Abstract
Autocrine motility factor (AMF) stimulates, via an autocrine route, the motility of cancer cells. The current study investigated the expression of AMF and its receptor, AMFR (gp78), in breast cancer and attempted to dissect a clinical link. Breast tumor tissues (n=120) and non-neoplastic normal tissues (n=32) were studied. AMF and AMFR distribution in tissues were assessed using immunohistochemistry and their transcripts were analyzed using RT-PCR and quantitative PCR. Median follow-up of the cohort was 10 years. Normal mammary epithelial cells, but not stromal and endothelial cells, weakly stained for AMF and AMFR. However, cancer cells showed stronger staining. Both AMF and AMFR transcripts were significantly higher in tumor than in normal tissues (p=0.003 and p=0.0001, respectively). High levels of AMF and AMFR were seen in patients who died of breast cancer (p=0.049, p=0.0435) and high AMF was also seen in patients who had local recurrence (p=0.039) compared with those who remained disease free. A significant correlation was seen between long-term survival and the AMFR:CK19 ratio, in which patients with high AMFR:CK19 ratio tumors had a significantly shorter survival (101.0 months, 80.6–121.4) compared with those with low ratio (136.0 months, 123.7–148.2), p=0.0331. In conclusion, AMF and AMFR are overexpressed in human breast cancer and are negatively associated with patients' clinical outcome. This strongly indicates that the AMF–AMFR complex plays an important role in the progression of breast cancer, as well as having a prognostic role.
A
AMF receptor (AMFR) is a 78-kDa glycoprotein (gp78) (Watanabe et al. 1991; Huang et al. 1995). The AMFR gene encodes a 323-amino acid polypeptide that has a single transmembrane domain and several putative glycosylation sites. The protein sequence has some homology to human p53. AMFR is distributed evenly across the membrane of normal cells, has a polar cap distribution in papilloma cells, and is discretely localized to the leading and trailing edges of migrating carcinoma cells. Epithelial cells, including cancer cells, had increased motility in response to AMF (Silletti et al. 1993). In gastric cancer, the level of protein expression was associated with the pathologic stage, grade, and degree of tumor penetration to surrounding tissues. Positive AMFR expression was significantly associated with poor prognosis (Hirono et al. 1996).
AMF also protects cancer cells from developing apoptosis and regulates cell growth by activating Apaf-1 and caspases (Haga et al. 2003) and upregulation of cyclin/cyclin-dependent kinase activities and down-regulation of p27Kip1 (Tsutsumi et al. 2003). The signaling pathways downstream of AMFR include protein kinase CK2 (Yanagawa et al. 2005), Rho family regulators, the rho GDP dissociation inhibitor beta, and kinesin motor 3A (Tsutsumi et al. 2002; Yanagawa et al. 2004). In addition, AMF also regulates cell–matrix adhesion via activation of focal adhesion kinase (Silletti et al. 1996).
AMF-induced motility also involves the loss of E-cadherin, potentially through the upregulation of E-cadherin transcription suppressor, SNAIL protein (Tsutsumi et al. 2004) and also involves 12-lipoxygenase (12-LOX) (Silletti et al. 1994). AMF may also act as an angiogenic factor (Funasaka et al. 2001). In the past decade, AMF and its receptor have been studied in a number of tumor types. In malignant melanoma cells (Timar et al. 2002), pancreatic cancer cells, oral squamous cell carcinoma cells (Niinaka et al. 2002), and liver cancer cells (Torimura et al. 2001), expression of AMF and AMFR was associated with invasion and progressive disease. Using an orthotopic model, it has been shown that pancreatic cancer cells, when transfected with AMF, grew large tumors with frequent liver metastasis (Tsutsumi et al. 2004). Together these findings indicate that AMF has a diverse role in the behavior of cancer cells, including regulation of cell motility, cell adhesion, and cell growth. The action of AMF, mediated by AMFR, involves a number of intracellular signaling pathways.
Several studies have reported that expression of AMF and/or AMFR has important clinical implications. AMFR has prognostic value in clinical gastric cancer (Hirono et al. 1996), non-small cell lung cancer (Takanami et al. 2001), melanoma (Timar et al. 2002), and colorectal cancer (Nakamori et al. 1994). For example, high levels of AMFR were associated with nodal metastasis and long-term and overall survival (Takanami et al. 2001). AMF expression is influenced by therapeutic agents such as the epidermal growth factor receptor (EGFR) antibody, Herceptin, which can inhibit the expression of AMF transcript and protein in cancer cells (Talukder et al. 2002). However, there has been no report on the expression of AMF and its receptor in human breast cancer cells or their role in determining clinical outcome in breast cancers.
Breast cancer is the leading female cancer in the UK and in the US, affecting 1 in 10 women. Metastasis is the main cause of death in patients with breast cancer (Ciatto et al. 1988). Development of metastasis involves a series of cellular events from cell–cell dissociation at the primary site, matrix degradation and invasion, intravasation and extravation, and resettling in a new site where cells will re-grow as secondary (metastatic) tumors, supported by angiogenesis (Stracke et al. 1991. During this complex process, cell motility is critical for a successful completion. A few protein factors are known to affect the motility of cancer cells and are referred to as motility factors or motogens (Gherardi and Stoker 1991). Well-documented motogens include hepatocyte growth factor (HGF; also known as scatter factor), autotoxin, motility-stimulating factor (MSF), and AMF. In vitro, these motility factors are known to increase cellular motility, disrupt cell–cell adhesion, and assist the breach of tissue barriers including the basement membrane. Some of these factors, predominantly HGF, are also highly active in inducing angiogenesis (Jiang et al. 2003a, 2005a). The clinical importance of some of the motogens in clinical breast cancer has been demonstrated in the past. HGF and its receptor are highly expressed in breast tumors and are linked to nodal spread and long-term survival of the patients (Yamashita et al. 1994; Toi et al. 1998; Parr et al. 2004). Although AMF has been shown to be important in certain solid tumors, little information is available with regard to AMF and AMFR in clinical breast cancer. Furthermore, it is not known if AMF and AMFR are linked to clinical outcome and survival in patients with breast cancer.
In the current study we examined the expression profile of AMF and its receptor at protein and mRNA levels in a cohort of fresh tissues from patients with breast cancer in order to establish the role of AMF and its receptor in the progression and long-term outcome of breast cancer. We report here that AMF and AMFR are highly expressed at protein and mRNA levels in human breast tumor tissues and have significant correlation with the aggressive nature of breast cancer and clinical outcomes of the patients.
