Abstract
Tartrate-resistant acid phosphatase (TRAP) is essential for elimination of Staphylococcus aureus, the main infectious agent responsible for osteomyelitis. This in vitro study investigated uptake and processing of fluorescence-labeled S. aureus by human osteoclasts and dendritic cells. The cells were stained for TRAP and the acidic compartment using a fluorescence-based protocol. In dendritic cells, TRAP and bacteria were colocalized. In osteoclasts, there was no colocalization of bacteria, TRAP, or the acidic compartment, indicating that there are three distinct vesicular compartments: the apical phago-lysosomal compartment, the basal secretory compartment, and the basolateral transcytotic compartment. Dissociation of the TRAP-containing transcytotic vesicles from the apical phago-lysosomal compartment may restrain osteoclasts from eliminating S. aureus.
T
Materials and Methods
Cells
Human osteoclasts and dendritic cells were generated in vitro from adherent peripheral blood mononuclear cells (PBMCs) (Filgueira et al. 1996; Filgueira 2004). Briefly, PBMCs were isolated from buffy coats of healthy blood donors [Australian Red Cross Blood Service (ARCBS), Perth, Western Australia]. Blood from 20 donors was processed for this study and cultured for 2 hr (25-cm2 culture flasks; Sarstedt, Nümbrecht, Germany) in RPMI 1640 Glutamax medium (Invitrogen; Auckland, NZ) containing 10% human serum (ARCBS) and antibiotics (Invitrogen). Subsequently, the adherent cells were washed thoroughly with PBS (Invitrogen), and all non-adherent cells were discarded. For dendritic cells, the adherent PBMCs were cultured in RPMI 1640 Glutamax containing 5% human serum, antibiotics, recombinant human granulocyte macrophage colony-stimulating factor (50 ng/ml; Leucomax, Schering-Plough, Baulkham Hills, NSW, Australia), and interleukin-4 (10 ng/ml; R and D Systems, Minneapolis, MN) (Filgueira et al. 1996). For osteoclasts, the adherent PBMCs were cultured in RPMI 1640 Glutamax containing 5% human serum, antibiotics, recombinant human macrophage colony-stimulating factor (10 ng/ml; R and D Systems), and receptor activator of NF-κB ligand (10 ng/ml; R and D Systems) (Filgueira 2004). Medium, including all supplements, was replaced once a week.
Surface Markers
Cultured human dendritic cells and osteoclasts were characterized for expression of surface markers for up to 6 weeks in culture using flow cytometry and fluorescence-labeled mouse monoclonal antibodies binding specifically to human CD3, CD8, CD11c, CD14, CD19, CD33, CD80, and MHC class II (BD Biosciences; San Jose, CA).
Phagocytosis
The phagocytic capacity of cultured human dendritic cells and osteoclasts was quantified using flow cytometry and fluorescence confocal microscopy. For that purpose, 2-week cultured cells (105 to 106 cells/ml) were incubated with heat-inactivated fluorescence-labeled (tetramethylrhodamine or AlexaFluor 488) S. aureus (104 to 107 bacteria/ml; Molecular Probes, Eugene, OR) for 4–96 hr under culture conditions before being fixed with 1% paraformaldehyde in PBS. Cells without bacteria were used as control populations.
Electron Microscopy
Cultured human dendritic cells and osteoclasts were characterized using surface scanning electron microscopy (SEM) and transmission electron microscopy (TEM). The cells were fixed with 2.5% glutaraldehyde in culture medium. For SEM, the cells were processed through critical-point drying and sputtered with 15 nm of gold. They were analyzed with a Philips SEM500 microscope (Philips; Eindhoven, The Netherlands). For TEM, the cells were postfixed with an aqueous solution of 1% OsO4 containing 1.5% K4Fe(CN)6, and embedded in epon. Ultrathin sections were contrasted with lead citrate and uranyl acetate and studied with a Philips CM100 microscope and a JEOL2000 microscope (JEOL USA Inc.; Peabody, MA) (Filgueira et al. 1989).
