Abstract
rck/p54, a DEAD-box RNA helicase, is closely associated with the basic modification of RNA molecules in the process of mRNA transport, RNA decay, and translation initiation. In the current study, Western blot analysis revealed that rck/p54 protein was ubiquitously expressed in mouse tissues. Interestingly, three different-sized rck/p54 proteins were detected by antibodies against mouse rck/p54, and these products were differentially expressed in the tissues. An immunohistochemical study revealed that rck/p54 was strongly expressed in basal cells of the crypt in the gastrointestinal tract and in neuronal bodies of the cerebral cortex, and was localized in epithelial cells of the convoluted tubules of the kidneys, suggesting that the heterogeneous rck/p54 may play pivotal roles in cells committed to become specialized in these tissues.
DEAD-
The RCK gene was cloned through the study of t(11;14)(q23;q32) chromosome translocation in the human B-cell lymphoma cell line RC-K8 (Akao et al. 1992), and its transcripts were found to be expressed ubiquitously in mouse and human tissues (Akao et al. 1995). The protein product (rck/p54) of this gene exhibits ATP-dependent RNA unwinding activity (Akao et al. 2003) and participates in mRNA masking (Minshall et al. 2001) and 5î-3î RNA decay (Cougot et al. 2004). These findings led us to speculate that rck/p54 plays important roles in various tissues. In the current study, we examined rck/p54 expression in mouse tissues by using anti-rck/p54 antibodies raised in our laboratory. Our results suggest that heterogeneous rck/p54 proteins were expressed in mouse tissues, which expression implies the specialized function of the cells belonging to each tissue.
C57BL/6 strain mice maintained in the animal facility of Gifu International Institute of Biotechnology were used in the present study, and all experiments were performed in conformation with animal experimental guidelines of Gifu International Institute of Biotechnology. Sacrificed mice were immediately immersed in liquid nitrogen for RT-PCR and Western blotting, or frozen in TISSU MOUNT (Shiraimatsukiki; Osaka, Japan) for immunohistochemistry.
Semiquantitative RT-PCR was performed as described previously (Hashimoto et al. 2001). Briefly, total RNA was isolated by using an RNAqueous-4PCR kit (Ambion; Austin, TX) according to the manufacturer's instructions. RNA samples were reverse-transcribed by using Super Script II RNase H-reverse transcriptase (Invitrogen; Carlsbad, CA) and oligo(dT) primer (Invitrogen). Prepared cDNA samples were purified by use of a PCR Purification kit (Qiagen; Hilden, Germany) and used for PCR. The PCR was performed according to the manufacturer's instructions. β-actin was used as an internal control. The primers were designed for parts of the cDNA sequence corresponding to the amino acid sequences detected by antibodies used in this study. Primers for mouse RCK were as follows: musRCK-S1 forward, 5î-ATGAGCACGG-CCAGAACAGAGAAA; and musRCK-R1 reverse, 5î-CGGTTTCTCGTCTTCTGCA-3î, musRCK-R2 reverse 5î-CTGACCTGTAGAGCAAGTTC-3î, and musRCK-R3 reverse 5î-CGCCACTATTAAACCTGGTG-3î. The PCR products were evaluated by agarose electrophoresis.
