Abstract
Self-renewing, multipotent neural progenitor cells (NPCs) reside in the adult mammalian spinal cord ependymal region. The current study characterized, in vitro, the native differentiation potential of spinal cord NPCs isolated from adult enhanced green fluorescence protein rats. Neurospheres were differentiated, immunocytochemistry (ICC) was performed, and the positive cells were counted as a percentage of Hoescht+ nuclei in 10 random fields. Oligodendrocytes constituted most of the NPC progeny (58.0% of differentiated cells; 23.4% in undifferentiated spheres). ICC and electron microscopy (EM) showed intense myelin production by neurospheres and progeny. The number of differentiated astrocytes was 18.0%, but only 2.8% in undifferentiated spheres. The number of differentiated neurons was 7.4%, but only 0.85% in undifferentiated spheres. The number of differentiated radial glia (RG) was 73.0% and in undifferentiated spheres 80.9%. EM showed an in vitro phagocytic capability of NPCs. The number of undifferentiated NPCs was 32.8% under differentiation conditions and 78.9% in undifferentiated spheres. Compared with ependymal region spheres, the spheres derived from the peripheral white matter of the spinal cord produced glial-restricted precursors. These findings indicate that adult rat spinal cord ependymal NPCs differentiate preferentially into oligodendrocytes and RG, which may support axonal regeneration in future trials of transplant therapy for spinal cord injury.
Keywords
P
NPC transplantation for spinal cord injury (SCI) is under intense investigation. However, the ideal source of NPCs for repair of the damaged spinal cord is an issue of ongoing debate, with some arguing that all stem cells are alike and that all can be triggered to differentiate into the appropriate phenotype in response to cues from the local microenvironment. On the other hand, the impetus to transplant region-specific stem cells is based on studies suggesting that the local microenvironment, or niche, that surrounds a stem cell dictates its behavior and fate. Because there is evidence to support both sides of the argument, discriminating between the innate capability of neural stem cells to determine their own phenotypic fate and the environment-specified differentiation of these cells is crucial (Goh et al. 2003; Wagers and Weissman 2004; Kulbatski et al. 2005). In vitro, it has been shown that NPCs isolated from the embryonic rat brain and spinal cord have distinct region-specific growth properties and trophic requirements that may influence the development of cell therapies for region-specific neurological disorders (Fu et al. 2005). Adult NPCs are most similar to the cell types they replace, they are rarely oncogenic, and there are fewer ethical road-blocks to their procurement. Environmental signals governing NPC survival and differentiation may be region specific within the CNS, influencing local NPCs to differentiate into specific phenotypes. Cerebral (Parmar et al. 2003) and retinal (Yang et al. 2002) transplantation studies suggest that regional cells are lineage restricted and may respond better to region-specific cues (Gotz 2003). Transplanted adult NPCs differentiate into neurons in the hippocampus but into astrocytes in the spinal cord, and hippocampal astrocytes instruct these NPCs to differentiate into neurons in vitro (Shihabuddin et al. 2000).
The aim of the current study was to morphologically characterize adult rat spinal cord-derived NPCs, based on immunocytochemical (ICC) and ultrastructural analyses of the cells in culture. We showed that spinal cord periventricular region NPCs from adult enhanced green fluorescent protein (EGFP) rats preferentially differentiate into oligodendrocytes and radial glia (RG) vs the typical astrocytic differentiation of brain-derived NPCs (Reynolds et al. 1992; Gritti et al. 1996; Lobo et al. 2003). We also found that only periventricular and not parenchymal spinal cord NPCs are multipotent, that the neurospheres and differentiated progeny produce myelin, and that NPCs within the neurosphere maintain their microenvironment by phagocytosing debris from degenerating cells and myelin. These characteristics are region specific and unique to spinal cord NPCs and may have important implications for future therapeutic strategies.
Materials and Methods
Harvesting and Culturing
All animal procedures were performed in accordance with the Guide to the Care and Use of Experimental Animals (Canadian Council on Animal Care) and approved protocols from the Animal Care Committee of the Research Institute of the University Health Network, Toronto, ON, Canada.
NPCs were harvested from adult male Wistar EGFP rats [Wistar—TgN(CAG-GFP)184Ys; The YS Institute Inc., Utsunomiya, Tochigi, Japan]. NPCs cultured from these rats have been shown to stably express the EGFP transgene long term both in vitro and in vivo (Mothe et al. 2005). As such, we have opted to use these EGFP transgenic animals for the current study because our goal is to characterize these NPCs for transplantation, under which conditions the cells can easily be tracked and distinguished from host tissue. Rats (2- to 7-months old) were sacrificed, and the cervical spinal cord was excised and washed in 4C Dulbecco's PBS (DPBS) supplemented with 30% glucose (Sigma—Aldrich; Oakville, ON, Canada). For harvesting of periventricular (ependymal region spinal cord) NPCs, the overlying meninges and blood vessels were removed, and the white and most of the gray matter surrounding the central canal were removed. The tissue immediately surrounding the central canal, including the closely apposed ependymal and subependymal regions, was harvested under sterile conditions. For harvesting of parenchymal (parenchymal spinal cord) NPCs, the overlying meninges and blood vessels were removed, and portions of the white matter were harvested under sterile conditions.