Confocal Fluorescence Microscopy
The essential part of this study was to investigate whether bacteria-containing endo-lysosomes were acidic and contained TRAP. For that purpose, bacteria-treated (tetramethylrhodamine-labeled) and untreated cells were incubated with 50 μM DAMP-HCl(N-(3-[2,4-dinitrophenyl)amino] propyl)-N-(3-aminopropyl)methylamine; D-1565, Sigma-Aldrich, St Louis, MO) under culture conditions for 1 hr, expecting DAMP to accumulate in acidic vesicles (Megumi et al. 1999). Subsequently, the cells were fixed with 1% paraformaldehyde in PBS. The fixed cells were spun onto glass slides (Star Frost; Knittel Gläser, Braunschweig, Germany) and permeabilized with 0.1% Triton X-100 (ICN Biomedicals; Aurora, OH) in PBS for 1 min. The cells were stained for TRAP activity using a fluorescence-based protocol (Filgueira 2004). Briefly, the cells were incubated for 15 min with ELF97 substrate (20 μM, E6569; Molecular Probes) in 110 mM acetate buffer (pH 5.2) containing 1.1 mM sodium nitrite and 7.4 mM tartrate (Sigma-Aldrich). The acidic compartment was visualized by using a rabbit anti-dinitrophenol antibody (DakoCytomation; Carpinteria, CA) and a secondary AlexaFluor 488-conjugated goat anti-rabbit IgG antibody (Molecular Probes) (Megumi et al. 1999). The nuclei were stained with DAPI (4',6-diamidine-2'-phenylindole dihydrochloride, 10 ng/ml; Roche Diagnostics, Mannheim, Germany). After mounting (Dako Fluorescent Mounting Medium; DakoCytomation), the samples were analyzed and documented using confocal microscopy (Bio-Rad MRC 1024, Coherent Enterprise argon ion 250-mW multi-line UV, American Laser 100-mW argon ion multiline laser, Melles-Griot 0.5-mW green helium-neon laser; Bio-Rad, Hercules, CA).
Results
Characterization of Cells
Dendritic cells increased expression of MHC class II and CD80 and decreased expression of CD14 to almost undetectable levels. Osteoclasts decreased expression of CD14 and MHC class II to almost undetectable levels. However, both cell populations expressed high levels of CD11c (Lader et al. 2001; Nunez et al. 2004). The cells were negative for markers of T-lymphocytes (CD3), B-lymphocytes (CD19), NK-cells (CD8, CD56), monocytes (CD14), and myeloid precursor cells (CD33). Osteoclasts tested positive for expression of transcripts for calcitonin receptor, TRAP, and cathepsin K using reverse transcriptionpolymerase chain reaction (RT-PCR) protocols and specific primer pairs. Dendritic cells and osteoclasts were also tested for their T-lymphocyte-stimulatory capacity in mixed lymphocyte reactions after 1, 2, 3, and 4 weeks in culture. Dendritic cells increased their T-lymphocyte-stimulatory capacity over time in culture, whereas osteoclasts clearly decreased their potency to stimulate allogeneic T-lymphocytes, down to control levels after 3 weeks in culture. The cells were also tested for resorption of dentine slides. Only osteoclasts were able to resorb dentine.
The cells were also processed for SEM and TEM, confirming clear-cut ultrastructural differences between the two cell populations generated from the same common adherent PBMC precursor cells (Figure 1). Dendritic cells were non-adherent or loosely adherent cells of 12 μm to 15 μm in diameter, of varying shapes, with extended membrane protrusions of different shapes. They contained one lobulated nucleus, a large nucleolus, and abundant organelles, including lysosomes. Osteoclasts were large, polarized, adherent cells of 20 μm to 50 μm in diameter and displayed a mushroom-like appearance. They contained one or multiple oval-shaped nuclei with large nucleoli and abundant organelles.