Mouse B-cell line NP3 and human cervical cancer cell line HeLa were grown in RPMI-1640 medium supplemented with 10% (v/v) heat-inactivated FBS (Sigma; St. Louis, MO) and 2 mM
To knockdown of RCK in HeLa cells by RNAi, HeLa cells were seeded in six-well plates at a concentration of 1-2 × 105/well (30-40% confluence) the day before the transfection. The potential siRNA target sequences were found on RCK by use of an siRNA Designer Computer System (Invitrogen). An siRNA for targeting RCK mRNAs was obtained from Invitrogen. The target sequence for this siRNA was 5î-AACAGATGAACCAGCTGAAAA-3î. The target sequence for eIF4E siRNA was 5î-CCCTTTCGCAGAAATGGCAACGAAT-3î. The siRNAs were used to transfect the cells (80 nM in a well) by using cationic liposomes TransIT-TKO (Mirus Bio Co.; Madison, WI) according to the manufacturer's Lipofection protocol. The transfection efficiency was evaluated by transfection of HeLa cells with a duplex siRNA-FITC; and we examined expression of interferon-induced OAS-1 mRNA by RT-PCR using 5î-CGATGTGCTGCCTGCCTTTGATGC-3î (sense) and 5î-GTCTCCACCACCCAAGTTTCCTGT-3î (antisense) as primers (data not shown). A nonspecific control duplex (NS, 57% GC content; Dharmacon, Lafayette, CO) was used as a control for nonspecific effects. Evaluation of the effectiveness of RNAi for RCK was made at 36 hr after transfection by Western blot and RT-PCR analyses.
Antibodies for rck/p54 were raised in our laboratory. Rabbit polyclonal antibody against the first N-terminal amino acids numbers 1-10 of mouse rck/p54 (anti-rck/p54N; Matsumoto et al. 2005), which are identical to those of human rck/p54, and that against C-terminal amino-acids, numbers 467-482 (anti-rck/p54CT), were developed in our laboratory (Figure 1A). Recognition of rck/p54 protein by the antibodies was confirmed by performing an ELISA (data not shown) and Western blotting (data not shown). Human monoclonal anti-β-actin antibody was purchased from Sigma.
To examine the rck/p54 protein expression by Western Blot analysis, fresh mouse tissues were homogenized in chilled lysis buffer containing 10 mM Tris-HCl (pH 7.4), 1% NP-40, 0.1% deoxycholic acid, 0.1% SDS, 150 mM NaCl, 1 mM EDTA, and 1% protease inhibitor cocktail (Sigma) and stood for 30 min on ice. After centrifugation at 14,000 rpm for 20 min at 4C, the supernatants were collected as protein samples. Protein contents were measured with a DC Protein assay kit (Biorad; Hercules, CA). One microgram of lysate protein for Western blot of rck/p54 was separated by SDS-PAGE on a 10% polyacrylamide gel and electroblotted onto a PVDF membrane (Du Pont; Boston, MA). After nonspecific binding sites had been blocked by immersion for 1 hr in 5% nonfat milk in PBS containing 0.1% Tween 20, the membrane was incubated overnight at 4C with anti-human rck/p54N or anti-human rck/p54CT antibody. The membranes were then washed three times with PBS containing 0.1% Tween 20, incubated further with HRP-conjugated donkey anti-rabbit Ig antibody (Amersham; Piscataway, NJ) at room temperature, and then washed three times with PBS containing 0.1% Tween 20. The immunoblots were visualized by use of an enhanced chemiluminescence detection kit (New England Biolabs; Beverly, MA).
