Abstract
A total of 18 histological samples containing both transitional cell carcinoma (TCC) and normal urothelial epithelium were analyzed for protein kinase C (PKC)-α and -βI expression, and for their phosphorylated substrates. The results showed an increased expression of PKC-α in 13 out of 18 samples and -βI in 11 out of 18 TCC samples when compared with normal urothelium. In addition, 11 out of 18 of the TCC tumors displayed heterogeneous expression of the PKC isoenzymes, with different levels of immunosignal in different areas of the tumor. Within the same sample, areas of highest PKC isoenzyme expression also showed highest classical PKC activity, as estimated by immunodetection of phosphorylated forms of PKC substrates. The areas of highest expression of PKC-α and/or -βI isoenzymes showed also the highest number of cells positive for Ki67, an indicator of proliferation. Immunofluorescence and Western blotting demonstrated that in cultured TCC cells, PKC-α was located in the cytoplasm, whereas PKC-βI was located primarily in the nucleus as a 65-kDa fragment and in the cytoplasm as a full-size 79-kDa protein. Our results indicate that increased expression of PKC-α and -βI leads to increased total classical PKC kinase activity and suggest that increased activity of the isoenzymes plays a role in accelerated growth of TCC. Furthermore, these results suggest that even in carcinoma tissue, PKC expression and activity are under strict control.
P
Chemical tumor-promoting agents have been identified as specific activators of PKC. For instance, phorbol 12-myristate 13-acetate (PMA) binds to the DAG-binding site of PKC with high affinity and promotes prolonged activation of PKC and subsequent degradation of the enzyme (Blumberg et al. 1984; Sharkey et al. 1984; Lu et al. 1998). It is not known whether tumor promotion results from PKC activation, or depletion due to chronic activation of the enzyme.
PKC isoenzymes have been shown to display variable expression profiles during cancer progression, depending on the particular cancer type. The most common isoenzymes displaying alterations in expression during cancer progression are α, β, and δ, but abnormal expression of other isoenzymes may also take place. Immunohistochemical studies have shown PKC-α overexpression in high-grade prostate and endometrial cancers, whereas low-grade tumors and normal epithelia of the respective organs show significantly lower expression (Martinez-Gimeno et al. 1995; Fournier et al. 2001; Koren et al. 2004). In contrast, breast, colon, hepatocellular, and basal cell cancers display downregulation of PKC-α expression (Tsai et al. 2000; Gokmen-Polar et al. 2001; Neill et al. 2003; Ainsworth et al. 2004; Kerfoot et al. 2004). PKC-β expression has been shown to be upregulated in colon and prostate cancers (Gokmen-Polar et al. 2001; Koren et al. 2004). To date, it seems that alterations in PKC isoenzymes during cancer progression are highly variable and depend on cancer cell type. Thus, it is not possible to reach general conclusions about isoenzyme expression patterns with respect to carcinogenesis in general. In fact, mutations in genes encoding PKCs are found very rarely in human cancers, and therefore expressional changes are not likely to represent primary events. Mutations of the PKC-α gene and change in the subcellular location of the corresponding protein have been detected in thyroid cancers, but the importance of these mutations to PKC-α activity remains unknown (Prevostel et al. 1995).
PKC-α and -βI have been linked also to urinary bladder transitional cell carcinoma (TCC). To our knowledge, there are three in vivo studies that included data concerning PKC-α and/or -βI isoenzyme expression in human urinary bladder carcinoma (Koren et al. 2000; Varga et al. 2004; Kong et al. 2005). These studies showed that of classical PKCs, only α and β are expressed in transitional epithelium. One of the studies showed that PKC-βI expression decreases with increasing grades of TCC (Varga et al. 2004). In addition, the studies showed some contradictory results concerning PKC-α expression. PKC-α expression has been reported to be elevated and to correlate with tumor grade (Varga et al. 2004; Kong et al. 2005) or to remain non-altered in TCC (Koren et al. 2000). However, expression studies have overlooked the genetic heterogeneity of tumors, which is well documented in TCC, and results in variable gene expression profiles as well as proliferative properties of cancer cells in different tumor compartments (Brown et al. 1990; Voorter et al. 1995; Diaz-Cano et al. 2000; Blanes et al. 2002).
We have recently shown that Go6976, which specifically inhibits PKC-α and -βI activity, downregulates invasion and migration of cultured high-grade TCC cells, thus linking the α and βI isoenzymes to the malignant phenotype of TCC cells (Koivunen et al. 2004). Furthermore, inhibition of PKC-α and -βI has been shown to inhibit cancer cell growth (Biswas et al. 2003; Tortora and Ciardiello 2003). The results of the present study on tissue specimens containing both TCC and normal urothelial epithelium demonstrate increased expression and activity of PKC-α and -βI isoenzymes in most proliferative areas of TCC when compared with normal epithelium. Areas with less proliferative activity showed non-altered or less expression of both isoenzymes when compared with normal epithelium. Our results thus demonstrate that PKC-α and -βI isoenzymes and their phosphorylated substrates are heterogeneously expressed within TCC tumors. These results suggest that PKC expression and activity are under control elicited by internal and/or external stimuli even in carcinoma tissue.
Materials and Methods
Antibodies
Mouse monoclonal antibodies (IgG1) against PKC-α (clone H-7) (Kerfoot et al. 2004) and PKC-βI (clone E-3) (Raghunath et al. 2003) were purchased from Santa Cruz Biotechnology (Santa Cruz, CA), and mouse monoclonal antibodies against Ki67 (clone 7B11) (Stearns et al. 2004) were purchased from Zymed Laboratories (San Francisco, CA). Rabbit anti-phospho-PKC substrate antibody was obtained from Cell Signaling Technology (Beverly, MA). This antibody detects phosphorylated serine residues when surrounded by arginine or lysine at the −2 and +2 positions and a hydrophobic residue at the +1 position. Thus, it binds specifically to phosphorylated substrates of the classical PKC isoenzymes (α, βI, βII, and γ) but not to those of novel or atypical isoenzymes (Nishikawa et al. 1997; Koivunen et al. 2004).
