Abstract
A golgin family protein, Mea2, is expressed at enhanced level in pachytene spermatocytes and is indispensable for mouse spermatogenesis. Because Trax was shown to interact with Mea2 in yeast two-hybrid, we investigated the localization of Trax in pachytene spermatocytes with immunofluorescent staining. Trax was found to accumulate in the Golgi complex of mid-late pachytene spermatocytes and intermingled with granular Mea2 signal in the central region. In a subline of the Mea2 mutant mouse, a truncated form of Mea2 devoid of the N-terminal region, ΔMea2, was expressed. It localized to the rim of Golgi complex and thus occupied a region separate from that of Trax.
T
ΔMea2H was a subline of T604 transgenic mouse derived from the proband of a fertile male homozygote. Unlike the original mouse line in which only 9% of homozygous males showed fertility, the reproductive capability of ΔMea2H males was improved significantly and they sired consistently at ages above 10 weeks. However, testis weight remained at 49% of the hemizygous males, and the epididymal sperm count in ΔMea2H remained at 29% of the hemizygotes on average (Table 1). Sperm production of ΔMea2H homozygous males exhibited variation from 3.5 × 105 to 2 × 107 inclusive of apparently exceptional infertile animals. On the other hand, sperm production of the homozygotes in the original line was below 3.5 × 105 in 61% of individuals.
Mea2 was reported to interact with Trax in yeast two-hybrid and in GST pull-down experiments (Bray et al. 2002). We observed a similar molecular interaction in yeast two-hybrid and in transiently transfected cells (unpublished observations). Therefore, localization of Trax in pachytene spermatocytes in relation to Mea2 and ΔMea2 seemed to be an interesting issue to study. An anti-mouse Trax antibody feasible for immunofluorescent detection in mouse testis tissues was prepared as follows.
A full-length cDNA fragment of mouse Trax was produced by PCR with primers (5'-GGATCCAT-GAACGGCAAAGAAGGACCA-3', 5'-CCAGGAGC-TCCTTAGAGAGCA-3') and the template of Trax cDNA plasmid clone (pI151). The fragment was used to create a GST–Trax fusion protein in pGEX-6P-1. The recombinant fusion protein bound to glutathione Sepharose beads (GS4B) was washed and treated with PreScission protease (Amersham Bioscience; Piscataway, NJ) at 4C overnight. The cleaved mixture was separated with 12% SDS-PAGE. Trax (33 kD) was recovered as a gel strip cut out according to colored size markers. The cut-out gel was homogenized with an equal volume of PBS and was used to immunize guinea pigs by injecting 75 μg of Trax protein with Freund's complete adjuvant every 2 weeks over a period of 12 weeks.
Reduced reproductive capacity in the male of ΔMea2H homozygous subline (means ± SD)

The recombinant Trax recovered from the SDS-PAGE gel strip described above was electro-eluted (Electro-Eluter model 422; Bio-Rad Laboratories, Hercules, CA) and coupled to actigel ALD (Sterogene Bioseparations; Carlsbad, CA) for affinity-purification of anti-Trax antibody. The antibody eluted with ActiSep elution medium (Sterogene Bioseparations) was stored at 4C in PBS containing 0.02% sodium azide.
With the affinity-purified anti-Trax antibody, a major band at 33 kD of the size of mouse Trax and a minor band at 25 kD were detected in the immunoblot of normal mouse testis tissue lysate (Figure 1A). A DDY mouse (SLC; Shizuoka, Japan) was used for fractionation of testicular cells (Bellvé 1993). When the fractions of pachytene spermatocytes, round spermatids, and elongated spermatids were examined in the immunoblot, the minor band was greatly reduced in pachytene spermatocytes (at 80% purity) in contrast to the gradual increase of the minor band in round and elongated spermatids (Figure 1B). In pachytene spermatocytes, 33-kD Trax protein alone seemed to be present and detected by the antibody.
