Abstract
An increased number of chromosome plates can be obtained by use of calyculin A (CLA). CLA is an inhibitor of protein phosphatases (type 1 and type 2A serine/threonine). Inactivation of these phosphatases leads to premature chromosome condensation (PCC) in all phases of the cell cycle; thus, it is possible to investigate both metaphase and G2-PCC chromosomes. Amniotic fluid (AF) cultures were treated with calyculin A (CLA). GTG banding was obtained. Using this method it is possible to investigate all cell cycle phases, GTG banding, chromosomal breaks, and rates of PCD on the same preparation. Analyses of AF cultures treated with CLA allow complex studies on fetal genetic material. This work presents potential usefulness of CLA for cytogenetic prenatal diagnosis.
Keywords
A
So far, premature chromosome condensation has been widely used in radiation biology because it can induce condensation of interphase chromosomes even in highly damaged cells after irradiation (Johnson et al. 1999; Kanda et al. 1999). When cells are irradiated, most of them arrest in G2 phase because of DNA damage, and these cells do not enter mitosis. In such situations it is difficult, or even impossible, to obtain a satisfactory number of mitotic chromosomes for analysis (Gotoh and Asakawa 1996). Using the PCC method for studying the initial effects of radiation damage and chromosome break rejoining, many investigators have found a linear dose response for the induction of chromosome breaks immediately after exposure to radiation (Suzuki et al. 1990; Durante et al. 1998). Gotoh and Asakawa (1996) developed a method for detecting sister chromatid exchanges in highly mutagenized cells using prematurely condensed chromosomes induced by CLA treatment.
It was previously observed that treatment with CLA produces high G2/M index in lymphocyte cultures in comparison to conventional colcemid blocking, and this method does not influence the banding pattern or chromosome number (Kowalska et al. 2003).
The aim of this work was to determine if CLA is useful in fetus examination during prenatal diagnosis and if it provides additional information about the pregnancy outcome.
Fetal cells from amniotic fluid (AF) cultures were of interest to us; thus, the project and methods had to be approved by our institutional committee on human experimentation (The Committee of Ethics at Silesian Medical Academy) who allowed using the remaining material after standard diagnostic procedure. Remaining cultures were used for this work only in cases when written consent was obtained from the patient. No standard cell lines were investigated.
AF in vitro cultures were set up according to the standard procedure (Rooney and Czepulkowski 2002). When prenatal diagnosis was completed, the remaining AF flask cultures were used for this study. The cells were either subcultivated or directly treated with CLA. The cultures with a normal karyotype result were chosen for this study. The cell medium was Amniomax Complete II (Invitrogen). When a culture showed the required density, CLA (100 nM) was added for 45–60 min. The cultures were treated with hypotonic solution and fixed according to a standard procedure. Chromosomes were stained by the use of standard GTG banding procedure (Rooney and Czepulkowski 2002).
CLA caused PCC in all treated cultures. It was possible to distinguish cell cycle phases (G1, S, G2/M). Data from several cultures are presented in Table 1 and Table 2. The cultures were not set up from a single cell line, but the cells belonged to different patients referred for prenatal cytogenetic testing in our laboratory. Clinical cytogeneticists know that old AF cultures tend to decrease mitotic index, and eventually the cells stop dividing. All analyzed cultures remained after routine diagnosis and were older than 3–4 weeks. This situation can be seen when one compares cultures 28/04P (3-week-old) and 37/04P (6-week-old) from Table 1. They both were subcultivated from the same patient, but within 3 weeks the G2/M index decreased from 19.80% to 5.30%. Moreover, it should be noted that the percentage of cells that did not respond to CLA treatment increased in these cultures from 28.80% to 86.00%. This suggests that most of the cells from the old culture (37/04P) might have left the cell cycle and entered the G0 phase (Durante et al. 1998) or even the apoptotic path.