Immunohistochemistry was performed as described previously (Miyaji et al. 2003). Eight-week-old male mice were used for the immunohistochemical study. Sections of 4-μm thickness made from paraffin-embedded tissues were mounted on poly-
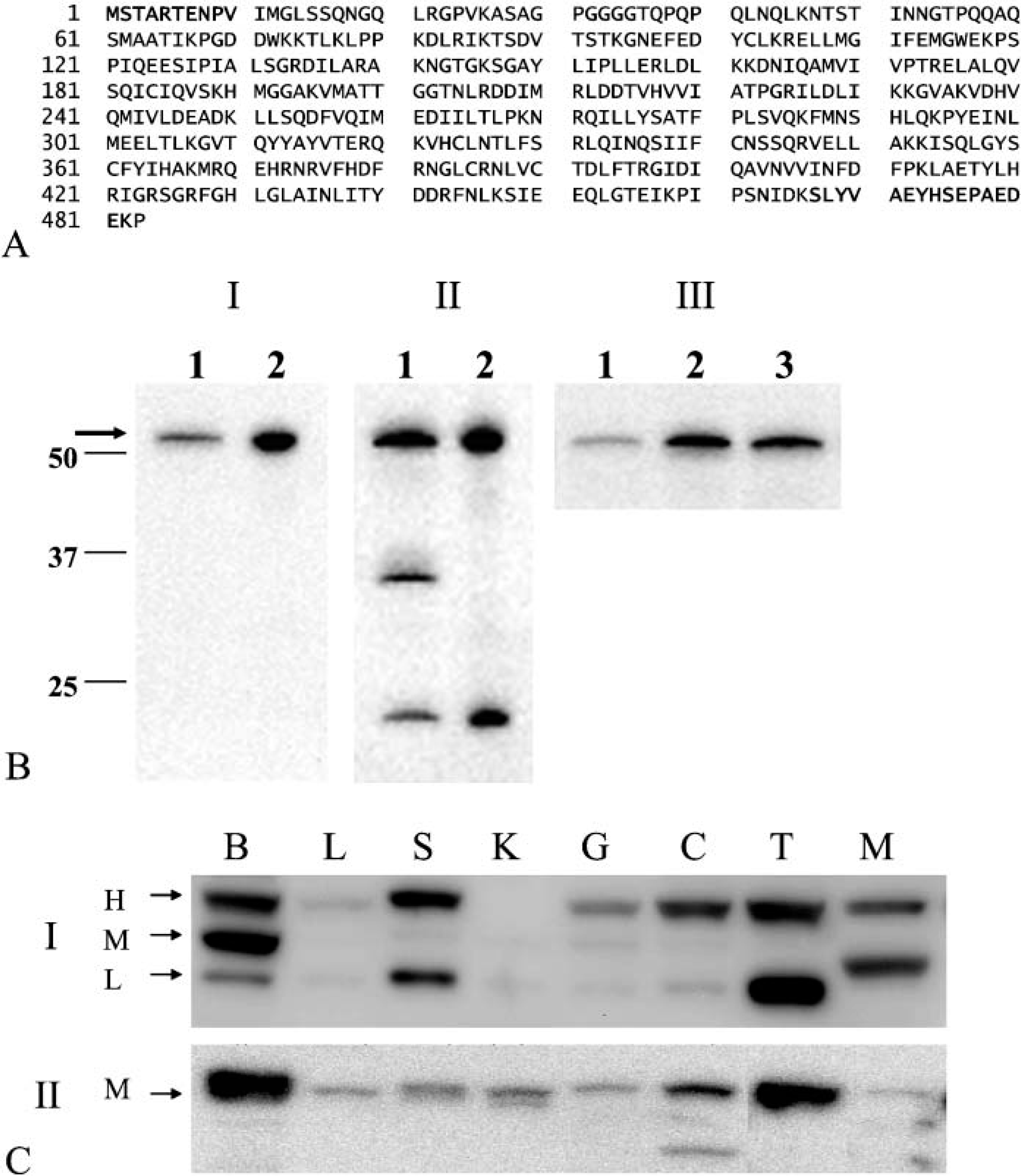
The transcripts of the mouse RCK gene were expressed in all tissues tested, and expression levels were almost the same among tissues tested, as evaluated by semiquantitative RT-PCR (Figure 2; Akao et al. 1995). No splice variant was detected by PCR using the other primer pairs (Figure 2B). The three different-sized PCR products (Figure 2B, Lanes 1-3) were confirmed to be the sequences of RCK cDNA by sequencing. For assessment of the expression of mouse rck/p54 protein, we evaluated the specificity of the two anti-rck/p54 antibodies used in this study by Western blot analysis in an RNAi experiment. The target sequences of the anti-rck/p54N (Figure 1A) were identical to the corresponding part of human rck/p54. First, we examined reactivity of the antibodies toward a mouse B-cell line, NP3, and human cervical cancer cell line HeLa. Both reacted with the same band corresponding to mouse or human rck/p54 (Figures 1B I and II). However, the anti-rck/p54CT reacted with two other molecules of approximately 30 kDa and 20 kDa. An RNAi experiment using siRNA directed against RCK in HeLa cells demonstrated that the level of rck/p54 expression in the siRNA-treated cells was significantly decreased compared with that in NS- or eIF4E-siRNA-treated cells, as evaluated by Western blotting using anti-rck/p54N antibody (Figure 1B III). Thus the specificity of both antibodies against mouse rck/p54 was assessed.
