Abstract
Nestin is considered a marker of neurogenic and myogenic precursor cells. Its arrangement is regulated by cyclin-dependent kinase 5 (CDK5), which is expressed in murine podocytes. We investigated nestin expression in human adult and fetal kidney as well as CDK5 presence in adult human podocytes. Confocal microscopy demonstrated that adult glomeruli display nestin immunoreactivity in vimentin-expressing cells with the podocyte morphology and not in cells bearing the endothelial marker CD31. Glomerular nestin-positive cells were CDK5 immunoreactive as well. Western blotting of the intermediate filament-enriched cytoskeletal fraction and coimmunoprecipitation of nestin with anti-CDK5 antibodies confirmed these results. Nestin was also detected in developing glomeruli within immature podocytes and a few other cells. Confocal microscopy of experiments conducted with antibodies against nestin and endothelial markers demonstrated that endothelial cells belonging to capillaries invading the lower cleft of S-shaped bodies and the immature glomeruli were nestin immunoreactive. Similar experiments carried out with antibodies raised against nestin and α-smooth muscle actin showed that the first mesangial cells that populate the developing glomeruli expressed nestin. In conclusion, nestin is expressed in the human kidney from the first steps of glomerulogenesis within podocytes, mesangial, and endothelial cells. This expression, restricted to podocytes in mature glomeruli, appears associated with CDK5.
Keywords
T
Materials and Methods
Antibodies
Primary antibodies were purchased as follows: mouse anti-nestin monoclonal antibody (MAb5326) and rabbit anti-nestin polyclonal antibody (Ab5922; Chemicon, Temecula, CA); mouse anti-SMA MAb (clone 1A4; Neomarkers, Fremont, CA); mouse anti-CD31 (clone JC70A) and anti-CD34 MAbs (clone QBEnd-10) from DakoCytomation (Glostrup, Denmark); mouse anti-vimentin MAb (clone V9; Sigma, St Louis, MO); and rabbit anti-CDK5 PAb C-8 and rabbit anti-vimentin (H-84) (Santa Cruz Biotechnology; Santa Cruz, CA).
Secondary antibodies were obtained from the following sources: double-labeling-grade tetramethylrhodamine isothiocyanate (TRITC)-conjugated donkey anti-mouse IgG and TRITC-conjugated donkey anti-rabbit IgG from Chemicon; fluorescein isothiocyanate-conjugated (FITC) goat anti-mouse IgG antibody from Sigma; double-labeling-grade horseradish peroxidase (HRP)-conjugated goat anti-rabbit IgG from Zymed (South San Francisco, CA); and rabbit anti-mouse IgG from DakoCytomation.
Tissues
Cases and controls were taken consecutively and retrospectively from the surgical and autoptic files of the department of Human Pathology and Oncology. Adult kidney samples were obtained from surgical nephrectomy for carcinoma (n = 10, age range 24–78 years, median age 60 years). Kidneys were fixed in 4% buffered formalin for 24–48 hr, and normal tissue, far from the neoplasia, was sampled and paraffin embedded.
Fetal kidneys were obtained from autopsies for spontaneous abortion or induced termination of pregnancy (TP) (n=23; gestational age range, 16–37 weeks; median gestational age 22 weeks). Macerated and malformed stillbirths were not included in the study. The cause of spontaneous fetal death was placental in origin and included abruptio placentae, true cord knot, acute massive placenta infarcts, acute chorioamnionitis; and conditions associated with prolonged stress such as fetal growth restriction and other morphological signs of chronic suffering, as well as maternal preeclampsia and eclampsia, were excluded from this study. TP stemmed from either minor malformations unrelated to alterations in the urinary system or maternal (personal) reasons. A complete autopsy was carried out between the second and twelfth hour after fetal expulsion.
Selected samples for biochemical analysis were immediately frozen in liquid nitrogen and stored at −80C. Other fresh tissues (only from adult kidneys) were sampled for immunofluorescence histochemistry, cryoprotected with Killik Frozen Medium (Bio Optica; Milan, Italy), and promptly frozen in liquid nitrogen. Ten-μm-thick sections were cut at −25C, air-dried, fixed in acetone at −20C for 10 min, and stored at −80C.
Immunohistochemistry
Immunohistochemistry to detect nestin was performed as previously described (Toti et al. 2005). Three- to 5-μm sections were cut from each specimen, mounted on electrostatically charged slides, and dried. Sections were dewaxed, rehydrated, and washed in TBS. TBS was used for all subsequent washes and for antibody dilutions. Nestin antigenic sites were unmasked by heating sections in 0.01 M citrate buffer (pH 6.0) with a microwave oven (two 5-min cycles at 750 W) and were subsequently rinsed for 10 min in 3% hydrogen peroxide to block endogenous peroxidase. Slides were then incubated for 1 hr at room temperature with anti-nestin MAb (working dilution 1:200). The ABC system (Vector Laboratories; Burlingame, CA) was applied on sections of fetal samples, and the reaction was developed with 1 mg/ml 3,3′-diaminobenzidine tetrahydrochloride (Sigma) dissolved in TBS containing 0.3% H2O2. Sections of adult kidneys were incubated with a bridge antibody anti-mouse IgG for 30 min at room temperature, and the reactions were developed by the alkaline phosphatase-anti-alkaline phosphatase technique (Dakopatts; DakoCytomation) with new fuchsin. Harris hematoxylin was used for nuclear counterstaining. Negative controls were prepared by substituting the primary antibody with the corresponding preimmune serum. Cognate sections were stained with hematoxylin and eosin to check for renal pathologies or malformations.