Table 1 presents the phase index in different AF cultures. The last two samples were lymphocyte cultures (110/03 and 114/03), and they were included in Table 1 to show that G2/M index in AF cultures can be as high as in lymphocyte cultures. Table 2 shows the G2/M and M index in AF cultures in the same patients. G2/M index was calculated from cultures treated with 100 nM CLA. M index was calculated in AF cultures treated with Colcemid (3.5 hr) and fixed on the same day as those treated with CLA. M index was treated as an internal control in this study.
Usefulness of calyculin A for prenatal diagnosis
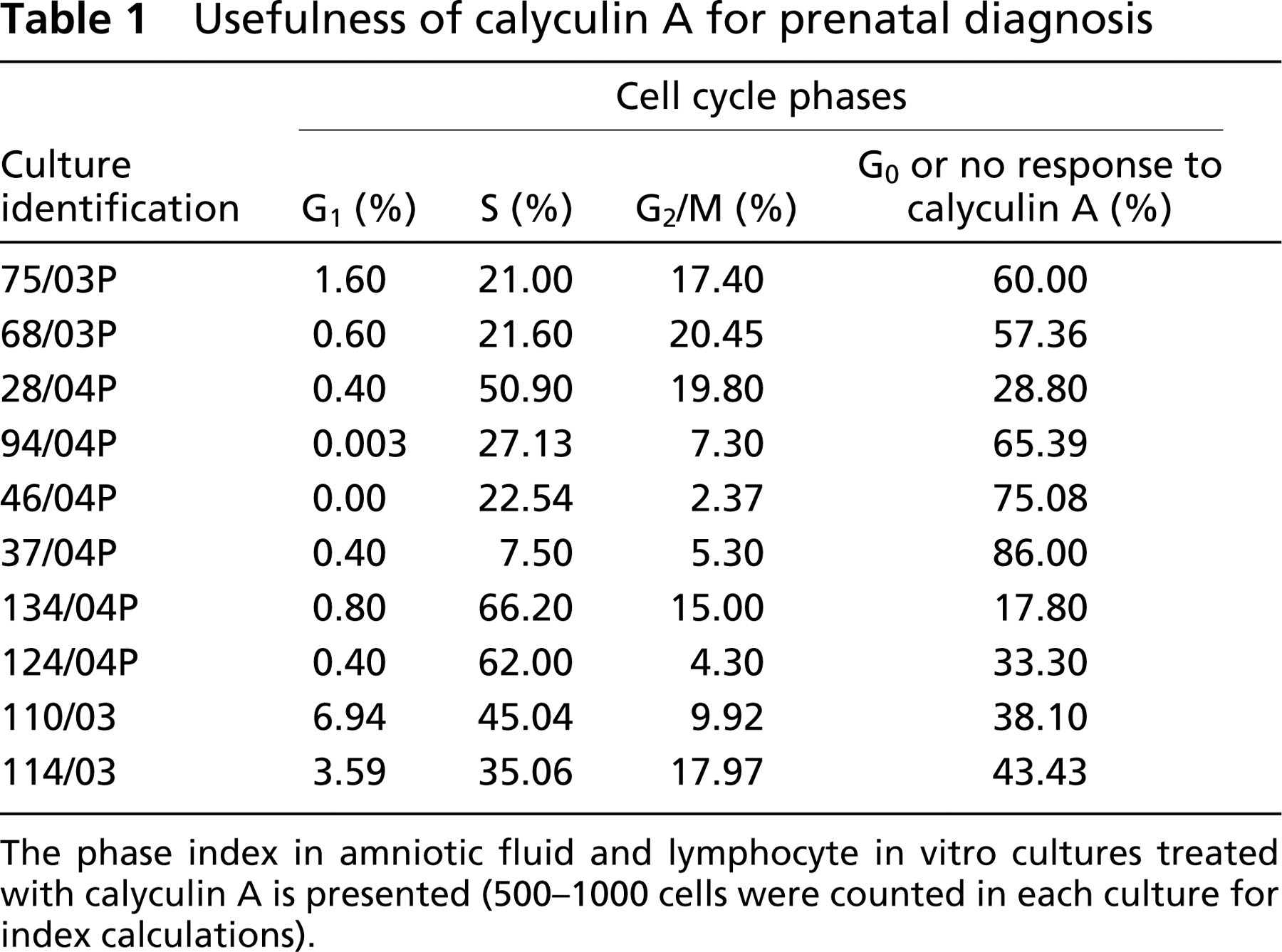
The phase index in amniotic fluid and lymphocyte in vitro cultures treated with calyculin A is presented (500–1000 cells were counted in each culture for index calculations).