Mouse testes embedded in OCT compound (Sakura Fine Technical; Tokyo, Japan) were frozen in liquid nitrogen vapor. Cryosections were fixed in cold methanol. For triple immunofluorescent staining, anti-Trax antibody (Ab) was detected with FITC-conjugated anti-guinea pig IgG Ab (Sigma-Aldrich; St Louis, MO), anti-C-terminal Mea2 Ab (Banu et al. 2002) with AlexaFluor 633-conjugated anti-rabbit IgG Ab (Molecular Probes; Eugene, OR). Immunostaining with anti-GM130 monoclonal Ab (Santa Cruz Biotechnology; Santa Cruz, CA) was performed after blocking endogenous mouse IgG (MOM immunodetection kit; Vector Laboratories, Burlingame, CA) and detected with biotinylated anti-mouse IgG Ab followed by Texas Red-avidin. A confocal laser scanning microscope (LSM5; Carl Zeiss Meditek, Jena, Germany) was used for observation. GM130 was employed as a marker for the cis-Golgi protein (Nakamura et al. 1995).
Testis of an 18-day-old mouse was enriched with mid-late pachytene spermatocytes, mostly devoid of spermatids, and thus provided a suitable material for examining localization of Trax with immunostaining. In C3H pachytene spermatocytes, Trax was accumulated in the central region of an enlarged Golgi complex where Mea2 was detected intermingled with Trax (Figures 2A–2C). GM130 was detected in the rim region of the Golgi complex in separation from Mea2 (Figures 2D and 2E). In pachytene spermatocytes of ΔMea2H homozygotes, Trax was observed to accumulate in the central region of the Golgi complex (Figure 2F) as in C3H. ΔMea2, on the other hand, was localized not in the central region but in the rim domain where GM130 was present (Figures 2G-2J).