Expression of rck/p54 protein in mouse tissues evaluated by Western blot analysis.
Interestingly, Western blot analysis using the anti-rck/p54N antibody demonstrated three different-sized bands (approximately 54 kDa in size) that differed in their expression patterns among the tissues (Figure 1C I). The liver and kidney expressed a lower amount of rck/p54 protein than the other tissues, whereas mRNA levels were almost the same among the tissues (Figures 1C I and 2A). We designated these three protein products as the H-type (the band for the highest molecular weight), M-type (that for the middle molecular weight), and L-type (that for the lowest molecular weight; Figure 1C I). On the other hand, Western blotting with the anti-rck/p54CT antibody against the last 16C-terminal amino acids of rck/p54 (Figure 1A) detected only the M-type in all tissues (Figure 1C II), which reactivity was confirmed by rehybridization with each antibody on the same filters (data not shown). Referring to the amino acid sequence of mouse rck/p54, an approximately 54 kDa-protein product (ACCESS NO. D50494) was predicted (Figure 1A). Given that the N- and C-terminal sequences with which the respective anti-rck/p54N and anti-rck/p54CT antibody reacted are specific to mouse rck/p54, we consider the M-type product to be identical to the mouse rck/p54 protein reported in NCBI (D50494), which was also judged to be that from the results of RT-PCR (Figure 2A) and sequencing of the PCR products. However, the amount of protein evaluated by Western blot analysis did not necessarily parallel the levels of mRNAs corresponding to the M-type in the tissues (Figures 1C and 2A). Basically, the M-type was present commonly in all tissues tested (Figure 1C), but there was a considerably small amount in the tissues except the brain and testis. The anti-rck/p54CT antibody, which detected the M-type as effectively as did the anti-rck/p54N, detected doublet bands of the M-type in the spleen and kidney samples, which may indicate the existence of a modified M-type rck/p54 in them (Figure 1C II). Because there is another ATG sequence at the position 33 nucleotides downstream of that for the M-type, the lower band of the doublet may be due to translational modification. Surprisingly, expression of the H-type was much better than that of the M-type. As to the H-type, we predict two possible cases: the 5î terminus of the cDNA corresponding to the H-type may be further upstream of that of M-type or the C terminus may be further downstream of the C terminus of the M-type. It is possible that the further upstream ATG may be used for translation of the H-type. On the other hand, the C terminus of the L-type was not identical with that of the M-type, because the anti-rck/p54CT did not detect it.
