Abstract
Breast cancer resistance protein (BCRP/ABCG2) is an ATP-binding cassette transport protein that is expressed in several organs including the liver. Previous studies have shown that ABC transport proteins play an important pathophysiological role in several liver diseases. However, to date, expression pattern and possible role of BCRP in human liver diseases and animal models have not been studied in detail. Here we investigated the expression pattern of BCRP in normal liver, chronic parenchymal and biliary human liver diseases, and parallel in different rat models of liver diseases. Expression was studied by immunohistochemistry and additionally by RT-PCR analysis in Thy-1-positive rat oval cells. Bile ducts, hepatic progenitor cells, reactive bile ductules, and blood vessel endothelium were immunoreactive for BCRP in normal liver and all types of human liver diseases and in rat models. BCRP was expressed by the canalicular membrane of hepatocytes in normal and diseased human liver, but never in rat liver. Remarkably, there was also expression of BCRP at the basolateral pole of human hepatocytes, and this was most pronounced in chronic biliary diseases. In conclusion, BCRP positivity in the progenitor cells/reactive ductules could contribute to the resistance of these cells to cytotoxic agents and xenotoxins. Basolateral hepatocytic expression in chronic biliary diseases may be an adaptive mechanism to pump bile constituents back into the sinusoidal blood. Strong differences between human and rat liver must be taken into account in future studies with animal models.
Keywords
T
In normal human tissues, BCRP is expressed on the apical membrane of trophoblast cells in the placenta; on the epithelium of the small intestine, colon, brain, prostate, and testis; ducts and lobules of the breast; and in the endothelium of veins and capillaries (Doyle et al. 1998; Maliepaard et al. 2001). In rat, Bcrp is expressed in roughly the same set of organs as the human BCRP gene (Tanaka et al. 2005). BCRP is also expressed in a wide variety of stem cells including those of hematopoietic origin and is the molecular determinant of the side population (SP) phenotype (Zhou et al. 2001; Scharenberg et al. 2002). SP cells have been identified by their property to effectively exclude the fluorescent vital dye Hoechst 33342 and form a distinct population, definable by flow cytometry (Goodell et al. 1996). BCRP exhibits transport activity for several exogenous substrates such as cytotoxic drugs, fluorescent dyes, and endogenous substrates such as folic acid, sulfated conjugates of steroids, bile salts, and porphyrins (Jonker et al. 2002; Chen et al. 2003; Suzuki et al. 2003; Janvilisri et al. 2005) By transporting cytotoxic agents from cancer cells, it can render these cells resistant to chemotherapy.
In normal human liver, BCRP is expressed by bile canaliculi, and this has led to the suggestion that BCRP most likely plays a role in the secretion of bile constituents from the hepatocytes into the bile canaliculus (Maliepaard et al. 2001).
According to Tanaka et al. (2005), normal rat liver expresses low levels of Bcrp mRNA determined by a branched signal amplification assay, whereas Shimano et al. (2003) concluded the opposite by Northern blot analysis. The latter publication also found that rat oval cells, which are the counterpart of human hepatic progenitor cells/reactive ductules, express Bcrp mRNA (Shimano et al. 2003; Roskams et al. 2004).
It is well known that ABC transporters such as the multidrug resistance transporter 1 (MDR1), the multidrug resistance-associated transporter 1 (MRP1), and MRP3 play an important role in uptake and secretion of bile constituents and in protection of hepatocytes and progenitor cells/reactive ductules against toxic substances (Borst and Elferink 2002; Ros et al. 2003a,b).
To the best of our knowledge, there are no studies on the protein expression of BCRP in human liver diseases and animal models. This prompted us to study BCRP expression in a wide variety of liver diseases by immunohistochemistry (IHC).
Materials and Methods
Human Liver Specimens
Human liver specimens taken from liver explants or needle liver biopsies taken for diagnostic purposes were used for this study as follows: normal liver (n=5), chronic viral hepatitis (n = 15), chronic biliary liver diseases [primary biliary cirrhosis (PBS), n=12, primary sclerosing cholangitis (PSC), n=2], (non)-alcoholic steatohepatitis [(N)-ASH], (n=13), and acute necrotizing hepatitis (viral, drug induced, auto-immune) (n=7). From each specimen a part was snap frozen in liquid nitrogen-cooled isopentane and stored at −80C, and another part was fixed in 6% formalin or in B5 fixative and embedded in paraffin for routine diagnosis.
For assessment of histopathology and fibrosis, sections were stained with hematoxylin-eosin, PAS diastase, Hall's bilirubin, and Sirius Red (Bancroft and Cook 1984). Degree of fibrosis in (N)-ASH, viral hepatitis, and chronic cholestatic liver diseases was scored according to the Brunt, Metavir, and Scheuer classification, respectively (Scheuer 1967; Desmet et al. 1994; Brunt et al. 1999). The necro-inflammatory activity (grade) in chronic viral hepatitis was scored according to the Metavir system (Bedossa and Poynard 1996) and in (N)-ASH according to Brunt et al. (1999). Pathological data from all patients are summarized in Table 1.
As control, we used five liver biopsies that showed no morphological abnormalities. Two biopsies were taken from normal-appearing liver at a distance from focal nodular hyperplasia and three were morphologically completely normal needle biopsies taken to exclude auto-immune hepatitis, storage disease, and congenital hepatic fibrosis.
Serum bilirubin, alkaline phosphatase, gamma glutamyl transferase, alanine aminotransferase, and aspartate aminotransferase were measured in the blood of all patients. In 16/49 patients, blood levels were measured at the day of biopsy and in 32/49 patients, the number of days between measuring blood levels and biopsy was 17 ± 7.6. No serological data were available from six patients.
Use of these tissues for the study of hepatic transport protein expression was approved by the Local Commission for Medical Ethics and Clinical Studies of the University of Leuven.
Rat Models
In parallel, different rat models were studied to investigate Bcrp protein expression.
Carbon Tetrachloride (CCl4) Model (Hepatitis Model). Eighteen rats were administered CCl4 as previously described (Cassiman et al. 2001; Dubuisson et al. 2002). Six rats were sacrificed after 96 hr, 2 weeks, and 6 weeks, respectively.
Bile Duct Ligation Model (Biliary Model). Six rats underwent ligation and sectioning of the common bile duct and were sacrificed after 4 weeks (Miyazaki et al. 1993).
