Abstract
An adenovirus vector was generated using a neuron-specific promoter synapsin I and enhanced green fluorescent protein (EGFP) reporter (AdSynEGFP). In addition, two modifications were identified that resulted in robust and reliable retrograde transport and EGFP expression after injection of the virus into three different brain regions in adult rats (medial prefrontal cortex, posterior thalamic nuclear group, and CA1). These are post-injection survival times of 14 days and addition of high concentrations of NaCl (≥600 mM) to the injection buffer. These modifications resulted in obvious improvement in the intensity of the EGFP signal and in the number of labeled cells. Use of anti-EGFP in immunofluorescence or immunoperoxidase processing further enhanced the signal so that Golgi-like filling of dendritic spines and axon collaterals was routinely achieved. Effectiveness of the AdSynEGFP for Golgi-like filling was confirmed in one rhesus monkey with injections in visual area V4. Because of the long-term viability of the infected neurons (at least up to 28 days in rats and 22 days in monkey), this AdSynEGFP is suitable for use in microcircuitry studies in combination with other fluorescently tagged elements, including anterogradely labeled extrinsic projections. The native EGFP signal (without antibody enhancement) may be sufficient for studies involving cultured cells or slices. (J Histochem Cytochem 54:539-548, 2006)
V
Of the many available vector types (e.g., adeno-associated virus, herpes simplex virus, lentivirus, pseu-dorabies virus, Sindbis virus), adenovirus vectors have several attractive properties. In particular, they can accept large genetic sequences up to 8 kb, they can be concentrated at up to 1013 pfu/ml easily, and they have a large host range that includes non-replicative cells (Quantin et al. 1992; Le Gal La Salle et al. 1993; Teschemacher et al. 2005). These important features make adenovirus a suitable vector system for introducing a gene of interest into cells. In the CNS, tracing studies that injected adenovirus in the adult rat brain have reported preferential infection of glia cells around the injection site (Akli et al. 1993; Le Gal La Salle et al. 1993; Hermens et al. 1997). Other studies using larger amounts of adenovirus injected into the striatum or cerebellar cortex successfully demonstrated transfected neurons in the appropriate efferent projecting regions in the cerebral cortex, thalamus, substantia nigra, or pontine nuclei (Ridoux et al. 1994; Kuo et al. 1995; Terashima et al. 1997), but the resolution of the signal was inadequate to visualize fine morphological details such as dendritic spines. This was most likely because these studies used adenovirus with LacZ as a reporter and X-gal staining. Otherwise, Golgi-like neuronal filling has been achieved in adult rat cortex by large injections of EGFP-adenovirus tagged with a palmitoylation site, but in the adult the number of neurons visualized has been small (Tamamaki et al. 2000).
Here we report two modifications resulting in improved transport of virus particles and visualization of neurons infected by adenovirus after in vivo injection in the adult brain. These are appropriate survival time (≥2 weeks postinjection) and high concentration NaCl in the injection buffer (≥600 mM). With these conditions, successful retrograde transfection was achieved in both adult rat and monkey brain after injection in several different regions. We used a replication-defective recombinant adenovirus as a vector for the expression of EGFP under the control of the neuron-specific promoter, synapsin (syn) I. Optimal visualization of morphological features also required amplification of the EGFP signals by immunocytochemical staining with an anti-EGFP antibody.
Materials and Methods
Preparation of Recombinant Adenovirus Vectors
The adenovirus vector is based on human adenovirus type 5, rendered replication incompetent by deletion of the E1 and E3 regions of the adenovirus genome. The adenovirus vector AdexSyn-EGFP-WPRE-BGHpoly (A) (AdSynEGFP) expresses enhanced green fluorescent protein (EGFP) under the control of a neuron-specific promoter, syn I. The AdSynEGFP was constructed as follows. The promoter region of mouse syn I, EGFP, WPRE, and BGHpoly (A) were amplified by polymerase chain reaction (PCR) with the following primers: syn, 5′-TAAGCTTCGTGGGAGGGAGTTTCGTT-3′ and 5′-TGCTCACCATGGTGGCGGCG TGGGGCAGGG-3′; EGFP, 5′-CGCCGCCACCATGGTGAGCAAGGGCGAGGA-3′ and 5′-GAGGTTGATTTATGGCTGATTATGATCTAG-3′; WPRE, 5′-AATCAGCCATAAATCAAC CTCTGGATTACA-3′ and 5′-ACAGCAGATCCGATGCGGGGAGGCGGCCCA-3′; BGHpoly (A), 5′-CCCCGCATCGGATCTGCTGTGCCTTCTAGT-3′ and 5′-TCCCCAGCATGCCTGCT ATTGTCTTC-3′. PCR products were purified by agarose gel electrophoresis, and 10 ng of each of two DNA fragments was used as a template for a second round of PCR using only two external primers (Horton et al. 1989,1990). After a third round of PCR, a fragment of syn-EGFP-WPRE-BGHpoly (A) was inserted between the PI-SceI and I-CeuI sites of the modified pBluescript II (SK+). To generate the AdSynEGFP, the fragment digested by PI-SceI and I-CeuI was inserted into BD Adeno-X genome (BD Biosciences; San Jose, CA), and HEK 293 cells were transfected with the PacI-digested BD Adeno-X genome according to the manufacturer's protocol. The AdSynEGFP was purified and concentrated by double-cesium step gradient centrifugation (Hashimoto and Mikoshiba 2003) and dialyzed in 50 mM Tris-HCl (pH 8.1). Dialysis was carried out with three different batches containing different NaCl concentrations (150, 600, 1000 mM NaCl) through Slide-A-lyzer Dialysis Cassette (Pierce; Rockford, IL). The viral suspensions were stored at 4C for up to 1 month after the date of purification. The titers of viral stocks were determined by plaque assay on HEK 293 cells and adjusted to 1.0 × 1012 pfu/ml before the injection.
