Abstract
Tissue microarray (TMA) is a powerful, high-throughput technique for in situ investigation of biomarkers in many tissue samples in a paraffin block by immunohistochemistry or fluorescence in situ hybridization (FISH), and has rapidly become the standard in marker studies. One of the difficult steps in the procedure is the sectioning of array blocks and mounting of sections using special slides and/or adhesive-coated tape, which demands specific experience and is time-consuming. We report an arraying method that allows melting of the receiving paraffin block and subsequent sectioning like any ordinary paraffin-embedded tissue block. The major difference from the standard microarray technique is the use of an agarose matrix in the recipient block. The agarose matrix allows melting of the paraffin without disturbing the array, resulting in perfect integration of the tissue cores. The agarose-paraffin TMA blocks limit tissue core loss during cutting, mounting, or immunohistochemical or FISH staining and better maintains the array.
P
The key element of TMA technology is the construction of an array of cylindrical tissue samples that are collected in recipient blocks consisting only of paraffin. It requires specific expertise to cut the resulting paraffin array blocks and mount the array sections with standard mounting procedures, because the tissue elements, which are relatively loosely positioned in the holes of the recipient block, do not easily retain the array configuration. This has led to the use of special slides and adhesive tape to maintain the tissue cores in place (Instrumedics; Hackensack, NJ). Melting would allow total integration of the cores in the receiving block, but this disturbs the array if the cores are not maintained in a stable matrix. An approach for integrating cores in the recipient array block (tempering) requires cycles of heating, pressing the surface of the block with a glass slide, and cooling (Hewitt 2004). We developed agarose-containing recipient blocks in which the agarose serves as matrix to maintain the array architecture during melting of the paraffin to achieve total integration of the array cores into the recipient paraffin block. The approach greatly facilitates sectioning and mounting of microarray sections, which is performed as for any other paraffin block.
Materials and Methods
Agarose-Paraffin Block Preparation
The agarose blocks were prepared and processed through a modified embedding cycle as summarized in Table 1.
Preparation and embedding of agarose blocks

Paraffin and Agarose-Paraffin Microarray Constructions
Paraffin and agarose-paraffin blocks were used as recipient blocks for TMA construction. A manual tissue array instrument (Beecher Instruments, Inc.; Sun Prairie, WI) was used to punch holes in the agarose-paraffin recipient block and to acquire tissue cores from the donor block by thin-walled needles with an inner diameter of 0.6 mm, 1 mm, and 2 mm, respectively, held in an X-Y precision guide. Paraffin blocks containing normal liver tissue were used as donor blocks for this study. Tissue cylinders with a diameter of 0.6 mm, 1 mm, and 2 mm were punched from the donor block and introduced into the recipient blocks with defined array coordinates containing 25 cores of 0.6 mm, 15 cores of 1 mm, and 9 cores of 2 mm. To allow comparison of tissue loss relative to standard paraffin recipient blocks or high-density array blocks, a standard paraffin recipient block and an agarose-paraffin recipient block containing 300 cores of 0.6 mm diameter were constructed as described above. To completely integrate the paraffin of the tissue cores into the recipient blocks, the array blocks were heated on a hot plate at 64C for 10 min and flattened by pressing the surface of a clean glass slide against surface of the block (Figures 1A1 and 1B1). Because the tissue cores in the standard paraffin block did not retain their original positions under these conditions (Figure 1E), these blocks were instead heated on a hot plate at 42C for 10 min and then flattened by pressing the surface with a clean glass slide (Figures 1C1 and 1D1). Before sectioning, both paraffin and agarose-paraffin array blocks were chilled to −10C for 30 min. Four-μm-thick sections were cut using an ordinary microtome and stretched on a water bath at 56C. They were then picked up on Superfrost slides (Microm; Walldorf, Germany) for immunohistochemical staining and FISH.
