Abstract
Spinal cord injury (SCI) initiates a cascade of biochemical changes that results in necrotic and apoptotic cell death. There is evidence that caspase-3 activation and apoptotic cell death occur within hours after SCI. However, the time course and cellular localization of activated caspase-3 has not been examined. Such information is essential because caspase-3–independent apoptotic pathways do exist. In this experiment, we describe the distribution of and cell types containing activated caspase-3 at 4 hr, 1 day, 2 days, 4 days, and 8 days following SCI in rats. Numerous caspase-3–positive cells were observed at 4 hr and 1 day postinjury and colocalized most often with CC1, a marker for oligodendroglia. Both markers disappeared near the injury epicenter over the next several days. Activated caspase-3 was again present in the injured spinal cord on postoperative day 8, which coincided with a reemergence of CC1-positive cells. Many of these CC1-positive cells again colocalized activated caspase-3. NeuN-positive neurons of the dorsal horn were occasionally immunopositive for activated caspase-3 at early time points. OX42-positive microglia/macrophages rarely contained activated caspase-3. The results indicate a biphasic pattern of caspase-3 activation during the first 8 days postinjury, suggesting that at least two mechanisms activate caspase-3 following SCI. This time-course study provides a framework for investigating and understanding the different signaling events contributing to this biphasic pattern of caspase-3 activation.
T
Although our understanding of apoptotic cell death following SCI is far from complete, much is now known about individual intracellular proteins with apoptotic or anti-apoptotic functions. Evidence suggests that the morphological and nuclear changes that occur during apoptosis are initiated and executed through activation of the evolutionarily conserved caspase family of cysteine proteases (Thornberry 1997; Thornberry and Lazebnik 1998). Recent findings have established that caspase-3 is activated in humans (Emery et al. 1998; Harter et al. 2001; Hentze et al. 2001) and in animal models (Yakovlev et al. 1997; Springer et al. 1999; Beer et al. 2000; Citron et al. 2000; Clark et al. 2000; Casha et al. 2001; Nottingham et al. 2002; Sullivan et al. 2002) following traumatic brain and SCI. Although activated caspase-3 has been localized to a variety of cell types post-SCI (Springer et al. 1999; Citron et al. 2000; Clark et al. 2000; Beattie et al. 2002) and was correlated with DNA fragmentation (Casha et al. 2001), the time course of caspase-3 activation has not been thoroughly examined.
The purpose of the present experiment was to perform a descriptive analysis of the spatiotemporal distribution of activated caspase-3 at acute times following SCI (up to 8 days postinjury), and to determine if activated caspase-3 colocalized with cellular markers for neurons, oligodendroglia, or microglia/macrophages. Because there is clear evidence for the existence of caspase-3–independent apoptotic cell death pathways (Li et al. 2001; van Loo et al. 2001; Cande et al. 2002; Arnoult et al. 2003), this study was conducted to elucidate the time course of caspase-3 activation in the acute periods following SCI. The results of this study serve as a springboard for quantifying the effectiveness of therapeutic interventions that limit the activation or activity of caspase-3.
Materials and Methods
Subjects
Thirty-five Long–Evans, young-adult female rats, weighing approximately 200 g, were purchased from Harlan Sprague Dawley (Indianapolis, IN). The rats were housed in the vivarium of the University of Kentucky College of Medicine on a 12:12 hr light:dark cycle with food and water available ad libitum until euthanized at 4 hr, 1 day, 2 days, 4 days, or 8 days postsurgery. Each of those five experimental groups was comprised of two rats that received sham surgery and five rats that received a spinal cord contusion. The animal unit of the College of Medicine at the University of Kentucky Medical Center is fully accredited by AAALAC. The care of animals used in this experiment complied with standards set forth in the US Public Health Service Policy on Humane Care and Use of Laboratory Animals and the National Institutes of Health Guide for the Care and Use of Laboratory Animals. All experimental procedures were approved by the University of Kentucky Institutional Animal Care and Use Committee and conform to guidelines of the Society for Neuroscience and the Society for Neurotrauma.
Surgery
All surgical techniques were performed under aseptic conditions. Rats were anesthetized with 40 mg/kg sodium pentobarbital (Nembutal; Abbott Laboratories; North Chicago, IL) administered intraperitoneally and dorsal incisions were made in the skin and underlying muscles. The muscles were retracted and a laminectomy was performed at thoracic (T) segment 10 (T10). The muscles of control rats (sham surgery) were closed with absorbable sutures, and the skin was closed with wound clips. Sham control rats were then placed into an incubator (35C) for 2 to 3 hr to recover from anesthesia. The vertebral column of the remaining rats was stabilized by clamping the vertebrae at T8 and T11. The spinal cord was subsequently injured by dropping a 10-g rod, with a tip that measured 2.5 mm in diameter, a distance of 12.5 mm onto the exposed dorsal surface of the spinal cord (NYU Impactor device; New York University, New York, NY). The incision site of those rats was closed in layers as described and the rats were allowed to recover from anesthesia. All rats were then returned to their home cages in the colony room until euthanized. Bladders of the spinally injured rats were manually expressed 2X per day until micturition was reestablished or until the rats were euthanized.