Producing Anti-EGFP Antibody
For the production of anti-EGFP antibodies, the full coding sequence of EGFP was amplified by PCR by using the pGG16 as a template (Tamamaki et al. 2000). Sequences of the primers used for the PCR were 5′-ATGGTGAGCAAGGGCGAGGA-3′ and 5′-TACATTGATGAGTTTGGACAAACCA-3′. The blunt-ended PCR product was cloned into pBluescript II (SK+) at the EcoRV site, and the plasmid was digested by BamHI and EcoRI and cloned into pGEX-4T1 (Amersham-Pharmacia Biotech; Piscataway, NJ). Glutathi-one-S-transferase (GST)-EGFP fusion protein was induced in Escherichia coli by adding isopropyl-1-thio-beta-D-galacto-pyranoside to the medium. EGFP was purified according to the protocol recommended by the manufacturer of the GST system (Amersham-Pharmacia Biotech). Purified EGFP was emulsified with complete Freund's adjuvant (Difco; Detroit, MI) and injected intracutaneously into two female rabbits (1 mg/animal). Three weeks after the first immunization, the same amount of EGFP emulsified with complete Freund's adjuvant was reinjected into the rabbits. Additional immunizations were performed every 2 weeks thereafter. After the third immunization, serum was taken from the rabbits. Antibodies were affinity purified with EGFP-conjugated Affigel 10 gel (2 mg EGFP/1 ml gel; BioRad, Richmond, CA). Crude-globulin fraction (25 mg) was applied to 1 ml of the antigen column, and the specific antibodies were eluted with 0.1 M glycine-HCl (pH 2.5) and mixed with 1 M potassium phosphate buffer (10:1) to achieve neutral pH. Antibody was stored at 4C with 0.02% NaN3.
Injection of Viruses, Tissue Fixation, and Immunohistochemistry in Rat
For virus, experimental protocols were approved by the Safety Division of the RIKEN Institute and were carried out in biosafety level 2 rooms in accordance with National Institutes of Health (NIH) Guidelines for Research Involving Recombinant DNA Molecules. All experimental animal protocols were approved by the Experimental Animal Committee of the RIKEN Institute and were carried out in accordance with the NIH Guidelines for the Care and Use of Laboratory Animals (NIH Publication No. 80-23; revised 1996). Every effort was made to minimize the number of animals used as well as any pain or discomfort experienced.
Wistar rats (n=32) weighing 200-300 g were deeply anesthetized with chloral hydrate (350 mg/kg body weight). The virus (1.5 μl of 1.0 × 1012 pfu/ml) was injected stereotaxically into the medial prefrontal cortex (coordinates: 2.2 mm anterior; 0.5 mm lateral to bregma; depth, 4.0 mm below the pia, within the gray matter), posterior thalamic nuclear group (coordinates: 4.16 mm posterior; 2.0 mm lateral to bregma; depth, 5.1 mm below the pia), or hippocampus (coordinates: 4.16 mm posterior; 2.0 mm lateral to bregma; depth, 3.0 mm below the pia) (Paxinos and Watson 2004) through glass micropipettes glued onto a Hamilton syringe.
Rats were allowed to survive for 2 to 28 days after the injection (Table 1). They were reanesthetized with chloral hydrate and perfused transcardially with 100 ml of saline, followed by 300 ml of 4% paraformaldehyde in 0.1 M phosphate buffer, pH 7.4. Brains were removed and placed for 2 hr in the same fixative. After cryoprotection with 30% sucrose in phosphate buffered saline (PBS), blocks were cut into 60-μm-thick frontal sections on a freezing microtome. Some of the sections were immediately mounted to observe the fluorescence intensity of EGFP. Other sections were incubated overnight at room temperature with 1.0 μg/ml of an affinity-purified rabbit antibody to EGFP in PBS containing 0.3% Triton X-100 and 1.0% normal goat serum (PBS-TG). After a rinse with PBS, sections were incubated for 1 hr with 5 μg/ml of Alexa-488-conjugated anti-rabbit IgG goat antibody (Invitrogen; Carlsbad, CA) in PBS-TG. Finally, sections were mounted on glass slides, coverslipped with IMMU-MOUNT (Thermo Shandon; Pittsburgh, PA), and observed with either a Zeiss Axioskop2-plus microscope (Zeiss; Oberkochen, Germany) or a Leica TCS SP2 AOBS confocal laser microscope (Leica; Heidelberg, Germany).
Number of days postinjection and numbers of rats injected with recombinant adenovirus
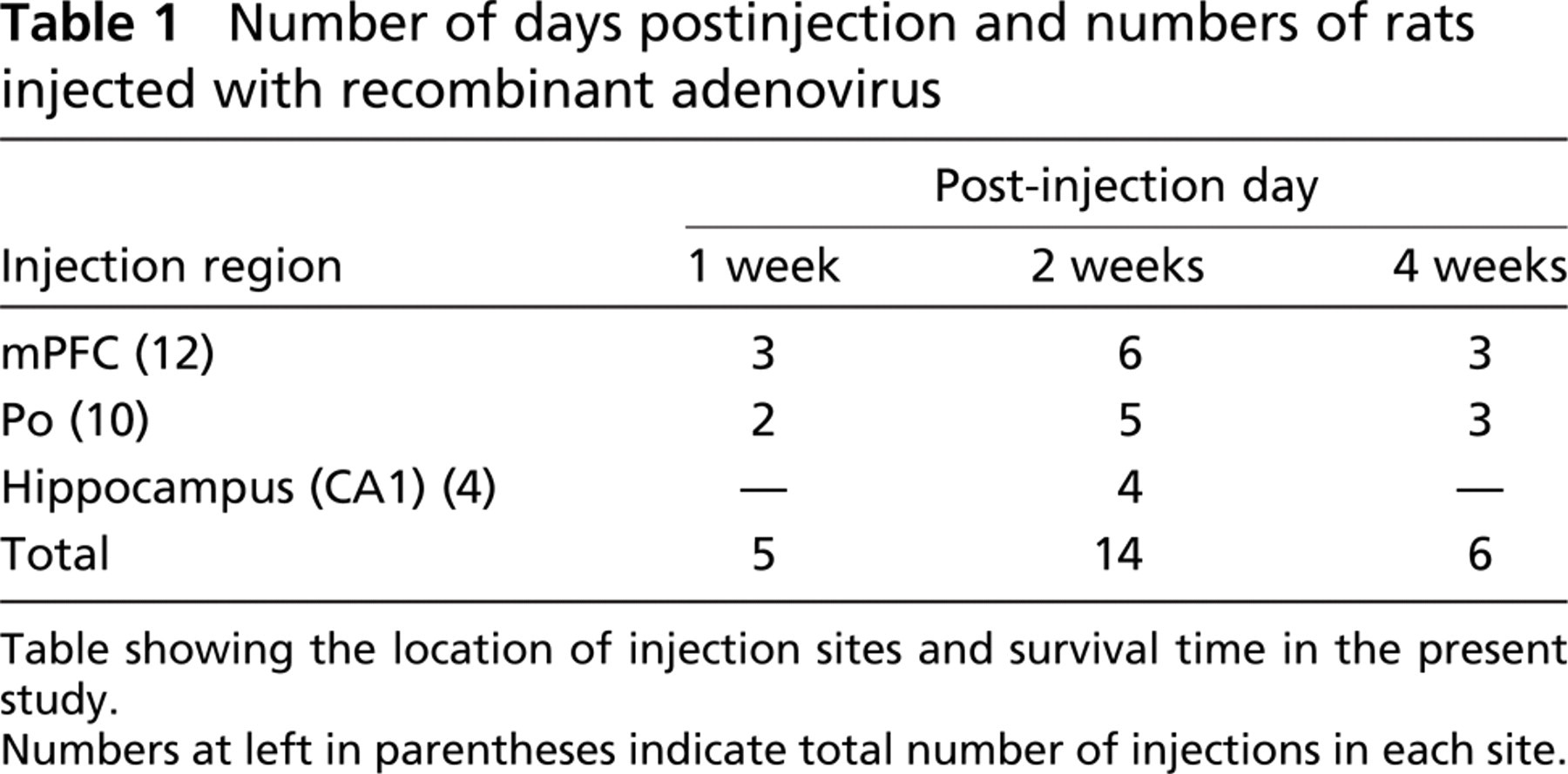
Table showing the location of injection sites and survival time in the present study.