Immunohistochemical Analysis
Immunohistochemical analysis was performed on formalin-fixed, paraffin-embedded tissue sections that contained human TCC and normal urothelium. Tumor grade and local invasion were analyzed by an independent clinical pathologist. Paraffin-embedded tissues were cut to 5-μm-thick sections and plated on glass slides, deparaffinized, and rehydrated. Antigen unmasking was performed by boiling the samples in a buffer containing 10 mM Tris and 1 mM EDTA. The sections were treated with H2O2 in PBS for 5 min to remove endogenous peroxidase. The sections were subsequently incubated in 1% BSA in PBS for 30 min to prevent nonspecific binding. Subsequent steps were performed using the Histostain-Plus broad-spectrum kit (Zymed Laboratories) following the manufacturer's guidelines. DAB color reaction was obtained using the Liquid DAB Substrate Bulk Kit (Zymed Laboratories). The sections were counterstained using hematoxylin.
Evaluation of Immunosignal in Tumor Specimens
The intensity of immunosignal in different areas of tumor was compared with that of normal urothelium (internal control) within the same sample. PKC-α was evaluated for cytoplasmic immunosignal, PKC-βI and phospho-PKC substrate for cytoplasmic immunosignal and positive nuclei, and Ki67 for the amount of positive cells for nuclear immunosignal. Tumor cells in invasive carcinomas were labeled “superficial” when located above the muscularis mucosa, and “deep” when located below the muscularis mucosa. Muscularis mucosa was detected in samples histologically by the presence of muscle fibers and its ectatic vascular plexus (Ro et al. 1987; Aydin et al. 2002). The results of the evaluation of immunosignals are presented in Table 1. No signs of necrosis were detected in morphological analysis of hematoxylin-eosin stains of the carcinoma samples.
Cell Culture
Cell lines 5637 (grade 2-3 TCC) and T24 (grade 3 TCC) were obtained from American Type Culture Collection (Rockville, MD). Cells were maintained in DMEM supplemented with 10% fetal calf serum, penicillin (100 U/ml), and streptomycin (100 μg/ml).
Immunofluorescence
The cells were cultured on glass coverslips and rinsed twice in PBS before fixation. The cells were fixed using 4% paraformaldehyde in PBS and permeabilized with 0.1% Triton X-100 in PBS at +20C and washed three times with PBS. To reduce nonspecific binding of antibodies, the cells were incubated in 1% BSA in PBS for 1 hr at +20C. Primary antibodies (anti-PKC-α or anti-PKC-βI and anti-phospho-PKC substrate) were diluted in 1% BSA in PBS, incubated on cells at +4C overnight, and washed three times in PBS. The samples were then incubated with secondary antibodies, goat anti-rabbit Alexa 488, and goat-anti-mouse Alexa 568 (Molecular Probes, Eugene, OR) at +20C for 1 hr, counterstained with Hoechst, washed three times with PBS, and mounted.
Four fields from two double-labeled samples (PKC-α and phosphorylated PKC substrates or PKC-βI and phosphorylated PKC substrates), thus a total of 8 fields, were photographed through a UV microscope, 20x objective and appropriate filters for Alexa 488, Alexa 568, and Hoechst. First, the total cell number was counted from all Hoechst-stained fields, and subsequently, expression of a particular isoenzyme (PKC-α or PKC-βI) and phosphorylated PKC substrates was estimated in each cell. The cells were labeled to have either increased or baseline expression. The evaluation of each immunolabeling and field was performed in a random order, after which the data of double labelings of each field were combined. Percentage of cells showing increased expression of isoenzymes and phosphorylated PKC substrates was calculated, as well as the percentage of cells that showed increased expression of both an isoenzyme and phosphorylated PKC substrates.
Nuclear Isolation
The cells were cultured on Petri dishes and washed twice with PBS before treatment with a buffer containing 10 mM Hepes, pH 7.9, 10 mM KCl, 0.1 mM EDTA, 1 mM dithiothreitol (DTT), 0.4% Igepal CA-630, and Complete Mini protease inhibitor tablets (Boehringer Ingelheim; Ingelheim, Germany) for 10 min at +20C. The cells were detached from the culture dishes with a rubber policeman, and centrifuged 13,000 × g at +4C for 3 min. The supernatant (cytosolic fraction) was collected, and the remaining pellet was treated with a buffer containing 20 mM Hepes, pH 7.9, 0.4 M NaCl, 1 mM EDTA, 1 mM DTT and Complete Mini protease inhibitor, and vortexed at +4C. The samples were then shaken on ice for 2 hr and centrifuged 13,000 × g at 4C for 20 min, and supernatant (nuclear fraction) was collected for measurement of protein concentration and Western transfer analysis.