Summary of antibodies used for immunocytochemistry
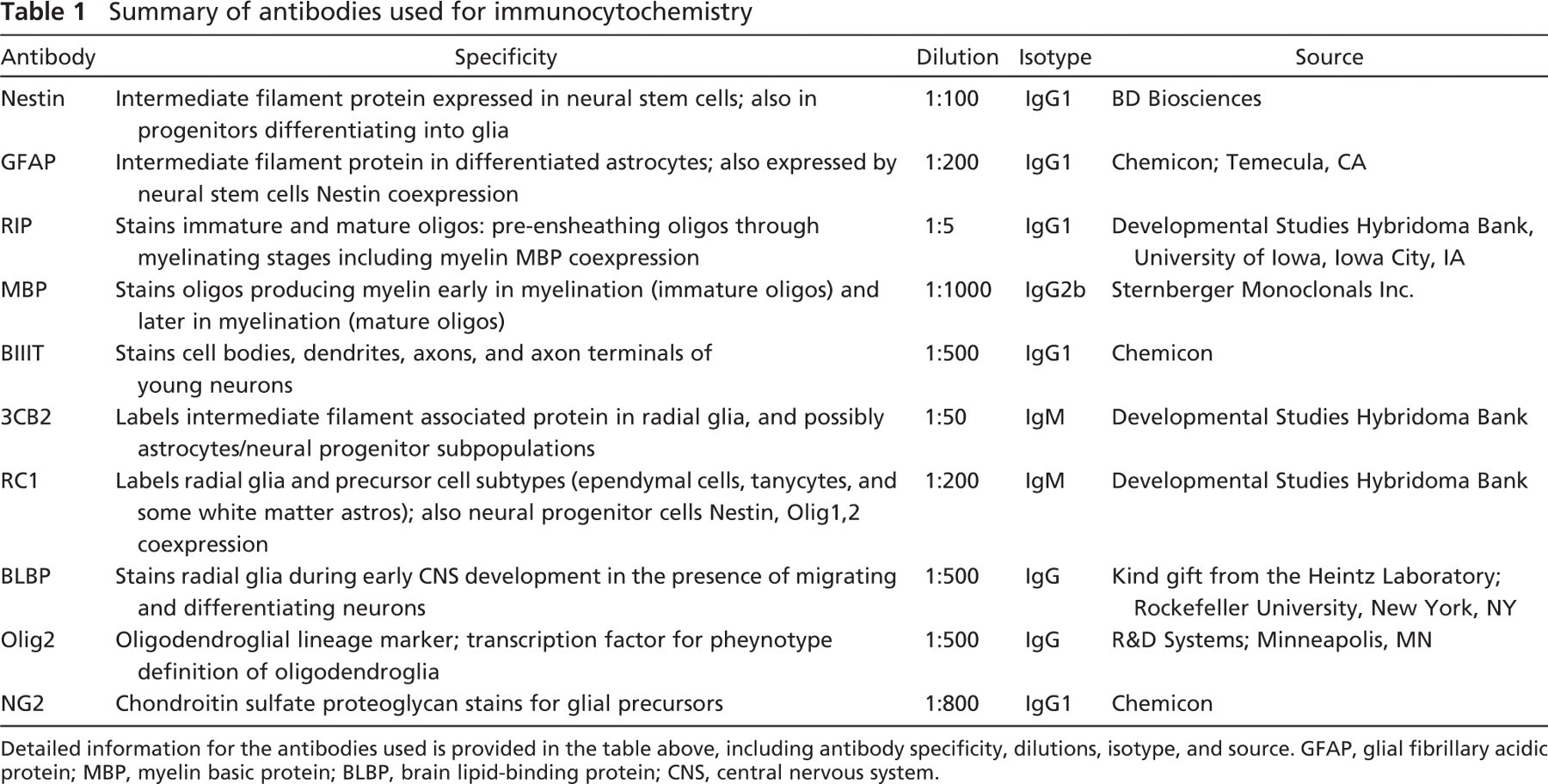
Detailed information for the antibodies used is provided in the table above, including antibody specificity, dilutions, isotype, and source. GFAP, glial fibrillary acidic protein; MBP, myelin basic protein; BLBP, brain lipid-binding protein; CNS, central nervous system.
Dissected tissue was placed in fresh DPBS—glucose, cut into 1-mm3 pieces, and then transferred to the papain dissociation system enzyme solution (Worthington Biochemical Corporation; Lakewood, NJ), consisting of 0.01% papain and 0.01% DNase I. The tissue was incubated in the enzyme solution at 37C for 1 hr with continuous agitation. Following incubation, the tissue was triturated with a 10-ml pipette and the enzymatic reaction stopped with ovomucoid protease inhibitor. Cell suspension was centrifuged for 5 min at 1000 rpm using a discontinuous density gradient to remove cell membrane fragments. Cells were resuspended in Neurobasal-A medium (Gibco—Invitrogen; Burlington, ON, Canada) supplemented with B27 (Gibco—Invitrogen), L-glutamine (Gibco—Invitrogen), penicillin/streptomycin (Gibco—Invitrogen), 20 ng/ml epidermal growth factor (Sigma—Aldrich), 20 ng/ml fibroblast growth factor-2 (Sigma—Aldrich), 2 μg/ml