Histology
After the appropriate survival period, the rats were deeply anesthetized with sodium pentobarbital (100 mg/kg) and perfused transcardially with ∼50 ml of 0.1 M phosphate-buffered saline (PBS), followed by 200 ml of 4% paraformaldehyde in PBS (pH, 7.2–7.4). A 2-cm segment of spinal cord centered on the lesion/laminectomy site was removed and placed into 25% sucrose-PBS at 4C for 5 days. Spinal cords were then rapidly frozen on dry ice and stored at −20C until they were embedded in tissue freezing medium (Triangle Biomedical Sciences; Durham, NC) and cut into 30-μm-thick transverse sections on a cryostat. Consecutive spinal cord sections were mounted in pairs on a series of charged glass slides (Superfrost Plus; Fisher Scientific, Pittsburgh, PA) so that adjacent pairs on each slide represented regions ∼1 mm (990 μm) apart in the spinal cord. After the sections air dried, the slides were stored at 4C until they were stained for myelin and Nissl substance or until fluorescence immunocytochemistry was performed.
Tissue sections from each subject were stained for myelin and Nissl substance for brightfield microscopy as follows: briefly, sections were dehydrated at room temperature (RT) in a graded ethanol series (5 min each) and cleared in Hemo-De (Fisher Scientific) for 5 min. Sections were then rehydrated in a reverse-graded ethanol series, followed by distilled water (5 min each). To stain myelinated fibers, the tissue sections were placed for 10 min into a solution that contained 0.16% Eriochrome cyanine-R (Sigma; St Louis, MO), 0.4% sulfuric acid, 0.4% iron chloride, and 0.12% hydrochloric acid and then rinsed under running tap water for 10 min. Sections were then differentiated in 1% ammonium hydroxide for ∼4 min and again rinsed under running tap water for 10 min. To stain neurons, the sections were placed in 0.1 M acetate buffer for 1 min and then stained in 0.03% neutral red (Sigma) in 0.1 M acetate buffer for 15 min. After staining, the slides were rinsed in distilled water (3 × 5 min), dehydrated in a graded ethanol series (2 min each), cleared in Hemo-De (2 × 2 min) (Fisher Scientific) and coverslipped with Permount (Fisher Scientific).
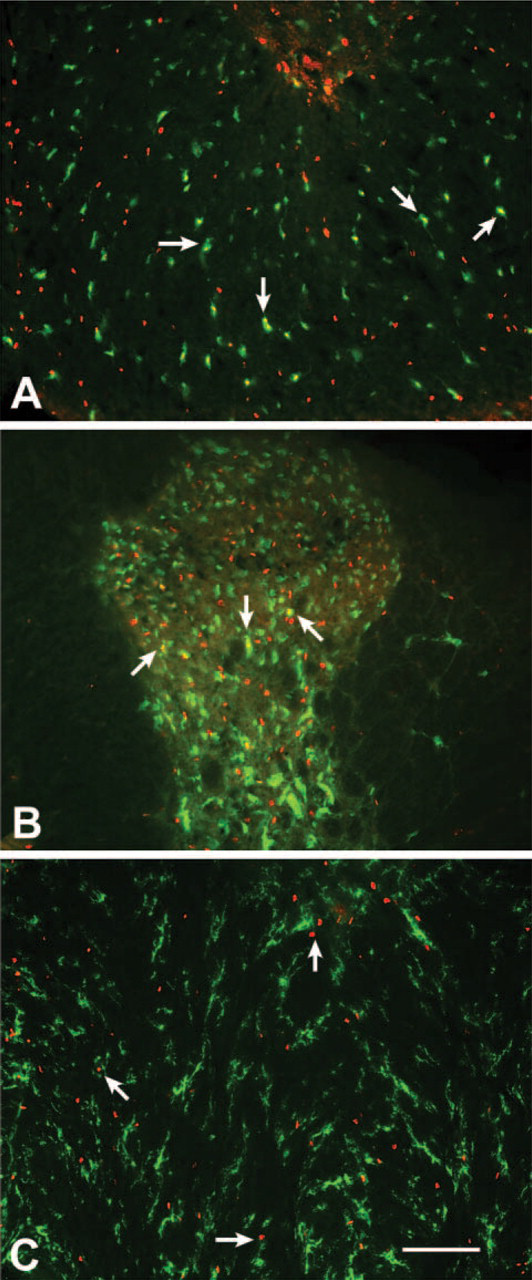
Cells that contain activated caspase-3 within the spinal cord of rats that received sham surgery. Photomicrographs provided are representative of the spinal cord of a rat that had one of the highest numbers of caspase-3–positive cells following sham surgery. These photomicrographs illustrate colocalization (yellow) of activated caspase-3 (red) and various cell markers (green) 1 mm from the center of the laminectomy site at 4 days postsurgery. Several cells were immunoreactive for both activated caspase-3 and the oligodendroglial marker, CC1, in the ventral funiculus
Adjacent slides were processed for fluorescence immunocytochemistry as follows: briefly, sections were rinsed for 1 hr at RT in several changes of PBS. Sections were then preincubated for 30 min at RT in a blocking solution comprised of 5% normal goat serum (NGS) and 0.05% Triton X-100 in PBS. The blocking solution was removed and sections were incubated in a humidified chamber overnight at 4C in PBS that contained 5% NGS and one of the following sets of primary antibodies: (a) rabbit anti-human/mouse activated caspase-3 IgG (polyclonal, 1:4000; R & D Systems, Minneapolis, MN) and mouse anti-APC/Ab7 (CC1) IgG (monoclonal, 1:250; Oncogene, Cambridge, MA), (b) anti-activated caspase-3 and mouse anti-neuronal nuclei (NeuN) IgG (monoclonal, 1:250; Chemicon International, Temecula, CA), or (c) anti-activated caspase-3 and mouse anti-rat CD11b/c (OX42) IgG (monoclonal, 1:4000; PharMingen, San Diego, CA). To test for nonspecific staining by the secondary antibodies, additional slides were processed in a similar fashion with the primary antibodies excluded. All slides were then rinsed for 1 hr at RT in several changes of PBS and incubated in the dark for 1 hr at RT in PBS that contained 5% NGS and the following fluorescent secondary antibodies: Cy-3-conjugated goat anti-rabbit IgG (polyclonal, 1:500; Jackson ImmunoResearch Laboratories, West Grove, PA) and AlexaFluor 488 goat anti-mouse IgG (monoclonal, 1:250; Molecular Probes, Eugene, OR). The secondary antibody solution was removed and the slides were rinsed in several changes of PBS for 15 min at RT. Tissue sections were immediately coverslipped with ProLong Anti-Fade (Molecular Probes) and stored at 4C in the dark to retard fading of the fluorescent labels.