Materials and Methods
Primary breast cancer tissues (n=120) and normal tissues that were away from tumor tissues and free from cancer cells (n=32) were collected immediately after surgery (with approval of the local ethical committee) and stored in the deep freezer until use. Patients were routinely followed clinically after surgery. The median follow-up period was 120 months. The presence of tumor cells in the collected tissues was verified by examination of frozen sections using hematoxylin–eosin staining. Details of the samples are given in Table 1. A rabbit anti-human AMF and a monoclonal antibody to AMFR were used as previously described (Nabi et al. 1990; Niinaka et al. 1998). Anti-cytokeratin 19 (CK19), anti-Ki67, and peroxidase-conjugated anti-IgG were from Santa Cruz Biotechnologies (Santa Cruz, CA) and Sigma-Aldrich Co. (Poole, UK), respectively.
RNA Preparation and RT-PCR
Frozen tissues were sectioned using a cryostat. Approximately 25 frozen sections from each tissue sample (120 tumor and 32 normal tissues) (8 μm in thickness) were combined and homogenized in an RNA extraction buffer (AbGene Ltd; Surrey, UK), using a hand-held homogenizer. Following a routine extraction procedure, RNA was washed and quantified using a spectrophotometer (Wolf Laboratories; York, UK). cDNA was synthesized using a first-strand synthesis with an oligo dt primer (AbGene Ltd). PCR primers for AMF are 5′-ttttgacaacttcgagcag-3′ and 5′-actgaacctgaccgtacaatg-taccagatacccagcag-3′; for AMFR, 5′-cctacacagcggtcagatag-3′ and 5′-actgaacctgaccgtacaagcagaagtttctccctctt-3′. PCR was performed using sets of primers with the following conditions: 5 min at 95C, 20 sec at 94C, 25 sec at 56C, 50 sec at 72C for 36 cycles, and finally 72C for 7 min. β-actin was amplified simultaneously using the following primers: 5′-gctgatttgatggagttgga-3′ and 5′-tcagctacttgttcttgagtgaa-3′. PCR products were then separated on a 0.8% agarose gel, visualized under UV light, photographed using a Unisave camera (Wolf Laboratories), and documented with Photoshop software (Adobe; San Jose, CA).
Quantitative Analysis of AMF and AMFR
Levels of AMF and AMFR transcripts from the above-prepared cDNA were determined using a real-time quantitative PCR based on the Amplifluor (Chemicon; Temecula, CA) technology modified from a previously reported method (Nazarenko et al. 1997; Jiang et al. 2003a). Briefly, pairs of PCR primers were similarly designed using the Beacon Designer software (version 2; Palo Alto, CA), but to one of the primers, a further sequence known as the Z sequence (5′-actgaacctgaccgtaca-3′), which is complementary to the universal Z probe (Intergen Inc.; Oxford, UK), was added. A Taqman detection kit for β-actin was purchased from Perkin-Elmer (Norwalk, CT). The reaction was carried out using IcyclerIQ (Bio-Rad; Hercules, CA) which was equipped with an optic unit that allows real-time detection of 96 reactions using the following conditions: 94C for 12 min, 50 cycles of 94C for 15 sec, 55C for 40 sec, and 72C for 20 sec. The levels of the transcripts were generated from a standard that was simultaneously amplified with the samples. CK19 was used as a control for difference in epithelial cellularity during the analysis, as CK19 has been shown to be a stable marker for epithelial cells and epithelial-derived cancer cells, including mammary epithelial cells and breast cancer cells (Bratthauer et al. 2003; Benoy et al. 2004). Primers for CK19 were 5′-caggtccgaggttactgac-3′ and 5′-actgaacctgaccgtacacactttctgc cagtgtgtcttc-3′, respectively (Jiang et al. 2004, 2005b). The levels of the AMF and AMFR transcripts are shown here as the number of copies and as the ratio of AMF or AMFR vs CK19.
Clinical information

Immunohistochemical Staining of AMF and AMFR
Frozen sections of tissues (32 paired normal and 32 matched tumor tissues, as well as dissected tumor tissues) were cut at a thickness of 6 μm using a cryostat. The sections were mounted on super frost plus microscope slides, air dried, and then fixed in a mixture of 50% acetone and 50% methanol for 15 min. Staining for each molecule was conducted on all slides at the same time in a single batch to avoid variance in experimental conditions. The sections were then placed in Optimax wash buffer (a Tween-20-containing washing buffer (Sigma) for 5 −10 min to rehydrate. Sections were incubated for 20 min in 10% horse serum of blocking solution and probed with the primary antibody (used at a dilution of 1:20 for anti-AMF and 1:10 for anti-AMFR, for 60 min). Primary antibodies were omitted in the negative controls. Following extensive washings, sections were incubated for 30 min in the secondary biotinylated antibody (multilink swine anti-goat/mouse/rabbit immunoglobulin; DAKO, Carpinteria, CA). Following washings, avidin–biotin complex (Vector Laboratories; Burlingame, CA) was then applied to the sections followed by extensive washings. Diaminobenzidine chromogen (Vector Laboratories) was then added to the sections that were incubated in the dark for 5 min. Sections were then counterstained in Gill's hematoxylin and dehydrated in ascending grades of methanol before clearing in xylene and mounting under a coverslip.
Statistical analysis was carried out using Mann–Whitney U test and significant difference was taken at p<0.05. Survival was analyzed using Kaplan–Meier survival curve on SPSS 12 (SPSS UK Ltd; Woking, UK).
Results
Distribution Pattern of AMF and AMFR in Mammary Tissues
We first stained normal mammary tissues to visualize the location and levels of AMF and AMFR proteins. As shown in Figure 1, normal mammary epithelial cells showed weak, predominantly cytoplasmic staining for AMF. The staining in stromal and endothelial cells was negative to weak (Figure 1, left panel). In tumor tissues, however, the staining of AMF was different (Figure 1, right panel). First, tumor cells showed a stronger staining for AMF in the cytoplasm and also in the nucleus. Two more features were seen in tumor tissues: staining of stromal cells and positively stained vascular endothelial cells (Figure 1, right panel). The endothelial cells showed strong cytoplasmic staining and to some degree nuclear staining. The positive staining was also seen in larger vascular vessels in tumor tissues.
AMFR was seen in both normal mammary epithelial cells (Figure 2, left panel) and tumor cells (Figure 2, right panel). Normal epithelial cells stained weakly positive for the receptor; however, tumor cells stained strongly for AMFR. It was interesting to note that endothelial cells from both normal tissues (Figure 2, left panel) and tumor tissues (Figure 2, right panel) stained very strongly for AMF receptor.