Numbers at left in parentheses indicate total number of injections in each site.
Injection, Tissue Fixation, and Immunohistochemistry in Monkey
One rhesus monkey (3.67 kg) was anesthetized with ketamine (11 mg/kg, i.m.) and nembutal (35 mg/kg, i.p.) and prepared for sterile surgery. An opening was made in the skull over the estimated position of the lunate sulcus, and a durotomy was performed to expose the prelunate gyrus. Two injections of virus (in 150 mM NaCl or 600 mM NaCl) were made in area V4 (7 mm apart). Volumes of 1.5 μl were injected through a Hamilton syringe. In between, a third injection (1.5 μl) was made of 10% biotinylated dextran amine (BDA, 1:1 mixture of 3000 and 10,000 MW in 0.0125 M PBS; Molecular Probes, Eugene, OR). After a postinjection survival of 22 days, the monkey was reanesthetized with ketamine and nembutal (overdose, 75 mg/kg, i.p.) and perfused transcardially in sequence with saline containing 0.1% sodium nitrite for 2 min, 41 of 4% paraformaldehyde, and chilled 0.1 M phosphate buffer with 10%, 20%, and 30% sucrose (500 ml each). The brain was removed, trimmed to a 4-cm block including the injection site, and allowed to sink in 30% sucrose for cryoprotection.
Two days later the tissue was sectioned at 60 μm. As for rat tissue, some sections were immediately scanned for fluorescence, but most of the tissue was reacted with antibody for immunofluorescence or 3,3′ diaminobenzidine tetrahydrochloride (DAB)-immunohistochemistry. For immunofluorescence, sections were incubated 40-48 hr in the cold room with 2.0 μg/ml of anti-EGFP in PBS containing 0.5% Triton X-100 (PBS-X) with 5% normal goat serum. After a rinse with PBS, sections were incubated for 1.5 hr at room temperature with 5 μg/ml of Alexa-488-conjugated anti-rabbit IgG goat antibody for EGFP and 10 μg/ml Alexa-594-conjugated streptavidin for BDA in PBS-X with 2% normal goat serum. For DAB-immunohistochemistry, tissue was reacted for 20-24 hr in avidin-biotin complex (ABC Elite Kits; Vector Laboratories, Burlingame, CA) at room temperature (one drop of reagent per 7 ml of 0.1 M PBS). In the final step, BDA was demonstrated by DAB-histochemistry with the addition of 0.03% nickel-ammonium sulfate. Processing for EGFP was continued using anti-EGFP (1 μg/ml in PBS-X with 2% normal goat serum), rabbit biotinylated anti-goat secondary antibody (1:200 in 0.1 M PBS-X with 2% normal goat serum; Vector Laboratories), and ABC complex as described for BDA. Finally, EGFP was demonstrated by DAB-histochemistry without nickel-ammonium sulfate.
Results
Pressure injections of AdsynEGFP consistently resulted in strong EGFP expression in retrogradely connected neurons, as assayed by injections in adult rats in three different structures, namely, medial prefrontal cortex (mPFC), posterior thalamic nuclear group (Po), and the CA1 hippocampal field. The quality of labeling was judged by (a) intensity of fluorescence, (b) expression of EGFP in minute structures such as dendritic spines and axon collaterals, and (c) the presence of infected neurons in structures known from other techniques to project to the injected region. In all cases, processing for immunofluorescence was more sensitive than EGFP fluorescence (compare Figure 1 and Figure 2 with Figure 3 and Figure 4). In addition, two technical parameters described below were found to be critical for optimum expression of EGFP. Initial results in monkey are also reported.
Technical Parameters
Optimal conditions were assayed by EGFP fluorescence in rats. This was strongly affected by the concentration of NaCl in the buffer solution (Figure 1). At 150 mM NaCl, only weak expression could be detected except for the region immediately around the injection. At 600 mM NaCl, by contrast, many neurons were prominent in regions projecting to the injection site, and these strongly expressed EGFP. At 1000 mM NaCl, however, no further change could be detected.
Expression was also clearly influenced by the post-injection survival time (Figure 2). At intervals of 2-7 days, EGFP fluorescence could be detected in only a few neurons projecting to the injection site. With 14-day survival, expression level was conspicuously stronger, and signal could be verified in many neurons. No further change was apparent up to 28 days postinjection.
Evaluation of Injection Sites
Injections were standardized to a volume of 1.5 μl of 1.0 × 1012 pfu/ml. Actual size of the injection site in histological tissue varied somewhat, partly depending on the different structures. For both rats and the monkey, the core injection size was judged as 0.8-1.0 mm in diameter. This was based on intensity of label around the injection track and comparison of fields of projecting neurons with previous reports in the literature. In the case of cortical injections, the injection site usually had a cylindrical shape, where 0.8-1.0 mm was the smaller diameter (parallel to the surface of the brain). The needle tip was aimed for the middle of the cortical thickness so that the injection might span layers 1-6. For some injections, especially those in rat hippocampus, there was evidence of glia labeling in the gray matter, but overall this was negligible or absent.