Overall intensity of immunosignal in various TCC tumor compartments when compared to normal urothelium in the same specimen
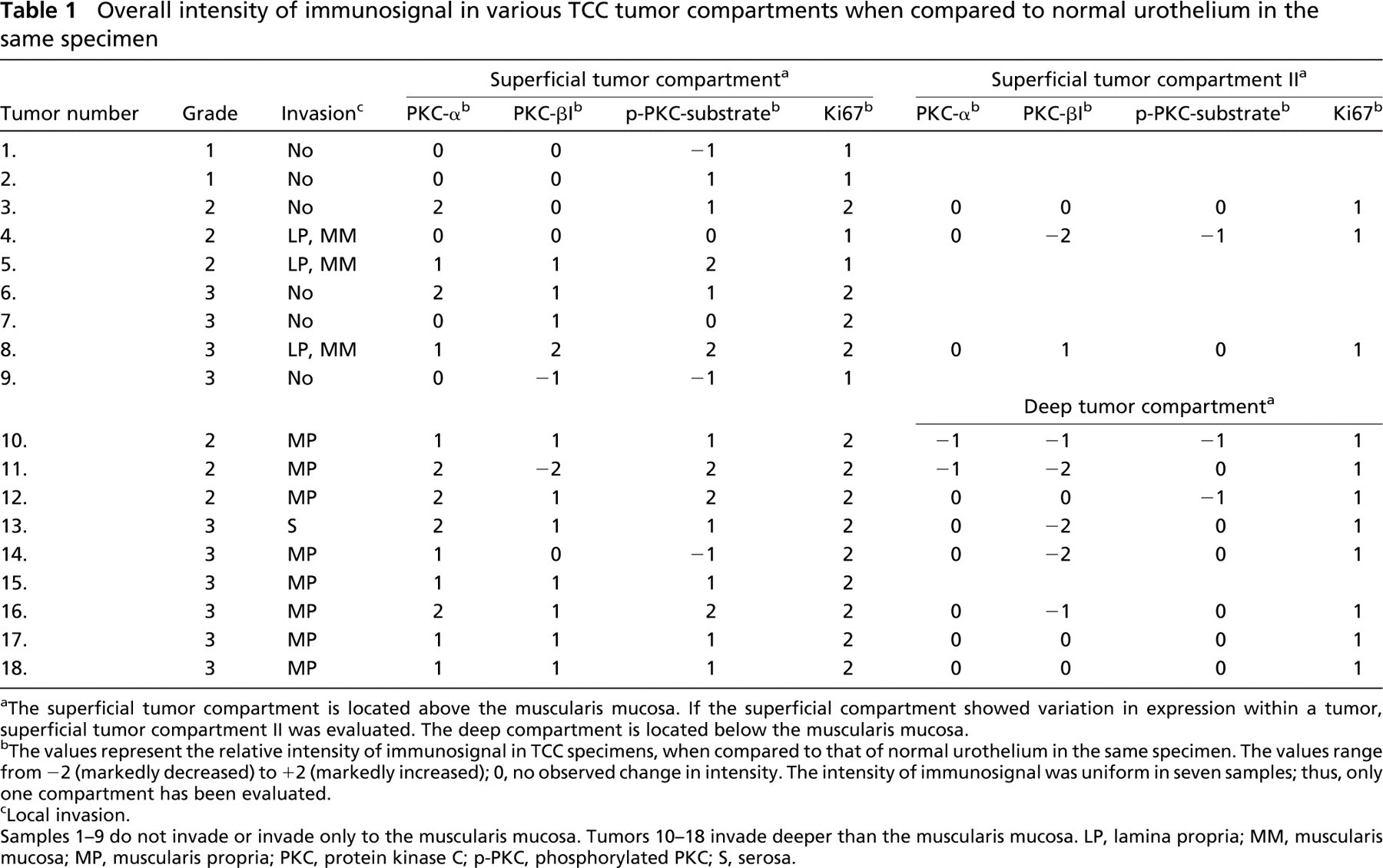
The superficial tumor compartment is located above the muscularis mucosa. If the superficial compartment showed variation in expression within a tumor, superficial tumor compartment II was evaluated. The deep compartment is located below the muscularis mucosa.
The values represent the relative intensity of immunosignal in TCC specimens, when compared to that of normal urothelium in the same specimen. The values range from −2 (markedly decreased) to +2 (markedly increased); 0, no observed change in intensity. The intensity of immunosignal was uniform in seven samples; thus, only one compartment has been evaluated.
Local invasion.
Samples 1-9 do not invade or invade only to the muscularis mucosa. Tumors 10-18 invade deeper than the muscularis mucosa. LP, lamina propria; MM, muscularis mucosa; MP, muscularis propria; PKC, protein kinase C; p-PKC, phosphorylated PKC; S, serosa.
Western Transfer Analysis
Protein concentration of nuclear and cytosolic fractions was measured using DC Protein Assay (BioRad; Hercules, CA), and equal amounts of protein (5 μg) were subjected to SDS-PAGE on 10% gel. The proteins were then electrophoretically transferred to polyvinylidene difluoride (PVDF) membrane and processed for immunoblotting. Membranes were first blocked with 5% BSA/PBS + 0.05% Tween-20 and immunolabeled with monoclonal anti-PKC-α or anti-PKC-βI antibodies. Anti-mouse horseradish peroxidase-conjugated antibody (Cell Signaling Technology) was used as a secondary antibody and detected with enhanced chemiluminescence (ECL) (Amersham Life Sciences; Little Chalfont, England).
Controls
Peptide competitions were performed for anti-PKC-α, anti-PKC-βI, and anti-phospho-PKC substrate antibodies, and the blocked antibodies were used as controls in immunohistochemical, immunofluorescence, and Western transfer analyses. Anti-PKC-α and anti-PKC-βI antibodies were preincubated with a ten-fold weight excess of the corresponding peptide provided by the manufacturer of the antibody (Santa Cruz Biotechnology) at +4C for 18 hr. Anti-phospho-PKC substrate antibodies were preincubated similarly with a phosphorylated peptide that corresponds to the phosphorylation pattern and amino acid sequence that this antibody detects (phosphorylated PKC substrate 3; H-Lys-Arg-Pro-phosphoSer-Gln-Arg-His-Gly-Ser-Lys-Tyr-NH2) (AnaSpec, Inc.; San Jose, CA).
Negative-control immunoreactions in immunohistochemistry and indirect immunofluorescence included the following: (a) primary antibody was replaced with 1% BSA in PBS; (b) primary antibody was replaced with 10% non-immune serum in PBS; (c) primary antibody was replaced with irrelevant mouse IgG1 (monoclonal mouse anti-Cre, Clone 7-23; Sigma, St. Louis, MO), normal mouse IgG (Santa Cruz Biotechnology), or normal rabbit IgG (Santa Cruz Biotechnology) using the same antibody concentrations as used with the primary antibodies; (d) primary antibody was preabsorbed with a 10-fold weight excess of corresponding blocking peptides (for anti-PKC-α and anti-PKC-βI antibodies), or preabsorbed with phosphorylated peptide corresponding to epitope recognized by anti-phospho-PKC substrate antibody (see peptide competitions above). In all negative controls, a faint uniform background immunosignal only was observed.
To gain additional evidence for anti-phospho-PKC substrate antibody performance in immunofluorescence, PKC activation and inactivation experiments for cultured TCC cells were performed. The TCC cells were cultured without serum for 18 hr and treated subsequently with 200 nM PMA (Sigma) for 20 min to activate PKC. Alternatively, the starved cells were treated with PKC-α-and -βI-isoenzyme inhibitor (1 μM Go6976; Calbiochem, La Jolla, CA) for 2 hr before and during the PMA stimulation. The treated cells were subsequently subjected to immunofluorescence labeling as described above.