Levels of AMF and AMFR Transcripts in Mammary Tissues
Quantitative analysis of transcripts has revealed that there were significantly higher levels of AMF transcript in breast tumors compared with normal non-neoplastic tissues, p=0.0004 (Figure 3, left). When normalized by CK19, the same statistical significance is seen (p=0.003) (Figure 3, left insert).
The levels of AMFR transcript were not different between normal tissues and tumors. However, after normalization with CK19, there was a significant difference between normal and tumor tissues where tumor tissues showed a significantly higher AMFR:CK19 ratio (p=0.0001).
AMF and AMFR and Pathological Features
The levels of AMF and AMFR were analyzed against tumor grade. As shown in Figure 4, significantly higher levels of AMFR transcript were seen in grade 2 (p=0.0042) and grade 3 (p=0.004) compared with grade 1 tumors. The same statistical difference was seen when AMFR transcript was normalized by CK19 (Figure 4, right insert). No significant difference was seen with AMF transcript in different grades, although CK19-normalized data showed a higher, but not statistically different, level in grade 2 and grade 3 tumors.
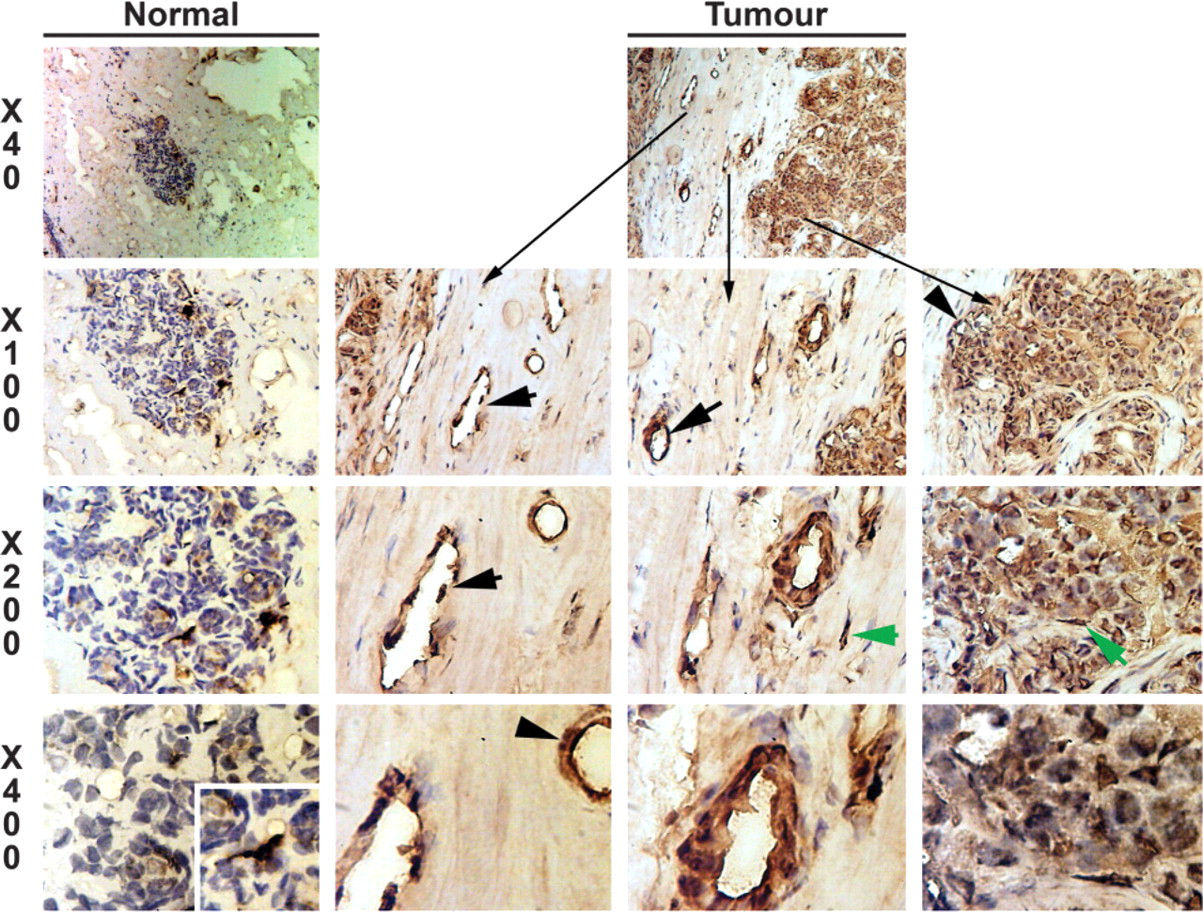
Immunohistochemical staining of autocrine motility factor (AMF). Left panel: normal tissues. Right panel(s): breast tumor tissue. Weak epithelial cell staining is seen in normal tissues, whereas stromal cells and endothelial cells show little staining. A small number of strongly stained cells are seen (left panel and insert in X400 of left panel). They probably represent the small population of stromal cells in the tissues. In tumor tissues, tumor cells show a strong staining of AMF. Interestingly, endothelial cells in microvessels (black arrows) and stromal cells (green arrows) in tumor tissues also displayed very strong staining.