Immunohistochemistry
To test the adhesion characteristics of the agarose-paraffin microarray sections during antigen retrieval and the histochemical reactions, five monoclonal antibodies requiring different pretreatment and incubation conditions were selected for immunohistochemical staining (Table 2). Deparaffinized sections were treated with 0.3% hydrogen peroxide in methanol for 15 min at room temperature to block endogenous peroxidase activity. The procedures of antigen retrieval used in this study are listed in Table 2. Pronase digestion (DAKO; Carpinteria, CA) was applied at room temperature for 15 min for antigen retrieval for the antiamyloid antibody. Microwave treatment with different buffers and different oven settings were used for the other antibodies. In brief, after preheating 1 liter of 0.01 M sodium citrate buffer (pH 6.0) or 0.1 mM EDTA buffer (pH 7.5) until boiling in a microwave oven, the sections were put into the boiling buffer and heated for another 15 to 40 min at 200 W. The slides were then chilled in cold running water for 10 min. After rinsing with PBS, the background reactivity was blocked with 10% normal goat serum for 30 min at room temperature. Sections were incubated with primary antibody for 1 hr or overnight. After rinsing with PBS, the slides were incubated with the DAKO EnVision+ system (mouse) for 30 min at room temperature. The peroxidase was revealed by immersion in DAB (DAKO), and the sections were counterstained with hematoxyline and mounted for microscopic examination.
FISH
To test the suitability of these tissue arrays for in situ hybridization, we performed FISH analysis using the PathVysion HER-2 Kit (Vysis; Downer's Grove, IL) according to the manufacturer's instructions with the following minor modifications of the pretreatment. Briefly, after drying the sections overnight at 55C and deparaffinization and rehydration in a graded series of ethanol, sections were incubated in 0.2 M HCl at room temperature for 20 min, followed by a rinse in distilled water and two rinses in 2X SSC (1X SSC is 0.15 M NaCl and 0.015 M sodium citrate) for 3 min each. The sections were then incubated in the pretreatment reagent provided in the kit at 80C for 3 min, again followed by a rinse in distilled water and two rinses in 2X SSC. Subsequent digestion was performed using the kit's protease I at a concentration of 0.05% at 37C for 100 min. After two rinses in 2X SSC at room temperature for 5 min each, the sections were dehydrated in a graded series of ethanol and air dried. Subsequent hybridization was performed according to the manufacturer's instructions.
Results
Paraffin Microarray and Agarose-Paraffin Microarray Constructions
In making an agarose-paraffin recipient block, a prolonged sequence of dehydration, clearing, and paraffin embedding was required for optimal consistency of the matrix. With slow dehydration starting from 30% ethanol and prolonged clearing and paraffin embedding cycles, the agarose blocks retained their original shape and provided an ideal matrix that did not interfere with standard microtomy. After construction of the array, melting of the paraffin at 64C for 10 min followed by cooling at room temperature resulted in perfect integration of the tissue cores without any disturbance of the array regardless of the number of tissue cores in the array block (Figures 1A1 and 1B1).
Tissue cores in the paraffin microarray block were seriously disturbed under these conditions (Figure 1E). In contrast, when the surface of the paraffin array blocks was heated on a hot plate at 42C for 10 min and then flattened, the tissue cores remained aligned (Figures 1C1 and 1D1).
Sectioning
After cooling at −10C for 30 min, both paraffin and agarose-paraffin array blocks could be sectioned without special adhesive tape as paraffin-embedded tissue blocks using standard microtomy. Sections of 4 μm could be cut without any difficulty, regardless of the diameter or number of tissue cores. The time required for cutting and mounting the sections did not differ between the two types of array blocks. However, stretching of agarose-paraffin ribbons of tissue sections on a water bath was significantly better than that of paraffin blocks. Because core loss tends to be a problem in TMA studies, we compared this between low- and high-density TMAs for both block types, conventional paraffin and agarose-paraffin. Repeated cycles (up to 10 sections) of cutting and mounting of agarose-paraffin blocks showed no loss on low-density TMAs (Figure 1B2; cores varying in size from 0.6 to 2 mm). In contrast, a maximum of 3 cores were lost on sections of a high-density agarose-paraffin TMA (Figure 1A2). The same procedure on conventional paraffin array blocks showed no loss of cores on low-density TMAs (Figure 1C2), but on the average, loss of 13 cores on sections of high-density paraffin TMAs. Some cores tended to show loss of part of the tissue (clearly visible in Figures 1D2 and 1D3 in the 2-mm cores). Furthermore, cores in conventional high-density paraffin TMAs tended to partially detach and fold over. Routine hematoxylin-eosin (H&E) staining did not result in the loss of additional cores in either array. The array architecture of the sections in agarose-paraffin blocks was perfectly maintained in terms of size and shape, but that in the paraffin array blocks was slightly disturbed, and because of the stretching without a matrix, the surface was increased by 40% (compare, for example, Figures 1A2 and 1C2).