Comparison of G2/M phase and mitotic (M) index in particular patients
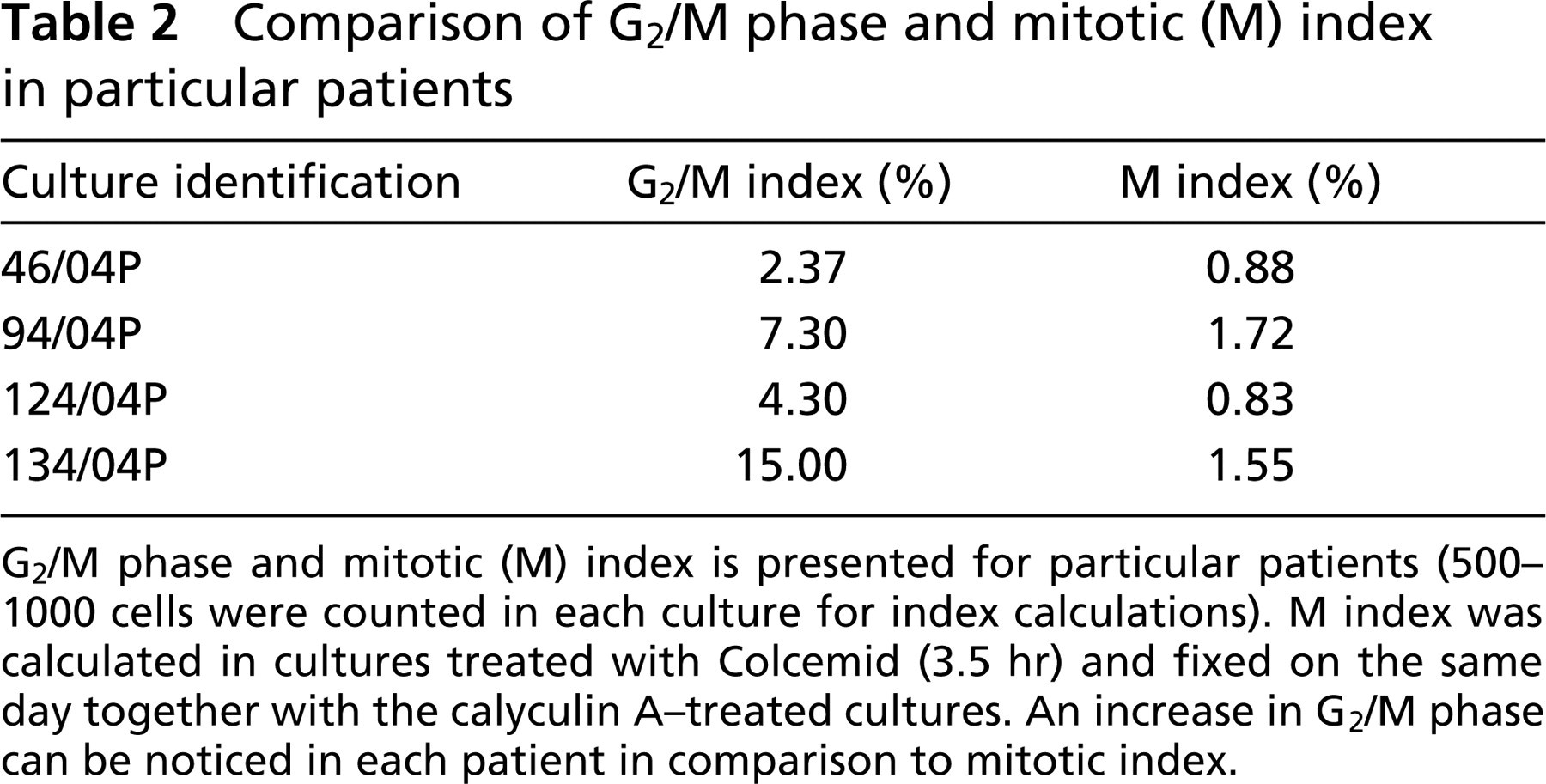
G2/M phase and mitotic (M) index is presented for particular patients (500–1000 cells were counted in each culture for index calculations). M index was calculated in cultures treated with Colcemid (3.5 hr) and fixed on the same day together with the calyculin A-treated cultures. An increase in G2/M phase can be noticed in each patient in comparison to mitotic index.