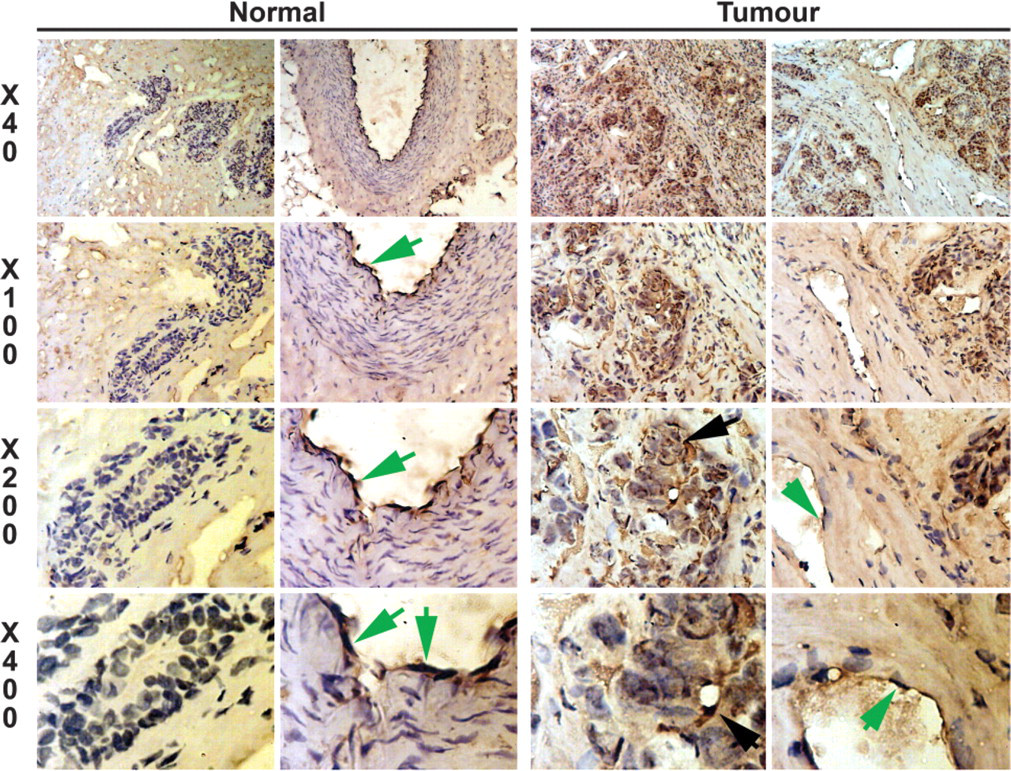
Immunohistochemical staining of AMF receptor (AMFR). Left two panels: staining of AMFR in mammary epithelial cells and vessels. Right two panels: staining of AMFR in tumor tissues. Normal epithelial cells showed a weak staining. Staining in breast cancer cells is increased (black arrows). Endothelial cells in normal and tumor tissues are positive for AMFR (green arrows in the righthand panel of both normal and tumor tissues).
AMF and AMFR and Predicted Prognosis
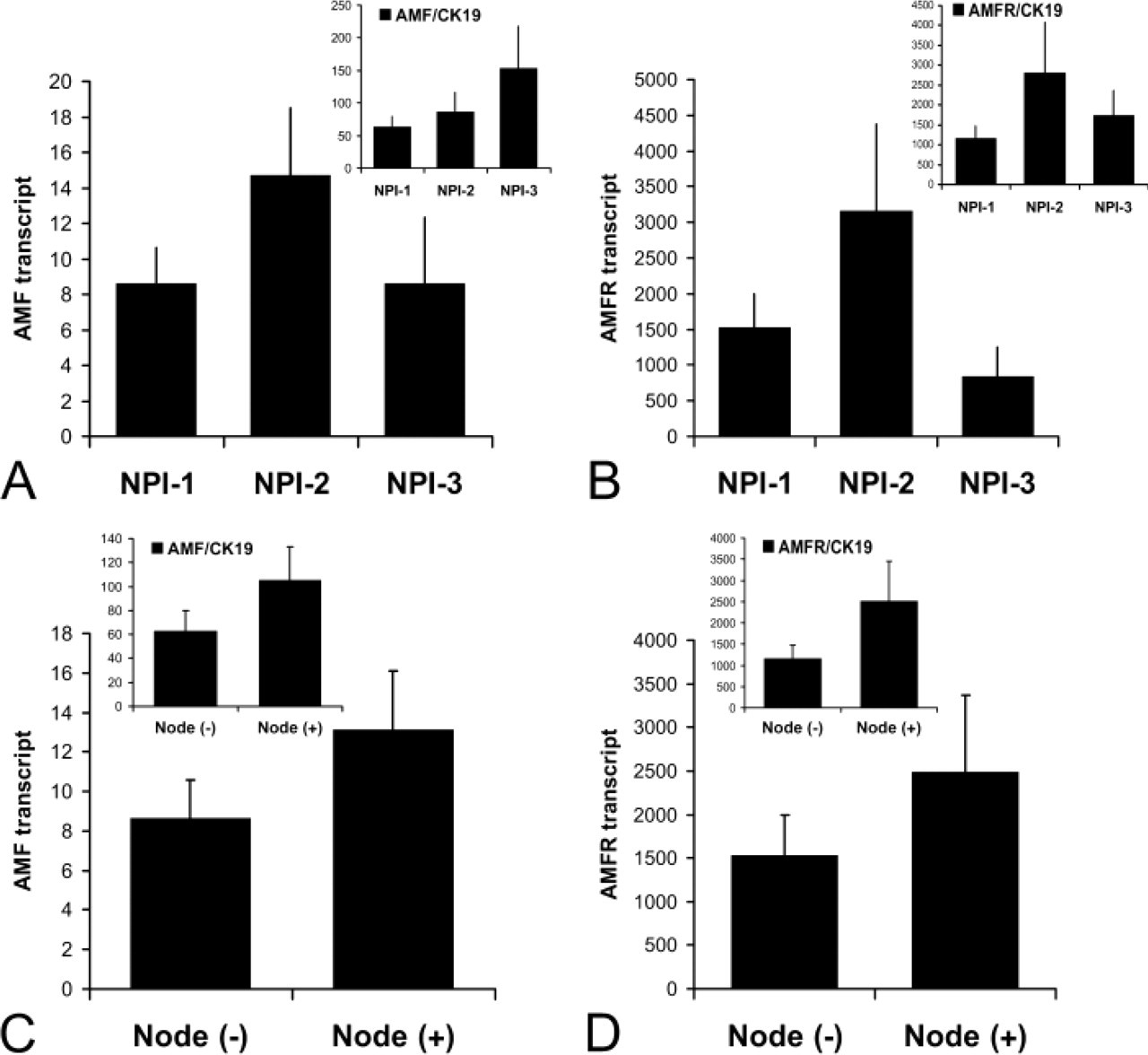
The levels of the transcripts were further analyzed against the Nottingham Prognostic Index [NPI = (0.2 X size) + grade + nodal status], where NPI <3.4, 3.4–5.4, and >5.4 represented good (15-year survival rate 80%) (NPI1), moderate (15-year survival 42%) (NPI2), and poor prognosis (15-year survival 15%), respectively. Neither AMF (A) nor AMFR (B) transcript showed a significant difference between patients with predicted clinical outcome (Figure 5, top). Normalization with CK19 did not reveal any further significant difference (Figure 5, top inserts).
AMF and AMFR levels were also compared with different nodal status of breast tumors (Figure 5, bottom). Although both molecules showed increased expression in node-positive tumors (AMF p=0.107, AMFR p=0.34) (Figure 5C for AMF and Figure 5D for AMFR), this was not significant. No statistically significant difference was seen after normalization with CK19 (p=0.11 for AMF:CK19 and p=0.18 for AMFR:CK19) (Figure 5, bottom inserts).