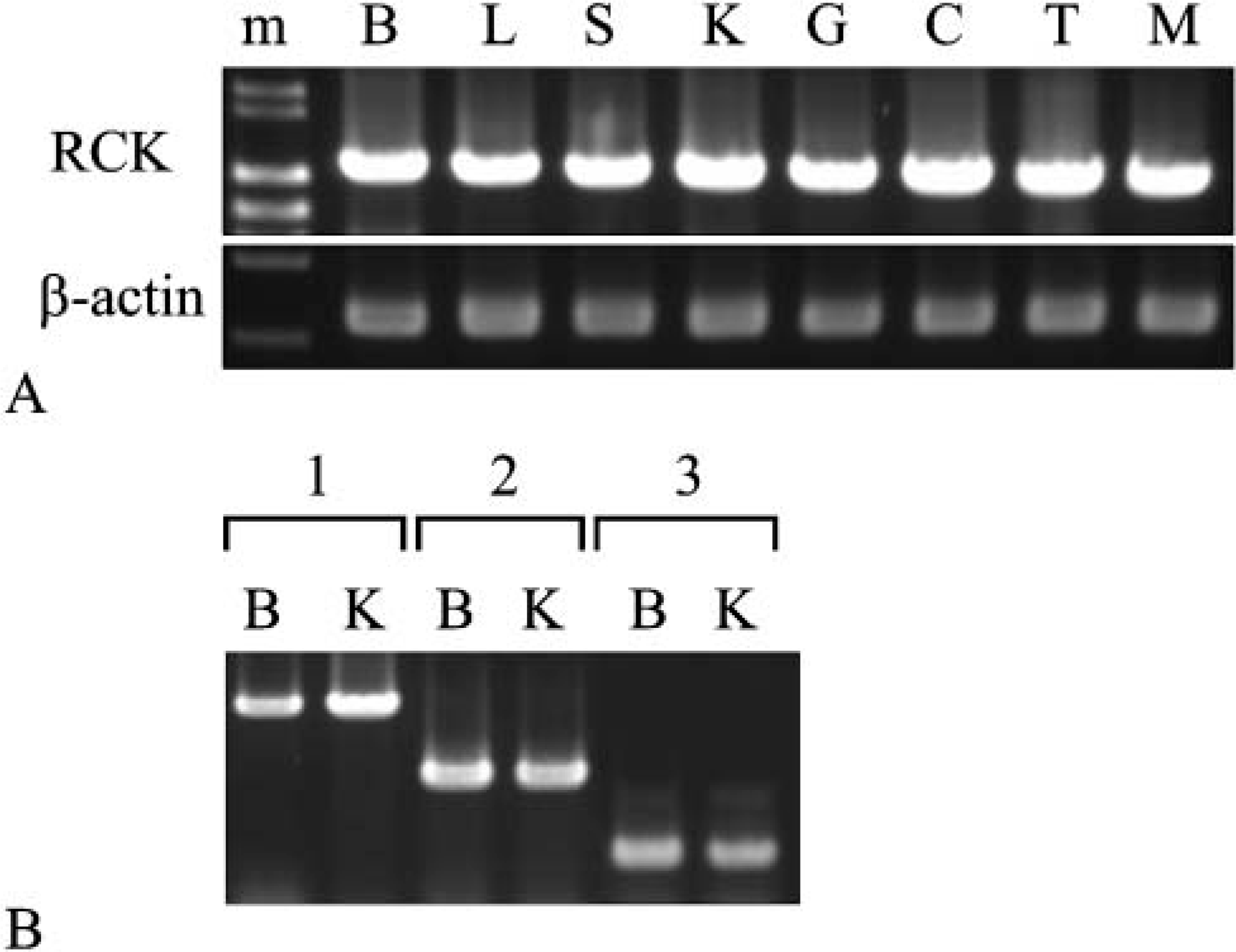
Expression of mouse RCK mRNA in mouse tissues evaluated by semiquantitative RT-PCR analysis. The PCR products of mouse RCK were obtained with three primer pairs. The mouse RCK cDNA region in
According to the tissue distribution of these types, the H-type was missing in kidney; and another band between L- and M-types was detected for skeletal muscle (Figure 1C, Table 1). When the colon sample was examined, the anti-rck/p54CT antibody detected a band indicating a molecular weight lower than that of the L-type in colon (Figure 1C II). Thus these results altogether strongly suggest the presence of heterogeneous rck/p54 proteins and that each protein was differently expressed in the tissues. It is not yet clear from what step in the gene expression these proteins originated.