Confocal Microscopy
A TCS 4D Leica (Wetzlar, Germany) confocal microscope was used to evaluate double-labeling experiments carried out on frozen sections of adult kidney and on formalin-fixed, paraffin-embedded sections of fetal kidney.
Frozen sections were incubated 2 hr at room temperature with anti-vimentin MAb (working dilution 1:50) or anti-CD31 MAb (working dilution 1:100) followed by FITC-conjugated anti-mouse IgG for 1 hr at room temperature; the second primary antibody, anti-nestin PAb (working dilution 1:200), was applied overnight at 4C followed for 1 hr at room temperature by the appropriate TRITC-conjugated secondary antibody (working dilution 1:50). Other experiments were carried out incubating sections with anti-nestin MAb (working dilution 1:100) or anti-CD31 MAb (working dilution 1:100) for 2 hr at room temperature, followed for 1 hr by TRITC-conjugated anti-mouse IgG (working dilution 1:50). Rabbit anti-CDK5 was then applied overnight at 4C, followed by HRP-conjugated anti-rabbit IgG for 1 hr at room temperature. Application of fluorescein-labeled tyramide amplification reagent (Perkin-Elmer; Boston, MA) catalyzed a HRP-driven reaction that resulted in the deposition of a fluorescent precipitate.
Sections of embryonic kidney were dewaxed, rehydrated, and processed as follows: they were incubated with rabbit anti-nestin PAb for 1 hr at room temperature followed by the ABC system according to manufacturer's instructions (Vector Laboratories); the second primary antibody, anti-CD34 MAb (working dilution 1:100) or anti-SMA MAb (working dilution 1:200), was applied overnight at 4C followed for 1 hr at room temperature by TRITC-conjugated anti-mouse IgG (working dilution 1:50). Final application of fluorescein-labeled tyramide amplification reagent (Perkin-Elmer) catalyzed a HRP-driven reaction that resulted in the deposition of a fluorescent precipitate on nestin antigenic sites.
SDS/PAGE Electrophoresis, Coimmunoprecipitation, and Western Blotting
To confirm our immunohistochemical results, Western blotting analysis with anti-nestin antibody was carried out on proteins of the intermediate filament-enriched cytoskeletal fraction (IFCF) of adult and fetal kidney, separated by SDS-PAGE. IFCFs were prepared as previously reported (Achtstaetter et al. 1986; Carapelli et al. 2004; Toti et al. 2005) and stored at −80C until use. Just before use, aliquots of IFCF were thawed and mixed with Laemmli sample buffer.
Immunoprecipitation experiments were carried out on total homogenates of adult renal cortex. Pieces of renal cortex were placed in homogenization buffer (50 mM Tris/HCl, 150 mM NaCl, 1 mM EDTA, 0.25% sodium deoxycholate, 1% NP-40, 1 mM PMSF, 2 μg/ml aprotinin, pH 7.4) and homogenized with a Dounce homogenizer. The suspension was centrifuged at 5000 × g for 10 min at 4C, and the protein content was measured with the microBCA protein assay reagent kit (Pierce; Rockford, IL). Samples were stored at −80C until use. Five hundred μl of homogenate was precleared with protein A-Sepharose (Sigma), centrifuged, and the pellet of Sepharose beads was placed in Laemmli sample buffer. Two μl of anti-CDK5 IgG or 3 μl of anti-vimentin PAb was added to the supernatant and incubated overnight at 4C, followed by 2 hr with the protein A-Sepharose. Immunocomplexes were washed three times with buffer, and the final pellet was resuspended in Laemmli sample buffer.
IFCFs, immunoprecipitates, and proteins released from the Protein A-Sepharose complexes used for the preclearings were separated by electrophoresis through a 6% polyacrylamide gel and transferred to nitrocellulose in a Bio-Rad Transblot apparatus (BioRad; Segrate, Italy). A 5% (w/v) solution of skim milk in TBS was used to quench nonspecific protein binding. Membranes were incubated overnight at room temperature with 1:2000 anti-nestin MAb in 5% (w/v) solution of skim milk in TBS. Specific bands were detected using an electro-chemiluminescense kit (Roche Diagnostics; Milan, Italy).