heparin (Sigma—Aldrich), and hormone mix. The hormone mix was composed of DMEM/F-12 (1:1), 0.6% glucose, 25 μg/ml insulin, 100 μg/ml transferrin, 5 mM HEPES buffer, 3 mM sodium bicarbonate, 30 nM sodium selenite, 10 μM putrescine, and 20 nM progesterone (all from Sigma—Aldrich). Cells were seeded in Nunc T25 culture flasks (VWR International; Mississauga, ON, Canada) and maintained at 37C in an incubator with 100% humidity and 5% CO2.
Neurospheres were passaged every 5-7 days. Passaging of neurospheres was accomplished by collecting the cell culture medium containing the free-floating neurospheres in a 50-ml centrifuge tube and centrifuging for 1 min at 1500 rpm. The supernatant was discarded, the pellet resuspended in fresh supplemented Neurobasal-A medium, and the cell suspension was triturated 40 times using a 1-ml pipette. Cells were seeded in Nunc T25 culture flasks (VWR International) and incubated at 37C in an incubator with 100% humidity and 5% CO2. Native EGFP expression in cultured cells was examined under a 488-nm excitation light microscope (Eclipse TE 300; Nikon, Tokyo, Japan).
Percent immunopositive cells in undifferentiated and differentiated periventricular spinal cord spheres
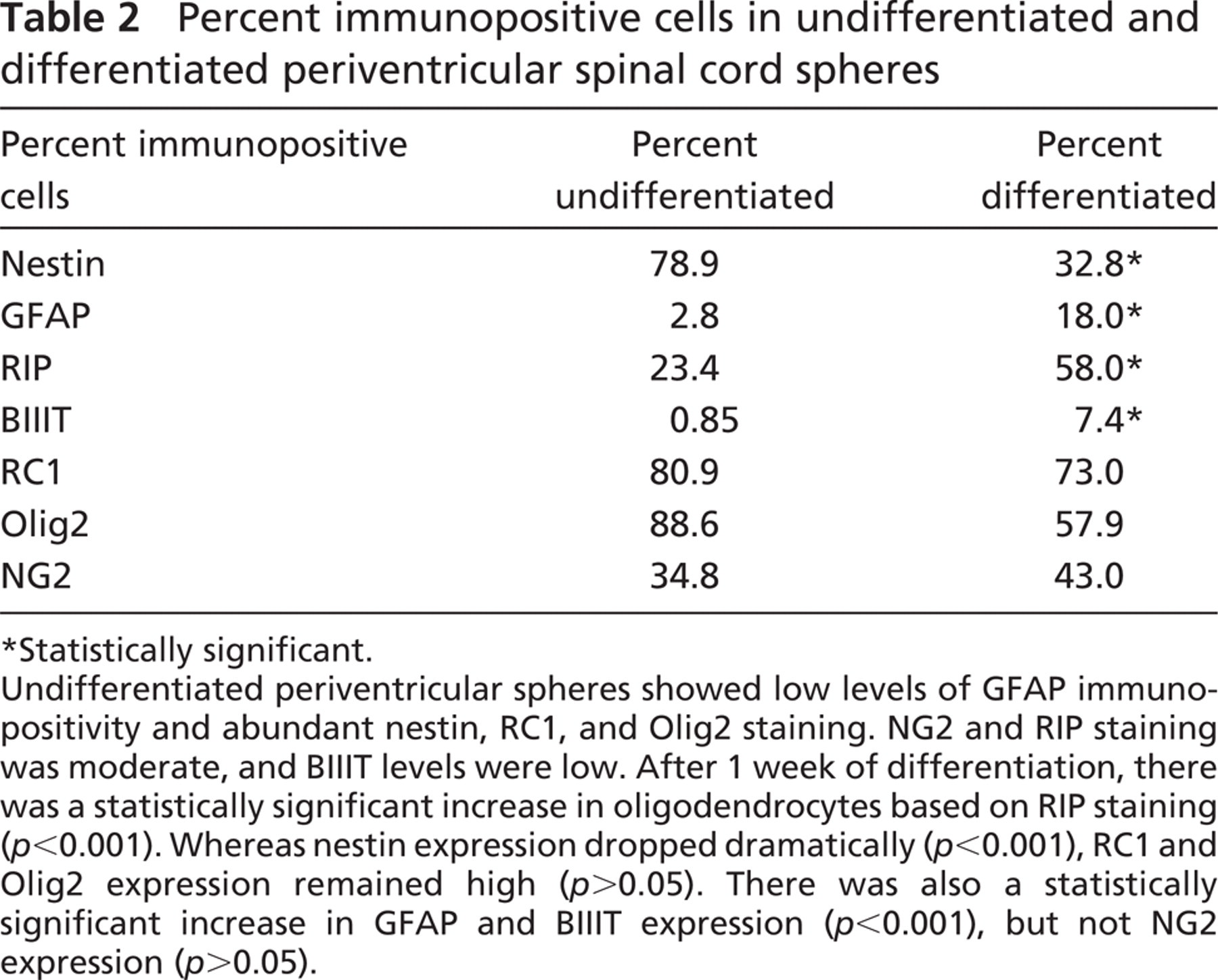
Statistically significant.