As a negative control for antibodies in Western transfer analysis, double sets of samples were loaded on opposite sides of the gel, subjected to SDS-PAGE, and transferred to PVDF membrane. The membrane was cut vertically between sample sets and incubated with anti-PKC-α or anti-PKC-βI antibodies, or with antibodies that were preabsorbed with the corresponding blocking peptides (see peptide competitions above). ECL detection was performed simultaneously on all membranes, using the same incubation times.
Results
PKC-α and -βI Expression in Different Compartments of TCC Tumors
A total of 18 TCC samples were selected for the study on the basis that all samples contained both the normal urothelial epithelium and TCC areas. PKC-α and -βI expression was addressed by IHC using monoclonal antibodies against isoenzymes α or βI. The results demonstrated that both normal urothelial cells and carcinoma cells showed cytoplasmic expression of PKC-α (Figures 1A-1F), but βI expression was predominantly nuclear (Figures 1G-1L). PKC-α expression was increased in superficial compartments of 13 out of 18 (13/18) samples when compared with normal urothelium in the same specimens. Furthermore, PKC-βI expression was increased in superficial compartments in 11/18 TCC samples (Table 1). Increased expression of PKC-α, or -βI, or both was detected in 14/18 samples. PKC-α/-βI immunoreactivity did not, however, correlate with tumor grade, even though two of the grade 1 samples displayed non-altered PKC expression.
Interestingly, PKC-α and/or -βI were expressed in a non-uniform manner in 11/18 samples: these samples contained clearly delineated areas of increased PKC-α/βI immunoreactivity and areas of non-altered or even decreased expression when compared with normal urothelium (Figures 1 and 2; Table 1). In samples that were invasive to lamina propria or deeper, two distinctive compartments could be separated: 8/9 tumors invading to muscularis propria showed heterogeneous expression of the PKC isoenzymes (Table 1). Immunohistological analysis of the invasive samples