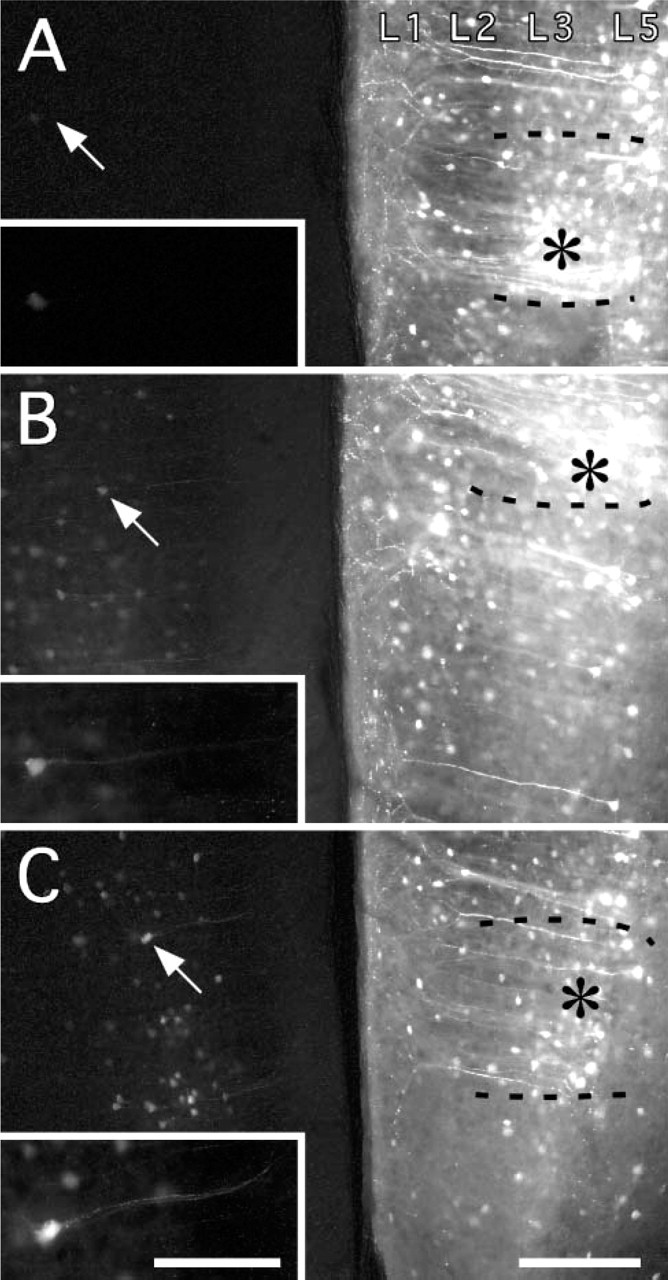
Intensity of enhanced green fluorescent protein (EGFP) fluorescence as a function of NaCl concentration (no immunoreaction). Injection of adenovirus into the right medial prefrontal cortex in three rats (2 week postinjection survival time). (
The connectivity of all three injected regions (mPFC, Po, and CA1) has been extensively investigated by conventional tracers, both anterograde and retrograde. Thus, in comparison with previous reports, it was feasible to evaluate the results in terms of whether expression occurred in all expected regions and with what density and pattern. Good accord was found between previously reported results based on conventional tracers and on the results from EGFP expression. These are briefly summarized below.
Evaluation of Projection Sites in Rats
The injections in mPFC (n=12) produced abundant EGFP-expressing neurons around the injection site. These could be identified as mainly pyramidal neurons with some interneurons. Labeled neurons in layer 1 could be inferred to be interneurons because of their location, and some putative interneurons could be confidently identified as such from their obvious bipolar dendritic arbor.
EGFP-expressing neurons were detected in distant sites (for comparison, see Takagishi and Chiba 1991; Conde et al. 1995). These included ventromedial and mediodorsal thalamic nuclei (Figure 3), CA1 in the ventral region of the hippocampus, the basolateral nucleus of the amygdala, and the nucleus of the horizontal limb of the diagonal band. Cortical areas with EGFP-expressing neurons included entorhinal, insular, cingulate, and perirhinal cortex (Figure 3C); secondary visual cortex (medially); and contralateral mPFC (Figure 1-Figure 3). Cortical neurons were pyramidal in shape in most areas, but extraverted neurons were recognizable in the cingulate, perirhinal, and entorhinal areas in the upper layers.
Injections in the posterior thalamic nuclear group (n=11) produced abundant transfected neurons in cortical areas, namely, somatosensory, frontal, and perirhinal cortex (Figure 4). Pyramidal neurons in layer 6 were densely labeled along with scattered neurons in layer 5, consistent with previous reports (Veinante et al. 2000). EGFP-expressing neurons were also found in the reticular nucleus of the thalamus, in the zona incerta, and in the deep layers of the superior colliculus as previously reported (Roger and Cadusseau 1984; Raos and Bentivoglio 1993).