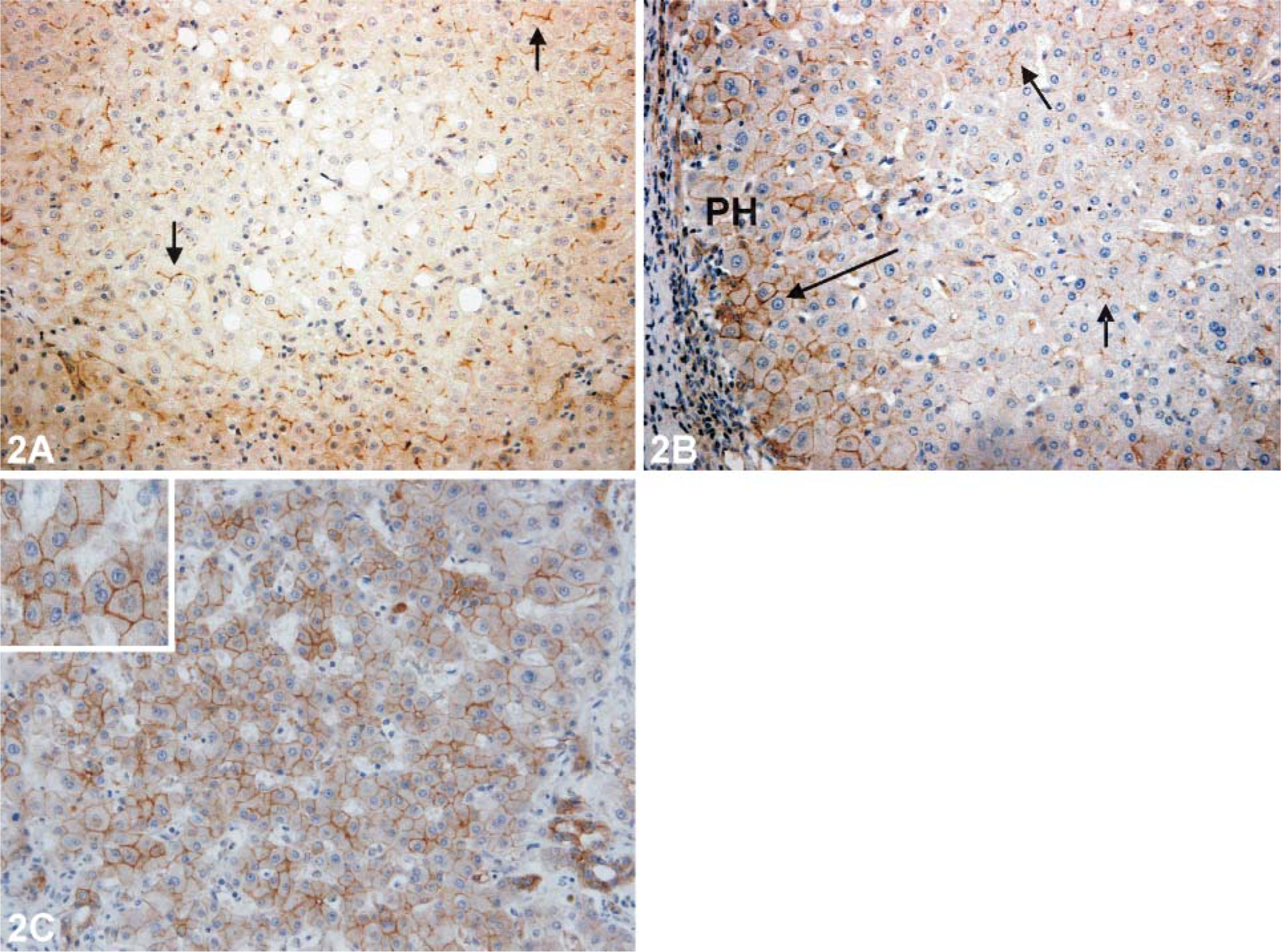
Pathological characteristics of patients with viral hepatitis, cholestatic liver diseases, and (N)-ASH
Degree of fibrosis in viral hepatitis, cholestatic liver diseases, and (N)-ASH was scored according to the Metavir, Scheuer, and Brunt classification of fibrosis/cirrhosis, respectively, wherea score = 0 is no fibrosis and a score = 4 is cirrhosis.
Grading of activity of viral hepatitis and (N)-ASH classified by Metavir and Brunt, respectively. G0, minimal; G1, mild; G2, moderate; G3, severe. NA, not applicable; (N)-ASH, (non)-alcoholic steatohepatitis.
Formalin Injection Rat Model (Biliary Model). Six rats underwent formalin injection into the bile duct as previously described by Houry et al. (1990) with minor modifications. Biliary tree injection with 150 μl of 2% formalin was followed by a second injection of formalin (200 μl, 2%) 2 months later. Six rats were sacrificed 5 months after the first formalin injection.
Choline-deficient-Acetylaminofluorene (CD-AAF) Model (Oval Cell Activation Model). Three rats were sacrificed on day 21 after feeding CD-AAF (Roskams et al. 1998). All animals received care in accordance with the guidelines established by the University of Leuven Ethical Committee.
Liver specimens were snap frozen in liquid nitrogen-cooled isopentane and stored at −80C. Three blocks were fixed in Bouin's solution, B5-fixative, and 6% formalin, respectively, and embedded in paraffin. RNA from Thy-1-positive rat oval cells was used for reverse transcriptase-PCR. These cells were isolated from rats 12 days after 2-acetylaminofluorene (2-AAF)/PHx-treatment as described previously (Petersen et al. 1998).
Western Blotting
Placenta, liver, and kidney were collected from a pregnant rat (E15) within 30 min of surgery, stored in homogenization buffer (0.25 M sucrose, 50 mM Tris-HCl, 5 mM MgCl2, pH 7.4), and immediately frozen in liquid nitrogen. Samples were homogenized, centrifuged (3000 × g, 15 min), and protein concentration in the supernatant determined by the standard Bradford method. Protein samples (50 μg/lane in reducing sample buffer) were loaded onto an 8% polyacrylamide gel (Invitrogen; Merelbeke, Belgium) and separated according to manufacturer's instructions. After blotting onto a PVDF membrane (Bio-Rad Laboratories; Nazareth, Belgium) and blocking of nonspecific binding sites, the membrane was incubated with primary rabbit anti-Bcrp antibody (M-70, sc-25822, dilution 1:1000; Santa Cruz Biotechnology, Santa Cruz, CA) followed by horseradish peroxidase (HRP)-conjugated secondary anti-rabbit antibody. Immunoreactivity was visualized by chemoluminescent detection using the ECL system (Amersham Biosciences; Diegem, Belgium). The membrane was reprobed with mouse anti-β actin (A-5441, dilution 1:5000; Sigma-Aldrich, St Louis, MO) followed by HRP-conjugated anti-mouse antibody to confirm equal protein loading. Experiments were performed in duplicate.