revealed a superficial compartment above the muscularis mucosa with increased expression of PKC-α (9/9 tumors) (Figures 1C and 1D) and/or PKC-βI (7/9 tumors) (Figures 1I and 1J) when compared with the expression of the PKC isoenzymes in normal epithelium in the same specimen (Figures 1A and 1B, PKC-α; Figures 1G and 1H, PKC-βI). In contrast, the deep compartment below the muscularis mucosa displayed non-altered (6/8) or decreased (2/8) PKC-α expression (Figures 1E and 1F, showing non-altered expression) and non-altered (3/8) or decreased (5/8) PKC-βI expression (Figures 1K and 1L, showing decreased expression) when compared with the normal epithelium within the same specimen (Figures 1A and 1B, PKC-α; Figures 1G and 1H, PKC-βI; Table 1).
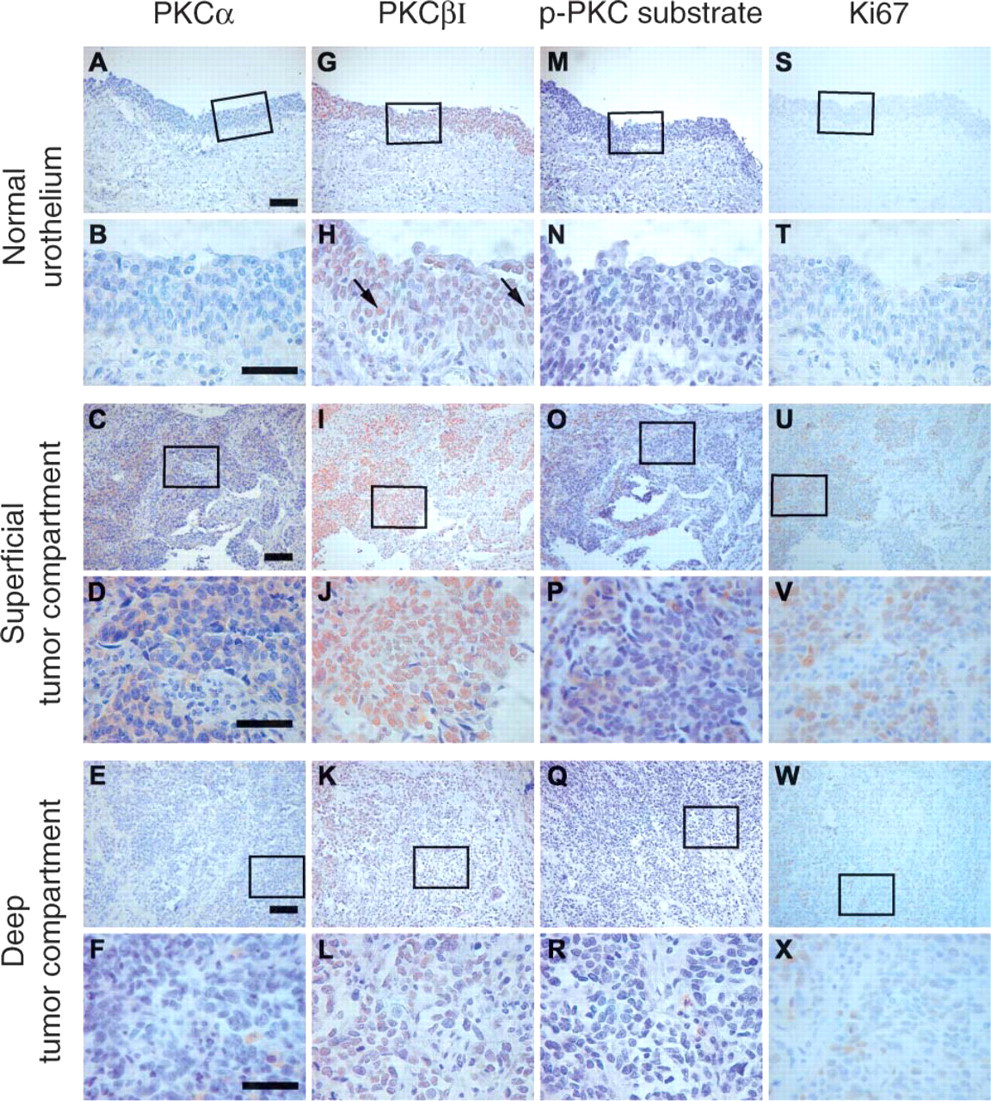
Expression of PKC-α, PKC-βI, phosphorylated PKC substrates, and Ki67 in normal urothelium and different compartments of transitional cell carcinoma (TCC) tumor. Adjacent tumor sections were labeled with antibodies against either PKC-α
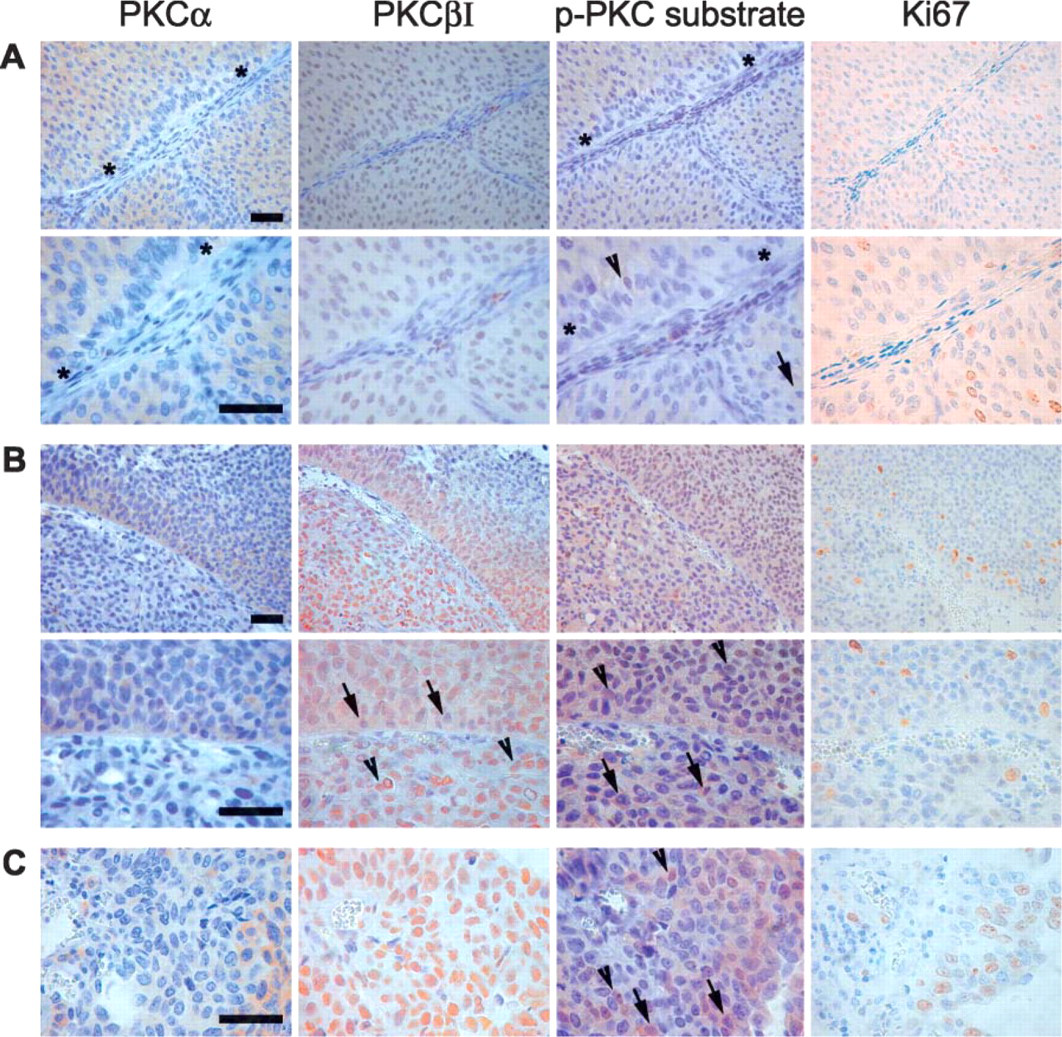
Intratumor heterogeneity of PKC-α, PKC-βI, phosphorylated PKC substrate, and Ki67 expression in three different tumor samples. Adjacent tumor sections were labeled with antibodies against PKC-α, PKC-βI, phosphorylated PKC substrates (p-PKC substrate), or Ki67. Sample
It is of interest that the material also included three samples in which there was heterogeneous expression of the isoenzymes and which did not invade through the muscularis mucosa. In addition, our results show that α and βI isoenzymes are variably expressed in single cells within small compartments of a tumor (Figure 2). Furthermore, cells that are in close proximity to the extracellular matrix (ECM) show less PKC-α expression (Figure 2A), suggesting a regulatory role for ECM contacts. All negative control immunoreactions resulted in a faint uniform background signal only (Figure 3A). These results suggest that even in carcinoma tissue, PKC isoenzyme expression is under strict control of specific internal and/or external stimuli.
