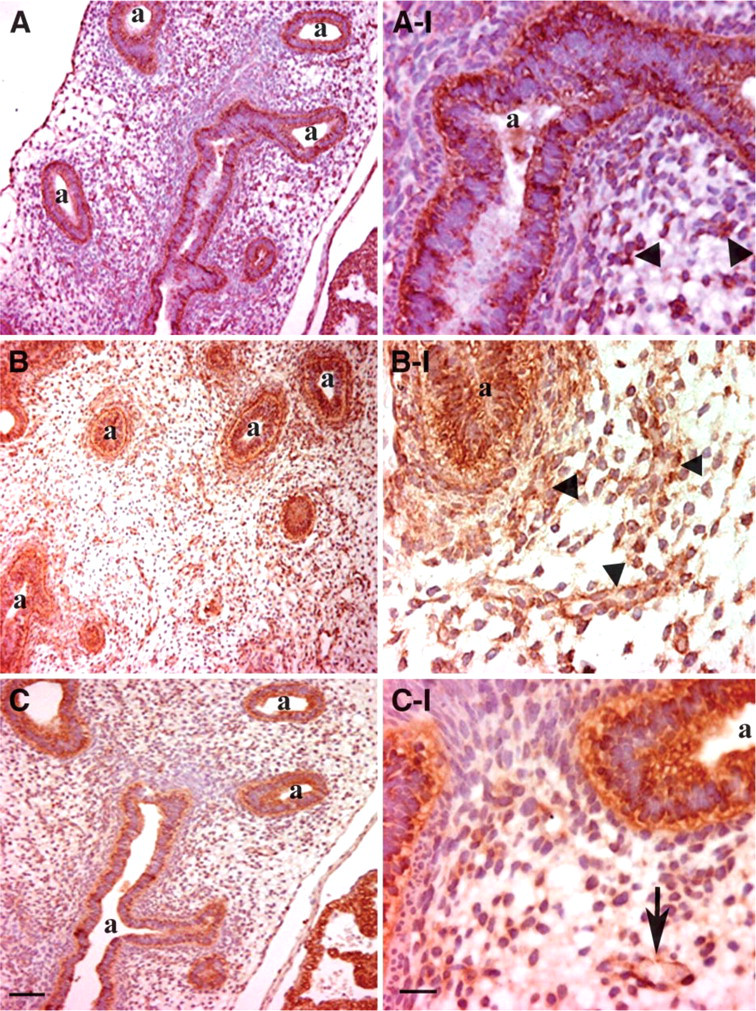
Immunohistochemical (IHC) analysis of HIF subunits in first trimester (10 weeks) human lung. Airway (a) epithelium shows positive brownish staining for HIF-1α (
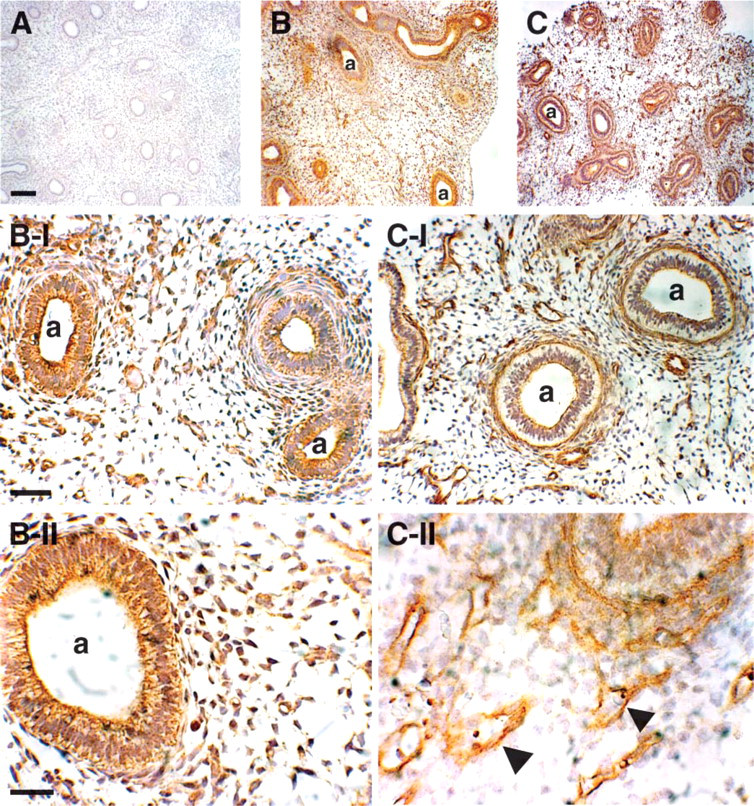
IHC analysis of vascular endothelial growth factor (VEGF) and VEGF-receptor 2 (VEGFR2) in first trimester (10 weeks) human lung. Strong positive brownish staining for VEGF (
Expression of HIF-1α did not decrease after week 10, and expression of molecules involved in degradation of HIF-1α did not increase. HIF-1α expression is mainly restricted to branching epithelium, whereas HIF-2α appeared also to be present in the vascular structures of the lung parenchyma. Their constitutive counterpart ARNT was present in both mesenchymal, most likely vascular, structures and epithelial structures, providing a basis for dimerization and further downstream activation of both HIF-1α and HIF-2α. The different spatial expression of HIF-1α and HIF-2α may reflect their different roles in pulmonary development. HIF-1α knockout mice suffer from severe cardiovascular defects and die in utero (Kotch et al. 1999; Compernolle et al. 2003), whereas HIF-2α knockout mice suffer from postnatal respiratory distress due to insufficient surfactant production (Compernolle et al. 2002). A heterozygous deficiency in HIF-2α protects mice from developing pulmonary hypertension (Brusselmans et al. 2003). It seems that HIF-1α is more important for organogenesis and the function of HIF-2α is the fine-tuning of pulmonary vascularization and remodeling. The fact that HIF-1α is found in the branching epithelium supports this as pulmonary branching morphogenesis depends on vascularization (van Tuyl et al. 2005).