Tissue sections were subsequently examined using a Zeiss AxioPlan microscope (Zeiss; Oberkochen, Germany). Myelin and Nissl staining were examined under brightfield, and images were captured with a Zeiss AxioCam color digital camera (X50). Colocalization of the fluorescent secondary antibodies was determined when paired images of each field were captured (X200 or X400) and superimposed using Photoshop (Adobe Systems; San Jose, CA). Immunoreactive cells were identified in gray (dorsal and ventral horn) and white matter. The analysis of immunoreactive cells in white matter was restricted primarily to the ventral funiculus, which contains descending supraspinal motor pathways affected by SCI in the rat.
Results
Activated Caspase-3 in Cells of Laminectomy Control Spinal Cords
Despite a normal gross morphology of the spinal cord and the absence of phagocytic microglia/macrophages, a few cells that contained activated caspase-3 were scattered throughout the gray and white matter of the spinal cord of all laminectomy control rats. CC1-positive cells were generally not immunoreactive for activated caspase-3 at the early time points postsurgery. However, some colocalization of the two antibodies was apparent 4 and 8 days after sham surgery (Figure 1A). A few NeuN-positive neurons of the dorsal horn were immunoreactive for activated caspase-3 at all time points (Figure 1B), whereas no NeuN-positive neurons of the ventral horn were observed to be caspase-3 positive (data not shown). Activated caspase-3 and OX42 were generally not colocalized following sham surgery, even though immunoreactive profiles for each marker were observed in close proximity to one another (Figure 1C).
Activated Caspase-3 and CC1 Immunoreactivity in Injured Spinal Cords
The radial and longitudinal expansion of the necrotic lesion following spinal cord contusion and the gradual increase in microglia/macrophage infiltration and activation (Figure 2) was similar to previous reports (Schwab and Bartholdi 1996). The presence of cells within the contused spinal cord that contained activated caspase-3 varied over time and appeared most abundant within cells in intact tissue that bordered regions of tissue necrosis, which appeared to decrease as the lesion area tapered with distance from the lesion epicenter. At 8 days postinjury, the necrotic lesion extended up to 4 mm rostral and 4 mm caudal to the injury epicenter. Therefore, the cell types that were immunoreactive for activated caspase-3 were examined within that 8-mm segment of spinal cord.
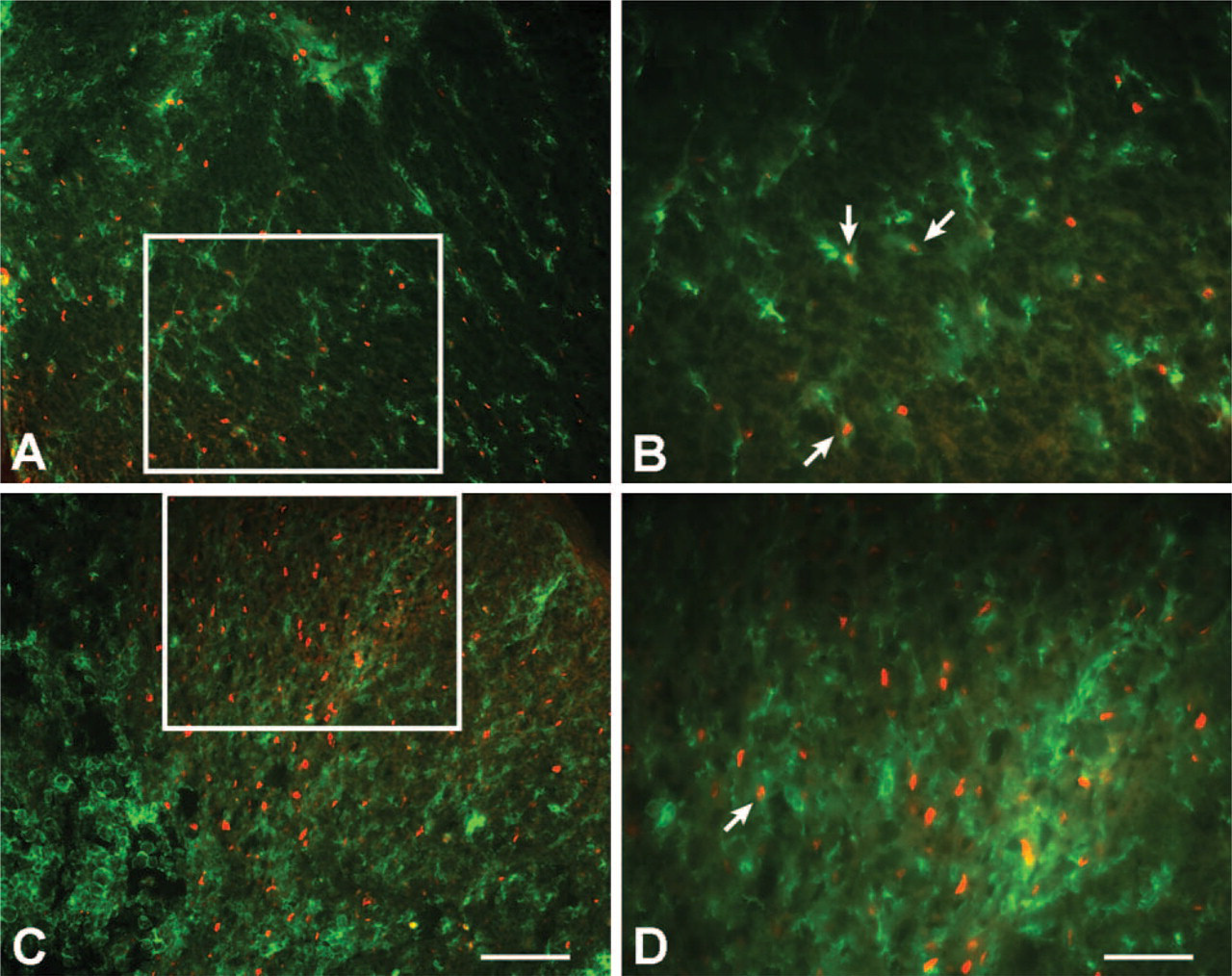
Within 4 hr of spinal cord contusion, cells that contained activated caspase-3 appeared more abundant, relative to time-matched sham controls, and were localized to areas of intact white matter for the entire 8-mm segment analyzed (Figure 3A). Several of these caspase-3–positive cells were also immunopositive for CC1 and presumed to be oligodendroglia. Cells that colocalized activated caspase-3 and CC1 continued to surround regions of tissue necrosis at 1 day postinjury relative to that observed in the spinal cords of time-matched controls (Figure 3B). However, the overall population of CC1-positive cells at 1 day postinjury appeared smaller than at the earlier time point (4 hr) for at least 2 mm rostral and caudal to the lesion epicenter. Both activated caspase-3– and CC1-positive cells continued to decline on postoperative day 2 (Figure 3C) and were sparse by postoperative day 4 (Figure 3D). Interestingly, CC1-positive cells were again abundant throughout the white matter near regions of tissue necrosis at 8 days post-SCI, and several cells colocalized activated caspase-3 (Figure 3E).