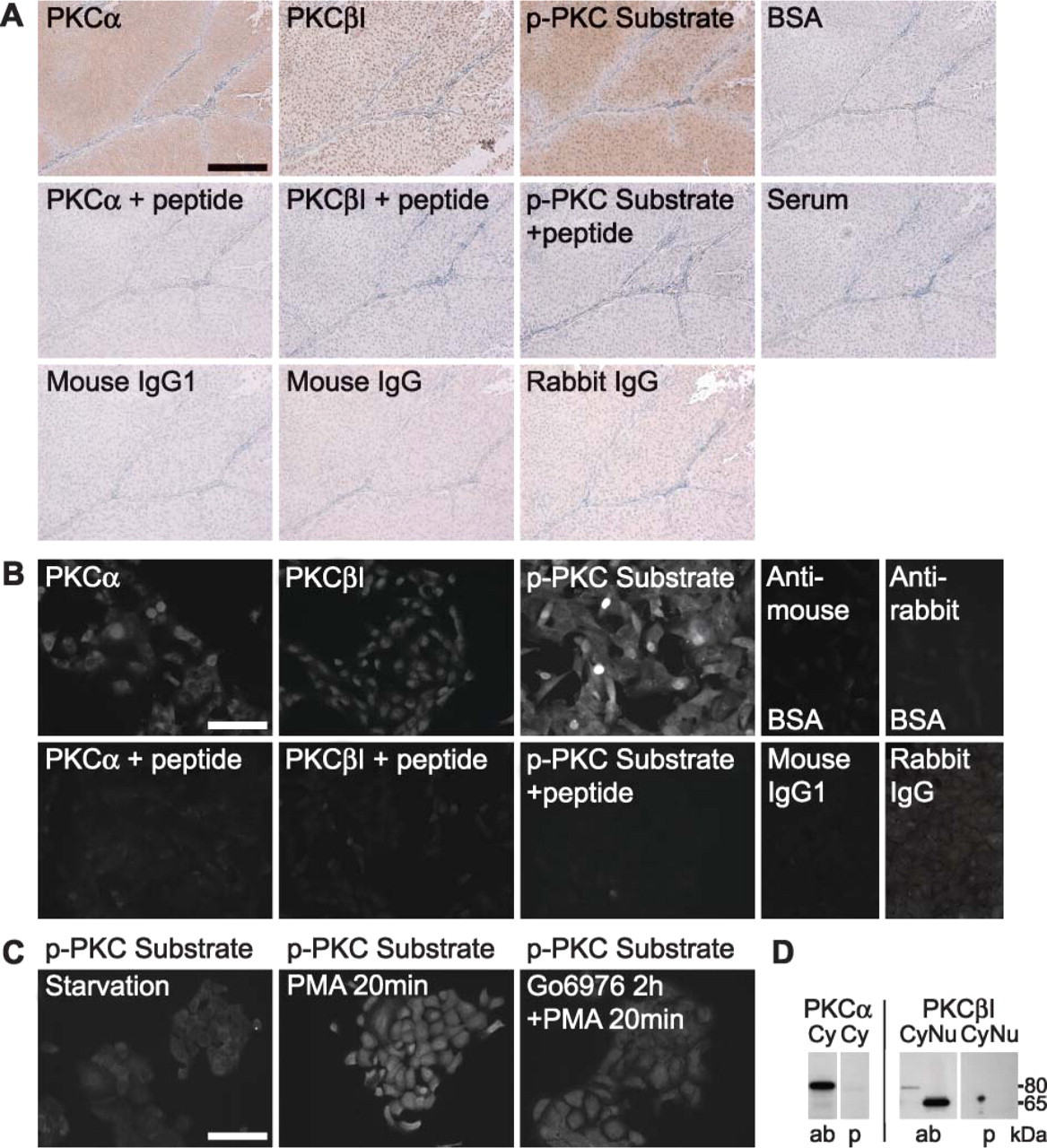
Controls for PKC-α, PKC-βI, and phosphorylated PKC substrate antibodies. Adjacent tumor sections were subjected to immunohistochemistry using antibodies against PKC-α, PKC-βI, or phospho-PKC substrates
PKC-α/-βI Substrate Phosphorylation in TCC
The activity of classical PKCs (α/βI) was studied using IHC and a novel antibody that detects endogenous levels of cellular proteins only when phosphorylated in a manner that is typical for cPKCs (phosphorylated cPKC substrates). The results demonstrated that increased cPKC activity is located in the same areas in which increased PKC-α or -βI expression was detected (Figures 1O and 1P and Figure 2; Table 1). In addition, the results show that the antibody detects phosphorylated PKC substrates in both the nucleus and the cytoplasm, which is in good agreement with the current knowledge of possible PKC substrate subcellular localization. All negative-control immunoreactions resulted in a faint uniform background signal only (Figure 3A). The data suggest that increased PKC-α and -βI expression leads to increased overall cPKC kinase activity.
Correlation of PKC-α and -βI Expression and Activity with Cell Proliferation in TCC
Because PKC has been linked to cell growth control, Ki67 expression was studied in the same tissue samples using IHC (Figures 1S-1X and Figure 2; Table 1). Ki67 is a nuclear antigen that is present in proliferating cells, because its expression takes place during the G1, S, G2, and M phases of the cell cycle, but not during G0. An elevated number of Ki67-positive cells (Figures 1U and 1V) was observed in the same TCC locations that displayed the highest expression of PKC-α (Figures 1C and 1D), -βI (Figures 1I and 1J), and phosphorylated PKC substrates (Figures 1O and 1P). Specifically, the expression of PKC-α was increased in 12/13, PKC-βI in 10/13, and phosphorylated PKC substrates in 10/13 areas harboring markedly increased numbers of Ki67-positive cells (Table 1).
In contrast, when the number of Ki67-positive cells was only moderately increased, the level of immunosignals for PKC-α and -βI and their substrates was low. Specifically, out of 16 evaluated areas that showed only moderate numbers of Ki67-positive cells, expression of PKC-α remained unchanged in 13/16 and decreased in 1/16 areas, expression of PKC-βI remained unchanged in 7/16 and decreased in 7/16 areas, and expression of phosphorylated PKC substrates remained unchanged in 9/16 and decreased in 5/16 areas of the evaluated areas. In all samples, normal urothelium showed no Ki67-positive cells (Figures 1S and 1T), which is in good agreement with previous reports (Limas et al. 1993).
Classical PKC Enzyme Activity and Expression of PKC-α and -βI Proteins in Cultured T24 and 5637 TCC Cells
T24 and 5637 TCC cells showed similar but not identical results. In general, PKC-α and PKC-βI expression levels varied between individual cells in both TCC line cultures. Double immunolabelings were performed using anti-phospho-PKC substrate antibodies with antibodies to PKC-α (Figure 4A), or to PKC-βI (Figure 4B) to reveal potential correlation between increased phosphorylation of PKC substrates and increased expression of PKC-α or -βI. Cells from double-labeled samples were counted and graded to have either increased or baseline expression of PKC-α, PKC-βI, or phosphorylated PKC substrates. The results showed that increased PKC-α and PKC-βI immunoreactivity correlated with increased phosphorylation of PKC substrates in the same cells.
More specifically, when expression of PKC-α and phosphorylated PKC substrates was measured in 5637 TCC cells, the results showed that 18.5% of 1537 cells counted had increased expression of PKC-α and 21.2% had increased expression of phosphorylated PKC substrates. Furthermore, 12.9% of cells displayed elevated expression of both PKC-α and phosphorylated PKC substrates. The measurement of expression of PKC-βI and phosphorylated PKC substrates in 5637 cells showed that 11.5% of 1406 cells counted showed increased expression of PKC-βI, 21.1% showed increased expression of of phosphorylated PKC substrates, and 9.3% had increased expression of both.