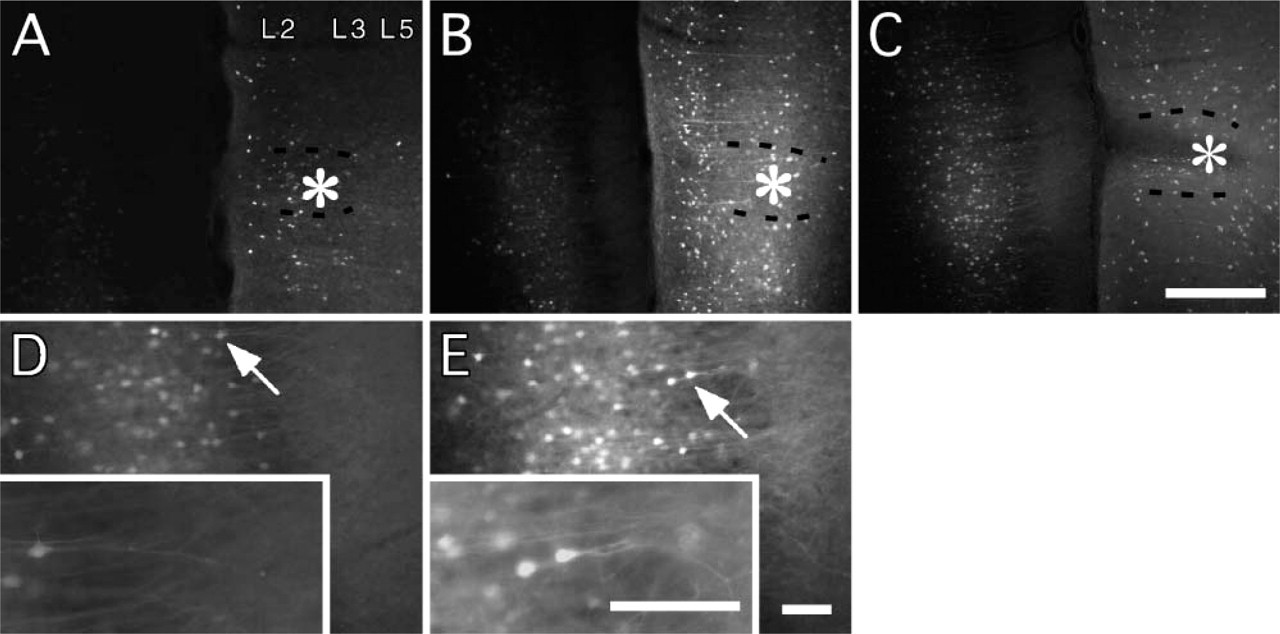
Intensity of EGFP fluorescence as a function of postinjection survival time (no immunoreaction) Adenovirus (600 mM NaCl) was injected into the right medial prefrontal cortex of three rats. (
Hippocampal injections were situated in the dorsal part of CA1 and adjoining dentate gyrus. EGFP-expressing neurons were obvious in the contralateral CA3 (Swanson et al. 1978; Zappone and Sloviter 2001). In the lateral entorhinal cortex, EGFP was strongly expressed by many neurons in layer 2 and a few in layer 3. The characteristic stellate dendritic tree of the neurons in layer 2 was easily recognized, as were the apical and basal dendrites of pyramidal neurons in layer 3.
EGFP expression was intense enough to visualize dendritic spines in the distant areas after using anti-EGFP antibody (Figure 3 and Figure 4). Although the visualization of spines was overall excellent, at the finest level we cannot be sure that all neurons were equally well labeled (Figure 1 and Figure 2). Some variability might be expected because of cell type specificity, even within the broad class of pyramidal projection neurons. In addition, the amount of virus might not be the same in all neurons. For example, neurons with terminations at the edge of an injection might have less virus than those projecting to the injection core.
Evaluation of Projection Sites in Monkey
Of the two injections in area V4 (mid- to ventral prelunate gyrus), the ventral one with 600 mM NaCl was effective, whereas the dorsal one with 150 mM NaCl was less so and resulted in little or no transfection of retrogradely projecting neurons. This is consistent with our results from the rat experiments. Many infected neurons occurred for 2-3 mm in the vicinity of the ventral injection site. Interneurons were less evident than after the cortical injections in rat but may have been overlooked because of lack of dendritic filling or because of the heavy labeling within the injection core.
EGFP-expressing neurons were detected in multiple distant sites, consistent with previous reports on connections to area V4 (Tanaka et al. 1990; Nakamura et al. 1993; Felleman et al. 1997; Amaral et al. 2003; Stepniewska et al. 2005). In particular, neurons were visible subcortically in the pulvinar nucleus (Figure 5), amygdala, and claustrum. Cortically, EGFP-expressing neurons occurred in visual areas V2 and V3; in temporal areas TEO, posterior TE, and perirhinal cortex (Figure 6); and in the