Abstract
We performed a comparative study of the enrichment of erythroblasts by a soybean agglutinin galactose-specific lectin method and a standardized magnetic cell-sorting (MACS) protocol. Blood samples, obtained from 11 pregnant women at between 11 and 40 weeks of gestation, were split and examined by each method in parallel. The number of erythroblasts recovered by the lectin method was approximately eightfold higher than the number obtained by MACS. Our data suggest that the lectin-based method may provide a better approach for the enrichment of rare fetal erythroblasts from maternal blood.
E
To date, the most successful approaches to the enrichment of fetal erythroblasts from maternal blood have been fluorescence-activated cell sorting and magnetic cell sorting (MACS). Consequently, the efficacy of these two methods in the enrichment of fetal erythroblasts and the detection of fetal aneuploidies was examined in a large-scale study funded by the National Institutes of Health, the so-called NIFTY study (Bianchi et al. 2002). This study indicated that neither method is suitable for potential clinical applications because both methods resulted in a poor yield of fetal erythroblasts (Bianchi et al. 2002).
A recent study has demonstrated a new enrichment technique that uses a galactose-specific lectin for the isolation of fetal erythroblasts from maternal blood (Kitagawa et al. 2002). This method is based on the observation that erythroid precursor cells express a galactose moiety that is associated with the development and maturation of these cells. By exploiting this feature, it has been shown that the binding of this galactose moiety to a lectin, in this case soybean agglutinin (SBA), can be used in the selective enrichment of erythroblasts from maternal blood samples (Kitagawa et al. 2002).
To evaluate this method, we performed a comparative study on 11 maternal blood samples collected at 11 to 40 weeks of pregnancy. One fraction of the blood sample was subjected to enrichment by our standard CD-71 MACS protocol (Troeger et al. 1999), and the other half was enriched by the lectin protocol (Kitagawa et al. 2002). All samples were processed within 6 hr after blood sampling.
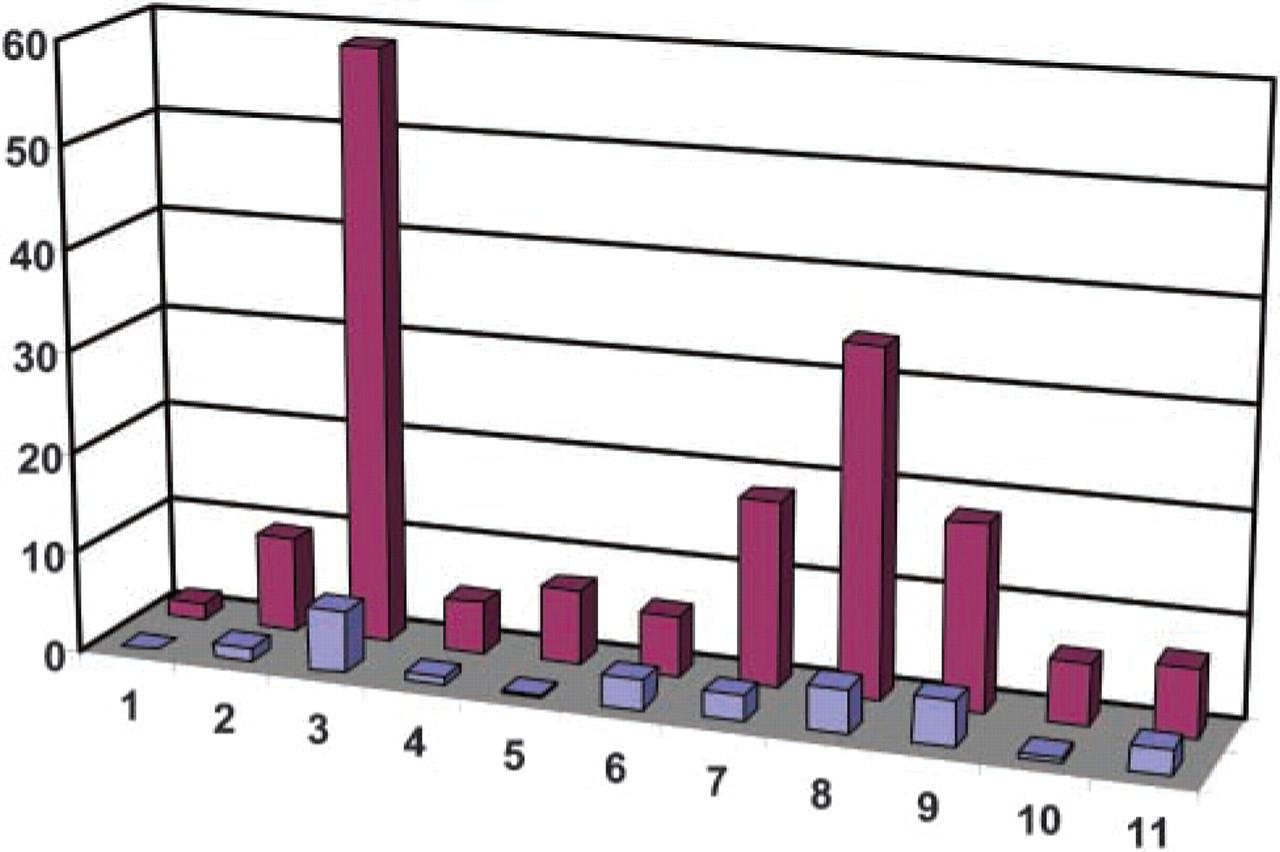
Erythroblast recovery following enrichment by SBA-lectin or CD-71 MACS. The y-axis refers to the number of erythroblasts recovered per milliliter of maternal blood. The x-axis indicates individual cases. Red bars indicate samples enriched by the SBA-lectin method, and blue bars indicate samples treated by MACS.
The lectin enrichment method was performed as described previously, except that a concentration of 100 μg/ml SBA (Vector; Geneva, Switzerland) was used (Kitagawa et al. 2002). The MACS/CD-71 enrichment was performed according to our established protocol (Troeger et al. 1999). In both instances, the enriched cells were stained with May-Gruenwald (Merck; Darmstadt, Germany)-Giemsa (Sigma; Buchs, Switzerland) and the number of recovered erythroblasts was scored. For the MACS/CD-71 enrichment procedure, two slides were evaluated, whereas in the SBA-lectin method, one chamber was scored. This was used to estimate the number of recovered erythroblasts per milliliter of maternal blood.