We were not particularly interested in raising the mitotic index but rather in obtaining additional information that might be gathered during G2/M chromosome analysis.
Unfortunately, GTG banding resolution of PCC chromosomes was very low. Nevertheless, it was possible to identify particular chromosomes (Figure 1). Chromosome breaks were very rare. Preliminary results showed b/c index (chromatid or chromosome breaks per cell index) lower than 0.5 b/c (Figure 2). None of the cultures was treated with a genotoxic agent, and those were cells from normal pregnancies selected for this study. It was interesting to observe quite high premature centromere division (PCD) rate (Figure 3). The rates of PCD in amniotic fluid cultures treated with CLA reached 16–17% in normal pregnancies. PCD was also detected in lymphocyte cultures treated with CLA. The PCD rate in lymphocyte cultures from normal individuals was ~10%.
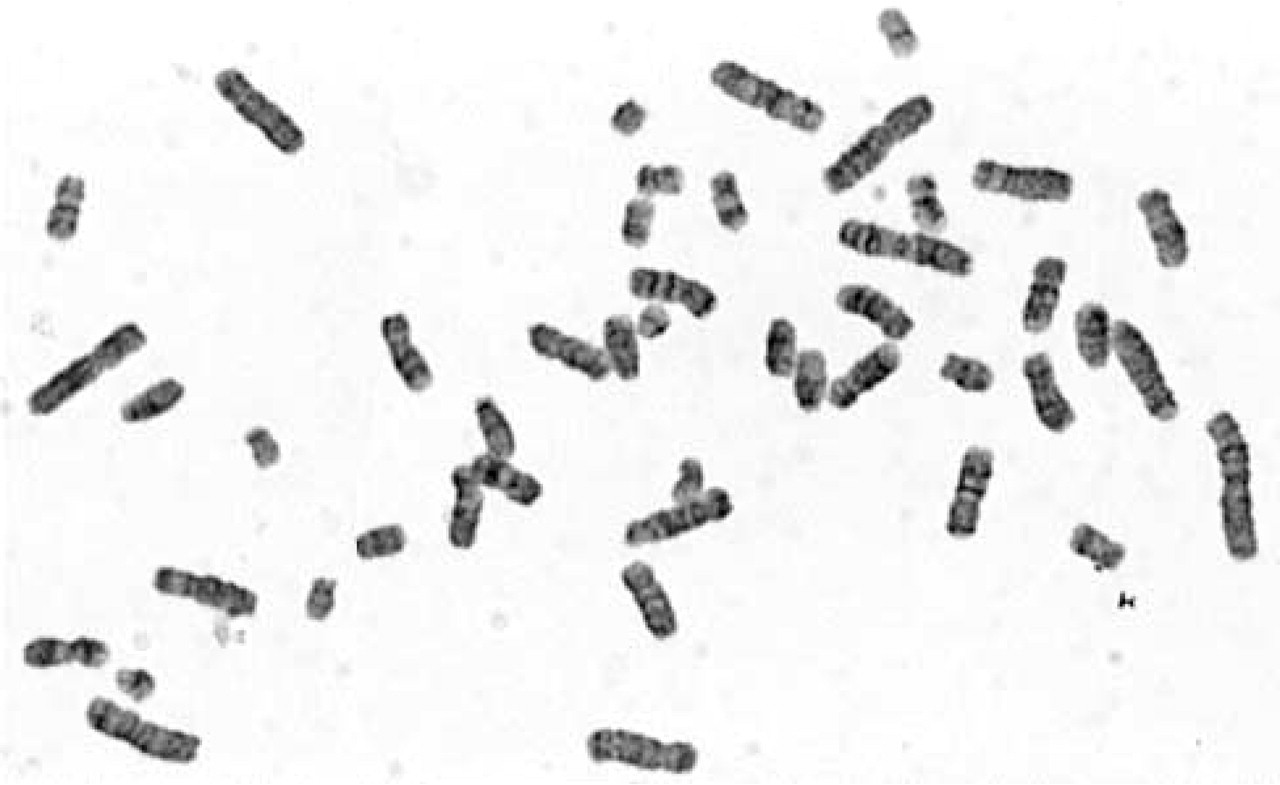
GTG banding on G2/M phase chromosomes obtained from AF in vitro culture treated with 100 nM CLA. The banding resolution was low; however, it was possible to identify particular chromosomes (GTG staining; magnification × 1000).