Undifferentiated periventricular spheres showed low levels of GFAP immunopositivity and abundant nestin, RC1, and Olig2 staining. NG2 and RIP staining was moderate, and BIIIT levels were low. After 1 week of differentiation, there was a statistically significant increase in oligodendrocytes based on RIP staining (
A limiting dilution assay was performed to verify the clonal derivation of the neurospheres by dissociating one to two spheres into single cells and replating the single cells in Neurobasal-A-supplemented media for 1 week. The assay was repeated in a total of 32 wells.
Quantitative Analyses of Average Sphere Volume and Propidium Iodide-positive Area Fraction
A 1-ml aliquot of neurospheres from passage 2 (P2), P3, or P4, free-floating in culture for 5, 6, or 7 days in vitro (div), was transferred to four-well Nunc dishes (VWR International). Neurospheres were counterstained with 10 μg/ml of Hoechst nuclear dye (Sigma—Aldrich) and incubated at 37C for 20 min. To assess cell viability, 1 μg/ml of propidium iodide (PI; Invitrogen Canada) was added to the Hoechst counterstained cultures. PI stained dead cells. A 4-μl aliquot of this suspension was transferred to a 1-mm2 coverslip placed upside down on a hanging-drop slide (Fisher Scientific; Ottawa, ON, Canada). Neurospheres were viewed with a confocal microscope (LSM-510; Carl Zeiss, Oberkochen, Germany), and representative images through a 1-μm plane of section in the central region of EGFP-expressing spheres were obtained. Corresponding Hoechst nuclear counterstained spheres and PI-stained images were also obtained. For some neurospheres, serial confocal images along the Z-axis were acquired at 5-μm intervals through the entire sphere.
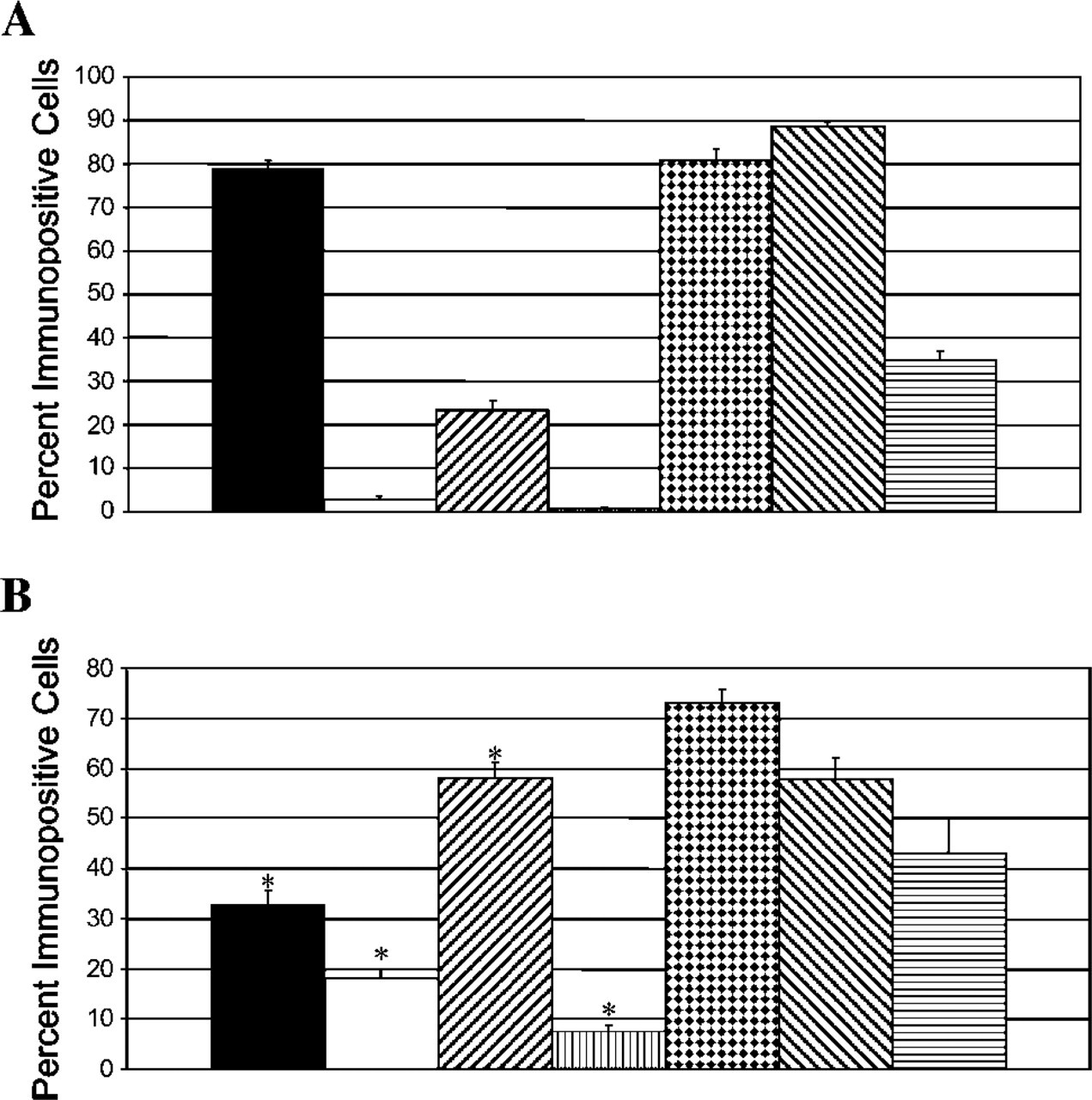
Percent immunopositive cells in undifferentiated and differentiated neurospheres. Undifferentiated neurospheres (
Quantitation was performed with the Image J analysis program (National Institutes of Health; Bethesda, MD). Average volume of a sphere for each passage and div was calculated from the mean diameter obtained by measuring intact representative spheres (
ICC
ICC was used to assess differentiated progeny as well as whole neurospheres. To characterize differentiated progeny, single neurospheres were transferred to Matrigel-coated multi-well culture plates (BD Biosciences; Mississauga, ON, Canada) in serum-free media containing 1% fetal bovine serum (FBS). After 7 div, cells were fixed in 4% paraformaldehyde for 20 min at room temperature, followed by three washes in 0.1 M PBS for 10 min each. Cells were blocked using normal goat serum with 0.3% Triton-X for 1 hr at room temperature (except for BIIIT staining where cells were exposed to Triton-X for only 5 min prior to blocking). Cells stained for Olig2 were blocked in normal donkey serum with 0.3% Triton-X and 1% BSA. Following the block, cells were exposed to the primary antibody overnight at 4C. The following day, cells were washed an additional three times in 0.1 M PBS for 10 min each, prior to adding the secondary antibody for 1 hr at 37C. This was followed by an additional three washes in 0.1 M PBS for 10 min each. Specific information about the primary antibodies used is listed in Table 1. Alexa 568 goat anti-mouse (1:500; Invitrogen Canada) was used as the secondary antibody in all cases except for Olig2 staining, for which Alexa 568 donkey anti-goat was used (1:500; Invitrogen Canada), and brain lipid-binding protein (BLBP) and NG2 staining, for which Alexa 568 goat anti-rabbit (1:500; Invitrogen Canada) was used. For double-labeling studies, Alexa 350 goat anti-mouse (1:100; Invitrogen Canada) was used. Hoechst (1 μ1/4 ml; Sigma-Aldrich, Oakville, ON, Canada) was included at the end of the ICC run to label nuclei, except in double-labeling studies.
A separate ICC study to verify the accuracy of cell counts was performed on periventricular whole neurospheres that were dissociated prior to differentiation for 1 week. To assess the expression pattern in undifferentiated whole neurospheres, neurospheres were dissociated into single cells and plated for 2 hr. In some cases, undissociated whole spheres were plated down for 2 hr. In all cases, cells were plated on Matrigel-coated multi-well culture plates (BD Biosciences) in serum-free media. Only the 1-week differentiated cells were exposed to 1% FBS for this study. All experiments were performed in triplicate to verify accuracy of results.
Quantitation of Differentiated Progeny
To determine the number of differentiated progeny generated, the number of immunopositive cells for each antibody was counted as a percentage of EGFP+ cells or Hoechst+ nuclei in 10 random fields of cells. Fluorescent cells were examined using a Nikon Eclipse TE 300 microscope, and images were captured with a CCD camera and Bioquant Imaging Software (R & M Biometrics Inc.; Nashville, TN). Data were collected and analyzed using SigmaStat version 3.1 software. Data are presented as mean ± SEM. Statistical differences between groups were evaluated with Student's
Electron Microscopy
Transmission electron microscopy (TEM) was used to analyze whole neurospheres (passage 4, div 7) as well as differentiated cells (passage 4, 7-day differentiation). Whole neurospheres were centrifuged and the supernatant removed prior to fixing. After fixation, whole neurospheres were set in 2% agar, cut into 1-mm3 sections on a glass slide, and placed back in a vial for further processing. Differentiated cells grown on Thermanox (VWR International Ltd.; Mississauga, ON, Canada) coverslips were processed in individual wells of four-well dishes and then transferred into polyethylene vials after dehydration. Neurospheres and differentiated cells were fixed in 2% paraformaldehyde and 2.5% glutaraldehyde (pH 7.3) for 20 min, postfixed in 1% OsO4 at room temperature for 1 hr, and stained en bloc with 2% uranyl acetate overnight at 4C in the dark, with washes in 0.1 M phosphate buffer in between each step. This was followed by progressive dehydration with 50%, 70%, 80%, 90%, and 100% ethanol, each of 10-min duration. Two additional 10-min washes with 100% ethanol were followed by washing with propylene oxide, progressive infiltration with Epon—Araldite resin, and overnight rotation with 100% resin. Differentiated cells were then embedded in fresh 100% resin in inverted polyethylene Beem capsules with tips removed. Whole neurospheres were embedded in regular Beem capsules. The samples were allowed to cure for 24 hr at 60C under a vacuum of 20 mmHg. For the differentiated cells, coverslips were peeled off, leaving the cells on the surface of the block.