Results
Adult Kidney
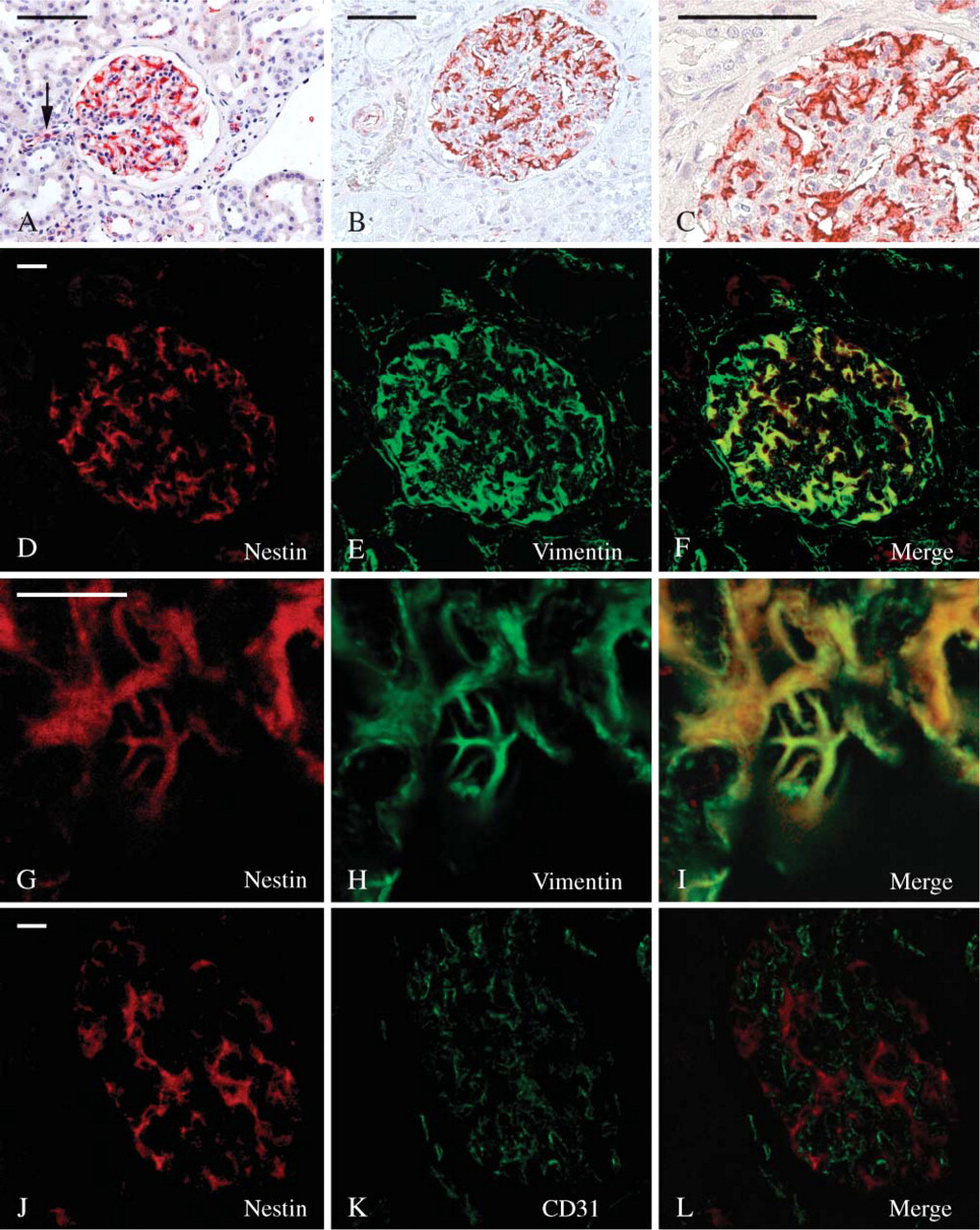
In adult kidney, nestin staining was restricted to glomeruli, smooth muscle cells of the arterioles, and some endothelial cells (Figures 1A-1C). The highest intensity of staining was referred to podocytes where nestin immunoreactivity was consistent (Figures 1A-1C) and to the tunica media of some arterioles. Even though with a variable degree of intensity, endothelial cells of arterioles and of some blood capillaries were nestin labeled as well (Figure 1A).
Glomerular nestin-expressing cells resembled podocytes as to their location and morphology. However, to confirm immunohistochemical data obtained on formalin-fixed, paraffin-embedded material, a series of double-immunofluorescence experiments were performed on frozen sections and analyzed with the confocal microscope. A well-known marker of podocytes, vimentin, was used along with nestin in double-labeling experiments. In the glomerulus, vimentin is expressed at high levels only within podocytes, mesangial, and endothelial cells being reported as showing possibly only a much weaker labeling (Bachmann et al. 1983; Holthöfer et al. 1984; Stamenkovic et al. 1986). Other podocyte markers like podocin, synaptopodin, nephrin, ZO-1, and podocalixyn could not be used for our purpose in vivo because they were all concentrated prevalently in the foot processes where intermediate filaments are not present, and their staining hardly overlapped with nestin. These experiences showed that all nestin-immunoreactive cells were, indeed, highly vimentin-expressing cells as well (Figures 1D-1F). Moreover, nestin/vimentin double-labeled cells clearly resembled podocytes, displaying long immunoreactive primary processes (Figures 1G-1I). Double-labeling experiments carried out using nestin and the endothelial marker CD31 (Naruse et al. 2000) showed that the relationships occurring between nestin-positive cells and endothelial cells were those expected between podocytes and the glomerular capillaries (Figures 1J-1L).
Because it has been reported that CDK5 is expressed in murine podocytes (Griffin et al. 2004), and that it plays a key role in the regulation of nestin organization (Sahlgren et al. 2003), we performed CDK5/nestin double-labeling experiments that showed a certain degree of colocalization of the two proteins (Figures 2A-2F). Moreover, because CDK5 has been previously reported in endothelial cells (Sharma et al. 2004), additional experiments have been carried out to rule out the possibility that CDK5 staining had to be referred in part to the glomerular endothelium. Indeed, confocal microscopy of CDK5/CD31 double-labeled sections showed two different staining patterns with no detectable colocalizations of the two antigens in the renal corpuscles (Figures 2G-2I). Remarkably, CDK5 fluorescence appeared frequently arranged parallel to the glomerular endothelium (Figure 2I).