When a total of 809 double-labeled T24 TCC cells were evaluated for expression of PKC-α and phosphorylated PKC substrates, the results showed that 36.8% of the cells had increased expression of PKC-α and 37.5% had increased expression of phosphorylated PKC substrates. Furthermore, 27.4% of cells showed increase in expression of both PKC-α and phosphorylated PKC substrates. When a total of 925 double-labeled T24 TCC cells were evaluated for expression of PKC-βI and phosphorylated PKC substrates, the results showed that 13.0% of the cells had increased expression of PKC-βI, 30.6% of phosphorylated PKC substrates, and 11.9% of both.
Taken together, increased PKC substrate phosphorylation seemed to result primarily from PKC-α activity, and to lesser extent from PKC-βI activity. Furthermore, it is of interest to note that the subcellular localizations of PKC-α and PKC-βI were markedly different: Indirect immunofluorescence studies showed that PKC-α was localized in the cytoplasm (Figure 4A). In contrast, PKC-βI localization was predominantly nuclear. Western blotting for cytoplasmic and nuclear extracts showed differences identical to those seen in immunofluorescence analysis (Figure 4C). Western blots also showed that PKC-α levels were higher in T24 cells than in 5637 cells. In addition, cytoplasmic full-size PKC-βI was more abundant in T24 cells than in 5637 cells. However, a 65-kDa PKC-βI fragment was found in nuclear extracts, and its expression was more abundant in 5637 cells.
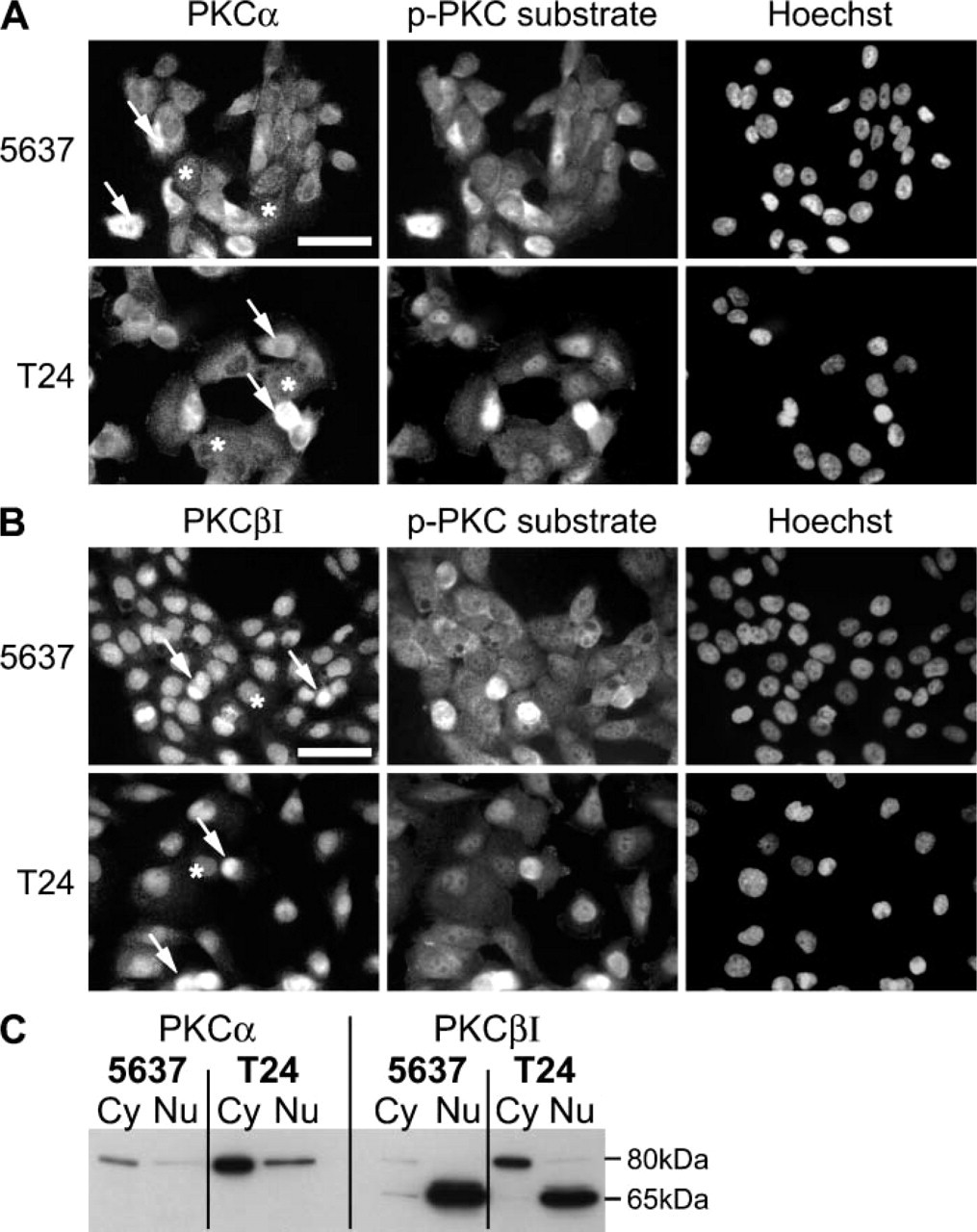
Subcellular localization of PKC-α, PKC-βI, and phosphorylated PKC substrates in 5637 and T24 TCC cells. PKC-α shows cytoplasmic localization with variable intensity
All negative controls used in immunofluorescence studies resulted in only a faint background fluorescence (Figure 3B). To further evaluate phospho-PKC substrate antibody performance, TCC cells were stimulated with the PKC activator PMA and subjected to immunofluorescence labeling. The results showed a prominent increase in immunofluorescence for phosphorylated PKC substrates, and the increase in immunosignal could be blocked by 2-hr treatment with the PKC-α/-βI inhibitor Go6976 before PMA stimulation (Figure 3C). These data strongly suggest that phospho-PKC substrate antibody detects specifically phosphorylated substrates of classical PKCs. In Western blots, preincubation of the antibodies with corresponding peptides resulted in almost complete loss of bands representing full-size PKC-α and PKC-βI, as well as the band representing the 65-kDa PKC-βI (Figure 3D).