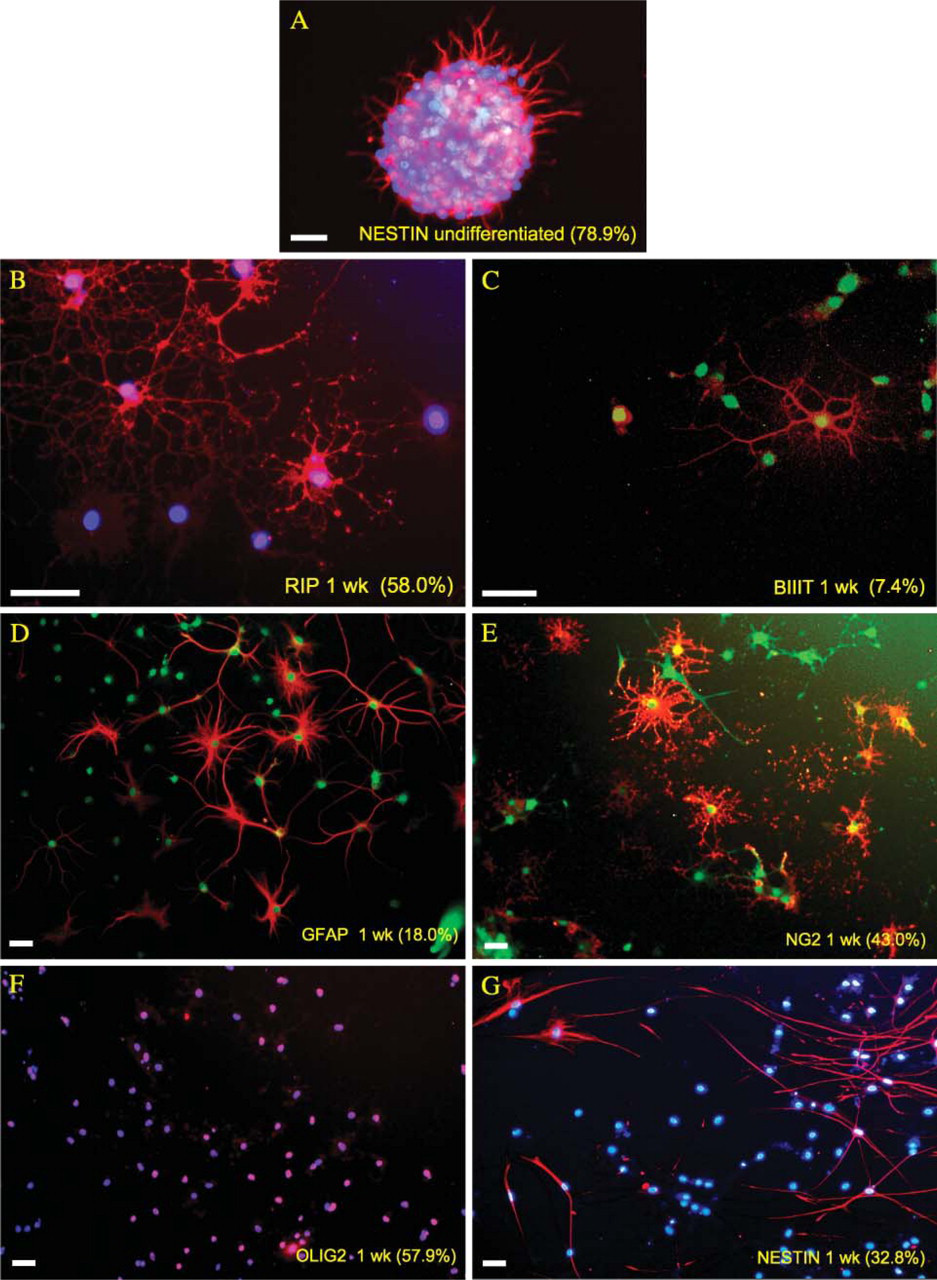
Mulitpotentiality of periventricular neural progenitor cells (NPCs). (
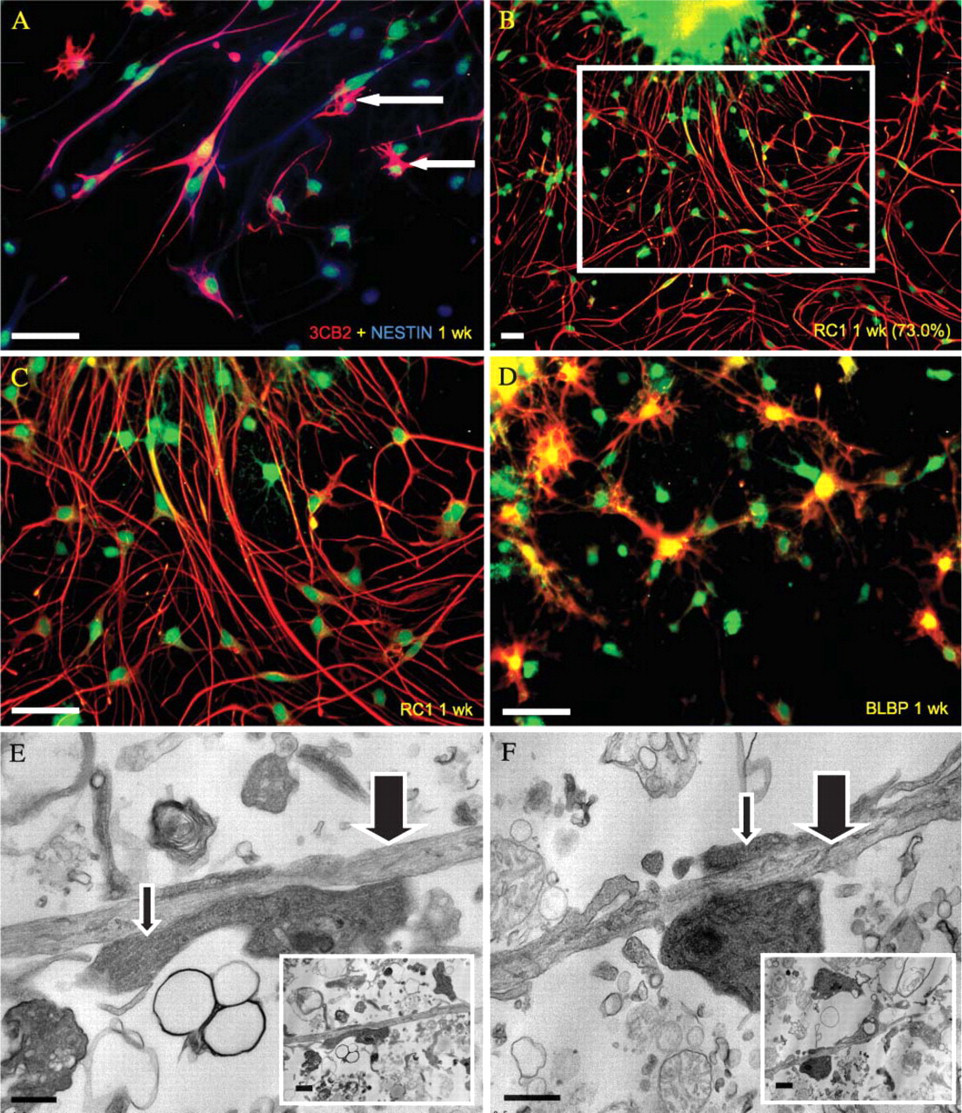
Radial glia 1 week post-differentiation. Identification of radial glia was based on radial, bipolar morphology, immunopositivity for several radial glial markers [3CB2, RC1, and brain lipid-binding protein (BLBP)], and transmission electron microscopy (TEM) imaging. (
Thick cross-sections (0.5 μm) of the whole spheres and differentiated cells were cut on a Reichert-Jung (Leica; Wetzlar, Germany) Ultracut E ultramicrotome and stained with 1% toluidine blue for orientation. Thin sections (70 nm) were cut with a diamond knife, mounted on copper mesh grids, and counterstained with 2% uranyl acetate and lead citrate for 15 and 5 min, respectively. The grids were then viewed and digitized images captured using a Jeol 1200EX electron microscope (Jeol; Tokyo, Japan).