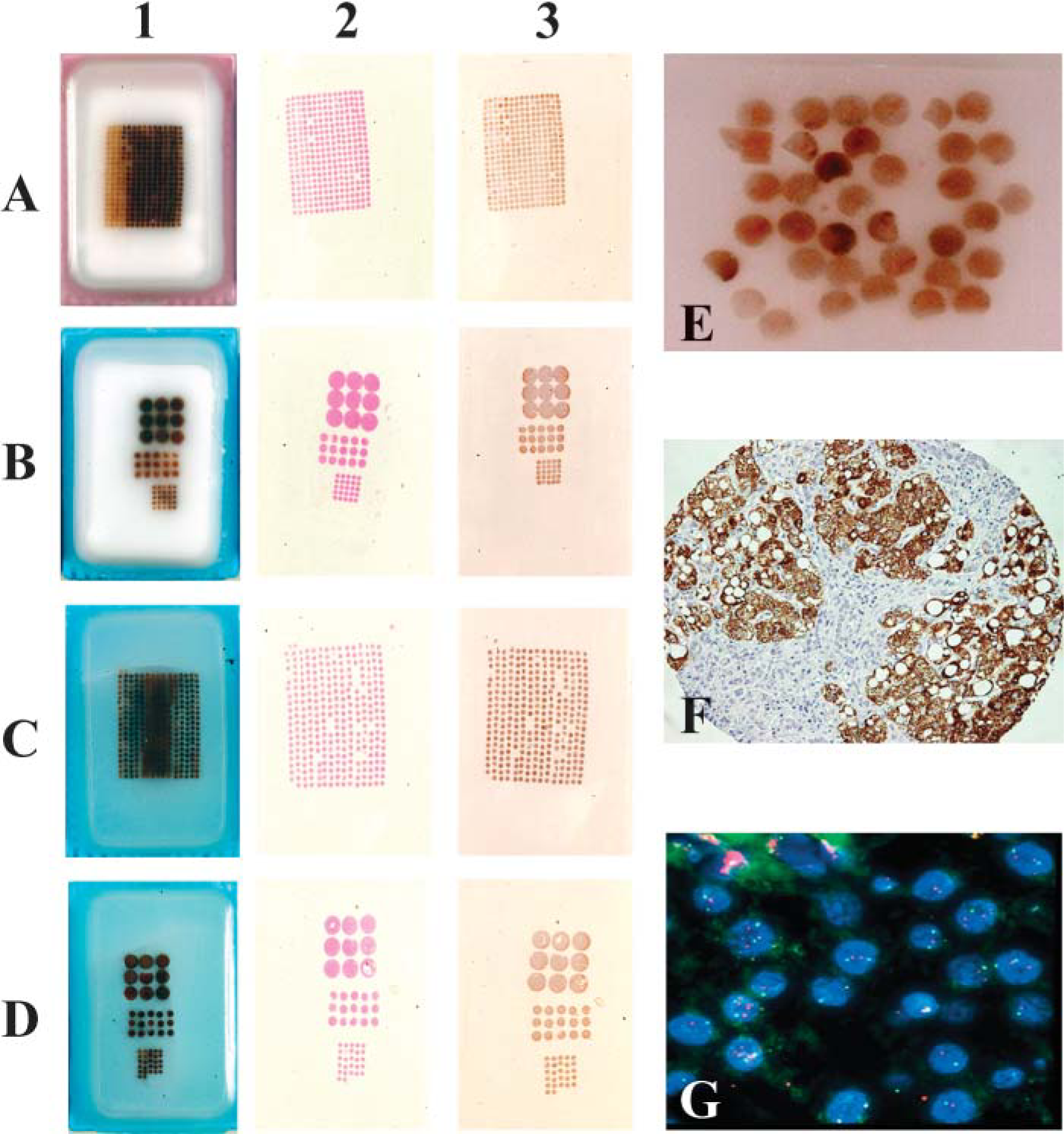
Tissue array construction and immunohistochemistry and fluorescence in situ hybridization (FISH) of tissue array sections.
