Abstract
Study of parental/meiotic origin of free trisomy 21 in nuclear families from Russia (70 cases), Ukraine (32 cases), and 22 from Germany revealed maternal nondisjunction in 77.3% (Germany), 93.8% (Ukraine), and 91.4% (Russia), paternal origin in 13.6%, 6.2%, and 8.6%, respectively. Maternal meiosis I errors were found in 84.4% (Ukraine), 77.1% (Russia), paternal origin in 3.1% (Ukraine), 2.9% (Russia). Maternal meiosis II errors occurred in 9.4% and 14.3% and paternal in 3.1% and 5.7% in Ukraine and Russia, respectively. No significant differences were found in maternal/paternal origin among Ukraine, Russia, Germany, and published data from other European regions.
T
Gomez et al. (2000) revealed that, in the Spanish population, trisomy 21 by origin was 80% maternal (90.6% meiosis I, 6.2% meiosis II, 3.1% maternal mosaicism), in 5.6% paternal (50% meiosis I, 50% meiosis II), and in 5.6% mitotic.
Muller et al. (2000) in the French prenatal trisomy 21 study reported 102 informative cases of maternal origin of trisomy 21 in 89.2% and increased paternal origin in 10.8%. In the case of maternal origin, meiosis I was detected in 77% and meiosis II in 23%. In paternal origin of trisomy 21, meiosis I was found in 44% and meiosis II in 56%. Their analysis did not support the hypothesis that an increased proportion of paternal meiotic disorders is caused by the selective loss of paternal origin fetuses.
The meiotic process is very sensitive to the effects of noxious endogenous and exogenous factors, as suggested by the cluster of Down syndrome incidence in Berlin in the first month following the Chernobyl accident (Sperling et al. 1994). Therefore, the impact of the Chernobyl accident on the increased prevalence of Down syndrome in Western Europe during the 1980s merits further investigation (Dolk and Nichols 1999).
Studies of parental and meiotic origin of trisomy 21 and case–control surveillance of these factors are important for further understanding of the pathogenesis of chromosome 21 nondisjunction (Macek et al. 2003).
Therefore, the aim of this study was to assess the parental and meiotic origin of free trisomy 21 in nuclear families in different regions of Ukraine and Russia comparing to Central Europe by means of quantitative fluorescent PCR (QF-PCR) to find out the possible differences due to different lifestyles and to load of exogenous noxious agents including Chernobyl radiation accident exposure. Cases with translocation trisomy 21 detected by cytogenetic examination were excluded.
From 208 referred nuclear families from Ukraine and Russia, 87 incomplete families and families with poor DNA quality samples were excluded as well as 19 families due to the non-informativity of the STR markers or inconclusive peak height ratio (PHR) in the range between 1.5 and 1.7 in more than two investigated markers. Finally, 102 nuclear families from Russia and Ukraine and 22 from Germany (Berlin region) were examined by QF-PCR. All families excluded from the current study will be analyzed in the near future with more STR markers.
DNA samples from Moscow, Rostov (Russia), and Kiev (Ukraine) were obtained by classical DNA extraction method. DNA from frozen blood samples or amniotic fluid referred from Donetsk (Ukraine) was extracted by commercial kit (PUREGENE Blood Kit; Gentra Systems, Minneapolis, MN) and QiaAmp DNA mini Kit (QIAGEN GmbH; Hilden, Germany), respectively.
QF-PCR (Pertl et al. 1999) was slightly modified (Macek et al. 2002). Each STR marker was analyzed in singleplex reaction with the exception of the first samples analyzed by sets of primers. Previously used dinucleotide marker D21S167 was eliminated due to its high peak height ratio variability. New STR markers (D21S1432, D21S1446, D21S1270, and D21S2052) were added. The following fluorescently labeled primers for the detection of trisomy 21 were used: D21S11 (Fam), D21S1414 (Fam), D21S1446 (Fam), D21S2052 (Fam), D21S167 (Fam), D21S1411 (Hex), D21S1412 (Hex), D21S1432 (Hex), and D21S1270 (Hex).