Results
Differentiation Pattern of Periventricular Neurospheres
Undifferentiated spheres were composed of 34.8% NG2-positive cells and 88.6% Olig2-positive cells (Table 2; Figure 1). After differentiation, NG2 and Olig2 positivity was 43.0% and 57.9%, respectively, but the changes were not statistically significant (
Tissue harvested from the parenchymal white matter of the cord did not produce multipotential NPCs, but rather glial-restricted precursors. The proportions of differentiated progeny for parenchymal white matter spheres were comparable to that of the periventricular spheres, in that the majority of cells were RIP+. However, there was no neuronal immunopositivity in either undifferentiated parenchymal white matter spheres or their differentiated progeny (data not shown).
Myelin Production and Phagocytosis of Debris
We found evidence of robust myelin production by adult spinal cord-derived NPCs, based on ICC staining for myelin basic protein (MBP) as well as TEM images of myelin profiles (Figure 4). Both periventricular as well as parenchymal white matter-derived spheres and differentiated progeny were immunopositive for MBP. Only the periventricular neurospheres and progeny were processed for TEM, and in both cases we saw distinctive myelin profiles (Figure 4). There was evidence of myelin production in the absence of axonal fibers as well as myelin wrapped around axonal fibers (Figure 4). Neurosphere cells appeared to be actively involved in maintaining their local microenvironment, based on electron microscopic evidence of phagocytosis of surrounding debris by viable NPCs (Figure 4).
Assessment of Neurosphere Diameter, Volume, and Viability Over Time
The average diameter of periventricular neurospheres growing in free-floating culture increased from P2 to P4. The average diameter of neurospheres at P4 was significantly higher than at P2 and P3 (
Discussion
Differentiation of Periventricular vs Parenchymal White Matter Spheres
Adult CNS neuroepithelial precursors express nestin (Hockfield and McKay 1985; Frederiksen and McKay 1988; Cattaneo and McKay 1990; Lendahl et al. 1990; Morshead et al. 1994) both in vivo and in vitro (Reynolds and Weiss 1992; Dahlstrand et al. 1995; Gritti et al. 1996; Weiss et al. 1996; Stemple and Mahanthappa 1997). During spinal cord development, nestin is expressed around the central canal and is progressively reduced with maturation (Tohyama et al. 1992). These nestin-expressing precursors proliferate and migrate after SCI (Frisen et al. 1995,1998; Johansson et al. 1999; Namiki and Tator 1999). In immature (Gilmore and Leiting 1980; Bruni and Anderson 1987) and adult mammals (Adrian and Walker 1962; Kraus-Ruppert et al. 1975), ependymal cells proliferate in the normal and injured spinal cord (Matthews et al. 1979; Vaquero et al. 1981; Wallace et al. 1987; Beattie et al. 1997; Namiki and Tator 1999; Takahashi et al. 2003; Mothe and Tator 2005), which is enhanced by growth factor infusion (Kojima and Tator 2000,2002; Namiki et al. 2000). Mouse spinal cord NPCs reside near the central canal because multipotent, self-renewing neurospheres are generated only if the central canal is cultured (Martens et al. 2000,2002). In contrast, the medial and lateral regions of the adult rat spinal cord yield spheres, although parenchymal spheres are more limited in their proliferation and differentiation potential (Yamamoto et al. 2001).
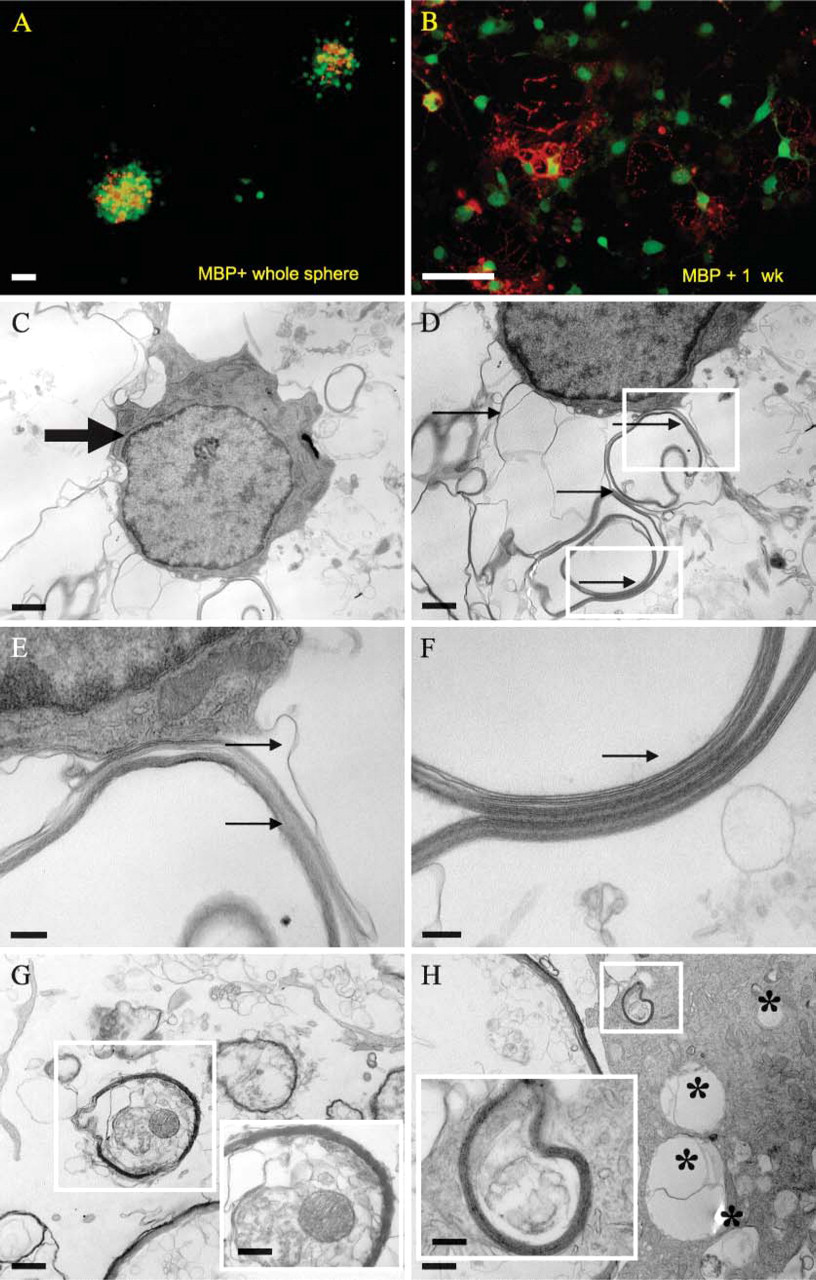
Myelin production and phagocytosis of debris. Production of myelin by undifferentiated whole central neurospheres as well as differentiated progeny was determined based on immunopositivity for myelin basic protein (MBP) (
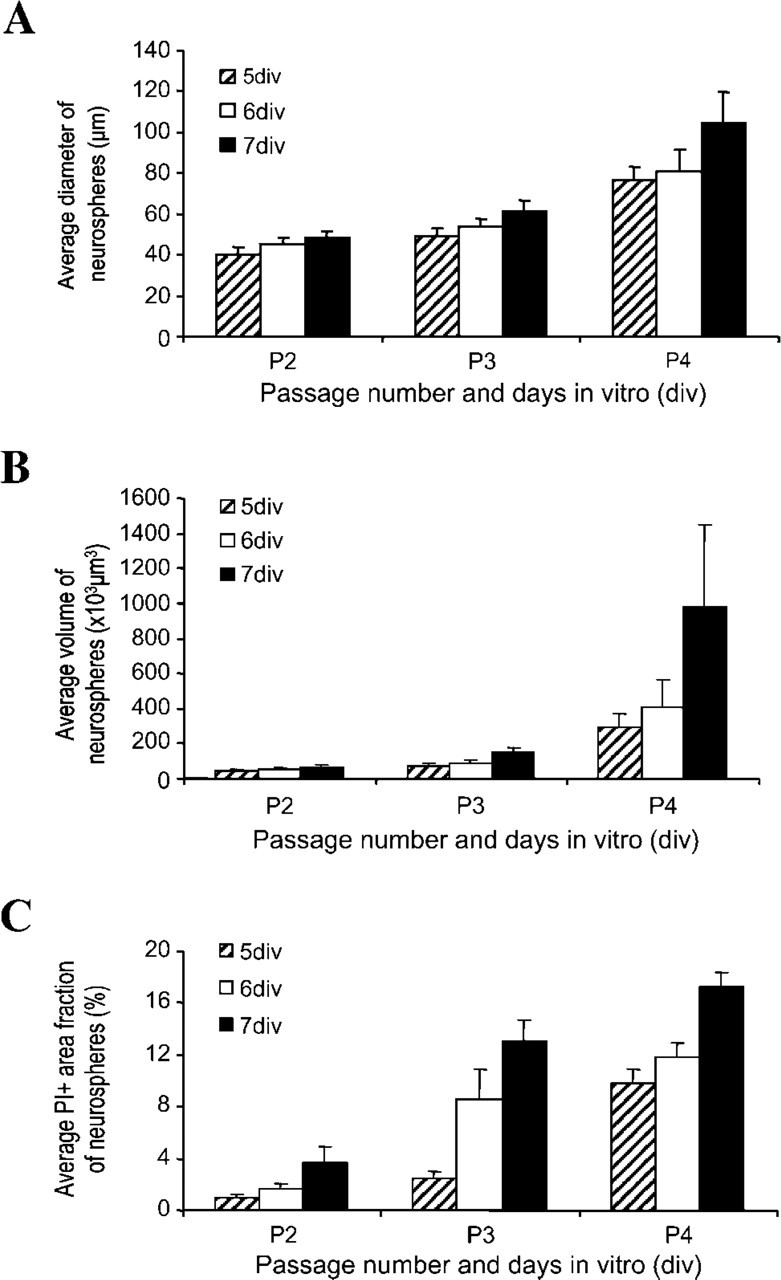
Neurosphere diameter, volume, and viability. Average diameter of central neurospheres in culture increases from passage 2 (P2) to passage 4 (P4) as shown in