Nestin expression in adult kidney. (
To confirm that the antibodies used in this study recognized actual nestin, we carried out Western blot analysis with anti-nestin MAb on renal IFCF of adult individuals from total homogenates of adult kidneys. Depending on samples, one or two nestin-immuno-reactive bands could be unveiled in the IFCF migrating at 240 and 280 kDa (Figure 3A), the lower band being constant. Moreover, to confirm that CDK5/nestin and nestin/vimentin colocalizations were due to their actual association, additional Western blotting experiments with anti-nestin MAb were carried out on CDK5 and vimentin immunoprecipitates. The presence of a double-nestin-immunoreactive band of the appropriate molecular mass (240 and 280 kDa) in the immunoprecipitates demonstrated that nestin coimmunoprecipitated with vimentin and CDK5, confirming on the one hand that nestin and vimentin coassemble together even in podocytes and suggesting on the other hand that the association of nestin and CDK5 observed in C2C12 myoblasts and ST15A neuronal precursors (Sahlgren et al. 2003) occurs in human podocytes as well (Figure 3B).
Fetal Kidney
Development of human metanephric kidney begins at gestational week 4–5 and is said to terminate after the 35th week of gestation (Nagata et al. 1993). In this period a nephrogenic zone located under the renal capsule continues to add new developing glomeruli, the older being located deeper in the kidney and the newer just under the capsule. For this reason, in a single section of fetal kidney it is possible to appreciate all the stages of glomerular development in a sort of gradient of maturation going from the surface to the depth of the organ. The steps of glomerular maturation are morphologically divided into the following stages: vesicle (V), S-shaped body (S), capillary loop (C), and maturing glomerulus (M) (Naruse et al. 2000; Pavenstädt et al. 2003). At low magnification, nestin staining on fetal kidney sections clearly appeared located in glomeruli at all the maturation stages (Figure 4A). Although less constantly, a certain degree of staining was apparent even at the V stage (Figure 4B). Then, at the S stage, nestin labeling became consistent and intense within the cell layer