Immunohistochemistry using the anti-rck/p54N antibody demonstrated specific staining in the cytoplasm (Figure 3; Akao et al. 1995), which was especially evident in sites such as cells in splenic germinal centers and at the bottom of gastric glands (Figure 3, S-r and G-r, respectively), as well as in spermatogenic cells in the testis (data not shown; Matsumoto et al. 2005). Interestingly, rck/p54 was also expressed strongly in postmitotic neuronal cell bodies and also showed positive expression in epithelial cells of the proximal and distal convoluted tubules in the kidneys (Figure 3, B-r and K-r, respectively). The immunoreactivity in basal cells of the gland and in lymphocytes of the germinal center was stronger than that in surface cells and in cells outside of the germinal center, which suggests that rck/p54 may contribute to cell proliferation (Akao et al. 1995; Nakagawa et al. 1999; Hashimoto et al. 2001).
Summary of rck/p54 expression in mouse tissues
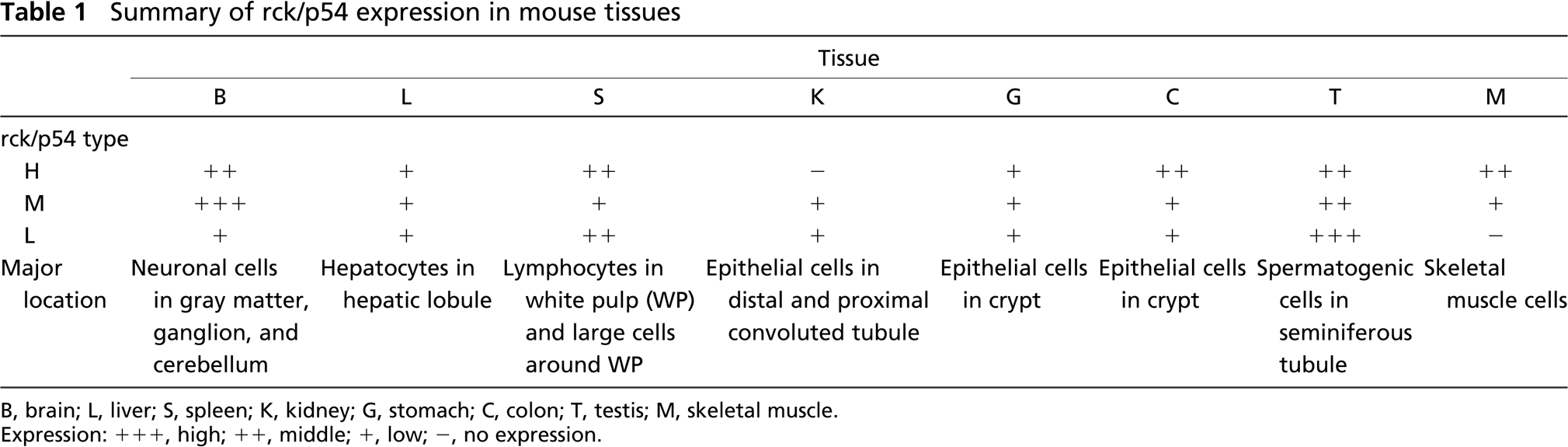
B, brain; L, liver; S, spleen; K, kidney; G, stomach; C, colon; T, testis; M, skeletal muscle.