We confirmed here that adult rat spinal cord periventricular region neurospheres are multipotent, producing neurons, astrocytes, and oligodendrocytes, whereas spheres derived from the parenchymal white matter are glial-restricted progenitors, producing only astrocytes and oligodendrocytes, but not neurons in vitro. We showed for the first time that the native differentiation pattern of periventricular spinal cord NPCs is region specific because most of the progeny differentiate into oligodendrocytes. Adult forebrain (Reynolds and Weiss 1992; Gritti et al. 1996), fetal brain (Lobo et al. 2003), and embryonic spinal cord NPCs (Chow et al. 2000) differentiate predominantly into astrocytes.
Myelin Production by Neurospheres and Progeny
It has been previously shown that neurospheres of rat (Mokry et al. 1996) and human embryonic brain NPCs (Yang et al. 2003) can produce myelin in vitro. We showed for the first time that adult rat spinal cord-derived neurospheres have the capacity to produce myelin in vitro. Given the high expression of oligodendrocyte markers by these neurospheres and the distinctive myelin profiles seen under TEM, it is reasonable to hypothesize that transplantation of adult rat spinal cord neurosphere transplants may improve axonal and functional recovery by remyelination of demyelinated or regrowing axons. This argument is further strengthened by our findings that these NPCs differentiate primarily into functional (myelin-producing) oligodendrocytes. Although the current study does not show in vivo data, additional transplantation experiments are currently underway in our laboratory to determine whether the in vivo differentiation pattern of spinal cord NPCs is the same as in vitro.
Recently, embryonic stem cells were driven along an oligodendrocytic lineage and promoted functional recovery after transplantation into the injured rat spinal cord (Keirstead et al. 2005; Nistor et al. 2005). Therefore, remyelination post-SCI is crucial; however, among the drawbacks of transplanting embryonic stem cells are the ethics of their procurement and their inherent oncogenic potential. Adult spinal cord NPCs may be ideal candidates for SCI therapy based on their inherent ability to become myelin-producing oligodendrocytes, without the increased risk of tumorigenicity seen with embyronic stem cells.
Oligodendrocyte Progenitor Cells (OPCs) as Multipotent NPCs
There is accumulating evidence that OPCs, which have until recently been regarded as lineage-restricted precursors, can behave as multipotent neural stem cells under particular conditions (Kondo and Raff 2000,2004; Gaughwin et al. 2006). Olig2 is a transcription factor required for the development of motoneurons and oligodendrocytes during early embryogenesis (Takebayashi et al. 2002), and a distinct oligodendrocyte—motoneuron precursor is thought to act as an intermediate precursor between stem cells and differentiating oligodendrocytes (Rowitch et al. 2002). Recently, it has been shown that progenitor cells expressing Olig2 in the embryonic spinal cord give rise to motoneurons, oligodendrocytes, astrocytes, and ependymal cells (Masahira et al. 2006).
Based on the high levels of NG2 and Olig2 expression of our spinal cord spheres, it is likely that a large proportion of these spheres are OPCs. Because we did not sort our cells after isolating the spinal cord tissue, it is highly unlikely that we have a pure population of progenitors, but rather that our cultures represent a heterogeneous population of spinal cord progenitor cells including NPCs and OPCs at variable stages of maturation. However, because our undifferentiated neurospheres express high levels of nestin and Ki-67 (data not shown), the majority of the cells within the spheres are immature/undifferentiated proliferative cells. Moreover, Olig2 expression itself suggests cellular heterogeneity because Olig2 expression in the embryonic spinal cord is specific to a domain that contains a heterogeneous population of progenitors. These include stem cells and glial progenitors, including those that coexpress RG markers (Liu and Rao 2004).