Activated Caspase-3 and NeuN Immunoreactivity in Injured Spinal Cords
Caspase-3–positive cells were found throughout all 8 mm of gray matter tissue examined but appeared most abundant proximal to the lesion epicenter. There was some colocalization of activated caspase-3 and NeuN in the dorsal horn at 4 hr and 1 day postinjury (Figures 4A and 4B). Although activated caspase-3 reappeared in white matter on postoperative day 8 (see Figure 3E), there was little colocalization of NeuN and activated caspase-3 in the gray matter at this time point (data not shown). Activated caspase-3 was not observed in any NeuN-positive neurons of the ventral horn, regardless of time postinjury (Figures 4C and 4D).
Activated Caspase-3 and OX42 Immunoreactivity in Injured Spinal Cords
Cells immunoreactive for activated caspase-3 or OX42 were often located in close proximity to one another at the time points examined following spinal cord contusion (Figure 5). Although rare, a few instances of antibody colocalization were observed in cells proximal to the lesion epicenter, primarily at 4 hr postcontusion when few activated phagocytic OX42-positive microglia/macrophages with a rounded morphology were detected in the spinal cord (see Figure 1D). At this early time point, the OX42-positive cells containing activated caspase-3 had a ramified morphology, but the processes appeared shorter than in the spinal cord of control rats.
Discussion
Previous research has demonstrated that apoptotic death of neurons and glia following spinal cord contusion occurs in experimental models (Crowe et al. 1997; Liu et al. 1997; Shuman et al. 1997; Yong et al. 1998; Li et al. 1999; Springer et al. 1999) and in humans (Emery et al. 1998). The results of the present descriptive study illuminate the time-course of caspase-3 activation in the spinal cord of adult rats following a “mild/moderate” contusion to the low-thoracic spinal cord. Although our goal was to map the temporal and spatial distribution of cells expressing activated caspase-3, in the absence of cell counts the interpretation of the findings should be considered provisional and will require confirmation by quantitative analysis. Regardless, the results of the present study are consistent with previous data from our laboratory (Nottingham et al. 2002) and suggest that activated caspase-3 is a biochemical mediator of at least two phases of oligodendroglial death during the first week following SCI. Briefly, activated caspase-3 was scattered within the gray and white matter of the contused spinal cord as early as 4 hr postinjury. Cells that contained activated caspase-3 were also observed at 1 day postinjury, but were dramatically reduced during the next several postoperative days. By postoperative day 8, activated caspase-3 was again present within cells of the gray and white matter of the contused spinal cord, which is consistent with published cell-counting data for this later time point (Nottingham et al. 2002).