IHC
Four-μm-thick frozen sections of human liver were cut, dried overnight at room temperature, fixed in acetone or 4% paraformaldehyde, and finally washed in PBS immediately before use.
Five-μm-thick sections were made from the human paraffin-embedded biopsies, subsequently deparaffinized in xylene, and rehydrated. Endogenous peroxidase activity was blocked using 0.3% H2O2 in methanol for 20 min. Before staining, paraffin sections were pretreated in citrate buffer (pH 6.0) for 30 min in a hot water bath at 98.5C.
To detect BCRP expression, we used two recently developed monoclonal antibodies: BXP-21 and BXP-34. Development of these antibodies has been described previously (Scheffer et al. 2000; Maliepaard et al. 2001). Specificity of BXP-21 for BCRP has been confirmed in several cell lines by Western blotting (Maliepaard et al. 2001; Diestra et al. 2002; Yoshikawa et al. 2002; Xia et al. 2005) Frozen human sections were incubated for 30 min at room temperature with a 1:10 dilution of BXP-21 and BXP-34, whereas paraffin sections were incubated with a 1:5 dilution of BXP-21 and BXP-34. For the human frozen sections we used a three-step immunoperoxidase procedure: a peroxidase-labeled rabbit anti-mouse IgG (Dako; Glostrup, Denmark) was followed by a peroxidase-labeled swine anti-rabbit IgG (Dako) incubation. Secondary and tertiary antibodies were diluted (1:50 and 1:100, respectively) in PBS, pH 7.2, containing 10% normal human serum. Paraffin slides were incubated with anti-mouse peroxidase-conjugated EnVision antibody (Dako) for 30 min at room temperature.
Five-μm-thick serial sections were made from the paraffin-embedded tissue of four patients. On these sections, IHC was performed with monoclonal antibodies against CK7 (dilution 1:50), CK19 (dilution 1:20), and BCRP (BXP-21; dilution 1:5). The same two-step EnVision method (Dako) as above was applied.
Four-μm-thick frozen sections of rat liver were cut, dried overnight at room temperature, fixed in acetone for 10 min, and finally washed in PBS immediately before use. Frozen rat sections were blocked with normal goat serum (dilution 1:5 in PBS) for 7 min and subsequently incubated with a 1:10 dilution of the rabbit polyclonal anti-Bcrp, M-70 antibody followed by incubation with anti-rabbit peroxidase-conjugated EnVision (Dako). Serial sections were stained with a 1:300 dilution of a mouse monoclonal anti-OV-6 antibody (Dr. S. Sell; Albany Medical College, Albany, NY) followed by incubation with a peroxidase-labeled goat anti-mouse IgG and with a peroxidase-labeled rabbit anti-goat-conjugated IgG. Secondary and tertiary antibodies were obtained from Sigma and diluted (1:100) in PBS containing rat liver powder. Negative controls consisted of omission of the primary antibody.
Semi-quantitative Scoring and Statistical Evaluation
Basolateral BXP-21 distribution pattern was semi-quantitatively assessed as 0 (no basolateral expression), + (periseptal basolateral expression), and ++ (diffuse basolateral expression). Statistical analysis was performed using StatView 5.0.1 (SAS Institute; Cary, NC). To determine the association between the presence of basolateral BXP-21 expression and serological markers, we performed ANOVA followed by Fisher's protected least significant difference test. To determine associations between the presence of basolateral BXP-21 expression and pathological parameters, we performed Fisher's exact test. Bonferroni correction for multiple comparisons was applied by multiplying the p value by the total number of tests; p values <0.05 were considered statistically significant.
Double-fluorescence Immunostaining
Double staining was performed with a sequential fluorescent method on 5-μm-thick human cryostat sections. Slides were fixed in paraformaldehyde 4% for 10 min and finally washed in PBS immediately before use.
Polyclonal carcinoembryonic antigen antibody, dilution 1:300, was incubated at room temperature for 45 min followed by tetramethylrhodamine isothiocyanate (TRITC) or fluorescein isothiocyanate (FITC)-labeled swine anti-rabbit antibodies (Dako). Subsequently, the monoclonal BXP-21 antibody, dilution 1:5, was incubated for 45 min at room temperature followed by FITC- or TRITC-labeled rabbit anti-mouse antibodies (Dako). Antibodies were diluted in PBS. All incubation steps were followed by a wash in three changes of PBS. Sections were mounted with para-phenylene-diamine. Controls consisted of omission of the first step and were consistently negative. No nonspecific labeling could be detected.
Double staining was detected using confocal laser scanning microscopy (410 inverted laser scan microscope; Carl Zeiss Inc., Göttingen, Germany).
RT-PCR
To eliminate genomic DNA contamination, oval cell RNA was treated with RNase-free DNase I (Life Technologies; Merelbeke, Belgium) and reverse transcribed according to manufacturer's protocol. Negative control was no template and no reverse transcription. Positive control was rat brain tissue. cDNA samples were subjected to PCR amplification with specific primers. Cycling parameters were as follows: denaturation at 94C for 1 min, annealing at 55C for 30 sec, and elongation at 72C for 30 sec, for a total of 35 cycles. PCR primers used for amplification were Bcrp (forward: 5′-AAT CAG GGC ATC GAT CTG TCA-3′ and reverse: 5′-CAG GTA GGC AAT TGT GAG GAA GA-3′). Bcrp PCR product was calculated to a length of 93 bp as confirmed on 1.0% agarose electrophoresis stained with ethidium bromide.
Results
Human Specimens
On paraffin-embedded biopsies of normal human liver tissue, we observed BXP-21 positivity on the canalicular surface of hepatocytes, the endothelium of small veins and capillaries, and in nerve bundles. Arteries showed a varying and weak reactivity of BXP-21. The canalicular and blood vessel expression is in agreement with Maliepaard et al. (2001). Epithelium lining the bile ducts showed clear apical BXP-21 positivity. Compared with the BXP-21 antibody, staining of these cell types was much less intense when using the BXP-34 antibody. Staining problems of BXP-34 on paraffin-embedded material have been reported previously (Maliepaard et al. 2001; Diestra et al. 2002). Modifications of the staining protocol did not enhance staining intensities of BXP-34. Thereafter, we used only the BXP-21 antibody. Staining pattern on frozen normal liver biopsies was identical to that on paraffin-embedded biopsies.
In all human liver diseases, the same structures as seen in normal liver were BXP-21 positive. In addition, we observed positivity in hepatic progenitor cells/reactive bile ductules on paraffin-embedded biopsies and on frozen sections fixed with 4% paraformaldehyde for 10 min. Progenitor cells/reactive ductules were negative on acetone-fixed frozen biopsies.