Phagocytosis of Staphylococcus aureus
In addressing the aim of this study, dendritic cells and osteoclasts (105 to 106 cells/ml) cultured for 1 to 4 weeks were incubated with heat-inactivated fluorescence-labeled S. aureus (104 to 107 bacteria/ml) for 4–96 hr under culture conditions. Cells without bacteria were used as control populations. The cells were fixed with 1% paraformaldehyde in PBS. Phagocytosis of bacteria by the cells was quantified using fluorescence microscopy and flow cytometry (Figure 2), which indicated that dendritic cells and osteoclasts had similar capacities for bacterial uptake. After a 4-hr incubation of 5 × 105 cells with 5 × 106 bacteria, all cells had taken up bacteria. Bacterial load increased after longer incubation time without changing subsequent results.
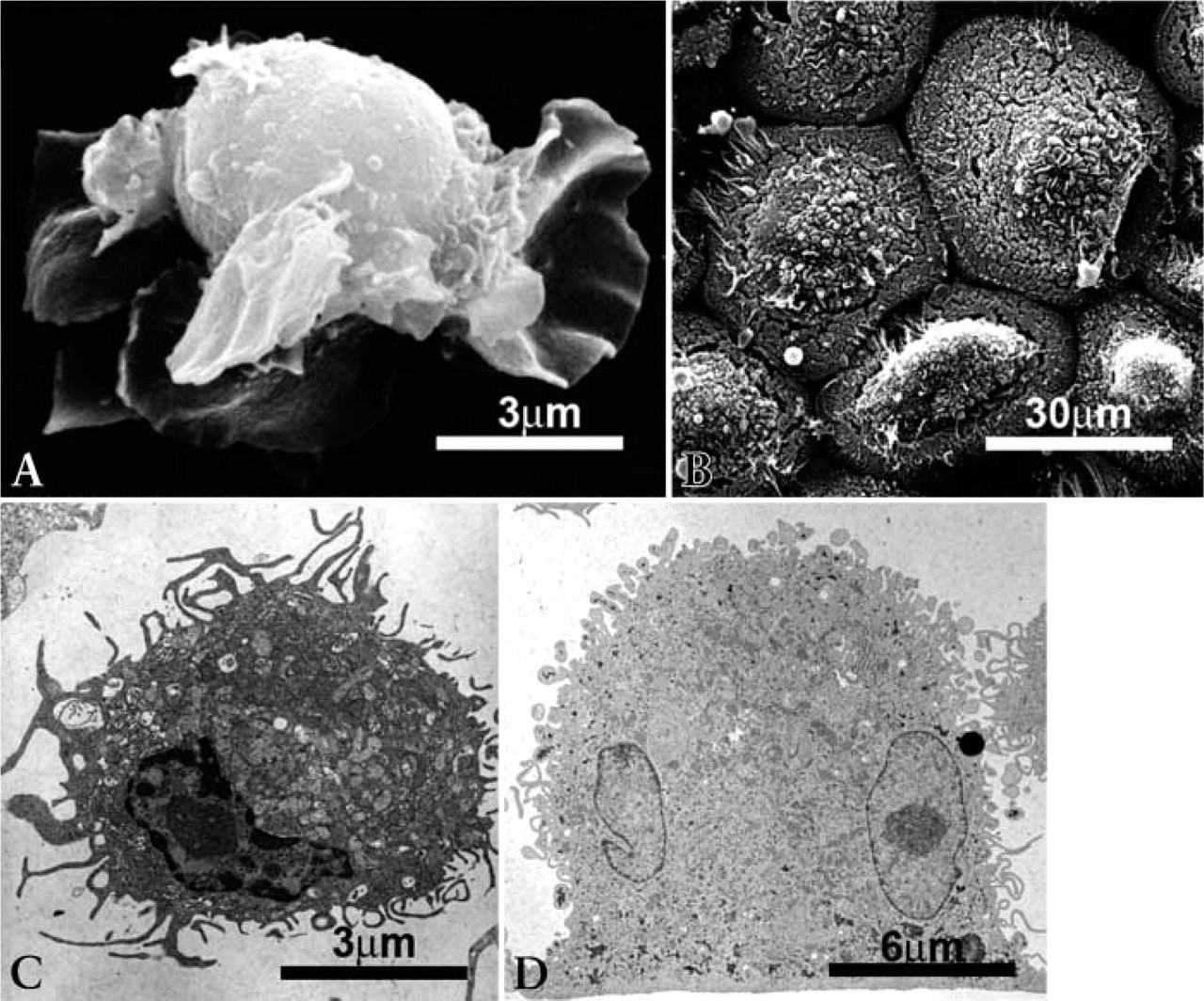
Surface scanning electron microscopy (SEM:
Intracellular Bacteria and Vesicular Compartments
The next step of the study was to investigate whether bacteria-containing endo-lysosomes were acidic and contained TRAP. For that purpose, acidic vesicles and TRAP-containing vesicles were stained in bacteria-treated (tetramethylrhodamine-labeled) and untreated cells using a fluorescence-based protocol (Megumi et al. 1999; Filgueira 2004).
In dendritic cells, TRAP activity was clearly colocalized with bacteria in endo-lysosomes (Figure 3A). Surprisingly, using the described four-color staining protocol, no overlapping of TRAP-positive, acidic, or bacteria-containing vesicles was detected in osteoclasts (Figures 3B and 3C). These results were repeatedly confirmed using cells from different blood donors and cultured for different time periods (1 to 4 weeks) and different incubation times (4–96 h). Similar results were also documented when using fluorescence-labeled Escherichia coli (data not shown).
Discussion
Staphylococcus aureus is the most frequent bacterial causative for bone infections (Mandal et al. 2002). Studies using animal models have shown that lysosomal expression of TRAP in macrophages plays a crucial role in eliminating bacteria (Hayman and Cox 2003). Because osteoclasts express TRAP, one would expect them to be optimally equipped for the phagolysosomal killing of the bacteria. However, little is known about the role of human osteoclasts in the context of bacterial osteomyelitis. Therefore, this study investigated uptake and processing of S. aureus by human osteoclasts in comparison with autologous dendritic cells.