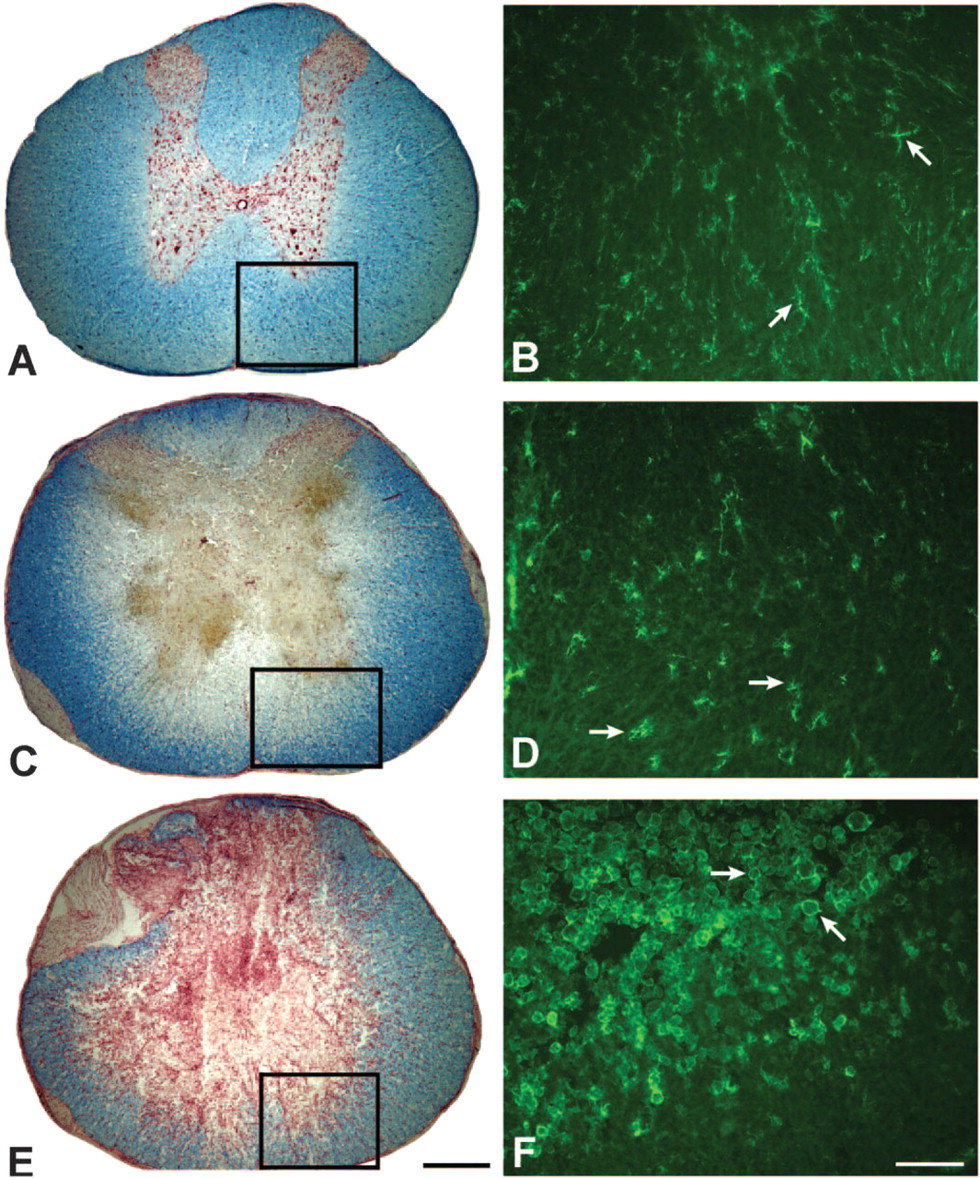
Time-course of lesion development and OX42 immunoreactivity at the lesion epicenter. The left-hand column of photomicrographs depicts tissue integrity following sham surgery
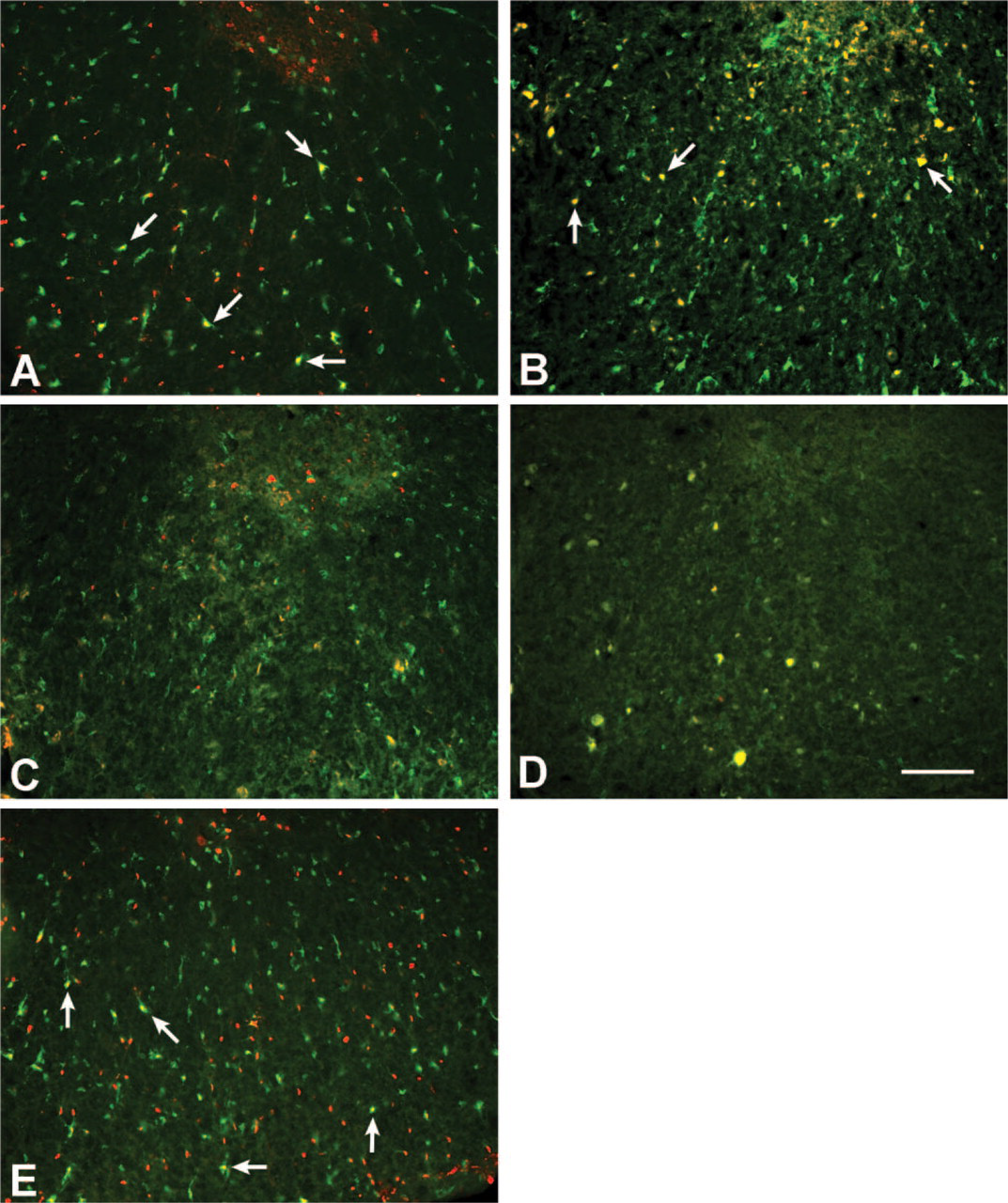
Photomicrographs that illustrate colocalization (yellow) of activated caspase-3 (red) and CC1 (green) in the ventral funiculus at different time-points following spinal cord contusion. Rats received a contusion injury to the low-thoracic spinal cord followed by a 4 hr-
Although caspase-3 activation may not necessarily indicate cells that will eventually die by apoptosis (Zeuner et al. 1999; Nicotera et al. 2000; Racke et al. 2002), it is noteworthy that the time-course of caspase-3 activation following spinal cord contusion reported here parallels previous reports of DNA fragmentation, as identified by terminal deoxynucleotidyl transferase-mediated dUTP nick end labeling (TUNEL) or Hoechst staining (Crowe et al. 1997; Liu et al. 1997; Shuman et al. 1997). However, the potential contribution of a caspase-3–independent apoptotic process cannot be overlooked and, therefore, our findings underscore the importance of correlating caspase-3 activation with other markers of apoptosis.
Activated caspase-3 in the contused spinal cord was primarily colocalized with CC1-positive cells, a marker for oligodendroglia (Bhat et al. 1996). Interestingly, the time-dependent decrease and resurgence of activated caspase-3 following SCI paralleled a time-dependent loss and reemergence of CC1-positive