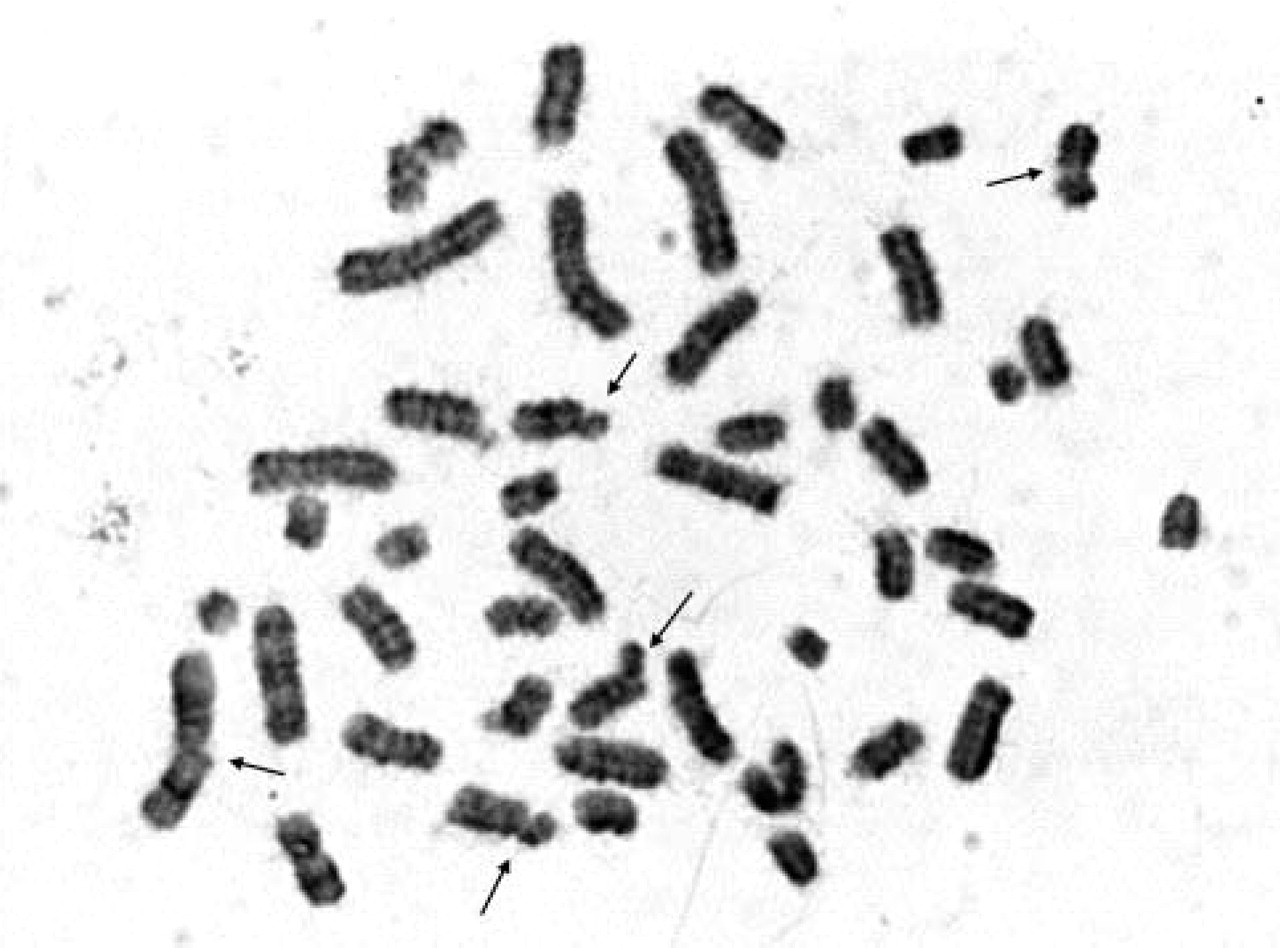
Chromosomal breaks can be observed in cultures treated with CLA. The b/c index in cultures that show normal karyotype is low. DNA damage can be assessed by calculating b/c index. By use of this technique chromosome breaks can also be seen in cells that would not be able to enter mitosis due to DNA damage. PCC is a method to omit the cell-cycle checkpoints and to visualize chromosome breaks (GTG staining; magnification × 1000).
GTG banding, chromosomal breaks, and PCD can all be investigated on the same preparation. After CLA treatment, cells that would not enter mitosis due to DNA damage can be seen among cells classified as category G2/M. This category is represented by both G2 and M fully condensed chromosomes. Induction of chromosomal breaks by use of agents such as bleomycin is not necessary.
Analysis of cultures treated with CLA allows complex studies on fetal genetic material. GTG banding does not show very good quality; however, after routine prenatal diagnosis there are always cells available for additional studies. This technique is not presented as an alternative chromosome investigation. DNA damage can be assessed in cytogenetic samples by calculating b/c index. Chromosomes can be identified, and the breaks can be localized on particular chromosomes. Increased number of chromosomal breaks might correlate with the efficiency of fetal DNA repair enzymes. It was previously shown that b/c index in chorion villi culture obtained after spontaneous miscarriage was extremely high. The only culture that allowed chromosome analyses was treated with CLA and the b/c index was 3.3 (Srebniak et al. 2003). It is interesting to determine whether a fetus with de novo chromosome aberration or multiple abnormalities also has an increased b/c index and whether there is any correlation between the number of damaged cells and pregnancy outcome. It also might be interesting to learn whether fragile sites can be observed by use of this method; therefore, extensive studies of known cases are necessary. However, analysis of chromosome damage in primary AF cultures rather than in subcultivated flask cultures is recommended. Therefore, extra cultures might be set up along routine analysis. Chromosome damage might be induced during intensive handling of long-term cultures, and false-positive results might be seen.