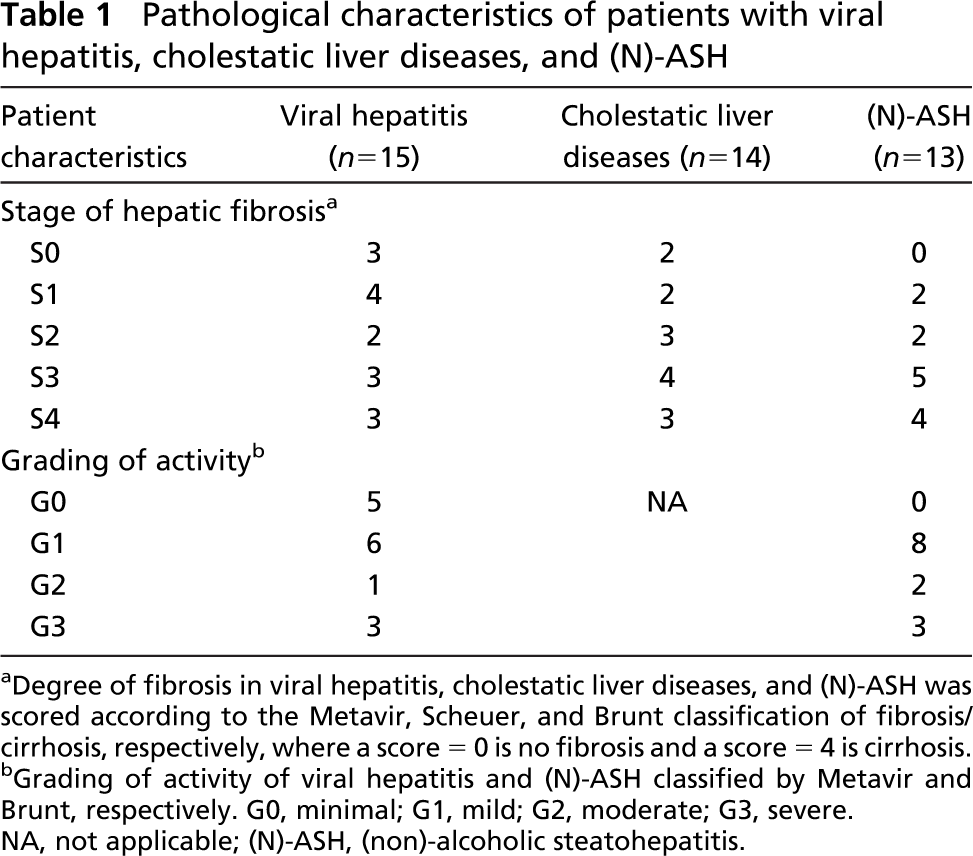
To confirm BXP-21 positivity of progenitor cells/reactive ductules, we performed stainings for BXP-21 and the progenitor cell/ductular markers CK7 and CK19 (Figure 1).
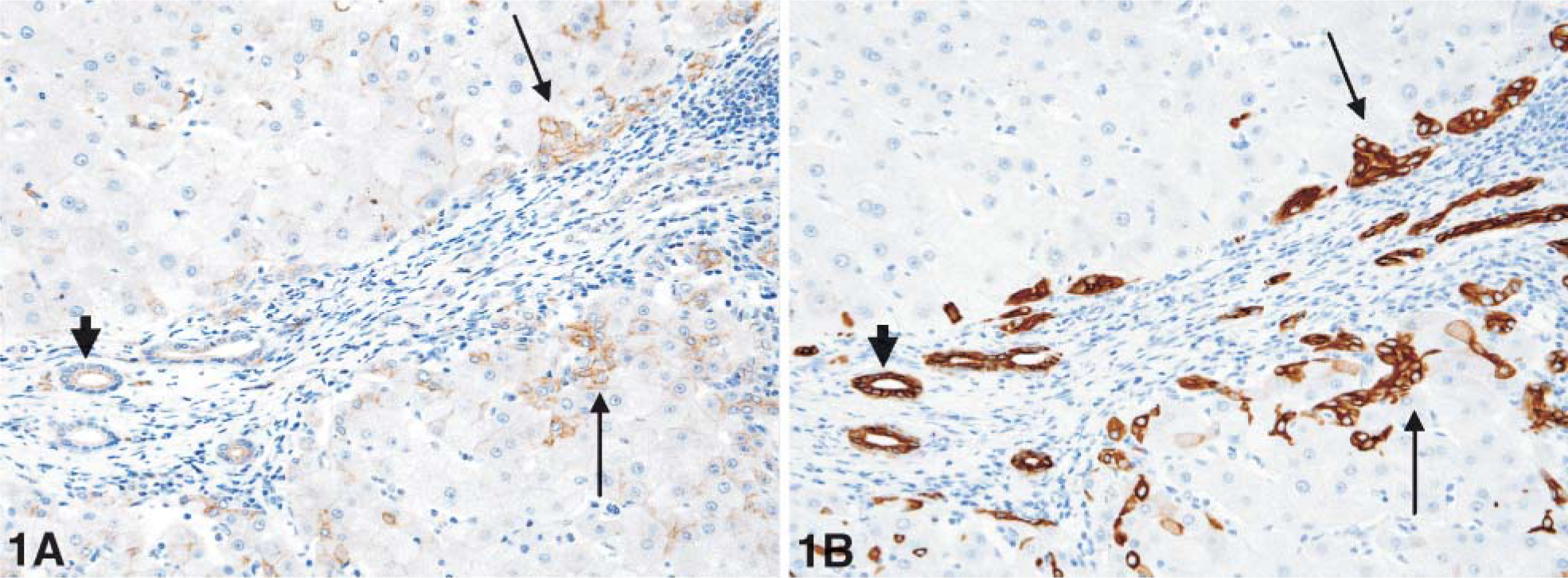
In several cases we also observed expression of BXP-21 at the basolateral pole of hepatocytes. Extent of this basolateral pattern was scored as absent, focal periseptal, or as diffusely present in the parenchyma (Figure 2; Table 2). Basolateral BXP-21 expression was more frequently seen in chronic biliary than in parenchymal (i.e., viral hepatitis, (N)-ASH, and acute necrotizing hepatitis) liver diseases and was neither correlated with fibrosis stage nor with inflammatory activity within the group of chronic viral hepatitis (Table 2). Extent of basolateral BXP-21 expression was not correlated with serological markers.
Basolateral expression of BXP-21 was confirmed by double-fluorescence immunostainings for BXP-21 and pCEA, which is an established canalicular marker (Figure 3) (Lau et al. 2002).