PCR was carried out in a total of 15 μl of reaction mix containing 1 × reaction buffer PE, 1.5 mM MgCl2 PE, 200 μM each of dNTP, and 1 U Ampli Taq Gold polymerase PE (all from Applied Biosystems, Foster City, CA), 5 pmol of each primer, and ~50 ng of DNA. The amplification was performed in PTC–200 DNA Thermal Cycler (MJ Research, Inc., Waltham, MA) started with 12 min of initial denaturation at 94C followed by 30 cycles of denaturation at 94C for 60 sec, annealing at 55–60C for 60 sec, elongation at 72C for 60 sec, and the final extension at 72C for 10 min. The fragment analysis of fluorescently labeled PCR products was performed on ABI Prism 310 Genetic Analyzer (Applied Biosystems) following manufacturers recommendations, and electrophoresis results were analyzed with the GeneScan Analysis Software.
For the assessment of trisomy, the PHR was evaluated. In triallelic trisomies it was ~1:1:1. The trisomy diagnosis is safe. It reveals nondisjunction in meiosis I. Parental and meiotic origin is reliably ascertained if parental heterozygosity confined to different electrophoretic localizations is preserved.
If parental heterozygosity was reduced to homozygosity, the error occurred during meiosis II indicating diallelic type of trisomy with PHR 2:1.
Monoallelic and diallelic cases with PHR were in the range of 1.4–1.7, or in the case of non-informativity, more STR markers were used. All diagnoses were confirmed by two repeated examinations.
For parental and meiotic origin detection, QF-PCR analysis of both parents is necessary, and the conclusions must be confirmed by at least two informative STR markers.