oligodendroglia in the spinal cord white matter within 2 mm of the lesion epicenter. Specifically, the initial upregulation of activated caspase-3 at 4 hr and 1 day postinjury was accompanied by a subsequent decrease in the number of CC1-positive oligodendroglia. Both cellular markers were sparse for the next several days postinjury. On postoperative day 8, activated caspase-3 was again present when CC1-positive cells were again abundant near the lesion epicenter. At this time-point, colocalization of activated caspase-3 and CC1 was again observed (see also Nottingham et al. 2002). Recent studies have also demonstrated a time-dependent loss and reemergence of oligodendroglia following SCI (Frei et al. 2000; McTigue et al. 2001). Previous reports have indicated that the number of mature oligodendroglia of the spinal cord dramatically decreases 3–4 days postinjury, especially near the lesion epicenter (McTigue et al. 2001). Several recent reports have suggested that oligodendroglial progenitor cells of the spinal cord increase after SCI (Ishii et al. 2001; McTigue et al. 2001; Jones et al. 2002; Jones and Tuszynski 2002). Death of mature oligodendroglia and subsequent proliferation and maturation of oligodendroglial progenitors may explain the observed loss and reemergence of CC1-positive cells 8 days post-SCI. However, injury-related changes in antigen expression or antigen binding by CC1 are possible alternative interpretations, which has been reported for neuronal markers following cerebral ischemia (Unal-Cevik et al. 2004).