Serial paraffin sections of a patient with chronic hepatitis C stained for BXP-21 (
Immunohistochemical (IHC) staining for BXP-21 on human paraffin sections reflecting the extent of basolateral BXP-21 expression. (
Rat Specimens
To confirm specificity of the polyclonal antibody M-70, we tested it on Western blot of rat placenta, liver, and kidney. The M-70 antibody reacted with a protein band with an apparent molecular mass of 72 kDa in all three samples, confirming the specificity of this antibody (Figure 4). Equal protein loading was demonstrated by β-actin detection on the same blot.
Using this antibody for IHC staining of normal rat liver, we observed positivity of the interlobular bile ducts and the endothelial layer of small veins, arterioles, and capillaries in normal rat liver. In sharp contrast with human liver, rat hepatocytes were consistently negative.
As shown previously (Roskams et al. 1998), oval cells were mainly organized in reactive ductules in the biliary model, whereas in other models they also occurred as singular cells. Oval cells were always positive for M-70. Again, hepatocytes were always negative. To confirm Bcrp positivity of oval cells in rat, we performed serial stainings for M-70 and the oval cell marker OV-6 (Figure 5).
Correlation between basolateral BCRP expression and pathological parameters a
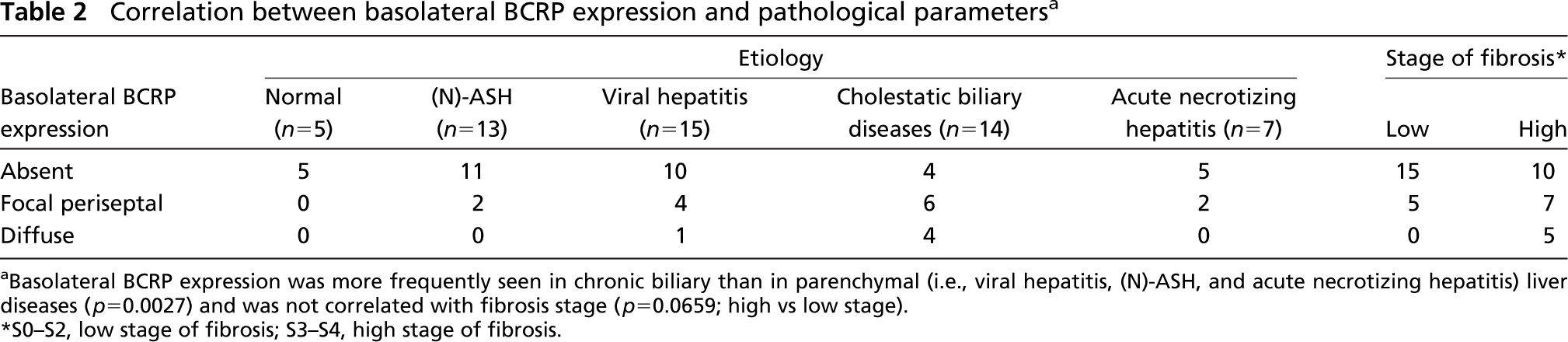
Basolateral BCRP expression was more frequently seen in chronic biliary than in parenchymal (i.e., viral hepatitis, (N)-ASH, and acute necrotizing hepatitis) liver diseases (p=0.0027) and was not correlated with fibrosis stage (p-0.0659; high vs low stage).
S0-S2, low stage of fibrosis; S3-S4, high stage of fibrosis.
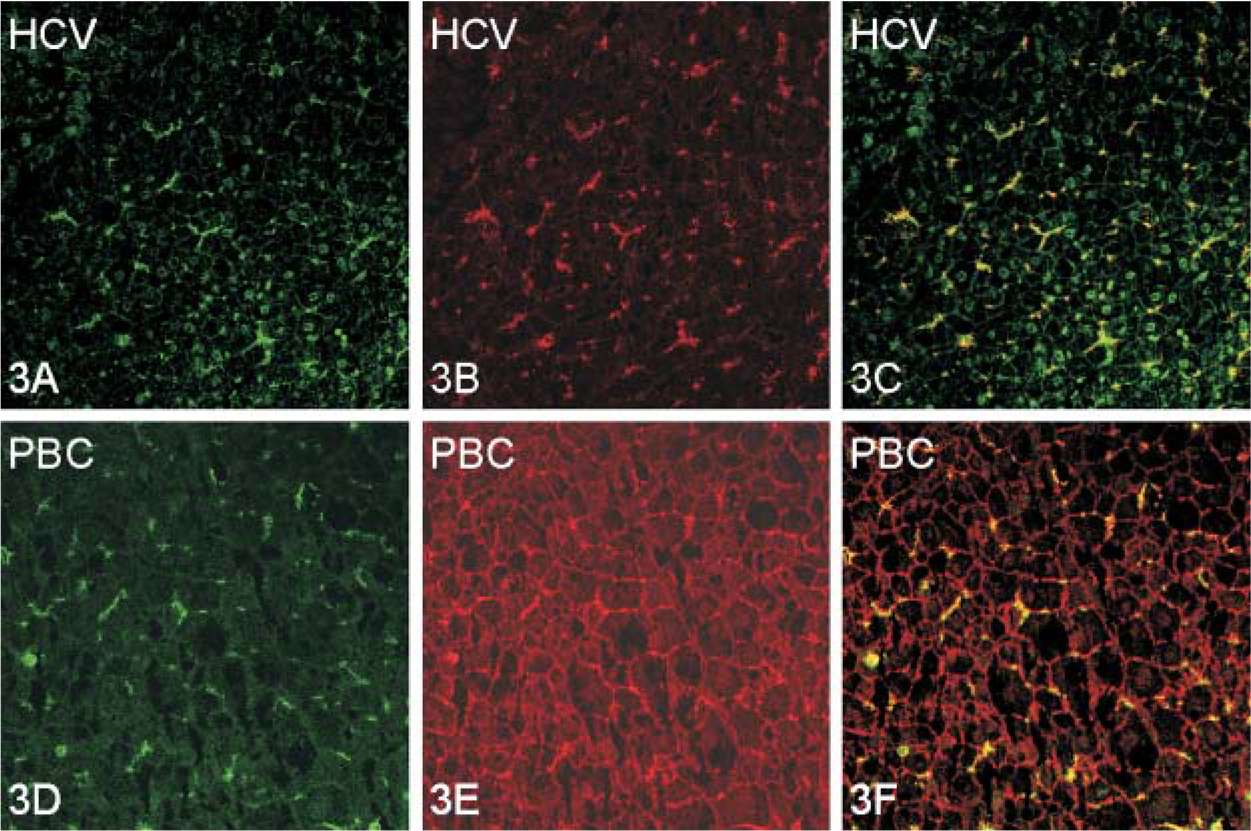
Double immunofluorescence staining for BXP-21 and canalicular marker pCEA in chronic hepatitis C and end-stage primary biliary cirrhosis (PBC). Liver biopsy of patient with chronic hepatitis C: (
This was further supported by the observation that Thy-1-positive oval cells contain Bcrp mRNA (Figure 6).