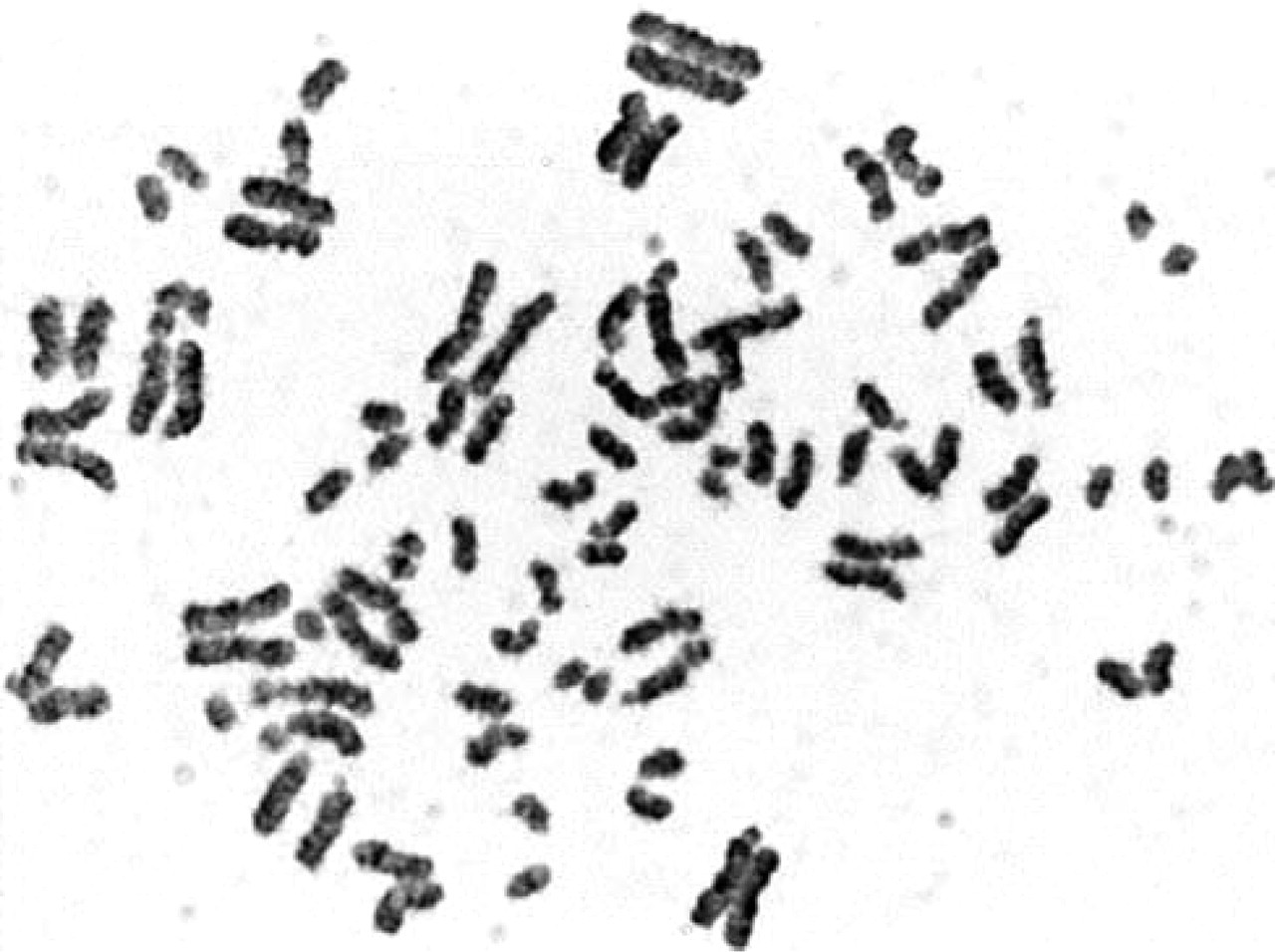
A metaphase showing PCD that was frequent in cultures treated with CLA (GTG staining; magnification × 1000).
PCD was quite often seen in cultures treated with CLA. About 16% of the G2/M phase chromosomes were affected in normal samples. It is known that increased PCD rate is detected in Roberts syndrome patients and also as a dominant abnormality in families with fertility problems (Gabarrion et al. 1986; Benzacken et al. 1996). It is interesting to investigate whether fetal abnormalities are correlated with increased PCD index or chromosome breaks and whether couples suffering from idiopathic infertility have increased PCD rates seen after CLA treatment. This work presents potential usefulness of CLA for cytogenetic prenatal diagnosis. Further studies are currently being conducted.