For the statistical evaluation of the differences in the prevalence of the parental and meiotic origin of trisomy 21 in Central Europe and different regions of Ukraine and Russia, homogeneity test in contingency tables and χ2 test were used.
Table 1 documents proportion of meiosis I and II and parental origin of nondisjunction in 22 nuclear families from Berlin, 32 from Ukraine, and 70 from Russia. Statistical analysis did not reveal significant differences in prevalence of meiosis I and II disorders in Kiev and Donetsk regions of Ukraine, as well as between Moscow and Rostov regions. No statistically significant differences were disclosed between proportion in meiosis I/II and maternal/paternal origin between Berlin, Ukraine, and Russia nuclear families with trisomy 21.
Meiotic and parental origin of trisomy 21 in Central and Eastern Europe
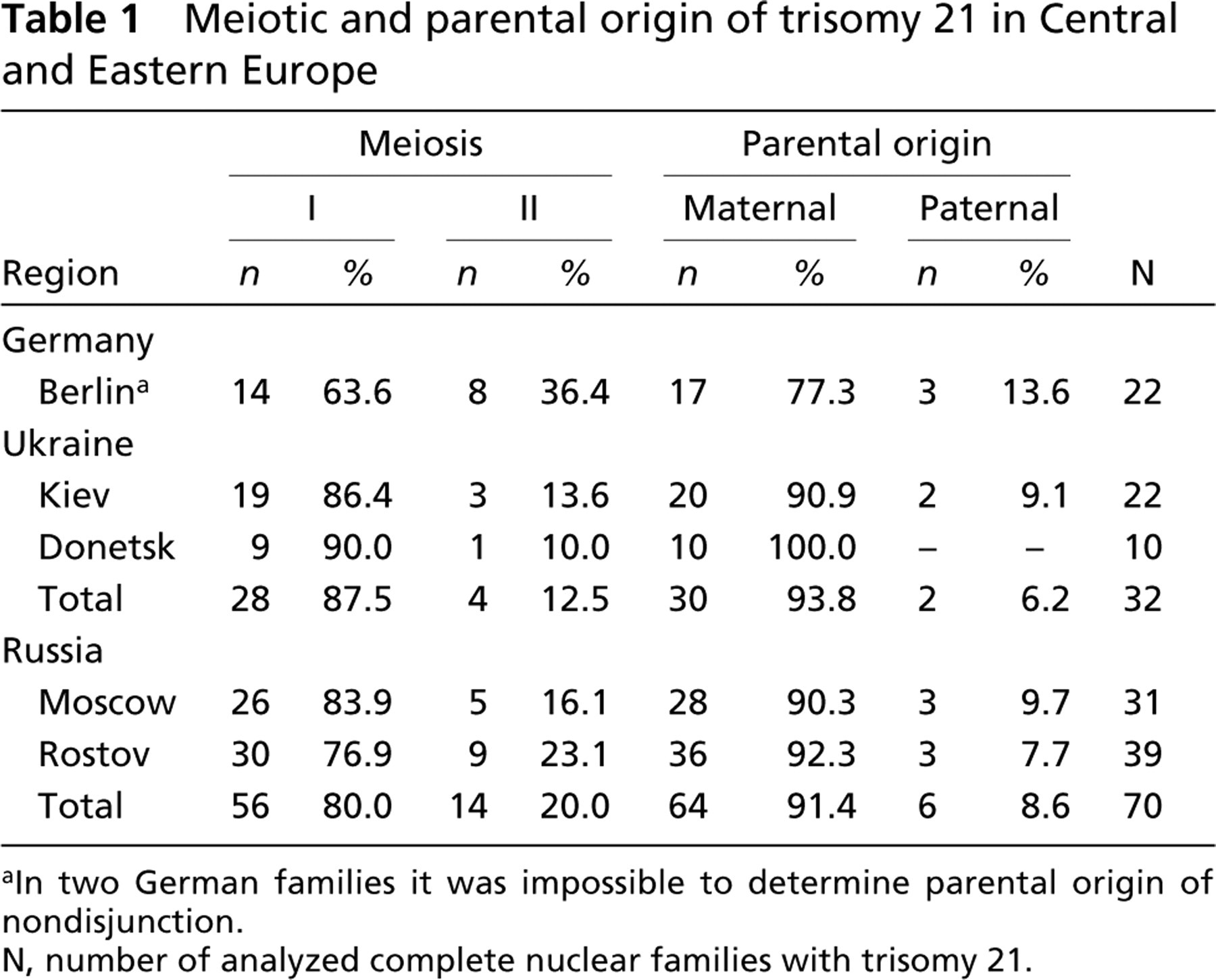
aIn two German families it was impossible to determine parental origin of nondisjunction.
N, number of analyzed complete nuclear families with trisomy 21.
Parental and meiotic origin of trisomy 21 in Russian and Ukrainian families ascertained by QF-PCR
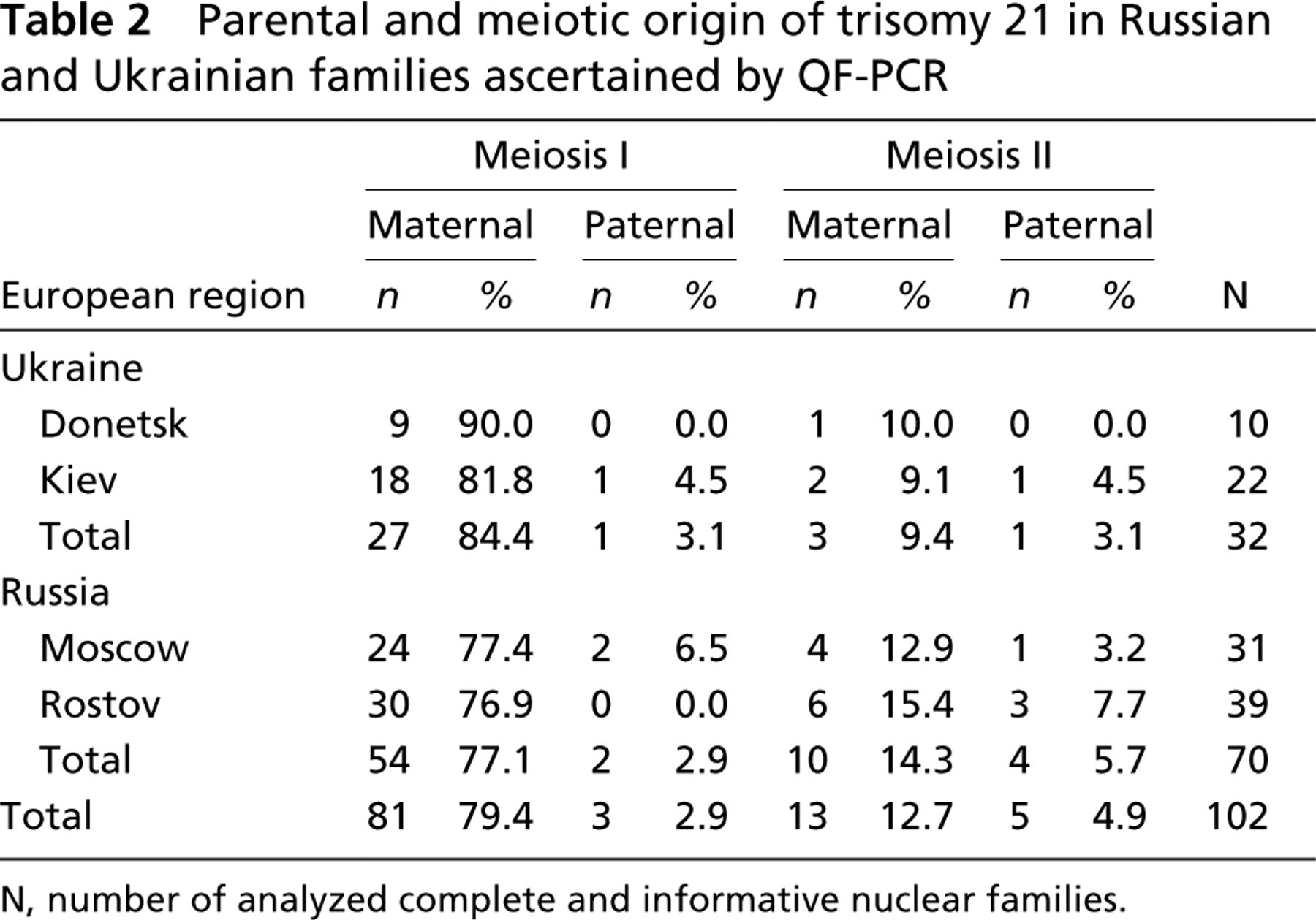
N, number of analyzed complete and informative nuclear families.
Table 2 documents maternal/paternal proportions in meiosis I and meiosis II in the studied regions of Ukraine and Russia. Statistical analysis did not disclose any significant differences between maternal and paternal proportions in meiosis I between cases in Ukraine and Russia. The same was true for maternal/paternal proportion in meiosis II.