Discussion
One of the important findings of the current study is that progenitor cells/reactive ductules in human liver strongly express BCRP. Because BCRP is known to export a variety of exogenous and endogenous toxic compounds from the cell (Staud and Pavek 2005), expression of BCRP by progenitor cells/reactive ductules might protect them from cytotoxic agents. Together with the expression of other hepatic transporters (Ros et al. 2003a,b), this could at least partly explain why, in almost all conditions of liver damage and cell loss, progenitor cells/reactive ductules are able to withstand this damage and even expand and contribute to the repair process by differentiating into hepatocytes and/or cholangiocytes (Roskams et al. 2003; Newsome et al. 2004). BCRP can be added to the list of hematopoietic stem cell markers also expressed by hepatic progenitor cells/reactive ductules (Petersen et al. 1998; Grompe 2003).
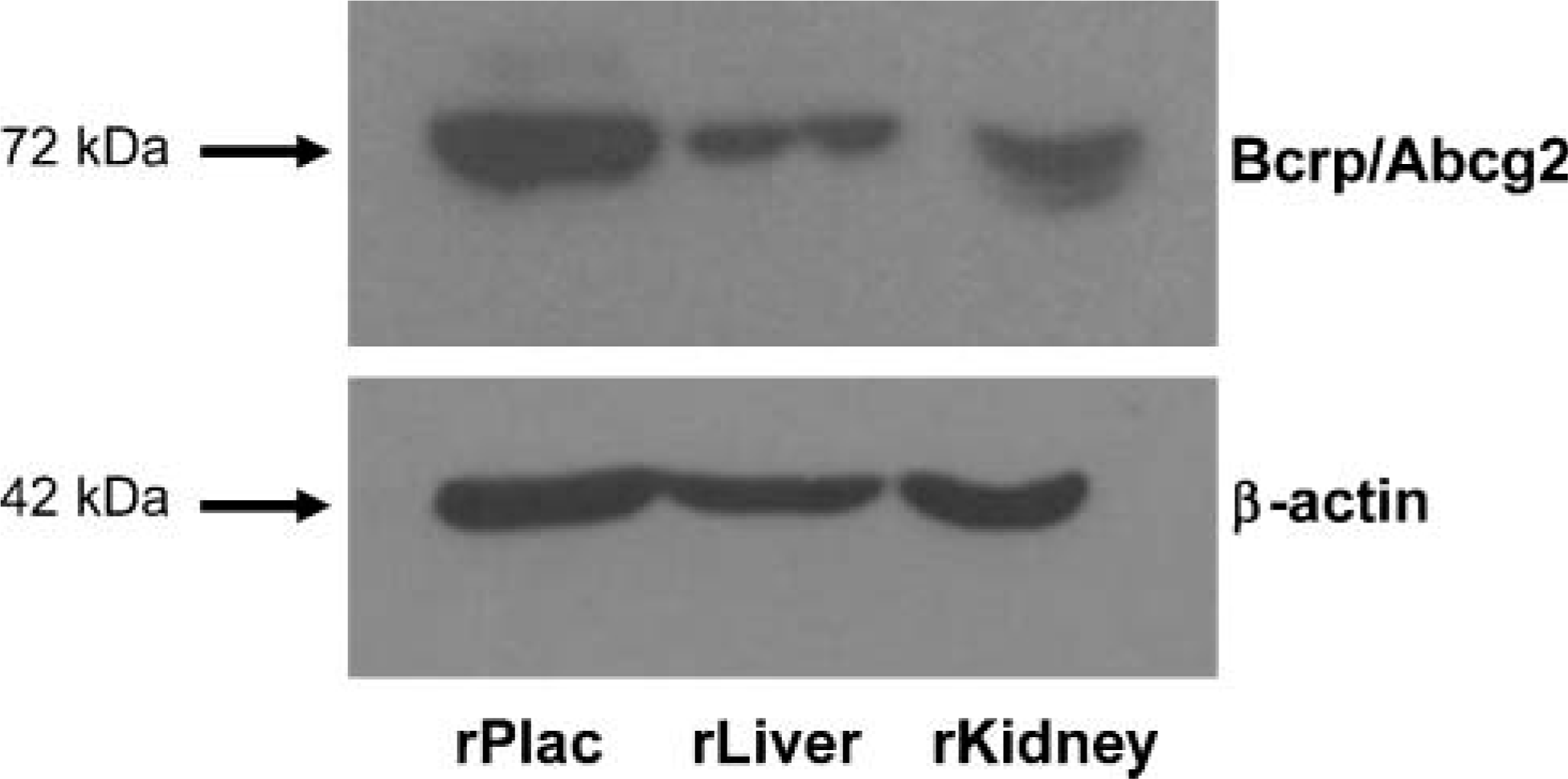
Identification of the specificity of the polyclonal M-70 antibody by Western blotting. By using M-70, a band of ~72 kDa corresponding to the predicted molecular mass of Bcrp was detected in Western blots of rat placenta (rPlac), liver (rLiver), and kidney (rKidney) (upper panel). Equal protein loading was determined by staining for β-actin (lower panel).
Remarkably, expression of BCRP by progenitor cells/reactive ductules could only be demonstrated on biopsies fixed with formalin-containing solutions and was not detectable on acetone-fixed frozen sections. Volk et al. (2005) recently observed a similar phenomenon during their study on the expression of MDR1, another ABC-transporter, in brain. Astrocytes were positive on formalin-fixed tissue but not on acetone-fixed tissue. They suggested that this was due to the fact that the antigenic epitopes might sterically be encumbered in different cell types and thus differ in their accessibility by the antibody or that the studied protein polymerizes into various complexes (di-, tetra-, hexamers) in different cell types, which variably react after acetone fixation with the antibodies.