Discussion
The current study shows for the first time, to our knowledge, that PKC-α and -βI expression and cPKC activity are increased in TCC tumors, particularly in the most-proliferative areas of the tumor, as demonstrated by immunolocalization of Ki67. Superficial compartments above the muscularis mucosa displayed the highest PKC-α/-βI expression (Figures 1C and 1D, 1I and 1J) and kinase activity (Figures 1O and 1P) and, in addition, the highest proliferative activity (Figures 1U and 1V). The muscularis mucosa is located in the lamina propria of the bladder wall, and invasion through this layer has been suggested to be a prognostic factor in TCC (Ro et al. 1987; Smits et al. 1998). Topographic evaluation of proliferation markers has shown that superficial compartments above the muscularis mucosa layer of muscle-invasive tumors have significantly higher proliferative activity than the deep compartments (Blanes et al. 2002), which is in good agreement with our results obtained with Ki67 immunolabeling. Previous reports have suggested that PKC-α expression is either non-altered or increased and that βI expression is decreased in TCCs (Koren et al. 2000; Varga et al. 2004; Kong et al. 2005). Our findings are consistent with previously reported results, because we show that in low-proliferation areas of the tumors, comprising most of the tumor volume, PKC-α expression generally remains non-altered when compared with normal epithelium (Figures 1A and 1B, 1E and 1F), and βI expression is decreased (Figures 1G and 1H, 1K and 1L). However, our results may explain why earlier studies have reported contradictory expression patterns of different PKC isoenzymes: the results of the present study depict restricted areas of high expression of selected PKC isoenzymes that apparently are not detected in tumor homogenates.
PKC-α and -βI have been linked to increased cell growth, inasmuch as introduction of PKC-α to cancer cells increases proliferation, and inhibition of PKC-α and -βI decreases cancer cell growth (Mandil et al. 2001; Biswas et al. 2003; Jiang et al. 2004). In addition, several studies have shown upregulation of different PKC isoenzymes in cancerous tissue. It is feasible to speculate that increased expression and activity of PKC-α and -βI play a role in the higher proliferation activity seen in superficial areas of TCC samples. Possible reasons for PKC-α/-βI down- or upregulation include varying tissue oxygenation status, because sustained anoxia has been shown to result in downregulation of PKC-α and -βI in brain and, in contrast, hypoxia has been shown to activate PKC-α in muscle tissue (Libien et al. 2005). In addition, PKC isoenzymes have been linked to cell cycle control, and it is possible that changes in PKC expression reflect changed activity of other cell signaling pathways affecting cell cycle progression. As an example, oncogenic Ras has been shown to upregulate PKC-α, c-myc oncogene induces increased PKC-β expression, and wild-type p53 has been shown to suppress PKC-α expression (Barr et al. 1991; Delage et al. 1993; Zhan et al. 2005). TCCs have previously been shown to be clonal, although different compartments may display distinct alterations in cell cycle-associated tumor suppressor status, such as type 1 neurofibromatosis gene defects in the superficial compartment and p53 in the deep compartment (Diaz-Cano et al. 2000). Thus, it is possible that variable PKC expression arises from differences in local growth environments, such as growth factors or oxygenation, or from tumor suppressor status that varies between compartments. Nevertheless, on the basis of our results and those of others, we consider that PKC-α and -βI play a significant role in the growth of TCC cells.
The results of the current study demonstrate that cellular localization in normal urothelial and TCC cells for PKC-α is cytoplasmic (Figures 1A-1F and Figures 4A and 4C) and that PKC-βI localizes either to the nucleus or to the cytoplasm (Figures 1G-1L and Figures 4B and 4C). We demonstrate that increased expression of PKC-α in the cytoplasm or membranes and classical PKC substrate phosphorylation correlate both in tumor samples and in TCC cultures. Furthermore, increased PKC substrate phosphorylation correlates with increased expression and nuclear localization of PKC-βI in tumor samples and in cultures with increased PKC-βI immunosignal. In normal epithelium, a significant number of cells were negative for PKC-βI nuclear localization, whereas in cancerous tissue, particularly in highly proliferative areas, nearly all nuclei were positive for immunosignal. In TCC cultures, nuclear PKC-βI appeared as an ~65-kDa fragment, whereas in the cytoplasm, it was present as a full-size 79-kDa form, as demonstrated by Western blotting (Figure 4C). There are earlier reports locating PKC-βI in the nucleus (La Porta et al. 1997; Svensson et al. 2000; Raghunath et al. 2003; Cogram et al. 2004), and it has been demonstrated that PKC-βI is transported to the nucleus and fragmented (Bastiaens and Jovin 1996). Others have shown a 65-kDa fragment for PKC-βI in the membrane fraction of the cells (al-Mazidi et al. 1998) and in whole-cell lysates (Pfaff et al. 1999; Redling et al. 2004). It is thus possible that PKC-βI is fragmented for a specific reason and that localization of the 65-kDa fragment in membranes (possibly nuclear membrane) indicates activation.
Taken together, our results provide direct evidence to associate high proliferation rate with increased expression and activity of classical PKCs in TCC in vivo. Our evidence suggests that PKC-α and -βI expression in TCC is under control, perhaps of some external or internal stimulus, and suggests that PKC-α and -βI expression and activity take part in increased growth of TCC. There are several ongoing Phase I-III studies focusing on the use of different PKC inhibitors in cancer treatment (PKC-α antisense oligodeoxynucleotide ISIS3521, broad-spectrum PKC inhibitors UCN-01 and PKC-412, and PKC-β inhibitor LY317615) (www.cancer.gov/clinicaltrials). The results of the current study may provide a rationale for further studies focusing on treatment of urinary bladder cancer with specific inhibitors targeting the isoenzymes, such as PKC-α/-βI inhibitor Go6976, ISIS3531, or LY317615.
Footnotes
Acknowledgements
This work was supported by Oulu University Hospital Grant H01139, by the Cancer Society of Finland, the Academy of Finland, the Finnish Cultural Foundation, AstraZeneca Finnish Research Foundation, the K. Albin Johansson Foundation, the Ida Montin Foundation, and the Cancer Society of Northern Finland.
We thank Marja Paloniemi for expert technical assistance, and Pekka Leinonen and Marja Nissinen for their careful review of the manuscript. Johanna Aaltonen is thanked for her support.