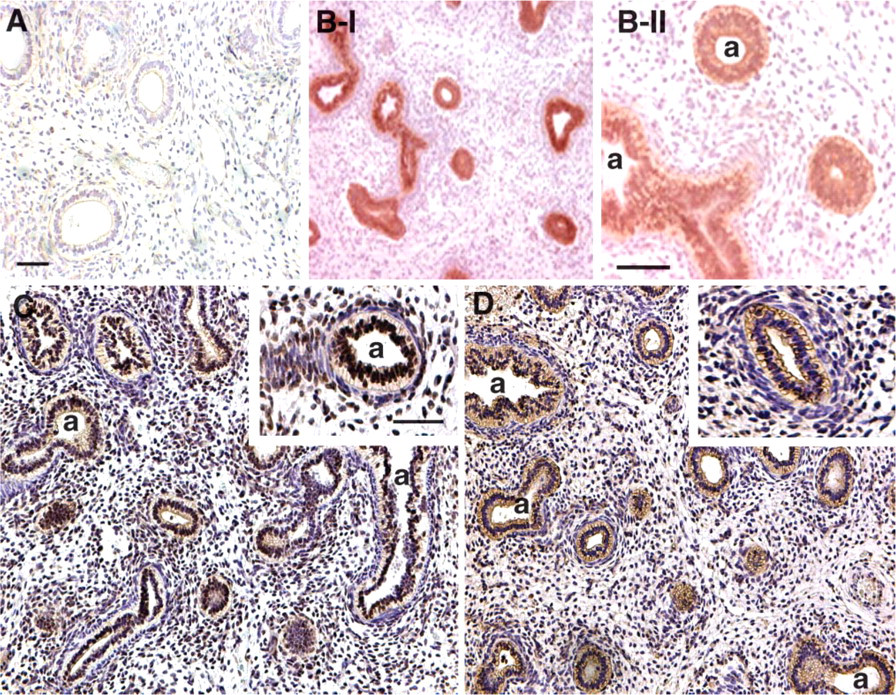
Immunolocalization of components of the VEC complex in first trimester (10 weeks) human lung. Positive brownish staining for von Hippel-Landau is evident in the airway (a) epithelium (
Components of the VEC complex (VHL, NEDD8, and Cullin2) are all present in the epithelium. VHL expression is restricted to epithelial structures and is not present in the mesenchyme. NEDD8 and Cullin2 were also detected in the branching epithelium but not in the immediate subepithelium. This means that all components for the assembly of the VEC complex are present in the epithelium, but not in the mesenchyme. The presence of this complex in the epithelium suggests a necessity for fine-tuning of HIF/VEGF expression and vascularization in this specific area. The absence of VEC complex in the mesenchyme allows HIF-2α to accumulate in the vascular cells in the mesenchyme. Our IHC data show that angiogenic factors such as VEGF are strongly expressed in the early pseudoglandular lung. There was no obvious decrease in VEGF expression after the rise in fetal oxygen saturation. It could be that relatively oxygen-rich blood is shunted away from the lung through the open foramen ovale and the ductus arteriosus, due to the high pulmonary vascular resistance. This mechanism ensures a low-oxygen environment in the developing lung, providing a good basis for angiogenesis.