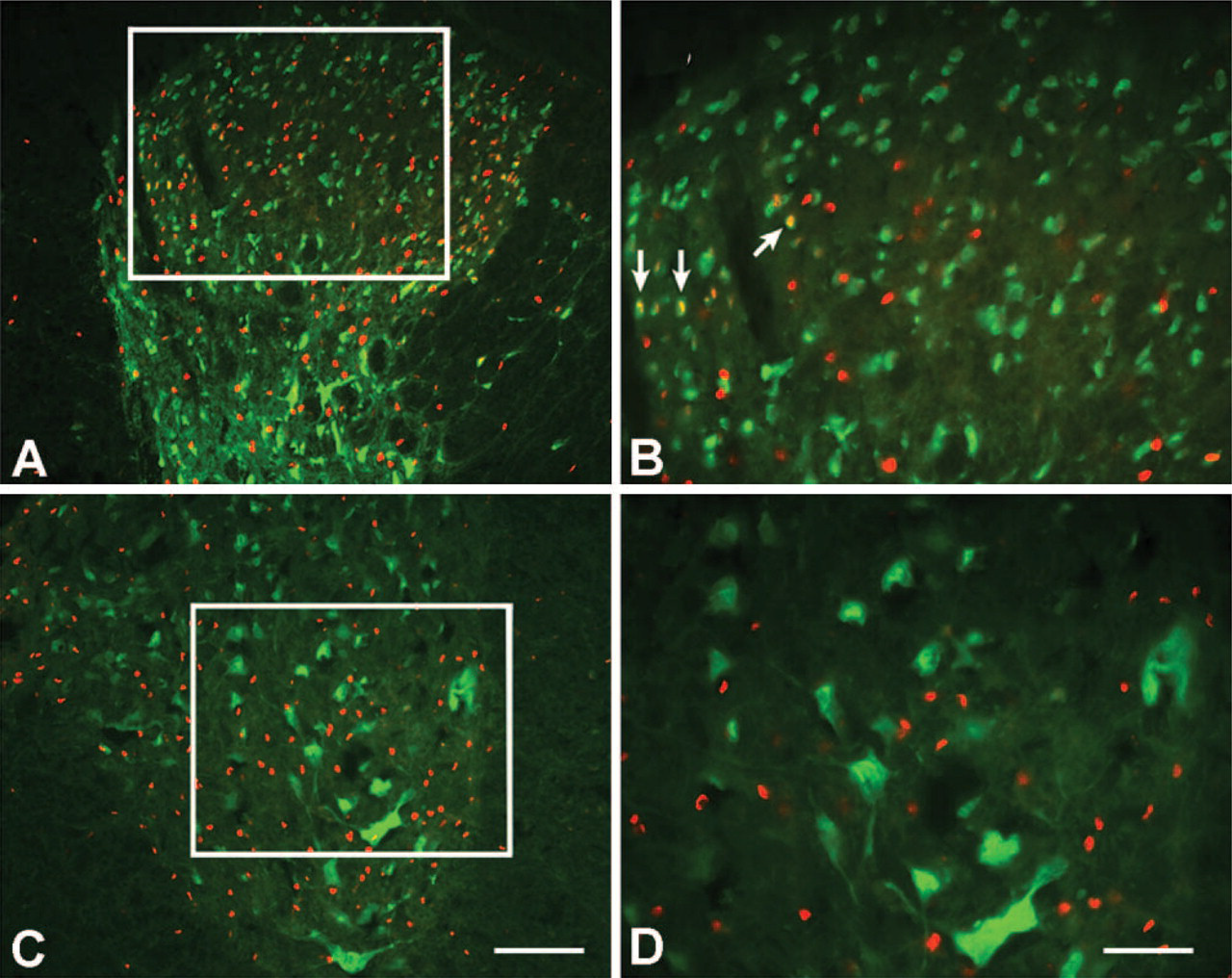
Colocalization (yellow) of activated caspase-3 (red) and NeuN (green) following spinal cord contusion. Antibody colocalization in cells of the dorsal horn at 4 hr postcontusion is illustrated in
Colocalization (yellow) of activated caspase-3 (red) and OX42 (green) following spinal cord contusion. Activated caspase-3 was generally not colocalized with OX42-positive cells with a rounded morphology (not shown) and was rarely colocalized to OX42-positive cells with a ramified morphology later than 4 hr postcontusion. Antibody immunoreactivity of cells in the ventral funiculus 1 mm caudal to the lesion epicenter at 4 hr postcontusion is illustrated in
A few NeuN-positive neurons of the dorsal horn were also immunoreactive for activated caspase-3, primarily at 4 hr and 1 day postcontusion. Activated caspase-3 and NeuN were primarily colocalized to the small neurons of the dorsal horn. In contrast, NeuN-positive motoneurons of the ventral horn were not immunoreactive for activated caspase-3 at any time-point examined. Although previous reports have suggested that motoneurons of the adult spinal cord undergo apoptosis post-SCI (Yong et al. 1998), which may be mediated by caspase-3 (Citron et al. 2000), experimental differences in injury severity or in the time-points examined may explain the different outcomes.