posterior superior temporal sulcus. In temporal areas, neurons were mainly in layers 5 and/or 6. No callosally projecting neurons were detected. Because ipsilateral infection was successful for neurons in the amygdala and claustrum (∼7-10 mm from the injection site), the apparent failure of infection of callosally projecting neurons may indicate some cell type specificity. Alternatively, the injection may have been in an acallosal subregion of area V4 (Van Essen et al. 1982).
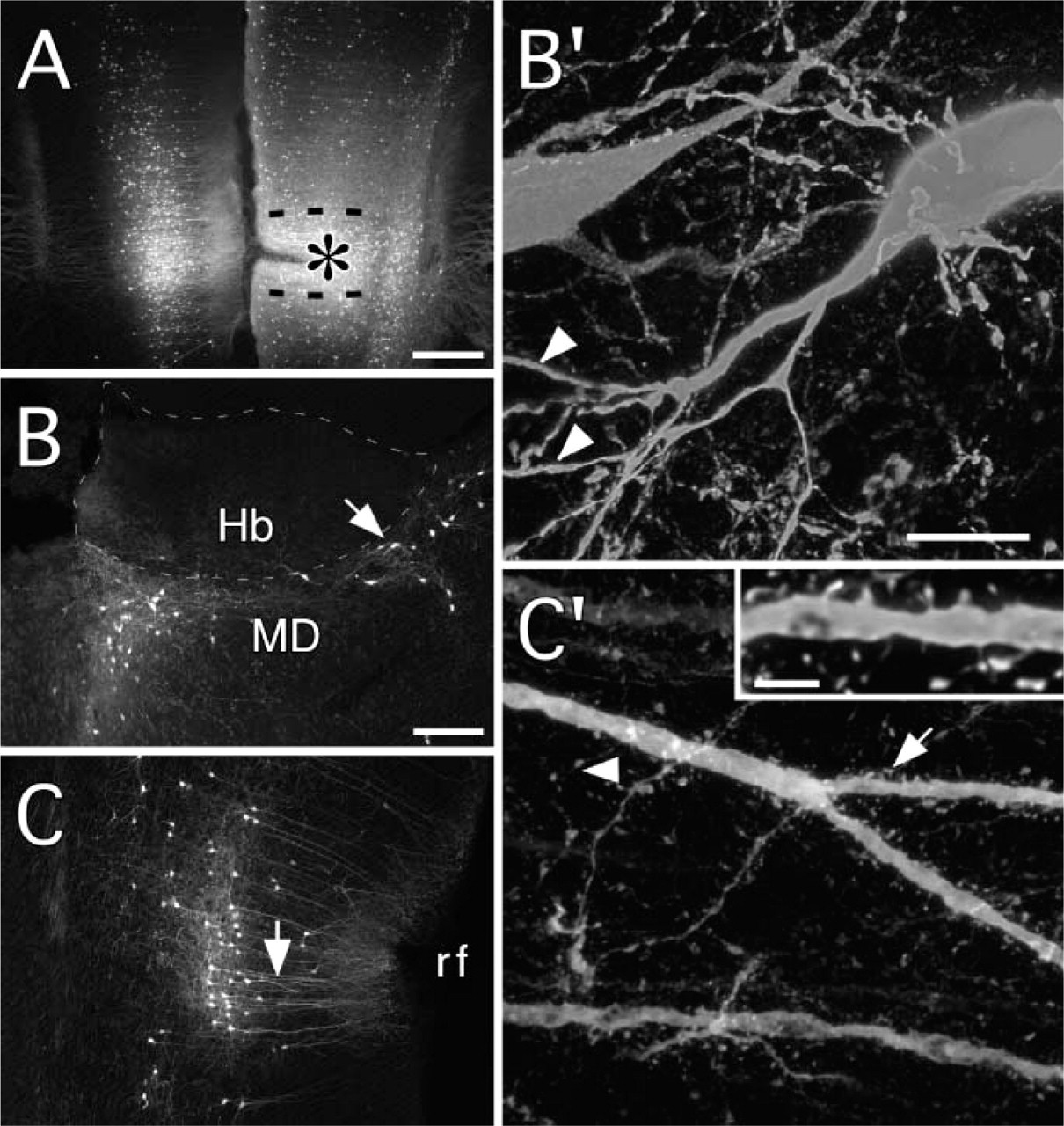
EGFP expression in thalamic and cortical neurons retrogradely infected after an injection of adenovirus in the right medial prefrontal cortex (survival = 14 days; 600 mM NaCl; processing by immunofluorescence). (
Axon Labeling
In addition to the Golgi-like dendritic labeling (Figure 3 and Figure 4), EGFP-expressing axons were also apparent as collaterals of the main axon (especially evident in cortical areas) and in the white matter. Clearly delineated axon bundles were evident in the vicinity of the injection sites (data not shown). We could not, however, detect clear evidence of what would be anterograde axons originating from neurons in the injection site. In the rat experiment, after thalamic injections no obvious fibers or terminations were seen in cortical layer 4. After the injections involving CA1, no obvious fibers or terminations were found in the hippocampal-recipient, deeper layers of entorhinal cortex. After cortical injections, no obvious EGFP-expressing fibers or terminations were found in the superior colliculus, caudate nucleus, claustrum, or amygdala, or in recipient cortical targets. In the monkey experiment, however, what appeared to be corticopulvinar terminations could be found in immunofluorescent and immunoperoxidase material. This could be an example of selective anterograde filling from neurons in the injection site or, alternatively, of EGFP expression through axon collaterals of transfected pyramidal neurons in layer 5 near the injection site or even in a distant cortical area. We favor the latter interpretation (of EGFP expression through axon collaterals), as this is most consistent with the other results. Thus, we suggest that axonal labeling is due to the Golgi-like labeling by EGFP of both the dendritic and axonal arbors. EGFP-expressing terminations in the pulvinar were considerably fewer than terminations labeled by true anterograde transport of BDA, also injected in area V4.
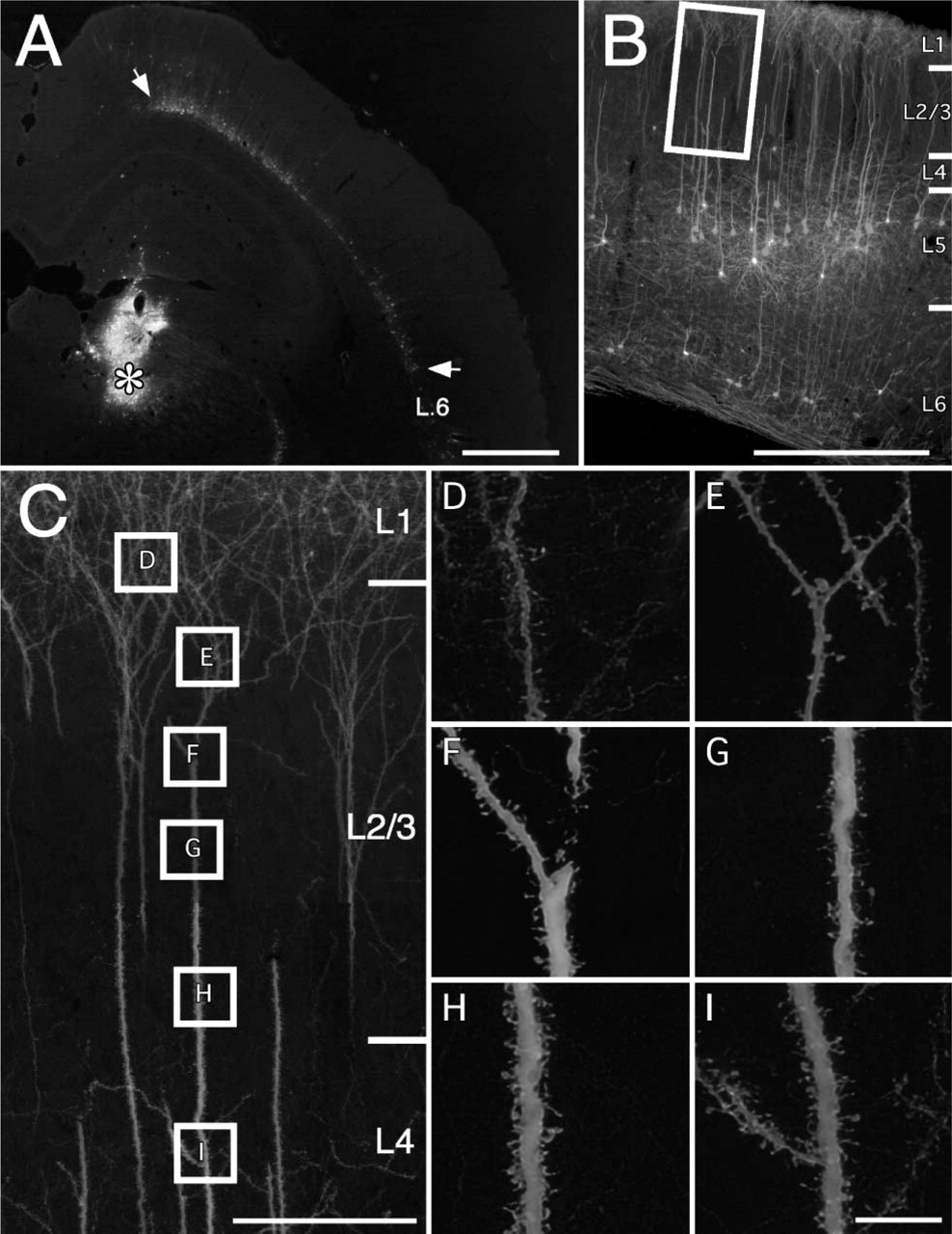