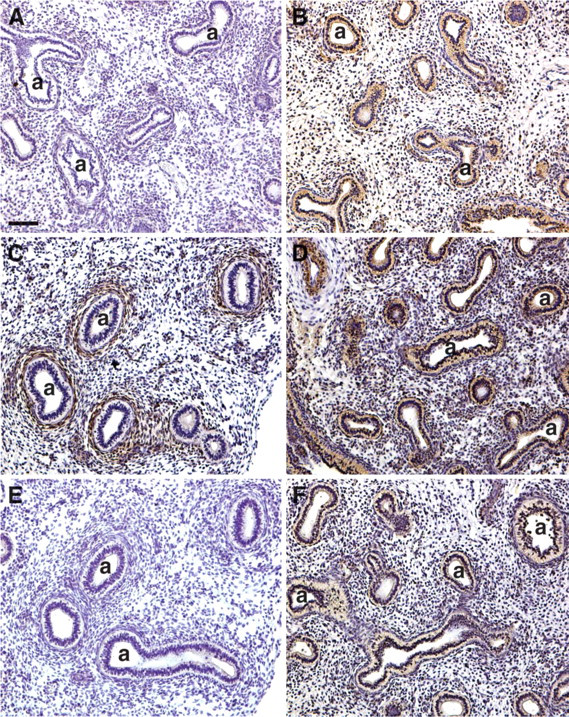
Immunolocalization of prolyl hydroxylases (PHDs) and seven in absentia homolog (SIAH) 1 in first trimester (10 weeks) human lung. Airway (a) epithelium stains positive for PHD1 (
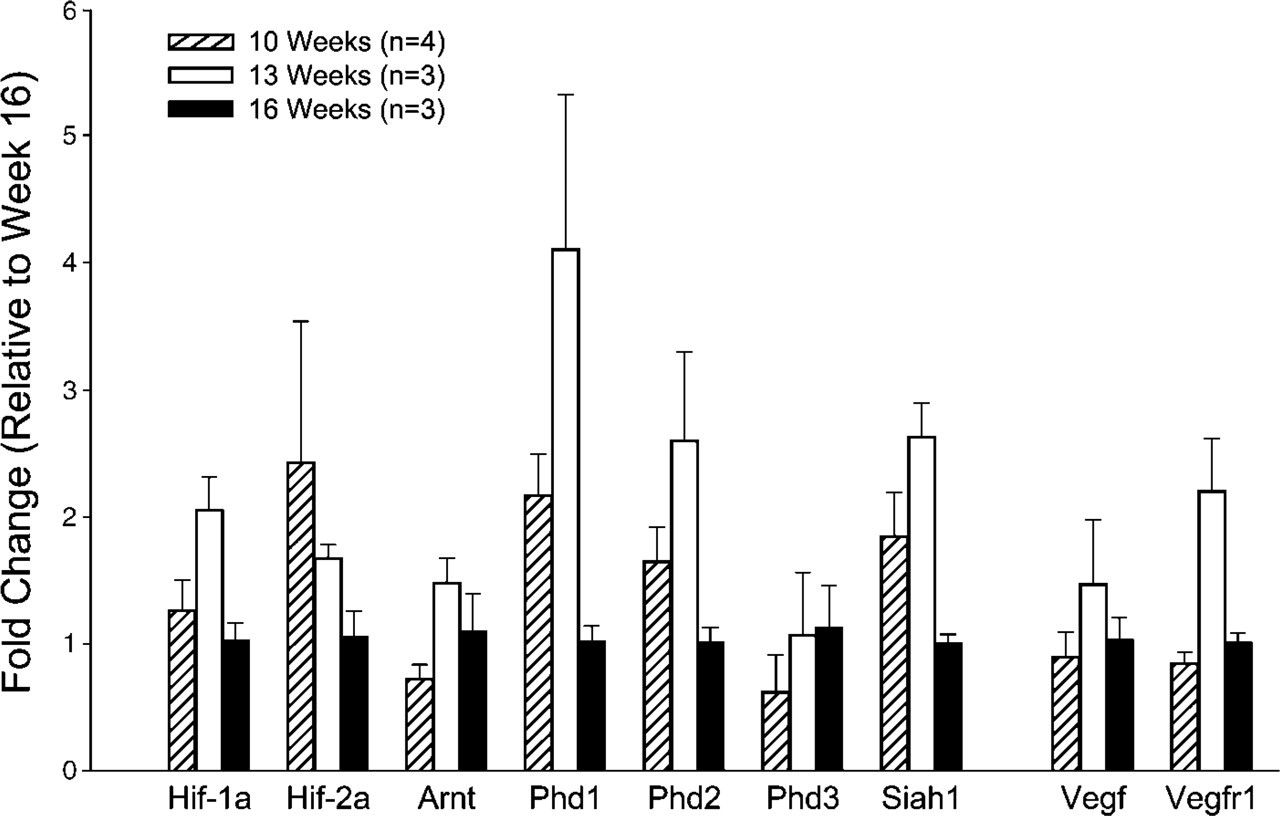
Gene expression of components of HIF/VEGF pathway in first trimester human lung. Expression of VEGF, VEGFR1, HIF-1α, HIF-2α, ARNT, PHD1, PHD2, PHD3, and SIAH1 mRNA in human lung as assessed by realtime PCR. Data are expressed as relative fold changes in expression when compared with human lungs of 16 weeks gestation (
Of the three PHDs, the main regulator of HIF-α hydroxylation (and thereby degradation) appears to be PHD2 (Berra et al. 2003; Appelhoff et al. 2004). In the developing human lung, PHD2 was differently expressed from PHD1 and 3; i.e., staining was only positive in the mesenchyme directly adjacent to developing airways, whereas the other two PHDs were expressed in the branching epithelium. This suggests that, in the areas with high HIF-1α expression, no PHD2 was present, making sure that HIF-1α is degraded at a slow rate. The mesenchymal expression of HIF-2α is in concordance with PHD2, suggesting that vascular development induced by HIF-2α is controlled by PHD2. PHD1 and PHD3 are under the control of their E3 ubiquitin ligase SIAH1 because their expression is restricted to airway epithelium. The fact that all these mechanisms are already present in the developing epithelium suggests that the development of the vascular bed surrounding the branching epithelium is a delicate and finely tuned process.
Continuous activation of HIF pathway even after opening of the intervillous space in the placenta suggests that the lung remains in a relatively low-oxygen environment during its development. This allows for vascular growth in the mesenchyme through downstream HIF effectors such as VEGF and VEGFRs.
Footnotes
Acknowledgements
This work was supported by the Canadian Institutes of Health Research (MOP-77751, MP, and MOP-14096, to IC) and the Sophia Foundation for Medical Research (SSWO #460, to FG). I.C. is a recipient of an Ontario Women's Health CIHR/IGH Mid-Career Award. M.P. is the holder of a Canadian Research Chair in Respiration.