Our results (Figure 1) indicate significant differences (p = 0.00) between the two enrichment methods, with almost eightfold more erythroblasts being recovered per milliliter of maternal blood by the SBA-lectin method (median 16; range 2–59) compared with our standard MACS approach (median 2; range 0–6).
Although the efficiency of the erythroblast recovery by MACS achieved in this study is comparable with data from other studies (Troeger et al. 1999), it is clear that the SBA-lectin method offers a better recovery. The present study also indicates that many erythroblasts are being lost during the MACS procedure. In this regard, it has previously been calculated that cell loss due to centrifugation can be as high as 2–4% and that the most significant loss (almost 10-20%) occurred during the transfer of enriched cells onto microscope slides by cytocentrifugation (Oosterwijk et al. 1998). It is also likely that target cells are lost during the MACS procedure, in that they may be retained by the column or lost during the washing steps. Therefore, it is possible that the lack of such steps may contribute to the overall improved recovery by the SBA-lectin method.
Another apparent advantage of the SBA-lectin method is that the enriched cells appeared to have a better morphology, facilitating easier recognition and analysis, than cells treated by the MACS protocol. This facet will have to be explored in a larger study.
A major drawback of all current enrichment methods, and especially that of the SBA-lectin method, is that the final enriched cell population is contaminated by a vast number of maternal cells. Therefore, although the recovery by the SBA-lectin method is greater than that achieved by the MACS method, the purity is significantly lower. This implies that a considerable effort must still be made to locate potential fetal cells after the lectin enrichment procedure, which renders the analysis tedious and time-consuming. It will therefore be necessary to employ systems that permit the automated recognition and recovery of potential fetal cells to obtain the optimal efficiency required for clinical applications. For this reason, studies are ongoing to explore the use of systems that facilitate automated recognition of rare events and their subsequent contact-free isolation by laser pressure catapulting (Hahn et al. 2002).
In summary, our study indicates that the lectin-based method is more efficacious than a MACS approach for the enrichment of erythroblasts from maternal blood and that this method may be a promising alternative for future investigations concerned with noninvasive prenatal diagnosis.