that eventually would differentiate into podocytes (Figure 4C). At the same stage, nestin was also clearly located within cells that populated the cleft of the S-shaped body, from which both mesangial and endothelial glomerular cells are known to differentiate. At the C stage, podocytes were the only glomerular cell elements incontrovertibly nestin immunoreactive (Figure 4D). However, a certain degree of nestin staining in presumptive mesangial cells was also seen at this stage. At the M stage, some cells of the parietal layer of the Bowman's capsule were nestin immunoreactive (Figure 4E), whereas all podocytes were morphologically well developed and maintained their intense nestin staining (Figures 4E and 4F). Outside the glomeruli, nestin was expressed by capillary endothelial cells (Figures 4B and 4D) and smooth muscle cells of the arterial wall (data not shown). To corroborate nestin expression in developing kidneys, the IFCFs of fetal kidneys were immunoblotted with a MAb against nestin. A double-immunoreactive band comigrating with nestin from adult kidneys was observed in several preparations (Figure 3C). When bands of the proper molecular mass were not detectable, they were always substituted by immunoreactive bands of lower molecular mass (data not shown), likely representing degradation products as previously reported (Sahlgren et al. 2001).
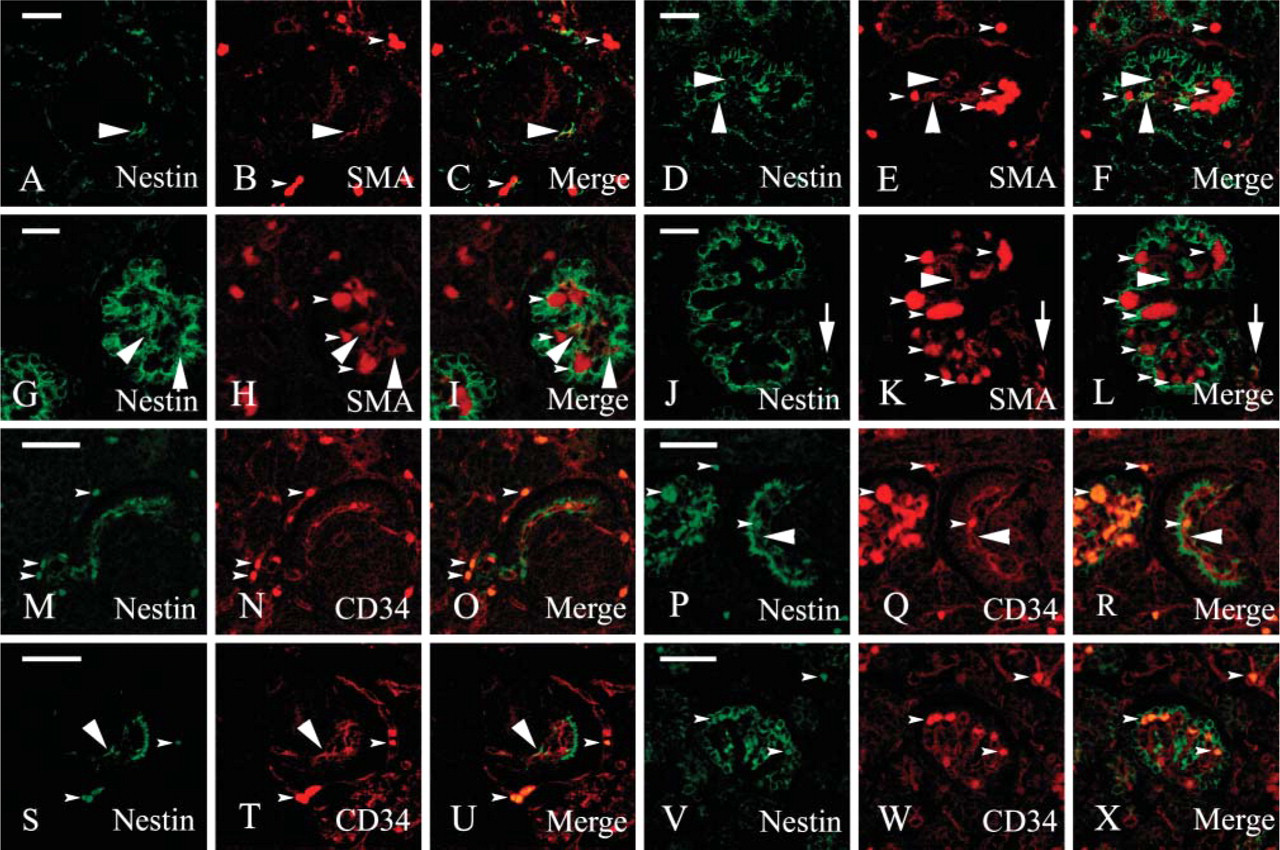
Confocal microscopy analysis of double-labeling experiments was carried out to identify nestin+ cells in the early stages of nephrogenesis. As a mesangial marker, we chose α-smooth muscle actin (α-SMA), which has been reported to be expressed in activated and fetal mesangial cells (Alpers et al. 1992; Naruse et al. 2000), whereas we used CD34 as a marker for the endothelium (Fina et al. 1990; Ito et al. 1995; Naruse et al. 2000). α-SMA was constantly expressed in the S (Figure 5B), C (Figure 5E), and M (Figures 5H and 5K) stages. Nestin/SMA double-labeled cells were visible at the S (Figures 5A-5C) and C (Figures 5D-5F) stages and within the smaller glomeruli (Figures 5G-5I) at the M stage. Mid-sized glomeruli did not show any overlapping between SMA and nestin stainings, whereas smooth muscle cells of the afferent and efferent arterioles expressed both antigens (Figures 5J-5L).