AMF and AMFR and Clinical Outcome and Long-term Survival
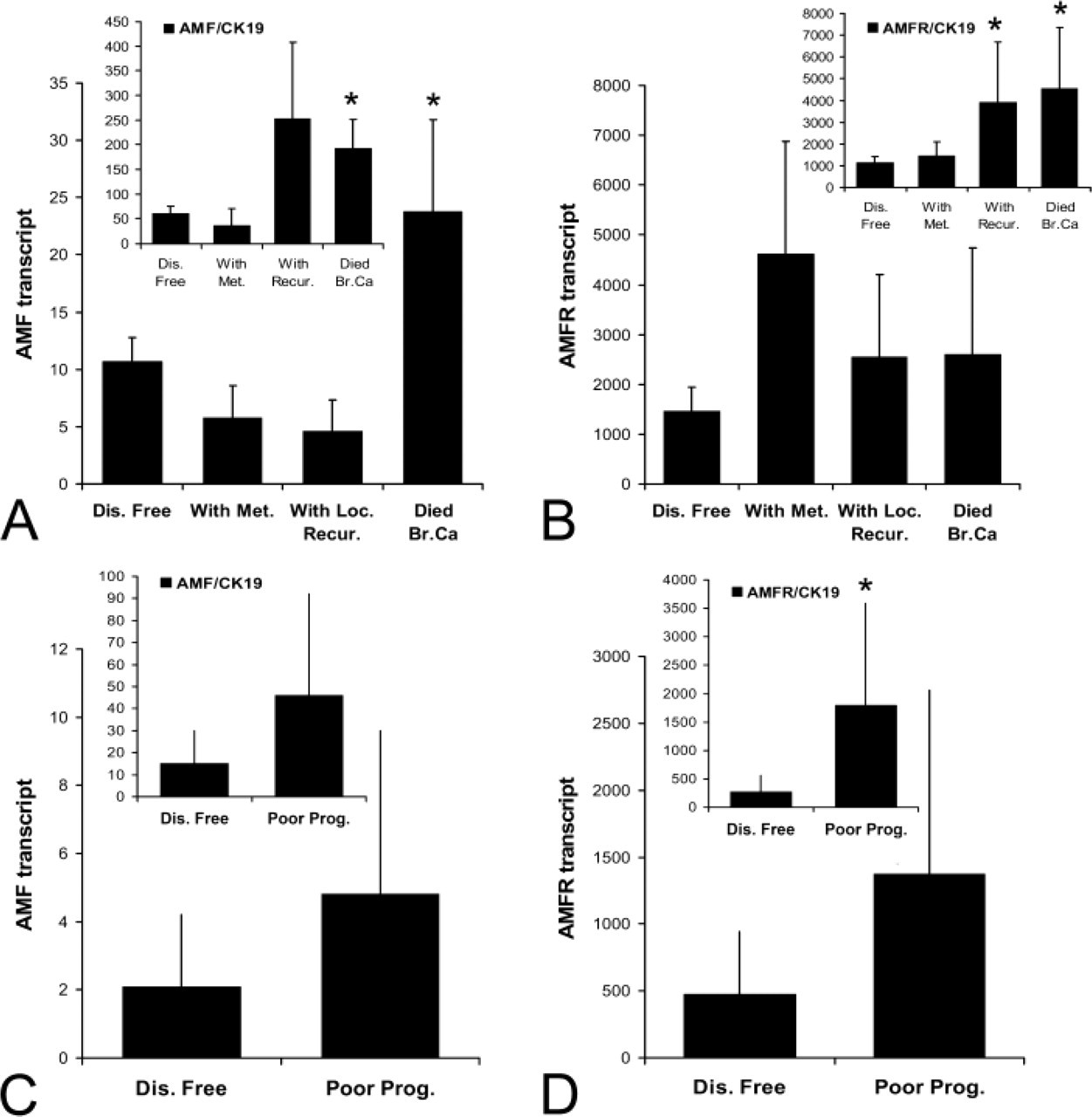
Over a median 10-year follow-up, patients were divided into those who remained disease free, those who developed metastasis or local recurrence, or those who died of breast cancer (excluding those who died of diseases unrelated to breast cancer). As shown in Figure 6A (left), patients who died of breast cancer had significantly higher levels of AMF transcript (p=0.049) and AMF:CK19 ratio (p=0.0069) (A insert) when compared with patients who remained disease free. Although patients with metastasis, recurrence, and mortality all had higher levels of AMFR transcript, these are not statistically significant (Figure 6). Interestingly, when normalized by CK19, a significant difference was seen in tumors from patients with local recurrence and patients who died of breast cancer, p=0.0435 and p=0.039 vs patients who remained disease free, respectively. We combined the three groups (with metastasis, recurrence, and mortality) to form a poor prognostic group (referred to as Poor Prog. in Figures 6C and 6D) and compared this group with those who remained disease free (Figures 6C and 6D). Higher levels of both molecules and their respective CK19 ratios were high in patients with poor prognosis.
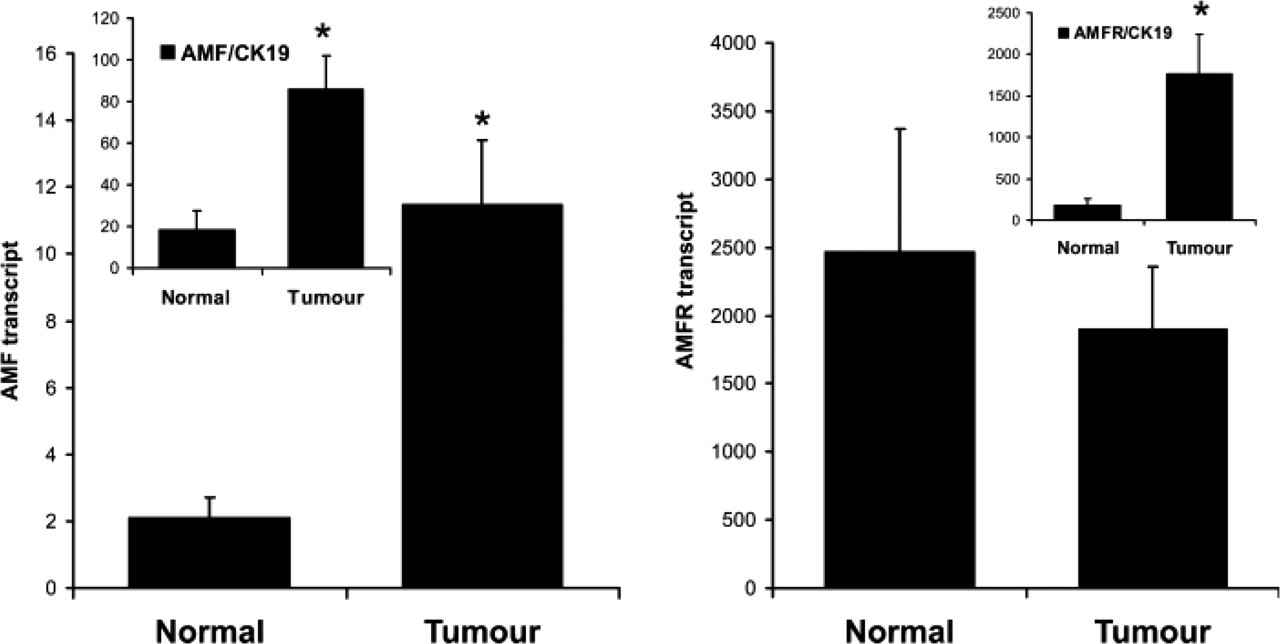
Quantitative analysis of AMF and AMFR transcript in mammary tissues. Left: AMF [inserts: AMF:cytokeratin 19 (CK19)]; Right AMFR (insert: AMFR:CK19 ratio). Tumor tissues had significantly higher levels of AMF and AMF:CK19 ratio compared with normal tissues (∗p=0.0004 and p=0.003, respectively). Although the measured transcript levels of the AMFR displayed no statistical difference between normal and tumor tissues, AMFR:CK19 ratio has shown a significant rise in tumor tissues (right panel insert, ∗p=0.0001 vs normal).