Expression of Bcrp mRNA in Thy-1-positive rat oval cells isolated from 2-AAF/PHx-treated rats is in line with the recent finding of Shimano et al. (2003) that oval cells in rats that underwent the 2-AAF/PHx protocol are positive when in situ hybridization for Bcrp was performed. In the present study we showed that oval cells also express Bcrp at the protein level by using the M-70 antibody. Specificity of the antibody has never been investigated. Here we show that the antibody was specific by Western blotting. Shimano et al. (2003) also found that the non-parenchymal cell fraction isolated from these rats contained a significant number of cells that excluded the Hoechst 33342 dye, which led them to conclude that oval cells have a SP phenotype. However, non-parenchymal cell fractions also contain endothelial cells of capillaries coproliferating with oval cells (Grant and Billing 1977; Freudenberg et al. 1989), and because we found that these endothelial cells are also BCRP positive, SP cells in the isolate may not only correspond to oval cells but also to endothelial cells. It has been reported that CD45-negative SP cells isolated from mice can differentiate into hepatocytes and bile duct cells in vivo (Wulf et al. 2003), and their counterpart from human liver appears to be capable of differentiating into hepatocyte-like cells in vitro (Hussain et al. 2005). In view of the distribution pattern of BCRP expression in the liver, it remains to be established whether these isolated CD45-negative SP cells correspond to hepatic progenitor cells, bile duct cells, or endothelial cells or to a mixture of these cell types.
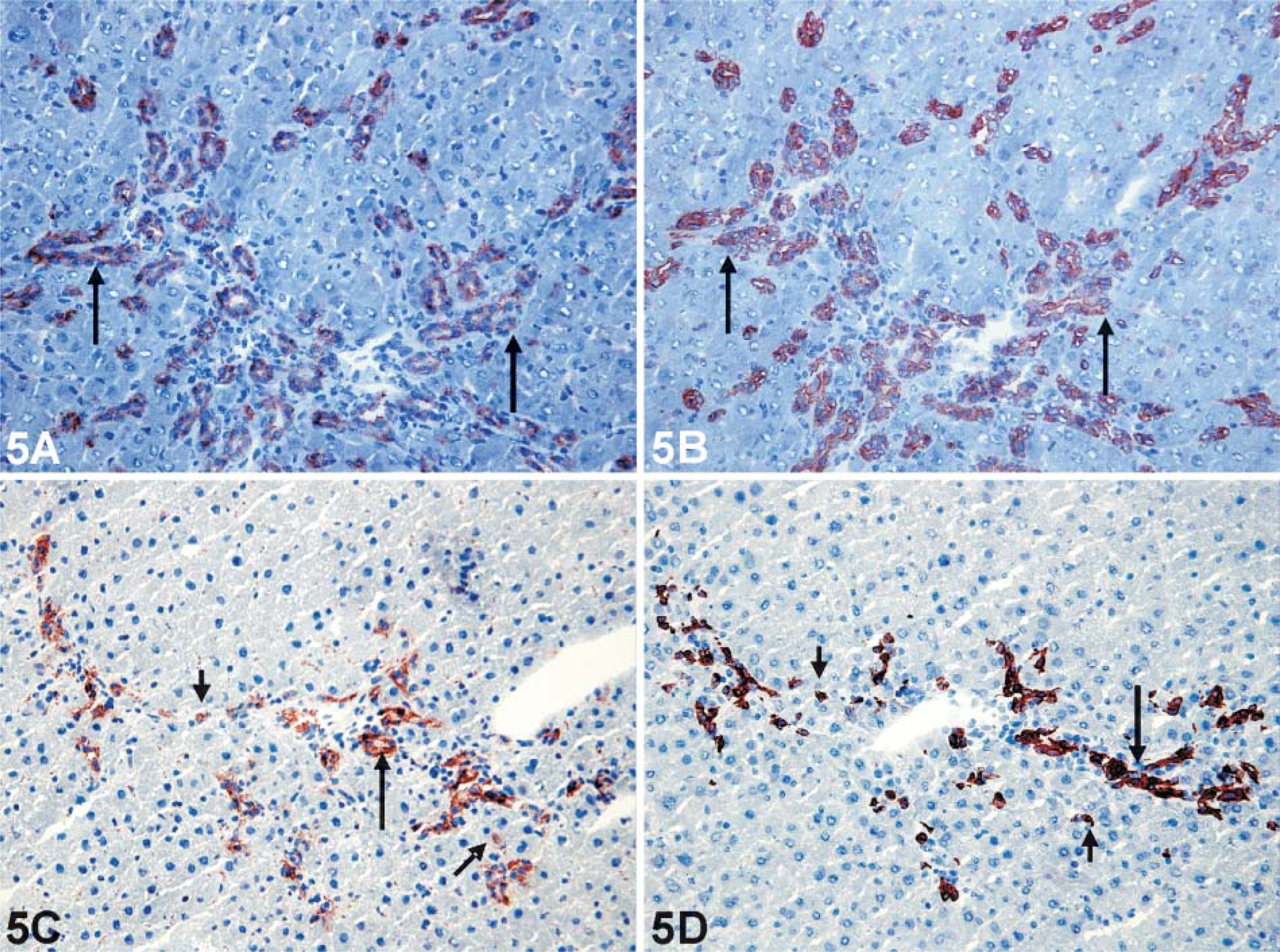
IHC staining for Bcrp with the M-70 antibody (
As in normal human liver, diseased human liver was characterized by BCRP expression at the canalicular pole of hepatocytes, which has previously led to the suggestion that BCRP most likely plays a role in the secretion of bile constituents from the hepatocytes into the bile canaliculus (Maliepaard et al. 2001). Indeed, BCRP can transport several biliary constituents such as bile salts, sulfated conjugates of steroids, and porphyrins (Jonker et al. 2002; Suzuki et al. 2003; Janvilisri et al. 2005). Surprisingly, we also observed a previously unreported expression of BCRP at the basolateral hepatocytic pole in some specimens. Using the same anti-BCRP antibodies, Xia et al. reported that IHC stainings on the Caco-2 human colonic adenocarcinoma cell line showed basolateral and apical staining 2 days after cell seeding and only apical staining in 5-day-old cultures (Xia et al. 2005). The presence of basolateral expression was strongly associated with biliary etiology of liver disease. Because the studied chronic biliary diseases are all characterized by bile duct damage and destruction leading to the accumulation of bile constituents in canaliculi and hepatocytes (Center 1999), basolateral expression of BCRP may represent a defensive mechanism: exporting toxic bile constituents back to the sinusoidal blood protects the hepatocytes from high, toxic levels of these compounds. The function of basolateral BCRP in chronic cholestatic conditions would thus be similar to that of other basolateral transporters such as MRP3 and MRP1 (Zollner et al. 2001; Ros et al. 2003a).