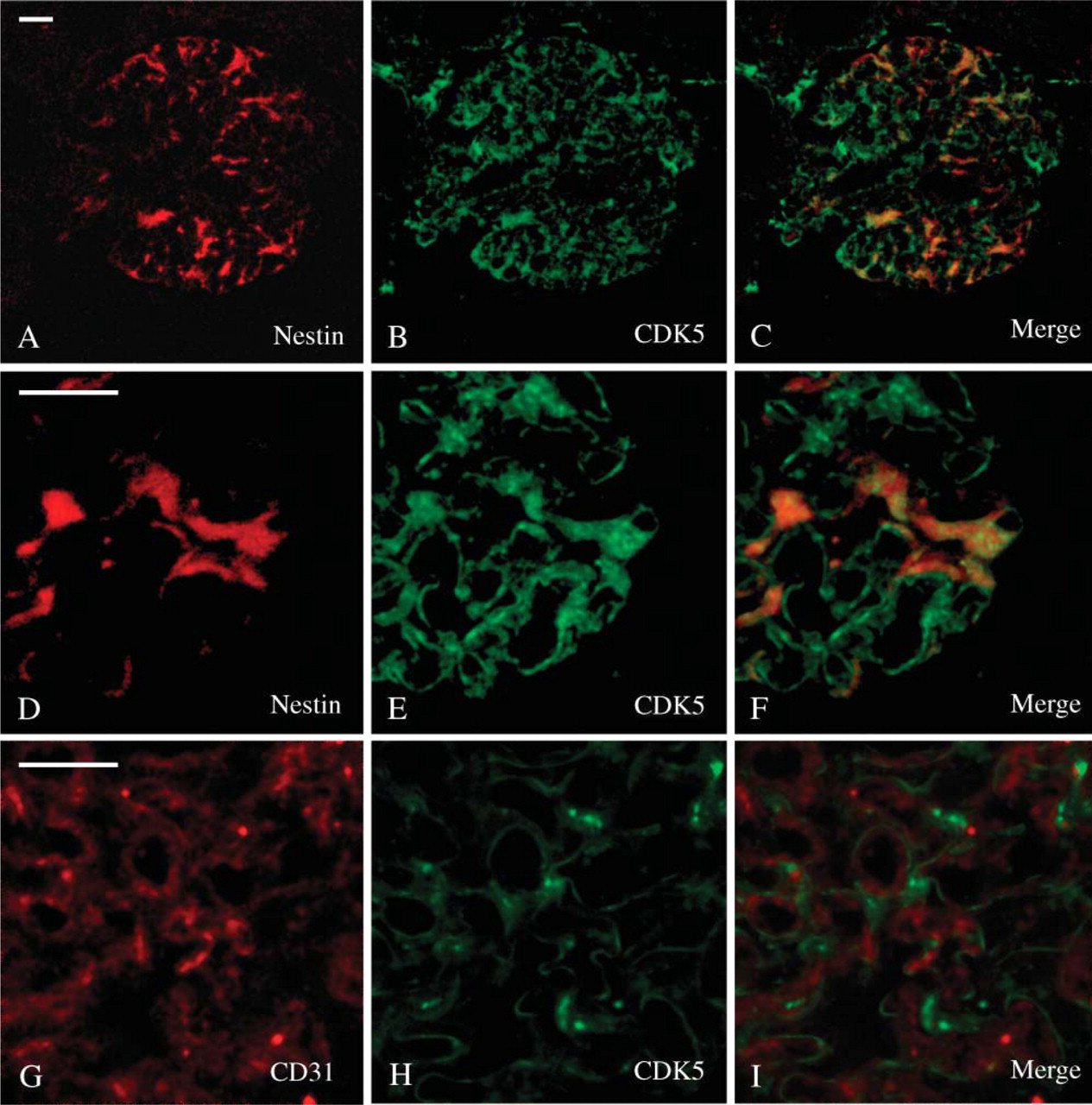
Nestin/CDK5 colocalization. Confocal microscopy of sections from adult human kidney. (
CD34 appeared early in the developing glomeruli at the S stage. It is believed that as soon as the lower cleft of the S-shaped body is formed, blood capillaries invade it or differentiate in loco from mesenchymal precursors (Naruse et al. 2000). In our preparations, CD34-positive blood capillaries were already visible at this stage (Figure 5N). Glomeruli at later stages of maturation displayed an increasing complex tuft of capillary loops (Figures 5Q and 5T). Double-labeled CD34/nestin cells were detected in the S-shaped bodies where all, or almost all, endothelial cells appeared nestin immunoreactive (Figures 5M-5O). At the C stage, only a few CD34-positive endothelial cells coexpressed nestin (Figures 5P-5U), and the few nestin-expressing cells that could not be referred to the layer of podocytes and did stain with the anti-CD34 antibody were most likely mesangial cells. Finally, at the M stage no double-labeled cells were detectable (Figures 5V-5X).
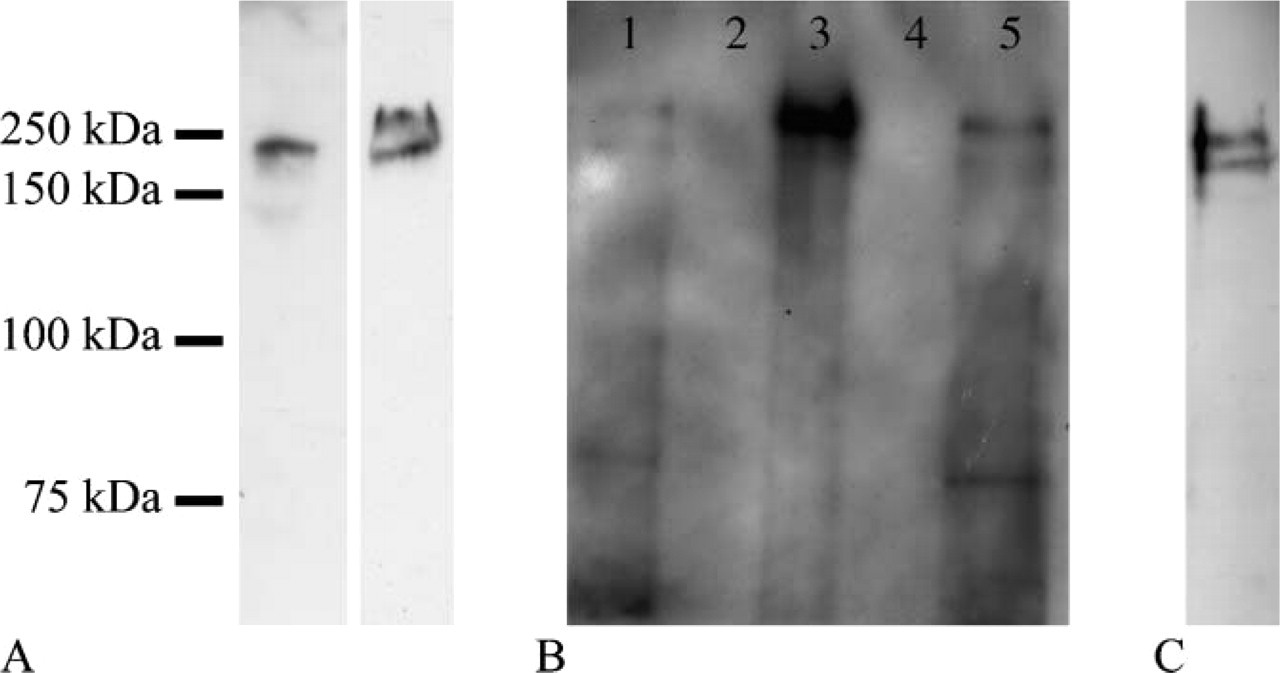
Western blottings with anti-nestin antibody. (
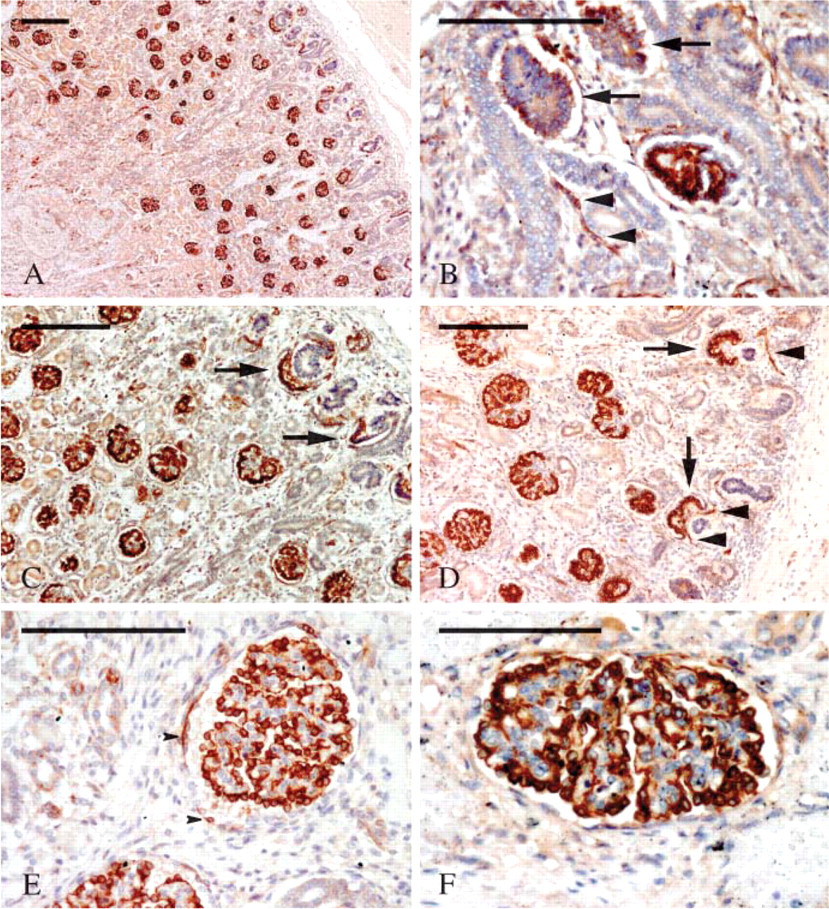
Nestin expression in developing kidney. (
Nestin expression in glomerular cells at various stages of development. (
Discussion
Nestin, a high molecular mass intermediate filament (IF), was first described as expressed in central nervous system stem cells (Lendahl et al. 1990). Since then, several other tissues and cells, including skeletal and cardiac myogenic cells (Sejersen and Lendahl 1993; Kachinsky et al. 1995), Leydig cells (Lobo et al. 2004), adrenocortical cells (Bertelli et al. 2002; Toti et al. 2005), adult and embryonic pancreas (Lardon et al. 2002; Klein et al. 2003; Delacour et al. 2004; Street et al. 2004), and many other cell types (for review, see Wiese et al. 2004) have been reported to contain nestin. On the whole, however, stable expression of nestin in cells from adult individuals is considered a rare phenomenon and a marker of stem or progenitor cells (Lendahl et al. 1990; Wiese et al. 2004). The present investigation, demonstrating