We investigated whether the distinctive localization patterns of Mea2 and ΔMea2 in relation to Trax observed above would hold true in the adult stage. Localization of Trax and Mea2 (ΔMea2) was examined with double immunostaining using rhodamine B-conjugated anti-rabbit IgG Ab (Biosource; Camarillo, CA) as the secondary antibody to detect the anti-Mea2 antibody. A bandpass filter BP505–530 was employed to obtain clear separation of FITC signal for Trax from the signal for Mea2. The same co-localization of Trax and Mea2 as shown in 18-day-old normal mice was invariably observed in the Golgi complex of normal spermatocytes in stages IV–XI (Figures 2K–2M). Trax and ΔMea2 in the mid-late pachytene spermatocytes of adult ΔMea2H homozygotes seemed to be separated into different regions of the Golgi complex (Figures 2N–2P) in the same manner as shown in 18-day-old mutants.
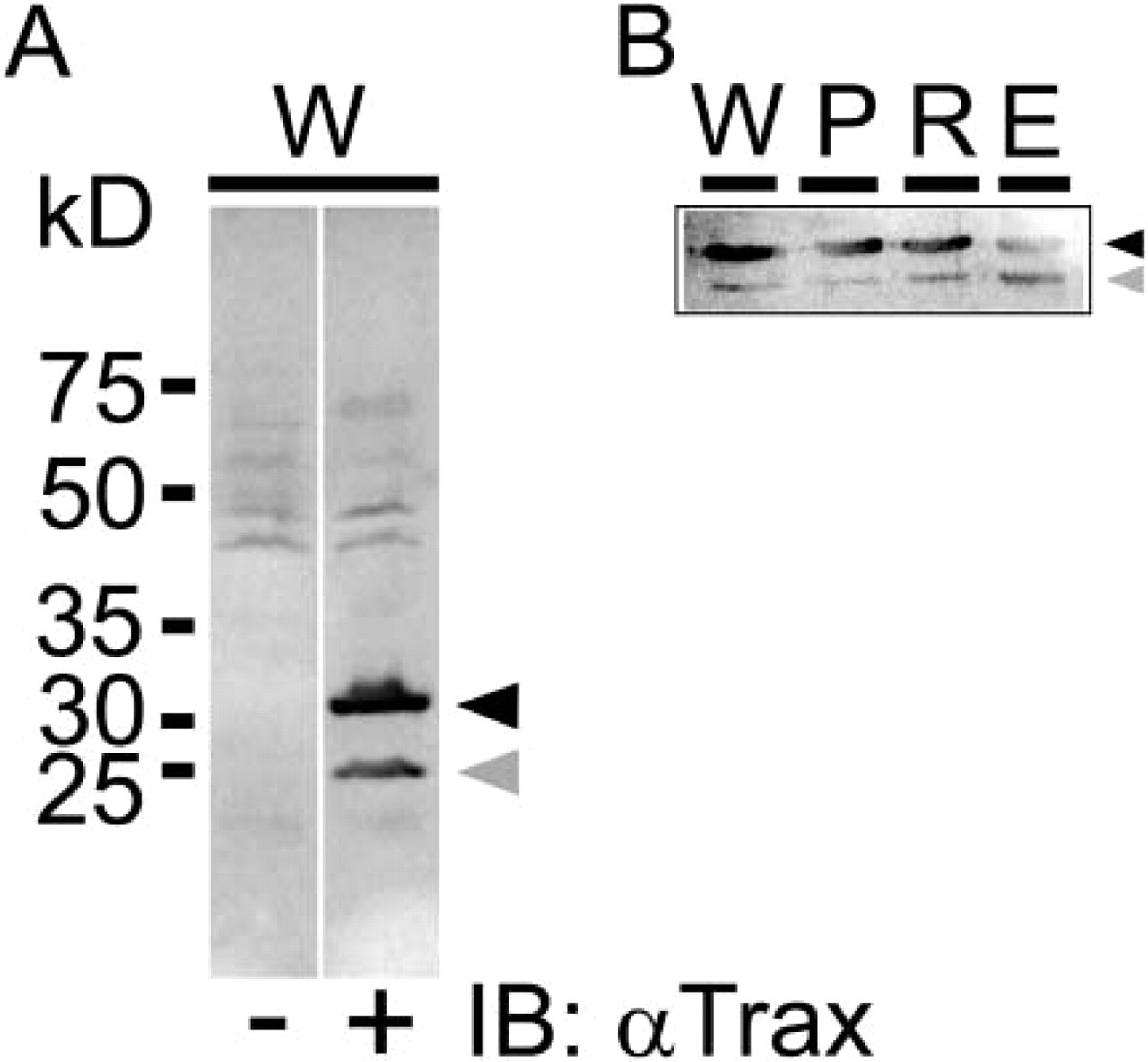
Specificity of anti-Trax antibody in testis tissue. (
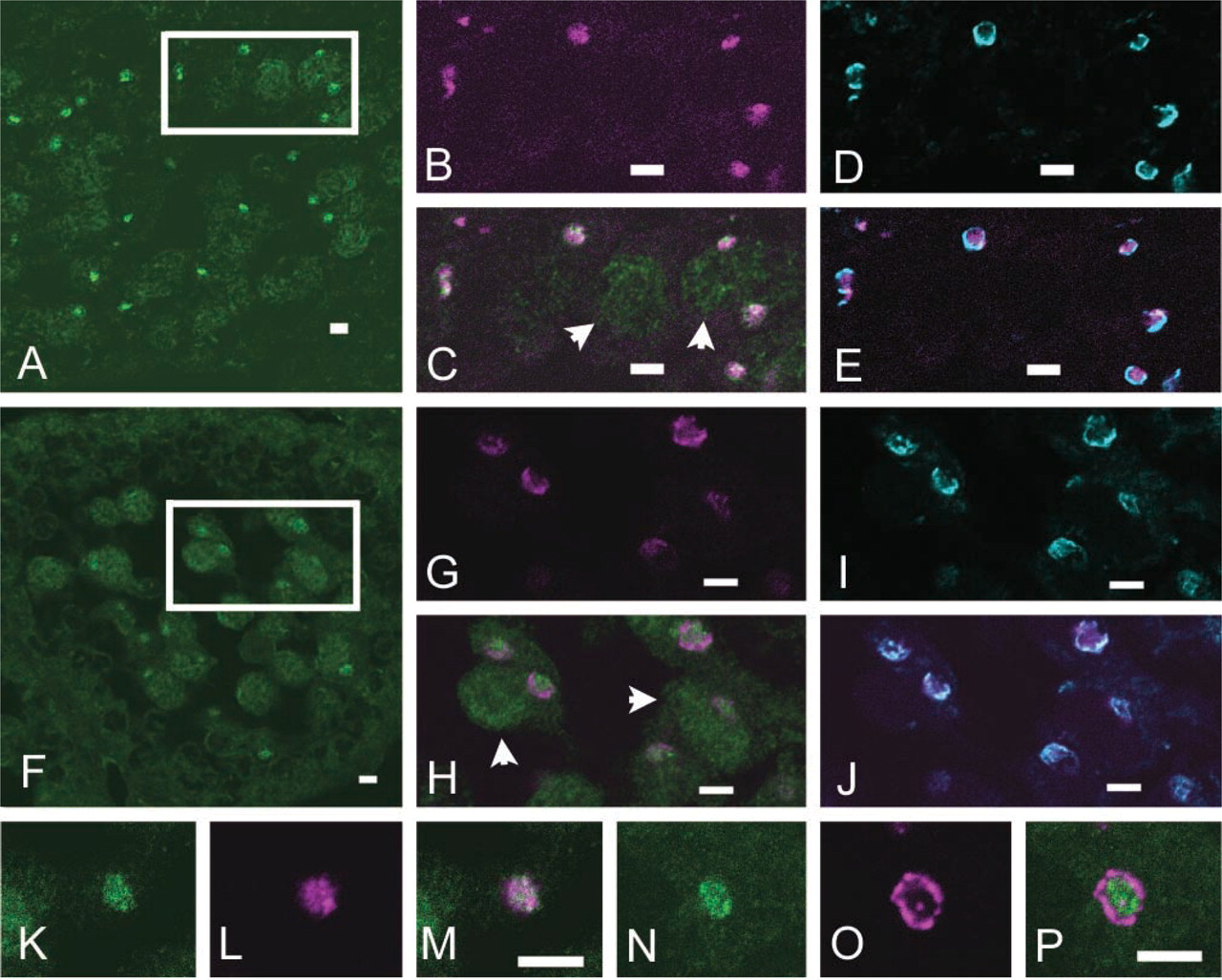
Immunofluorescent detection of Trax, Mea2, and ΔMea2 in Golgi complex of mid-late spermatocytes of prepubertal and adult mice. Wildtype (
The co-existence of Trax and Mea2 in the central region of Golgi complex in wild-type mid-late pachytene spermatocytes implied that the molecular interaction between the two molecules could occur in the organelle. On the other hand, Trax and ΔMea2, apparently localized in separate regions of the Golgi complex, might be restricted from interaction. Because spermatogenesis