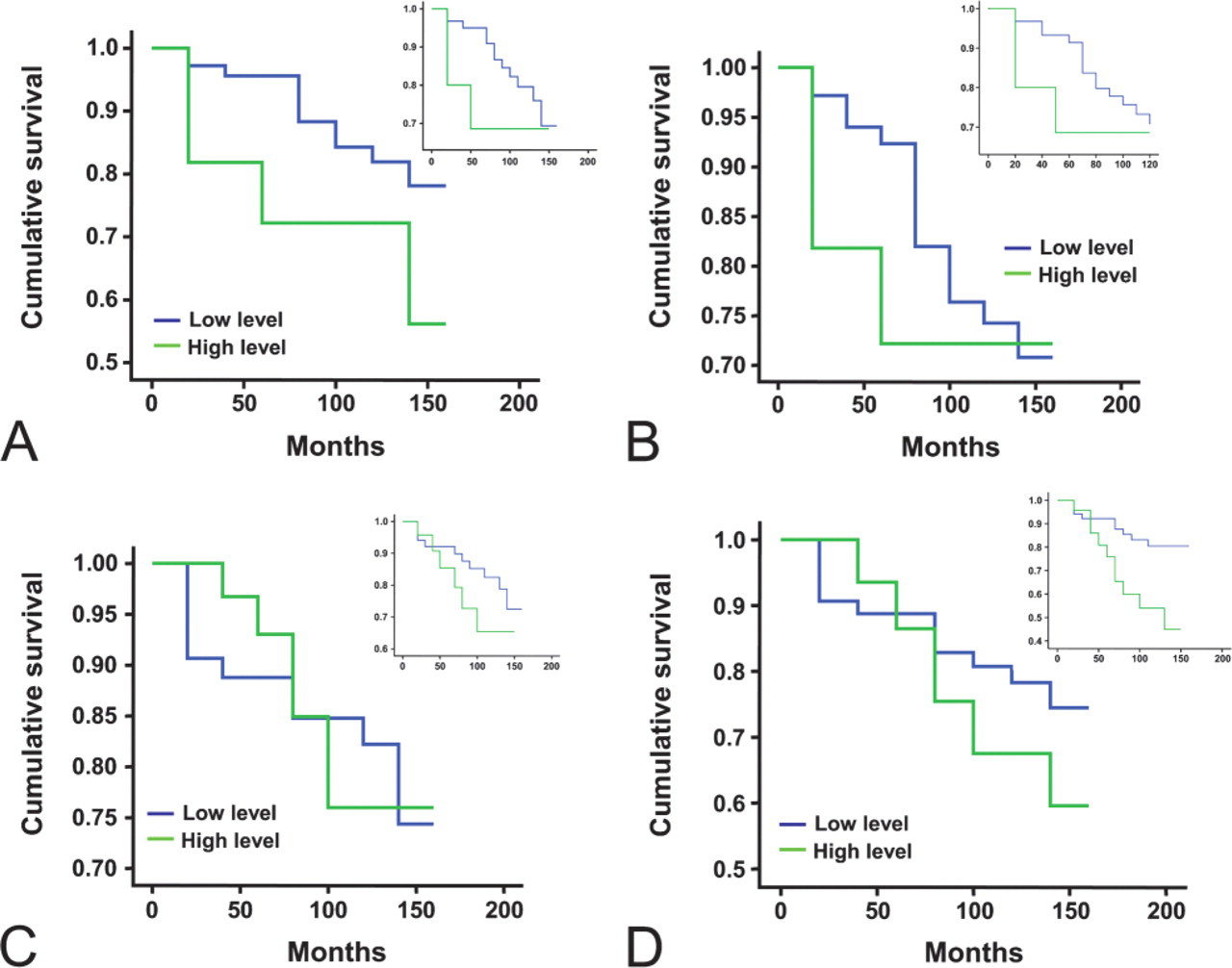
When long-term survival was analyzed using Kaplan–Meier survival method, patients with high levels of AMF transcript had a marginally shorter survival (115.6 months, 81.3–150, 95% CI) than patients with low levels (130.9 months, 122.4–239.4, 95% CI, p=0.10 (Figure 7). Normalized AMF:CK19 ratio also showed a similar trend (106.6 vs 135.6 months, for patients with high and low AMF:CK19 ratio, respectively, p=0.14, Figure 7A insert). There was no significant correlation between AMF with disease-free survival (120.0 vs 124.2 months for patients with high and low AMF transcript, respectively, p=0.40) (Figure 7). Similarly, no difference was seen with AMF:CK19 ratio (106.6 vs 129.9 months, p=0.31) (Figure 7B, insert).
AMFR transcript has also failed to show a significant correlation with overall survival (125.8 vs 133.2 months, high levels vs low levels, p=0.84)(Figure 7). AMFR:CK19 ratio has shown a closer, but statistically insignificant, link with survival (114.9 vs 135.8 months, p=0.13) (Figure 7C insert). AMFR had no significant correlation with the overall survival (115.7 vs 131.0 months, p=0.40) (Figure 7D). However, a significant correlation was seen with the AMFR:CK19 ratio, in which patients with high AMFR:CK19 ratio tumors had a significantly shorter survival (101.0 months, 80.6–121.4,95% CI) compared with those with low ratio (136.0 months, 123.7–148.2, 95% CI, p=0.0331). Furthermore, multivariant analysis failed to show that AMF and AMFR are independent prognostic factors.