Neurosphere Viability
The success of transplanted NPCs for SCI therapy depends on optimizing NPC viability within the neurosphere before transplantation. Our viability assays using PI staining showed that the ideal passage times to transplant rat spinal cord neurospheres are passages 2 to 4 inclusive, at 5 to 7 div, because of the progressive decrease in cell survival with subsequent passages. Although cell survival within the neurosphere decreases progressively with increasing passage times and div, the relative number of viable NPCs remains high. A consistent feature of spheres is that as NPCs proliferate and the sphere enlarges, cells within the center of the sphere are deprived of nutritional support, resulting in their death (Freyer and Sutherland 1986). TEM of passage 4, div 7 neurospheres showed characteristic profiles of degenerating cells and phagocytosis of cellular debris by adjacent healthy NPCs. This characteristic has been shown for neurospheres from the rat fetal brain (Lobo et al. 2003), human fetal brain (Bez et al. 2003), and human embryonic stem cell colonies (Johkura et al. 2004). We are the first to describe the phagocytic capability of adult mammalian spinal cord neurosphere NPCs, wherein viable cells maintain the equilibrium between ongoing cell proliferation, nutrient availability, and cell death. Regarding future SCI therapies, spinal cord neurosphere transplants may be particularly apt to “mop up” the injury environment by phagocytosing cellular and myelin debris, thus enhancing the hospitability to graft survival and integration following NPC differentiation in vivo. However, because in vivo experiments are beyond the scope of this paper, it is not known whether the phagocytic property of these cells is maintained after transplantation. Future studies will focus on whether spinal cord NPCs retain phagocytic capacity in vivo after transplantation.
RG as In Vitro Guidance Scaffolds and NPCs
During cortical development, RG direct migrating neurons, proliferate (Misson et al. 1988; Schmechel and Rakic 1979a,b), and give rise to astrocytes and oligodendrocytes (Choi and Kim 1985; Hirano and Goldman 1988). Their differentiation into astrocytes coincides with the cessation of neurogenesis (Culican et al. 1990; Edwards et al. 1990; Misson et al. 1991; Schmechel and Rakic 1979a,b; Voigt 1989; Yang et al. 1993). RG may also guide migrating glial precursors (O'Rourke et al. 1992; Diers-Fenger et al. 2001). Usually, RG are only present during CNS development; however, some RG remain into adulthood (Schnitzer et al. 1981; Liuzzi and Miller 1987). In lower vertebrates in which there is continual neurogenesis into adulthood, RG are always present (Rosen et al. 1994). In the adult rat, upregulation of RG markers in the ependyma following SCI likely plays a role in neural regeneration (Shibuya et al. 2003). It was recently shown that embryonic rat RG transplanted into the injured rat spinal cord formed bridges and promoted functional recovery (Hasegawa et al. 2005).
In vitro, NPCs migrate from neurospheres via chain migration (Lois et al. 1996; Jacques et al. 1998) and are guided by RG scaffolds in differentiating neurospheres from the embryonic rat (Espinosa-Jeffrey et al. 2002), mouse (Gregg and Weiss 2003), and fetal human brain (Caldwell et al. 2001), as well as mouse embryonic stem cell lines (Liour and Yu 2003). We showed for the first time that adult rat spinal cord neurospheres differentiate abundantly into RG. Moreover, we found that dissociated neurospheres do not differentiate into this radial morphology, supporting the transplantation of whole neurospheres rather than dissociated cells.
Developmentally, RG express BLBP only during neuronal migration, and BLBP transcription occurs in vitro in the presence of differentiating neurons (Feng et al. 1994; Feng and Heintz 1995), suggesting that the BLBP+ RG in our study are functioning to aid NPC migration in vitro. Our finding that 3CB2+ RG coexpress nestin suggests that they may have stem cell properties. Historically, RG were viewed as glial precursor cells, entirely distinct from the large population of neuronal precursors present during neurogenesis. However, it has recently been shown that RG comprise the majority of precursors in the ventricular zones in most areas of the CNS, especially in the brain (Malatesta et al. 2000,2003; Hartfuss et al. 2001,2003; Miyata et al. 2001; Noctor et al. 2001,2002; Tamamaki et al. 2001; Heins et al. 2002). Both RG in neonates and RG-derived cells in the adult lateral ventricle generate self-renewing multipotent neurospheres (Merkle et al. 2004). Additionally, there are RG that have been shown to differentiate into ependymal cells (Malatesta et al. 2003; Spassky et al. 2005). Our current findings that adult rat spinal cord neurospheres express high levels of RG markers, including nestin, and appear to function as guidance scaffolds based on preliminary evidence showing BLBP positivity and EM images, suggest a recapitulation of their developmental role in vitro. However, because we have not provided direct evidence of migration in the current study, our present results are only suggestive of an in vivo significance. Future studies involving in vitro time-lapse photography and lineage tracking are needed to provide a definitive answer.
In conclusion, we have shown for the first time that adult mammalian periventricular spinal cord NPCs, expanded as neurospheres, differentiate predominantly into functioning oligodendrocytes. In contrast, spheres harvested from the parenchymal white matter of the spinal cord are not multipotent but rather are glial-restricted precursors. Moreover, we found that periventricular neurospheres and differentiated progeny express high levels of several radial glial markers. This information, together with evidence from TEM images of presumed NPCs intimately apposed to radial processes and BLBP+ RG, suggests that adult mammalian periventricular spinal cord neurospheres produce RG that may act as guidance scaffolds. Our immunolabeling studies support previous work showing that RG are NPCs. We also showed evidence that spinal cord neurospheres have the capacity to maintain their microenvironment because healthy NPCs phagocytose surrounding debris from dead cells. Overall, our in vitro data support a region-specific transplantation paradigm for SCI treatment, wherein adult mammalian spinal cord neurospheres would be appropriate candidates for transplantation into the injured mammalian spinal cord.
Footnotes
Acknowledgements
This work was supported by grants from the Ontario Neurotrauma Foundation, Canadian Paraplegic Association (Ontario Branch), Physician's Services Incorporated, and the Christopher Reeve Paralysis Foundation (to CHT). Personal support was provided by an Ontario Student Opportunity Trust Fund/Vision Sciences Scholarship and Sandra and David Smith Graduate Student Award (to IK), and by an Ontario Neurotrauma Foundation Fellowship (to AJM). A.K. holds the Gloria and Seymour Epstein Chair in Cell Therapy and Transplantation at the University Health Network and University of Toronto.
We thank Huijie Jiang, XingHua Wang, and Dr. A. Keating for maintaining a steady supply of Wistar—TgN(CAG-GFP) 184Ys rats for these studies. We also thank Rita van Bendegem and Linda Lee for contributions to the immunocytochemical data, Sheer Ramjohn for contributions to the EM data, and A. Weisman for help with data analysis.