EGFP expression after adenovirus injection into the right posterior thalamic nuclear group (survival = 14 days; 600 mM NaCl; processing by immunofluorescence). (
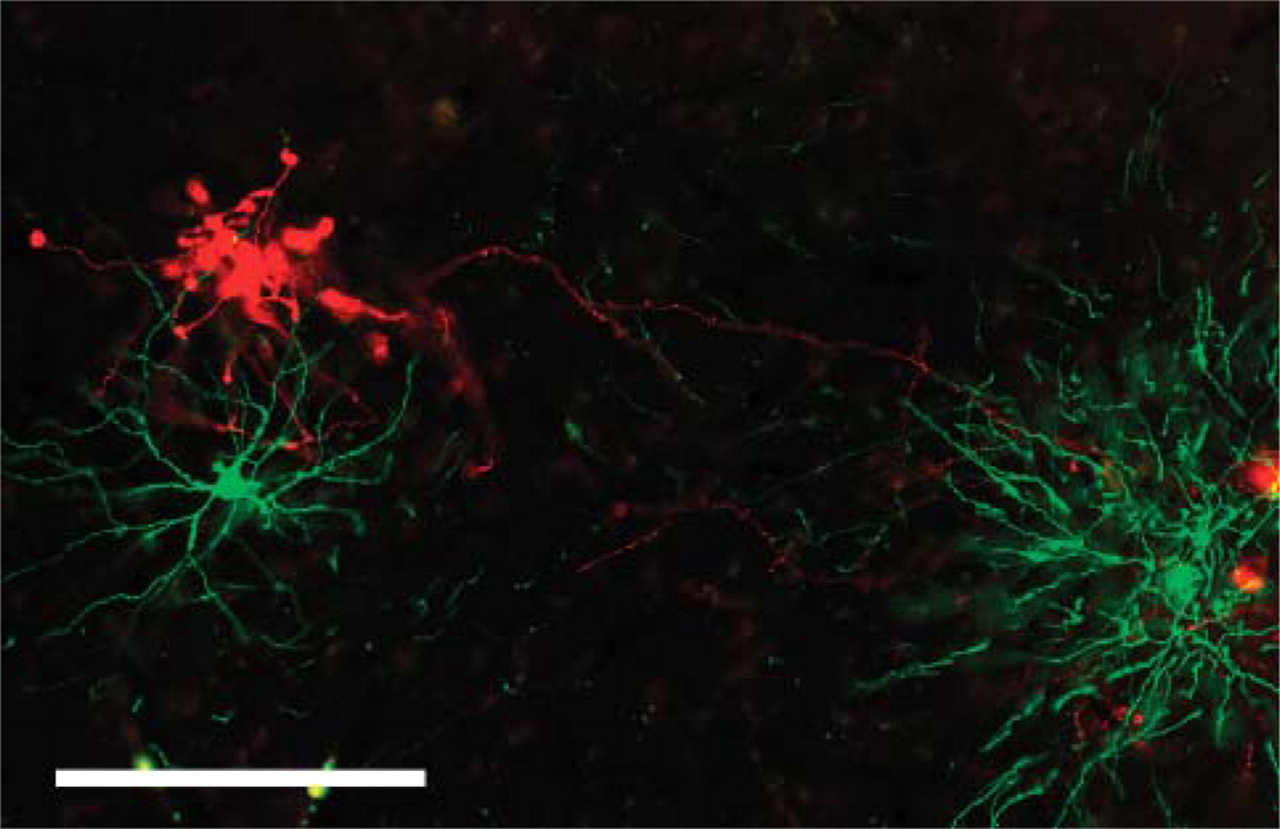
Double immunofluorescence for corticopulvinar terminations (in red), anterogradely labeled by a biotinylated dextran amine (BDA) injection in area V4 of monkey and two pulvinocortical neurons (in green), transfected by adenovirus, also injected in area V4. Bar = 200 μm.
Discussion
We have used a replication-defective recombinant adenovirus as a vector for the expression of EGFP under the control of the neuron-specific promoter syn I. The number of EGFP-expressing neurons and visualization of Golgi-like detail were significantly improved by using an antibody to EGFP (Tamamaki et al. 2000; Furuta et al. 2001; Dittgen et al. 2004), by using longer postinjection survival times, and by adding higher concentrations of NaCl (≥600 mM) to the virus solution. The enhanced labeling after antibody amplification, as compared with direct EGFP fluorescence, is expected. The effect of the high osmolarity NaCl is harder to evaluate, but we suggest this is not due to biological factors such as receptor-mediated endocytosis. For conventional tracers, putative disruption of synaptic or axonal membranes by detergents (FitzGib-bon et al. 1983), by N-methyl-D-aspartate (NMDA) (Jiang et al. 1993), or by low pH buffer (Kaneko et al. 1996) has been used to result in greater efficacy of uptake (e.g., horseradish peroxidase or tetramethyl-rhodamine-dextran amine). For viral particles, however, it is more likely that the high concentration of NaCl acts by countering the normally repulsive interaction of the negatively charged cell membrane with the negatively charged adenovirus particles. Similarly, complexes of adenovirus with cationic lipids have been reported to increase the efficiency of gene transfer (Fasbender et al. 1997). Another previously reported method for enhanced binding efficiency is a polylysine addition to the C-terminal end of the adenovirus fiber protein (Wickham et al. 1996). Enhanced attachment of viral particles may also explain the effectiveness of EGFP expression in mature neurons. Unlike in young animals, neurons in the adult brain have lower levels of coxsackievirus and adenovirus receptor, and this has been considered a problem leading to less effective infection by adenovirus at older stages (Fechner et al. 1999; Tomko et al. 2000).