Primary antibodies, antigen retrievals, and incubation times for primary antibodies Antibody
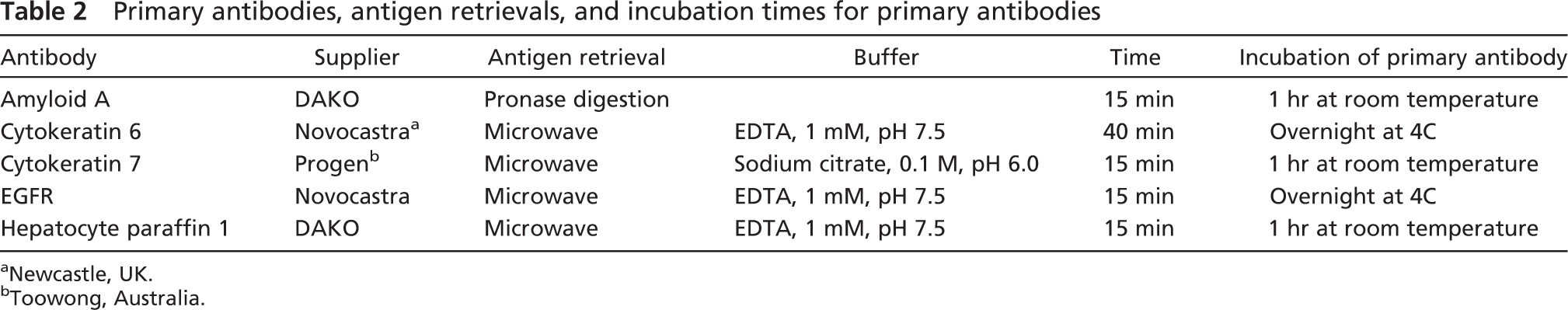
Newcastle, UK.
Toowong, Australia.
Immunohistochemistry
All incubations resulted in perfect labeling, similar to that of ordinary tissue sections. Relative to unstained or H&E-stained sections there was no additional core loss on the sections from either array type (Figures 1A3-1D3). The hepatocyte paraffin 1 immunostaining is shown as a representative example (Figure 1F).
FISH
Because FISH requires a more elaborate and potentially tissue-disintegrating pretreatment, we also subjected the sections to FISH analysis. Sections obtained from both array block types did not show any additional tissue core loss. The quality of the FISH labeling was as good as that for ordinary paraffin sections (Figure 1G).
Discussion
TMAs provide tremendous advantages in the analysis of molecular markers at the DNA, RNA, or protein level, compared with standard methods using whole tissue sections. They allow for screening of a great number of tumors on a single microscopy slide, thereby reducing reagent consumption and laboratory work. TMAs are an ideal tool for large-scale analyses, quality control, and standardization, and allow conservation of defined tissue collections. The major restrictions on more widely and easily producing TMAs are the cost of a fully automated arrayer, the complexity of mounting array sections, and the loss of tissue cores during the staining procedure. Inexpensive manual or automating arrayers were recently reported and found to make reproducible arrays (Matysiak et al. 2003; Hewitt 2004; Pan et al. 2004). Tempering of the paraffin block has to some extent facilitated section mounting and limited core loss (Hewitt 2004). This involves heating of the block three or four times for 1-8 hr at 35C in an oven and then cooling on a cooling plate (or in a−4C freezer) for 15 min. Howat et al. (2005) reported a resin TMA in which a glycol methacrylate resin array block was cut with a rotary microtome. Agarose was used as an intermediary recipient block to produce the resin array block through a long and complicated procedure. Following Howat's approach, we developed an agarose-paraffin recipient TMA block. The significant advantage of using agarose-paraffin as a recipient block is that the tissue cores are entirely integrated into the recipient block through melting of the paraffin, without disturbing the array. Agarose-paraffin blocks can be cut as easily as a usual paraffin block by using a standard microtomy with significantly less core loss relative to conventional paraffin TMA blocks after tempering. Most importantly, the tissue sections obtained stand up under the harsh pretreatments and lengthy incubations required for immunohistochemistry and FISH without significant loss of tissue, regardless of core size and core number. An additional advantage of the agarose-paraffin technique is perfect retention of the array alignment and size, issues important for automated reading of staining results. The quality of the immunohistochemical staining and FISH was comparable to that of whole tissue sections.
We conclude that agarose-paraffin blocks greatly facilitate the handling of TMA sections and merit becoming standard practice for this technique.