First, the cellular human in vitro model was established, and the cells were characterized for ultrastructure, phenotype, and function. Despite having a different aim and knowing that a comparison of mouse osteoclasts and dendritic cell differentiation has already been published (Miyamoto et al. 2001), the present study is the first to compare autologous human monocyte-derived dendritic cells and osteoclasts. As expected, the dendritic cells displayed all the characteristics of immature cells, able to take up and process antigens, including bacteria, and already able to have increased T-cell-stimulatory capacity (Filgueira et al. 1996). In contrast, although derived from the same blood precursor cell population, the osteoclasts showed distinct ultrastructural features different from the dendritic cells, including increased size, multiple nuclei, organized basal adherence (sealing zone and supporting acting ring), and polarization (Figure 1). The phenotype of the monocyte-derived osteoclasts corresponds to the features published by Lader et al. (2001), including expression of CD11c. In addition, the osteoclasts were able to resorb calcified extracellular matrix when cultured on dentine slides. In summary, the present study used a well-characterized autologous human in vitro cellular model, comparing human dendritic cells and osteoclasts.
Measurement of bacterial phagocytosis was essential for this study. Immature dendritic cells have already been shown to be potent phagocytes able to take up whole microorganisms, including bacteria (Filgueira et al. 1996; Nagl et al. 2002). However, little is known about bacterial uptake by human osteoclasts. Therefore, this study also explored the phagocytosis capacity of osteoclasts. For that purpose, the cells were incubated with fluorescence-labeled, heat-inactivated S. aureus and E. coli (data not shown) for up to 96 hr. Bacterial uptake was quantified using fluorescence confocal microscopy and flow cytometry (Figure 2). Both cell populations displayed similar phagocytosis capacity. Being polarized adherent cells, osteoclasts’ non-bone-related uptake is restricted to the apical membrane area. Finally, the present study indicates that human osteoclasts have phagocytosis features similar to those of other cells of the macrophage family.