in ΔMea2H mice was significantly restored, the restricted interaction between ΔMea2 and Trax in the Golgi complex seemed to have little influence on the survival of mutant spermatocytes and their differentiation to fertile sperm. However, considering that the spermatogenic potency in ΔMea2H mice was still partial, the possibility remained that the molecular interaction between Mea2 and Trax could be important for augmentation of reproductive potency through unknown mechanisms.
The NLS motif in Trax (Aoki et al. 1997) was presumably important for the nuclear transport of Trax. We actually recognized Trax signal in some nuclei of late pachytene spermatocytes (Figures 2C and 2H). The significance of the transient shift of subcellular localization from cytoplasm to Golgi complex and to nucleus in pachytene spermatocytes remains unknown.
Mea2 consists of N-terminal proline-rich and serine-rich domains and coiled-coil regions (Misumi et al. 1997). Our results implied that the N-terminal region, lost in ΔMea2, might contain a motif important for localization of Mea2 in the central region of the Golgi complex in mid-late pachytene spermatocytes. A small Golgi protein, GCP16, was recently shown to interact with the human orthologue of Mea2, GCP170/golgin-160, in the N-terminal region (Ohta et al. 2003) and could be a candidate protein to regulate localization of Mea2 within the Golgi complex.
Footnotes
Acknowledgements
Supported in part by a grant from Ministry of Health, Labor and Welfare, Japan. The animal experiment code of Kanagawa Cancer Center was observed.