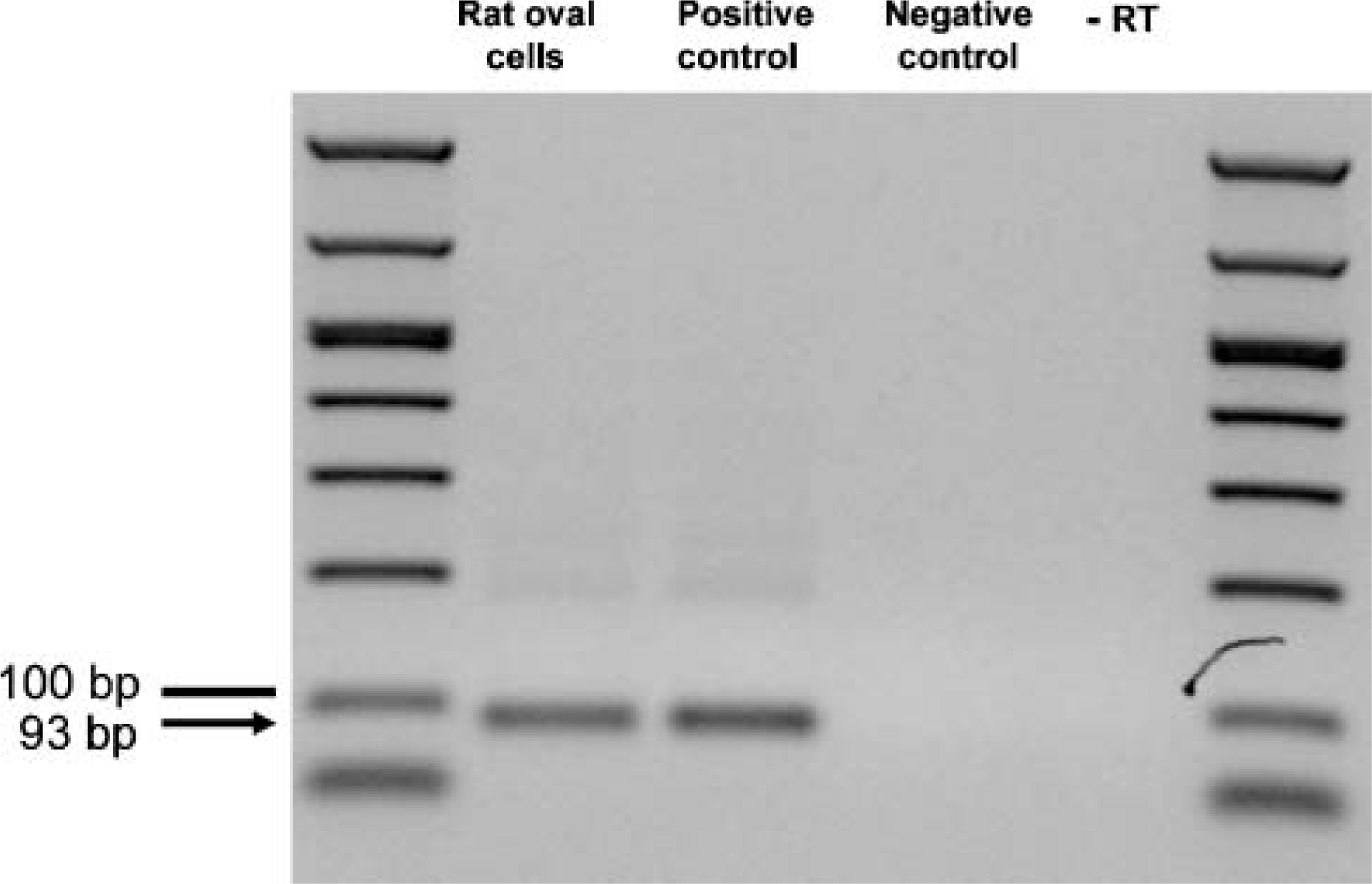
Bcrp mRNA expression is measured with RT-PCR in rat oval cells. PCR product was calculated to a length of 93 bp (arrow); the line is at 100 bp. Rat oval cells and positive control (brain tissue) showed Bcrp mRNA expression. Negative controls, no sample, and without reverse transcriptase were consistently negative. Identification of the PCR product (Bcrp cDNA) was achieved by sequencing.
The phenomenon of basolateral expression of a hepatic transporter that is normally only present at the apical pole has also been described previously for MRP2 in a case report of antidepressant-induced toxic liver disease and in human HepG2 cells treated with phorbol 12-myristate 13-acetate (PMA) and for MDR3 in PBC (Kubitz et al. 2001; Milkiewicz et al. 2001,2003). Similar observations have also been reported for dipeptidyl peptidase IV (DPP IV/Cd26), a canalicular cell surface ectopeptidase, in human cirrhotic liver and hepatocellular carcinomas (Matsumoto et al. 1992; Stecca et al. 1997). It should be mentioned that some reports on cholestatic animals and patients with obstructive jaundice and PBC III reported redistribution of canalicular export pumps like MRP2 into intracellular structures but never into basolateral membranes (Trauner et al. 1997; Kojima et al. 2003). These previous reports and the current study show that the type of transporter that is redistributed depends on the type of liver disease and probably not the consequence of a general alteration in diseased liver such as loss of polarity. It is not clear whether the redistribution of BCRP and these other transporters occurs prior to insertion of the transporter into the canalicular membrane or subsequently by lateral diffusion.
Although rat models and human diseased livers showed a strong expression of BCRP in bile ducts, ductules, and endothelia, hepatocytic expression observed in human normal and diseased liver was completely absent in all rat models and normal rat liver. This suggests that the hepatocytic expression of BCRP is much lower in the rat that in human liver. In agreement with these findings, Shimano et al. (2003) reported that Northern blot analysis of Bcrp mRNA showed no signal in parenchymal cells isolated from liver of rats that underwent the 2-AAF/partial hepatectomy protocol. Species differences in BCRP expression appear to be present in several organs because BCRP mRNA expression in placental tissue is much higher in humans than in rats (Tanaka et al. 2005). This species difference in expression may result in significant functional differences regarding excretion of exogenous and endogenous toxic substances, and this must be taken into consideration when developing rat models for pharmacokinetic and toxicological evaluation of novel and established drugs and other compounds.
In conclusion, BCRP positivity in the progenitor cells/reactive ductules could contribute to the resistance of these cells to cytotoxic agents. Basolateral hepatocytic expression in chronic biliary diseases may be an adaptive mechanism to pump bile constituents back into the sinusoidal blood. Strong differences between human and rat liver must be taken into account in future studies with animal models.
Footnotes
Acknowledgements
The authors thank Paula Aertsen, Martine Verhoeven, and Petra Windmolders for their excellent technical skills. T.R. is a basic researcher for the Fonds voor Wetenschappelijk Onderzoek (FWO)-Vlaanderen. L.L. and D.C. are postdoctoral researchers for the Fonds voor Wetenschappelijk Onderzoek (FWO)-Vlaanderen.