that nestin is constitutively expressed in adult human podocytes, should change this view. Moreover, as nestin has been recently shown in mouse and rat podocytes as well (Chen et al. 2006; Wagner et al 2006; Zou et al. 2006), its presence in human kidney confirms that it should play a fundamental role in the biology of the renal corpuscle. Because of its frequent species-specific variability, it is important to evaluate the pattern of expression of IF proteins in human samples. Even considering the same animal species, cells can change IF content once they are grown in culture (Franke et al. 1979; Virtanen et al. 1981). To mention a few examples, we recall membranous-epithelial (M-) cells that, in addition to cytokeratins, display vimentin in rabbits but not in other species (Jepson and Clark 1998; Carapelli et al. 2004) and pancreatic duct cells that express cytokeratin 20 in rats and pigs (Bouwens 1998; Bertelli et al. 2001; Bertelli and Bendayan 2005) but not in humans and mice (Bouwens 1998). However, many other cases could be listed and even podocytes differ in IF content from one animal species to another (Yaoita et al. 1999; Zou et al. 2006) showing, accordingly, that the molecular arrangement of the IF network can be quite diverse, depending on the animals. Nestin is expressed in undifferentiated and dividing cells in the central nervous system and in myogenic tissues (Lendahl et al. 1990; Sejersen and Lendahl 1993). Upon differentiation, nestin is downregulated and replaced by other IFs. Nevertheless, it is re-expressed upon injury and in regenerating phenomena (Frisen et al. 1995; Vaittinen et al. 2001). These features have led some investigators to propose nestin as a marker of multilineage progenitor cells (Wiese et al. 2004). Nestin expression has also been correlated to increased motility and tumor invasiveness (Dahlstrand et al. 1992; Thomas et al. 2004). It is evident that podocytes cannot be placed into such a framework. Podocytes are non-motile, postmitotic terminally differentiated cells that re-enter the cell cycle only in a limited set of diseases, namely, HIV glomerulopathy, collapsing glomerulopathy, and the cellular variant of focal segmental glomerulosclerosis (Pavenstädt et al. 2003).