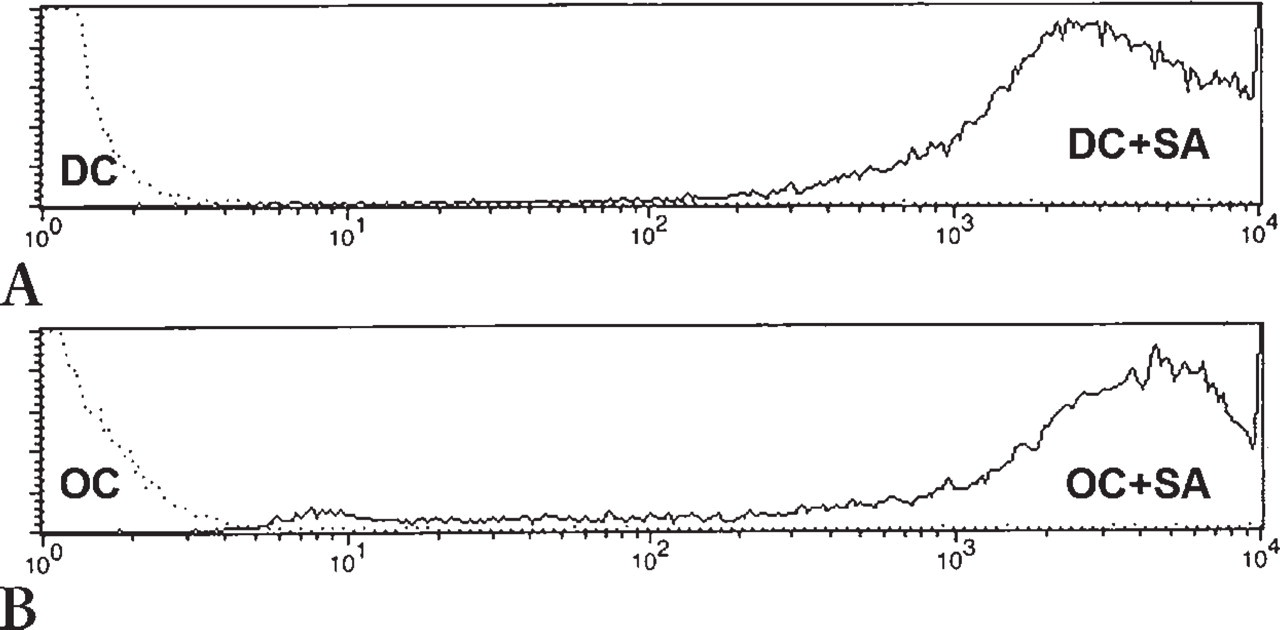
Uptake of S. aureus (SA) by dendritic cells (DC:
Processing of bacteria by dendritic cells has been investigated extensively in the past by many research groups (for review see Wick and Ljunggren 1999; Harding et al. 2003). Dendritic cells have been shown to kill phagocytosed bacteria (Filgueira et al. 1996; Nagl et al. 2002). Lysosomal TRAP therefore seems to play an important role in eliminating S. aureus (Räisänen et al. 2001; Hayman and Cox 2003). Those published data correlate well with the findings of the present study, which show that there is colocalization of TRAP and bacteria in the phagolysosomes of human dendritic cells. However, this study also indicates that processing of bacteria by osteoclasts differs clearly from what is known for dendritic cells and other macrophages.