We found that only a few OX42-positive cells appeared to be immunoreactive for activated caspase-3. When present, activated caspase-3 was most prevalent at 4 hr postinjury in OX42-positive cells that had a ramified morphology and shortened processes, the morphological characteristics of partially activated microglia/macrophages (Popovich et al. 1997; Shuman et al. 1997; Carlson et al. 1998). Our findings correspond with previous reports on spinal cords that received less severe contusions in which macrophages with apoptotic bodies were occasionally noted (Liu et al. 1997), and with previous descriptions of spinal cords that received more severe contusions where microglia/macrophage apoptosis was widespread (Shuman et al. 1997; Yong et al. 1998). Because macrophages can recognize and engulf cells undergoing apoptosis (Duvall et al. 1985; Savill et al. 1993), colocalization of OX42 and activated caspase-3 may indicate macrophages that devoured cells in which caspase-3 was previously activated and not necessarily indicate that the OX42-positive cells themselves expressed the activated form of this cell death protein (Shuman et al. 1997).
Although there is evidence of astrocyte cell death following SCI (Grossman et al. 2001), and subpopulations of astrocytes in dorsal horn of the uninjured spinal cord appear to express activated caspase-3 (Noyan-Ashraf et al. 2005), we have not found conclusive evidence of activated caspase-3 in astrocytes following mild to moderate SCI (Nottingham S and Springer J, unpublished data; however, see Yong et al. 1998). This may be due in part to the staining pattern of the astrocyte marker glial fibrillary acidic protein (GFAP). It is difficult to differentiate GFAP-positive cell bodies containing activated caspase-3 from GFAP-positive astrocytic processes encircling a caspase-3–positive cell (Nottingham S and Springer J, unpublished data). Although confocal microscopy might be more revealing, labeling of GFAP and activated caspase-3 was not pursued in the present study due to the extremely rare occurrence of potentially double-labeled cells. However, this observation does raise an important point with respect to the role of reactive astrocytes in formation of the glial scar, which is known to limit the potential for functional regeneration or sprouting. Specifically, the relative absence of caspase-3 colocalization in GFAP-positive cells following SCI might suggest that reactive astrocytes may be relatively more resistant to cell death signals in the injured spinal cord. As a consequence, a lack of apoptotic signaling may contribute to promote formation of the growth-inhibiting glial scar.
Cells that contained activated caspase-3 were present in the spinal cords of rats that received sham surgery and may reflect cellular responses to nonspecific trauma from the laminectomy procedure (e.g., hypoxia, edema). It is important to stress that the temporal distribution of activated caspase-3 within the spinal cords of sham controls was vastly different from the distribution of activated caspase-3 within the injured spinal cords. Following sham surgery, activated caspase-3 was observed in cells of the gray and white matter at all post-surgical time-points examined. In contrast, cells that contained activated caspase-3 in the injured spinal cord dramatically decreased and was sparse on post-operative days 2 and 4 in all subjects that received a spinal cord contusion. These data suggest that the cellular mechanisms activated in response to a “mild stressor” (sham surgery) or to a rapid traumatic impact to the spinal cord may differentially regulate caspase-3 activation.
Overall, the results of this experiment indicate that during acute times following SCI, caspase-3 activation occurs primarily in oligodendroglia in areas of relatively intact white matter. Some small neurons of the dorsal horn were immunopositive for activated caspase-3 postinjury, whereas neurons of the ventral horn were not immunopositive for activated caspase-3 under the conditions of the present experiment. These findings suggest that caspase-3–mediated apoptosis is differentially regulated in neurons, which may reveal neuronal differences in the time-course of caspase-3–mediated cell death or in the biochemical mediators of apoptosis. Finally, activated caspase-3 was rarely localized to OX42-positive microglia/macrophages and was most abundant at 4 hr postinjury. Caspase activation can alter cell structure (Kothakota et al. 1997; Rudel and Bokoch 1997), so the early localization of activated caspase-3 to OX42-positive cells that appeared to have shortened processes may indicate a role for activated caspase-3 in the morphological transition of microglia/macrophages from a non-reactive to an activated phagocytic phenotype.
In conclusion, the results of this study are the first to report the pattern and cellular localization of activated caspase-3 in the injured spinal cord. These findings are particularly relevant given the fact that not all apoptotic cell death is caspase-3 dependent (Li et al. 2001; van Loo et al. 2001; Cande et al. 2002; Arnoult et al. 2003). Relatively little is known about the survival signals important to mature oligodendroglia or to newly differentiating oligodendroglial progenitors of the adult nervous system, and whether those factors are still present or re-expressed following SCI. Therefore, the development of strategies that inhibit factors responsible for initiating and executing oligodendroglial apoptosis postinjury will be critical for promoting oligodendroglial survival and, hopefully, functional recovery.
Footnotes
Acknowledgements
This research was supported by the National Institutes of Heath, Public Health Service (PHS) Grants NS-40015, NS-46380, a grant from the Kentucky Spinal Cord and Head Injury Research Trust, and an endowment from Cardinal Hill Rehabilitation Hospital. M.L.M. was supported by PHS Training Grant AG-00242.