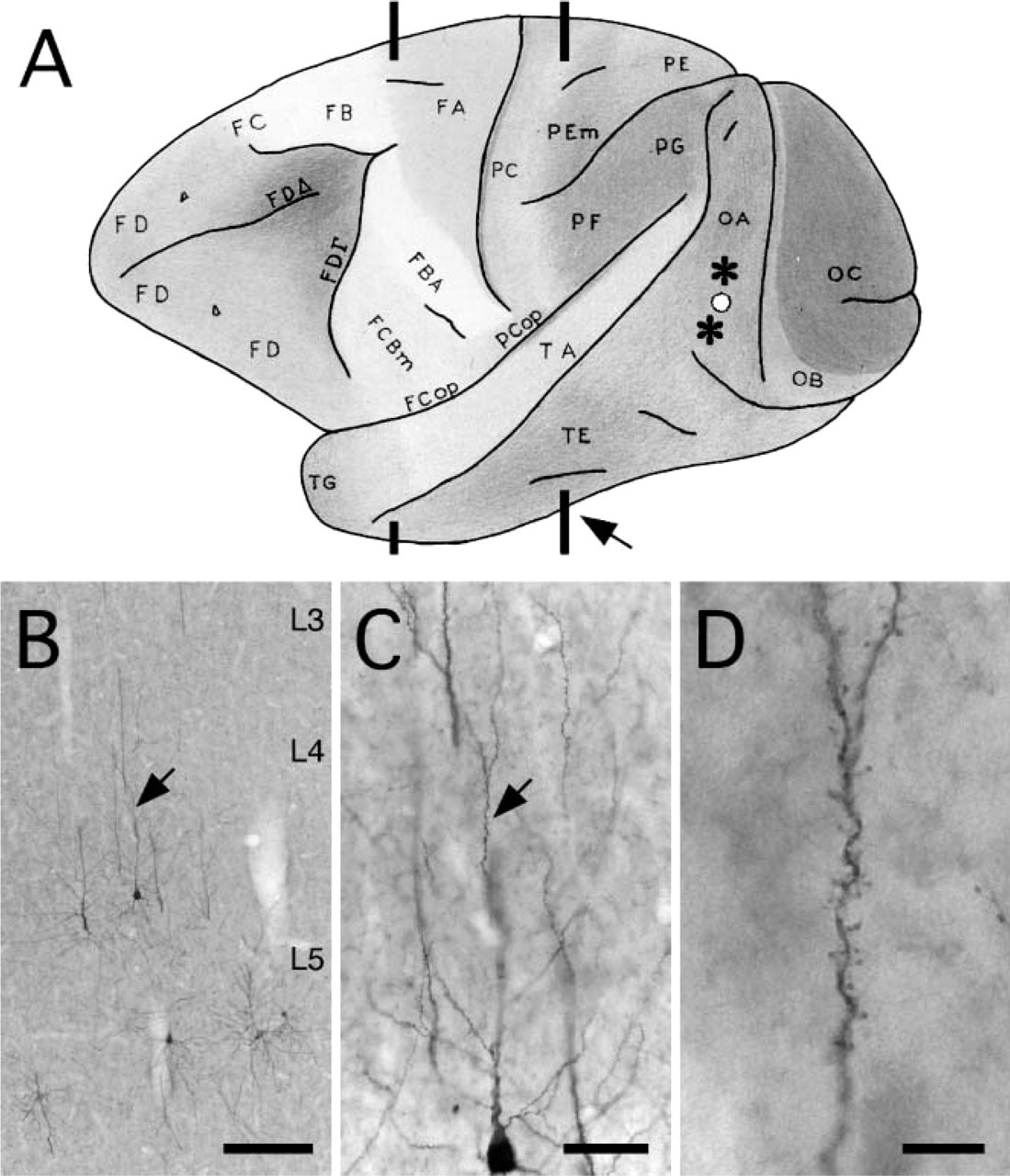
(
This AdSynEGFP is suitable for several applications. First, AdSynEGFP can be considered for the same connectivity purposes as standard retrograde tracers such as HRP, WGA-HRP, or fluorescent dyes. We note, however, that although we found good agreement between the occurrence of EGFP-expressing neurons and what would be expected from previous reports in the literature, it is still possible that the adenovirus is infecting only subpopulations of afferent neurons. In particular, in our single experiment in monkey, we found no evidence for callosally projecting neurons after an injection in area V4. Although some parts of V4 are acallosal (Van Essen et al. 1982), from the size and location of the injection we might have predicted some callosal labeling. Thus, as retrograde tracer, this AdSynEGFP should be regarded as complementary to standard tracers and used for the express advantage of the consistent Golgi-like filling.
Second, because a major advantage of this AdSyn-EGFP is that it produces Golgi-like detail in connectionally identified neurons, it can be used in microcircuitry studies in combination with other immunofluorescently tagged elements including extrinsic projections anterogradely labeled by BDA Figure 5; and see as examples, Wouterlood et al. 2003; Freese and Amaral 2005; Grinevich et al. 2005). In the case of cortical studies, there is a major need to define connectional inputs in relation to postsynaptic neurons that are specifically identified by layer, dendritic morphology, and projectional targets. For long distance connections and in vivo experiments in particular, conventional techniques have often not provided Golgi-like resolution. However, electron microscopy and/or intracellular filling, both of which can offer excellent resolution, are labor intensive and generally result in very small numbers of sampled neurons.
Third, these improvements (high osmolarity NaCl and longer postinjection survival) can be generally applied to adenovirus vectors to achieve consistent and reliable transfection of retrogradely connected neurons. Thus, exogenous genes can be delivered into predetermined areas in vivo to retrogradely infect neurons in distant interconnected sites. These distant, projecting neurons should remain healthy, in contrast with those at the injection site itself that are liable to experience inflammation or other cytotoxicity induced by endocytosis in the environment of high titer adenovirus. Viability and high-expression level of neurons infected by our AdSynEGFP have been ascertained as at least 28 days in rats and 22 days in monkey.
Finally, although antibody amplification is needed to achieve Golgi-like filling, direct EGFP fluorescence is sufficient for identifying infected cells (Figure 1 and Figure 2) including portions of their dendritic arbor and would be suitable for prolonged gene expression in cultured neurons or cultured slices, as well as in vivo applications.
Footnotes
Acknowledgments
This work was supported by research funds from RIKEN Brain Science Institute, Saitama, Japan.
The authors thank Dr. Teiji Chihara and the chemical analysis team of RIKEN for methodological discussion, Dr. Mitsuhiro Hashimoto for help with adenovirus purification and helpful discussions, Ms. Hiromi Mashiko, Ms. Yoshiko Abe, Mr. Adrian Knight, and Mr. Daniel O. Potapov for excellent technical assistance; Ms. Michiko Fujisawa for help with manuscript preparation; and Drs. Noritaka Ichinohe and Toshio Miyashita for critical reading of the manuscript and helpful discussion.