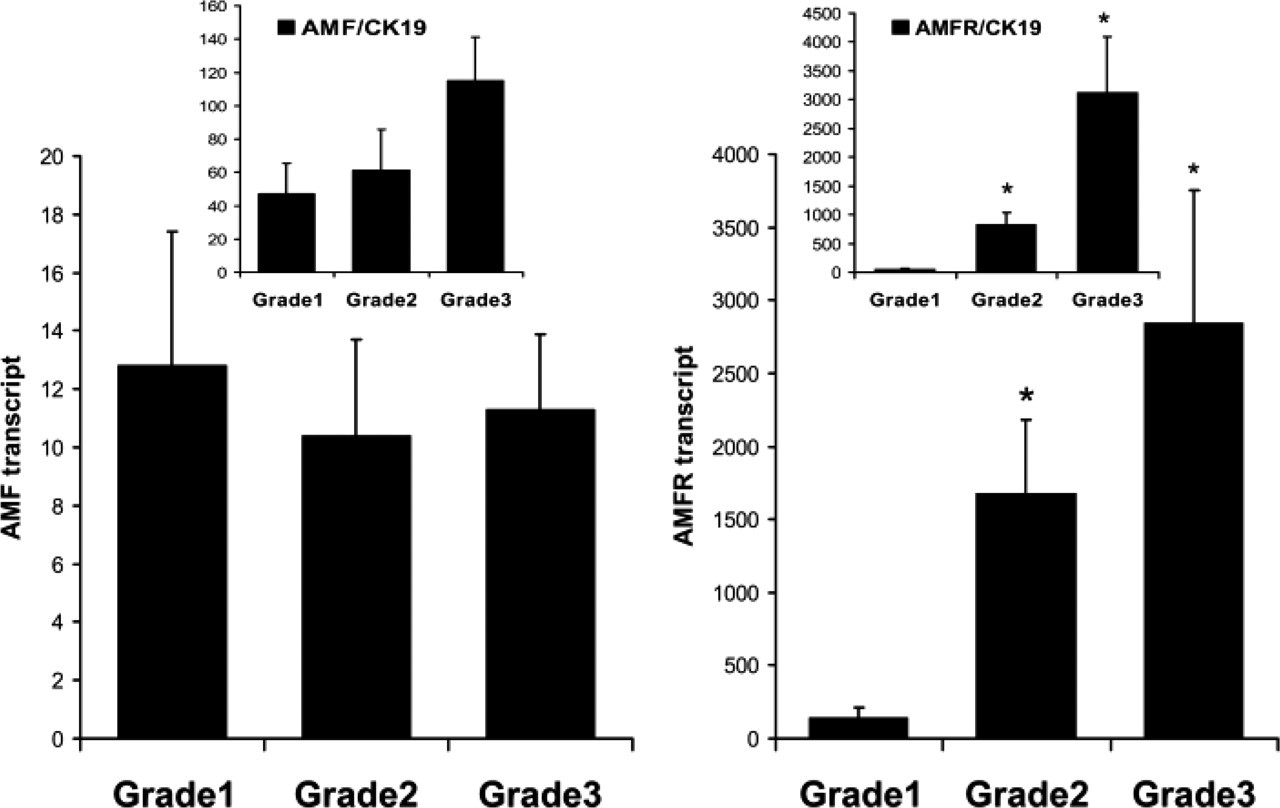
Levels of AMF (left) and AMFR (right) transcripts and their correlation with tumor grade. Inserts: AMF:CK19 and AMFR:CK19 ratio, respectively. No significant correlation was seen between AMF or AMF:CK19 and tumor grade. However, significantly higher AMFR and AMFR:CK19 ratio was seen in grade 2 and grade 3 tumors (∗p<0.01 vs grade 1 tumors).
Levels of AMF and AMFR transcripts, predicted prognosis (
We have also analyzed the potential correlation between AMF and other upstream or downstream regulators of AMF. A significant correlation was found between levels of AMF transcript and a transcription regulator Twist (r = 0.241, p<0.05), von Willebrand Factor (vWF, r = 0.56, p<0.01), TEM-6 (r = 0.22, p=0.05), Rho-C (r = 0.377, p<0.05), Cox-2 (r = 0.399, p<0.05), and MMP-7 (r = 0.427, p<0.01).
The relationship between AMF, AMFR, and estrogen receptor (ER) status was also analyzed. ER-negative tumors had marginally higher levels of AMF transcript (15.5 ± 4.0) than ER-positive tumors (10.6 ± 2.2), p=0.28. However, ER-negative tumors had significantly higher levels of AMFR transcript (2817.0 ± 729) than ER-positive tumors (666.0 ± 332), p=0.0085.
Discussion
The current study has reported for the first time that AMF and AMFR are aberrantly expressed in human breast cancer and have significant bearings on the clinical outcome of the patients.
The role of cellular motility in the invasion and aggressiveness of cancer cells has been long recognized (Hart and Fidler 1980; Gherardi and Stoker 1991). A number of protein factors are known to be motility/migration factors in cancer cells, most notably HGF, IL-6, PDGF, TGFβ, TNF, etc. These factors largely act on cells via a paracrine pathway, i.e., they are made by cells other than cancer cells such as stromal cells and white cells in the blood. The potential roles of these cell motility factors were recognized in the past decade. A couple of protein factors have been known to act on cancer cells by an autocrine pathway, i.e., the factors are made by cancer cells themselves and act directly on the source cells. The notable factors include AMF and autotoxin (Nabi et al 1990; Silletti et al. 1991; Murata et al 1994).
Since the identification of AMF more than a decade ago, the cellular and molecular roles of the factor have been widely studied. By acting on its specific receptor, AMFR (gp78), AMF stimulates the motility of a number of cancer cells via signaling pathways including Rho/rac complex, cytoskeletal-associated proteins, and cell–matrix adhesion machinery. Although expression of the factor and its receptor has been investigated in relation to clinical information in some human cancers, none has involved human breast cancer. The current study has shown that both AMF and AMFR were significantly overexpressed in human breast tumor tissues compared with normal mammary tissues. The raised levels of expression are also significant after normalization by CK19. This evidence indicates that aberrant high levels of the cellular motility factor and its receptor are a significant event in human breast cancer. Expression of CK19 is relatively stable in epithelial cell and its derived cancer cells. The use of CK19 as a means of normalizing the epithelial cell and cancer cells is yet to be generally accepted.