Due to their unique function of bone resorption, osteoclasts have distinct, functionally, and ultrastructurally separated vesicular compartments (Figure 4). First, there is one compartment for the supply of enzymes for bone resorption, secreting the vesicular lysosomal content through the basal ruffled border into the resorption lacuna (Väänänen et al. 2000). The acidic vesicles form part of this basal secretory compartment (Megumi et al. 1999; Hollberg et al. 2002). Second, there is the vesicular compartment for the transcytosis of resorbed bone material through the cell from the ruffled border to the functional secretory domain (Väänänen et al. 2000). TRAP itself is located in lysosomal storage granules, which fuse with the transcytotic compartment and help with further degradation of resorbed material (Väänänen et al. 2000). Third, there is the apical endocytotic compartment, where phagocytosed material, including bacteria, is located, as shown by the present study. In addition, this study indicates that TRAP-containing vesicles do not fuse with the apical phagosomes (Figure 3).
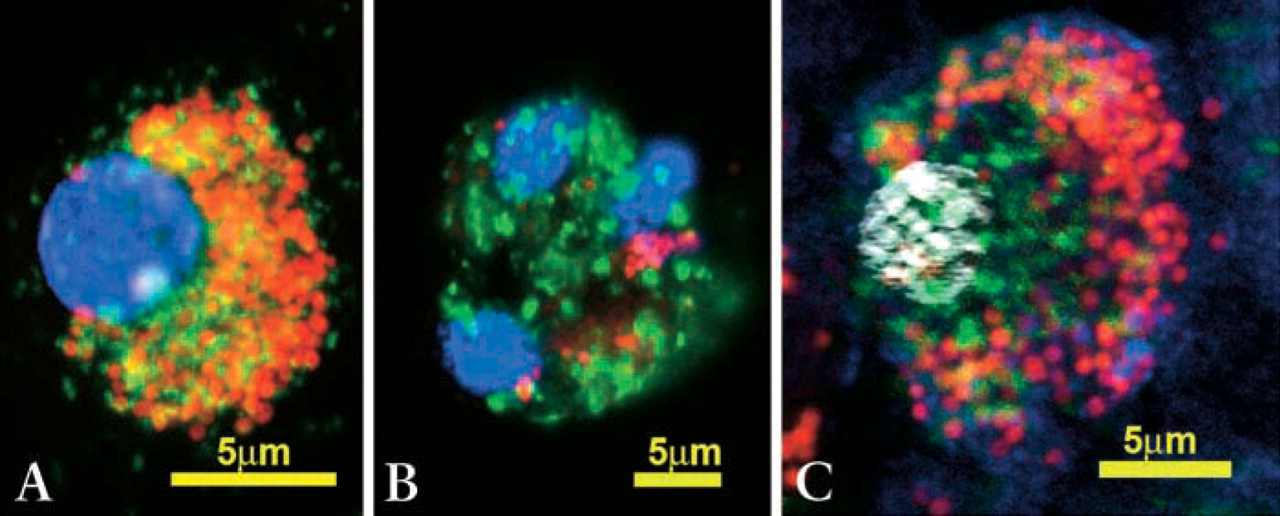
Confocal fluorescence microscopy of fluorescence-labeled intracellular S. aureus (red) after phagocytosis by dendritic cells (
Separation and sorting of the vesicles of the diverse compartments requires a fine-tuned transport function supported by the cytoskeleton. With this in mind, there has been the question of whether S. aureus may influence the cytoskeleton of osteoclasts and, by doing so, prevent fusion of TRAP-containing vesicles with phagosomes. Confocal fluorescence microscopy and staining for microtubules and actin filaments revealed no evidence for bacterial action on the cytoskeleton of osteoclasts (data not shown). Consequently, separation of the TRAP-containing transcytotic compartment from the apical phagocytotic compartment seems to be a constitutive property of osteoclasts.