Nestin has been functionally linked to CDK5. CDK5 seems to play a fundamental role in triggering the exit of neurons from the cell cycle and in inducing differentiation (Cicero and Herrup 2005). In addition, CDK5 is important in regulating the differentiation of muscle cells and the rearrangement of the nestin network (Lazaro et al. 1997; Sahlgren et al. 2003). To carry out its kinase activity, CDK5 requires an activator, p35, which has been reported to associate with nestin as well (Sahlgren et al. 2003). When activated, CDK5 phosphorylates nestin and, in this way, probably regulates nestin disassembling because phosphorylated nestin is detected exclusively in the Triton X-100 soluble fraction (Sahlgren et al. 2003). Our results demonstrate for the first time that in vivo nestin and CDK5 are co-expressed and associated in adult human podocytes. Moreover, our data show that, whereas CDK5 is diffuse throughout the entire cytoplasm, pedicels included, the nestin network is located in the cell body and in the primary processes, but not in the foot processes. This is in accordance with the notion that the IF network does not extend into foot processes (Drenckhahn and Franke 1988; Pavenstädt et al. 2003). CDK5 in podocytes likely plays multiple roles because it is probably responsible for the differentiation and exit from the cell cycle (Griffin et al. 2004). However, an additional and pivotal role could be the regulation of IF network extension, preventing it from invading foot processes. In support of this view, nestin disassembling has been reported as regulated by the active complex CDK5/p35 (Sahlgren et al. 2003), which has been spotted mainly gathered at the podocyte cell processes (Griffin et al. 2004).
Nestin, however, is not capable of forming IFs on its own, as it must copolymerize with other type III IF proteins (Sjoberg et al. 1994; Steinert et al. 1999). In accordance with the well-known expression of vimentin in podocytes (Holthöfer et al 1984; Stamenkovic et al. 1986; Moll et al. 1991), our immunocytochemical and biochemical results confirm that the nestin partner in human podocytes is vimentin. The persistence of nestin expression in adult postmitotic terminally differentiated cells, in contrast to the more general rule that would restrict nestin to undifferentiated and dividing cells, implies an involvement of nestin in peculiar roles specific for podocytes. The possibility of a structural strengthening of the podocyte IF network is not convincing because nestin, far from reinforcing vimentin IFs, has been shown to make them less stable (Steinert et al. 1999). Nestin/vimentin heterodimers, however, have been proposed to be located at the periphery of a core of vimentin homodimers with the long nestin carboxyl terminal that sticks out of the filament. The importance of this arrangement would reside in the ability of the carboxyl domain to interact with microfilaments and microtubules cross-linking the three distinct components of the cytoskeleton (Herrmann and Aebi 2000). These interconnections could result, therefore, in a net advantage for the mechanical stability of podocytes. Another possibility is that nestin may serve a regulatory function for the assembly state of vimentin because it has been demonstrated in mitotic cells where vimentin IFs, in spite of their phosphorylated state, disassemble only when nestin is coexpressed (Chou et al. 2003). In this respect, based also on our results, a functional chain linking CDK5, nestin, and vimentin could be envisioned in podocytes: active CDK5/p35 complex within foot processes would phosphorylate nestin which, in turn, could regulate vimentin disassembly, preventing the IF network from extending into the pedicels. This view is supported by the observation that, in puromomycin-induced nephrosis, nestin and vimentin are both overexpressed (Zou et al. 2006) and that foot process effacement is characterized by the presence of IFs within the cytoplasmic sheet that, replacing pedicels, covers the basement membrane (Kubosawa and Kondo 1994). At any rate, the proper architecture of the IF network seems essential to maintain foot process integrity. This is confirmed by experiments of nestin silencing with siRNA where the effect of nestin knock-down resulted in a marked decrease of podocyte processes (Chen et al. 2006).
The present investigation shows that nestin IF is expressed in human podocytes starting from the very beginning of their differentiation. The pattern of nestin expression during human glomerulogenesis shares some resemblances with that which has been observed in rat and mouse (Chen et al. 2006; Wagner et al. 2006; Zou et al. 2006) but also shows significant differences. In contrast to mouse developing kidney where nestin has been detected within podocytes only in mature glomeruli (Chen et al. 2006), in humans nestin appears to be expressed from the first stage of glomerulogenesis (V stage). Throughout the following stages of development, nestin staining becomes more intense, localizing preferentially in the epithelial cells of the visceral layer. This behavior seems closer to what has been reported in rat developing kidney (Zou et al. 2006). On the other hand, whereas nestin expression in human differentiating glomerular endothelial cells is in accordance with mouse glomerulogenesis (Chen et al. 2006), our finding of a further expression in mesangial precursors seems to be unique to human immature glomeruli.
In Western blotting, nestin is usually reported as migrating as a double-immunoreactive band at 220 and 240 kDa (Sultana et al. 1998; Sahlgren et al. 2001; Lobo et al. 2004; Wiese et al. 2004). However, our evaluation gives considerably higher molecular masses, 240 and 280 kDa. Our values, in accordance with other studies (Messam et al. 2000; Chou et al. 2003; Toti et al. 2005; Zou et al. 2006), point toward nestin heavy posttranslational modifications as possibly serving particular functions in podocytes and in a few other particular cell types. Otherwise, considering that the molecular mass predicted by the amino acid sequence should be only 177 kDa and that posttranslational modifications should add 100 kDa to the molecule, the existence of alternative splicings of nestin gene is a possibility that should also be considered. However, more studies are required to clarify this point.
In conclusion, the present investigation has shown, for the first time, that human podocytes express nestin and that this is associated in vivo with CDK5, which is also located within the foot processes where nestin is not detectable. Moreover, we have demonstrated that, during glomerulogenesis, nestin is expressed in differentiating and mature podocytes, in some endothelial cells and, for the first time, in mesangial precursors.
Footnotes
Acknowledgements
This work was supported by PAR2004 (quota servizi) (to EB) and PRIN2004 (to PT).