Levels of AMF and AMFR transcript and clinical outcomes over a 10-year period. (
The source of AMF in mammary tissues is interesting. In normal mammary tissues, AMF staining is seen in epithelial cells but very little in stromal cells. There is an overall increase in staining in cancer cells, but it is also increased in vascular endothelial and in stromal cells. This observation is interesting. First, increased expression of AMF in cancer cells may provide an augmented signal in stimulating, in an autocrine fashion, cellular motility of cancer cells. Second, stromal cells contribute to the AMF seen in breast cancer, indicating that paracrine pathway may also be important in tumors. Third, vascular endothelial cells in breast tumors stained positive. This is particularly interesting because AMF has recently been shown to be potentially angiogenic (Funasaka et al. 2001). Finally, there is a significant correlation between AMF and vWF and TEM-6, both being potential angiogenic markers that are aberrantly expressed in the same cohort of breast tumors (Davies et al. 2004; Martin et al. 2005a). Although correlation tests of this nature are somewhat indirect and should be interpreted with care, it nonetheless supports the concept that AMF may act as an angiogenic factor. The observation thus indicates that, by expressing AMF, endothelial cells may trigger an autocrine angiogenic process. This connection of AMF with angiogenesis warrants further scientific investigation.
AMF has recently been reported to be able to down-regulate the levels of E-cadherin in epithelial-derived cancer cells (Tsutsumi et al. 2004) via which AMF will assist cancer cells to dissociate from each other and gain motility. The study has suggested that AMF may down-regulate E-cadherin expression, by upregulating the E-cadherin gene promoter repressor, the Snail (Tsutsumi et al. 2004). Although our current study has failed to demonstrate a significant correlation between AMF and snail, we have found that AMF transcripts are significantly correlated with the other transcription repressor, the Twist, which has been shown to repress the expression of E-cadherin and was aberrantly expressed in the same cohort of tumors (Martin et al. 2005b). These data provide indirect evidence that aberrant AMF expression is linked to abnormal expression of E-cadherin. Finally, the study has also demonstrated that AMF transcripts are significantly correlated with that of Rho-C. Rho family members have been involved in downstream signaling of AMF receptor (Tsutsumi et al. 2002; Yanagawa et al. 2004). Rho family members have been widely involved in the regulation of the cytoskeleton system and cell motility (Hall et al. 1983; Ridley et al. 1992). They have been shown to act as downstream signaling events after stimulation by a few motility factors including that of HGF, AMF, and motility stimulating protein. The family members are aberrantly expressed in a number of solid tumors including breast cancer. We have recently reported that Rho-C and Rho-G, together with Rho-GDIs, are highly aberrant in human breast cancer. These signaling intermediates are highly linked to the disease progression in breast cancer. These data will provide additional support that both complexes do link in human breast tumors (Jiang et al 2003b). AMF has been previously reported to be able to activate 12-lipoxygenase, which results in production of 12-(S)-hydroxyeicosatetraenoic acid [12-(S)-HETE], in melanoma cells (Silletti et al. 1994). 12-HETE is a known powerful eicosanoid that regulates cell motility in cancer cells (Silletti et al. 1997). The current study has further shown a significant correlation between AMF transcript and that of cyclooxygenase-2 (COX-2), which is highly expressed in aggressive breast cancers. COX-2 protein was raised in breast tumors and was associated with the size of breast tumors, high proliferation rate, and nodal metastasis (Ristimaki et al. 2002). It is also associated with a poor clinical outcome of the patients (Ristimaki et al. 2002; Jiang et al. 2003c). Inhibitors/antagonists to COX-2 have been shown to be beneficial to patients with cancers including breast cancer. Taken together, AMF may play a key role in the aberrant expression of molecules that are central to motility and to the aggressive nature of breast cancer cells and may indicate its potential value in molecular targeting. Furthermore, the motility nature of AMF on cancer cells suggests that induction of cell motility may be the main aspect for AMF to cause tumor progression in breast cancer.
AMF and AMFR and long-term survival. Shown are Kaplan-Meier survival curve for overall survival (
From a clinical point of view, perhaps the most interesting observation in the current study is the relationship between high levels of AMF and clinical outcome. Patients who died of breast cancer had significantly higher levels of AMF and AMFR, compared with patients who remained disease-free. Furthermore, patients whose tumors expressed high levels of AMF and AMFR had significantly shorter survival than those with low levels of expression of AMF. Collectively, these findings suggest that AMF is a potential prognostic, although not independent, factor in patients with breast cancer. However, a combination of serum level of AMF, tissue level of the transcript, and its receptor at molecular levels, together with immunohistochemical analysis of AMF and AMFR, may provide a very useful approach in assessing the aggressiveness of breast cancer, disease progression, prognosis, and response to treatment. It would be very valuable indeed to conduct a study on matched samples of serum and tumors in the same patients with conventional biochemical and modern molecular techniques.
No difference in expression levels of AMF and AMFR in node-negative and node-positive tumors was seen. Together with the angiogenic effect and motility-inducing function of AMF, this suggests that AMF is primarily involved in acting on angiogenic process and stimulating the aggressiveness of cancer cells but is little involved in lymphatic and nodal spread of breast cancer cells. Finally, the aberrant nature of AMF/AMFR complex in breast cancer strongly points to them as putative therapeutic targets. Although specific means have yet to be developed for therapeutic targeting of these molecules, agents that can affect these molecules have been reported. For example, the EGFR antibody, Herceptin, can inhibit AMF expression from cancer cells (Talukder et al. 2002). Part of the mechanisms of the action of the agent may be to counteract the over-expression of AMF/AMFR in clinical breast cancer. However, AMF has important physiological functions including migration of cells, metabolism, and embryo implantation. Targeting AMF would have to be considered with caution.
In conclusion, AMF and AMFR are overexpressed in human breast cancer, thus creating an autocrine loop of stimulation. In addition, stromal cells and endothelial cells in mammary tumors, but not in normal tissues, also expressed AMF. High levels of expression of AMF and AMFR in breast tumors are associated with a high incidence of breast cancer-related death and shorter survival of patients. This strongly indicates a prognostic value of AMF and AMFR in breast cancer.
Footnotes
Acknowledgements
The authors acknowledge Breast Cancer Campaign (WGJ) and NIH (Grant CA-51714) (AR) for supporting their work.
We thank Mr. Gareth Watkins for his support in preparation of tissue sections. No conflict of interest is declared.