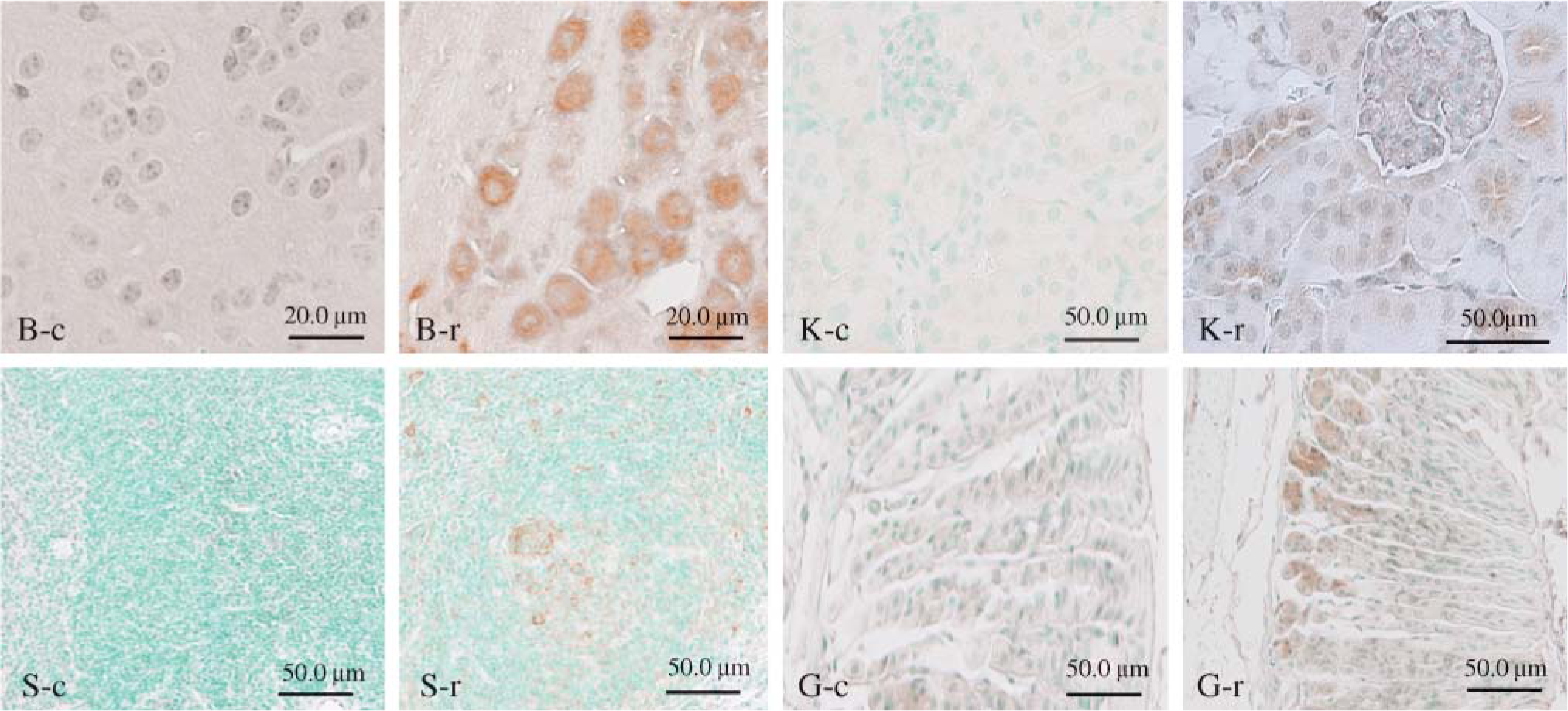
Immunohistochemistry for rck/p54 in various mouse tissues (B, brain; S, spleen; K, kidney; G, stomach). Each tissue is immunoreactive with anti-rck/p54N
Thus presently we demonstrated three major rck/p54 products in mouse tissues, which finding suggests the possibility of posttranscriptional and protein modification. Importantly, the tissue expression of rck/p54 evaluated by Western blot analysis, as was summarized in Table 1, would also include that in the vascular system and immune cells in the connective tissue, and therefore the data do not necessarily indicate expression in the major component cells in the tissues listed in Table 1. Nevertheless, the results on the tissue distribution of rck/p54 protein suggest that this protein serves a variety of functions, one as an RNA structure modifier in each cell lineage committed to differentiation in a given tissue (Table 1). In particular, it should be noted that rck/p54 was present not only in cells that proliferate, but also in postmitotic cells such as neurons and secretory cells in the kidneys. Recently, it was reported that rck/p54 functions as a translational repressor by enhancing decapping, leading to quality control of mRNAs in a cell (Coller and Parker 2005). Further study to determine the roles of rck/p54 in specific cell lineages is obviously needed.
Footnotes
Acknowledgements
Supported by a grant-in-aid for scientific research (to YA, No. 14657061) from the Ministry of Education, Science, Sports, and Culture of Japan.