These results are completely based on the QF-PCR analysis of the parental and meiotic origin of trisomy 21 with preservation of strict criteria to determine exactly the reliable conclusions. This study confirms that QF-PCR is also a very effective method for population studies of meiotic and parental disorders for aneuploidy analysis with very high throughput of 48 samples per 24 hr by ABI Prism 310 Genetic Analyzer.
The analysis of these preliminary data documents that prevalence of maternal meiosis disorders in Ukraine (93.8%) and Russia (91.3%) does not differ from the data ascertaining maternal origin of extra chromosome 21 (Antonarakis 1991; Petersen et al. 1992; Antonarakis et al. 1993; Yoon et al. 1996). The same follows also from the comparison of the proportions of meiosis I and meiosis II in studied populations from France and Spain. The same is true for the comparison of paternal origin—6.2% from Ukraine and 7.8% from Russia, respectively.
If these observations are confirmed by further analyses of Russian and Ukraine nuclear families with trisomy 21 in more than 150 families, it will suggest that different lifestyles, noxious exogenous factors load, and Chernobyl radiation exposure have no impact on proportions of parental and meiotic origin of trisomy 21 in postnatal and prenatal (second trimester) phase of development.
Footnotes
Acknowledgements
This study was supported by contract of EU Copernicus-2-ICA-2-CT-2000–10012; Czech Ministry of Health 00000064203 and IGA NE-6462–3, NR/7962–3; Czech Ministry of School and Educations No. 111300003 and LN00A079.