Most importantly, the physiological separation of apical endocytotic and TRAP-containing transcytotic vesicles may impede the action of TRAP on bacteria and restrain osteoclasts from eliminating endocytosed S. aureus. Finally, the separation of the vesicular compartments in osteoclasts may be responsible for the characteristics of osteomyelitis, where precursor cells of the macrophage family are recruited to the site of inflammation (Kataoka et al. 2000). The bone environment and certain staphylococcal factors direct differentiation of the precursor cells toward osteoclasts with their distinct function of bone resorption (Meghji et al. 1998), resulting in bone destruction instead of the elimination of bacteria.
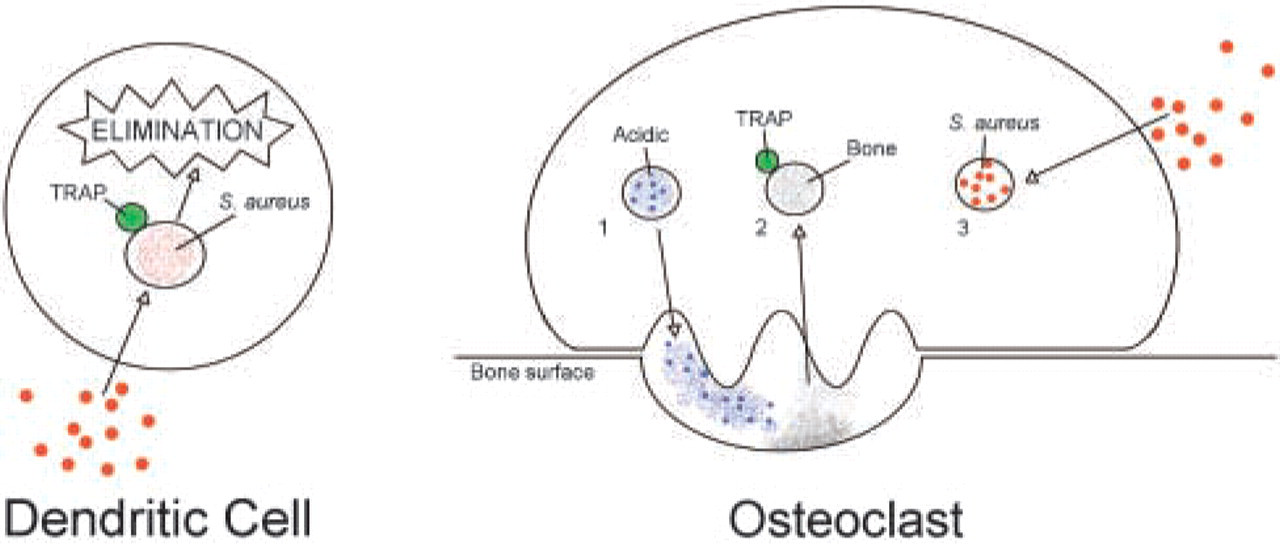
Comparison of bacterial processing by human monocyte-derived dendritic cells and osteoclasts. In dendritic cells, phagosomes fuse with TRAP-containing lysosomes, resulting in killing and degradation of the bacteria. In osteoclasts, because of physiological and functional separation of the basal acidic compartment (1) and the TRAP-containing transcytotic compartment (2) from the apical phagosomal compartment (3), bacteria may not be killed and disrupted, resulting in bacterial survival and possibly intracellular bacterial growth.
Footnotes
Acknowledgements
This study was supported by the S.E. Ohman Medical Research Fund (Western Australia), the Dudgeon Smith Medical Research Fund (Western Australia), and by the AO Research Fund (Switzerland).
We thank Miriam Erni, Urs Ziegler, and Peter Groscurth (Institute of Anatomy, University of Zurich, Switzerland), Paul Rigby and Kathryn Heel (Biological Imaging and Analysis Facility, University of Western Australia), and Guy Ben-Ary (Image Acquisition and Analysis Facility, University of Western Australia) for excellent technical support and advice. Electron microscopy was done at the Institute of Anatomy (University of Zurich) and at the Centre for Microscopy and Microanalysis, University of Western Australia.
