Abstract
We assessed the feasibility of fetal RHD and RHCE genotyping by analysis of DNA extracted from plasma samples of RhD-negative pregnant women using real-time PCR and primers and probes targeted toward RHD and RHCE genes. We analyzed 45 pregnant women in the 11th to 40th weeks of pregnancy and correlated the results with serological analysis of cord blood after delivery. Non-invasive prenatal fetal RHD exon 7, RHD exon 10, RHCE exon 2 (C allele), and RHCE exon 5 (E allele) genotyping analysis of maternal plasma samples was correctly performed in 45 out of 45 RhD-negative pregnant women delivering 24 RhD-, 17 RhC-, and 7 RhE-positive newborns. Detection of fetal RHD and the C and E alleles of RHCE gene from maternal plasma is highly accurate and enables implementation into clinical routine. We recommend performing fetal RHD and RHCE genotyping together with fetal sex determination in alloimmunized D-negative pregnancies at risk of hemolytic disease of the newborn. In case of D-negative fetus, amplification of another paternally inherited allele (SRY and/or RhC and/or RhE positivity) proves the presence of fetal DNA in maternal circulation.
The Rh blood group system is the most polymorphic of the human blood groups, consisting of at least 45 independent antigens, and is second to ABO as the most clinically significant in transfusion medicine (Avent and Reid 2000). Hemolytic disease of the newborn (HDN) is, in 50% of cases, caused by maternal anti-D (IgG) antibody crossing the placenta and binding to fetal red blood cells (RBCs), followed by their destruction, which results in anemia (Avent 1998; Avent and Reid 2000). The recent discovery of cell-free fetal DNA in maternal peripheral blood (serum or plasma) has opened up new possibilities for non-invasive prenatal diagnosis (Lo et al. 1997). With the use of real-time PCR technology, it is possible to successfully determine the Rhesus D status and sex of the unborn infant and other fetal-derived, paternally inherited alleles (Lo et al. 1998a,b; Hahn et al. 2000; Chiu et al. 2001; Costa et al. 2001; Zhong et al. 2001; Finning et al. 2002; Honda et al. 2002; Legler et al. 2002; Hromadnikova et al. 2003).
In this prospective study, we assessed the feasibility of fetal RHD, RHC, and RHE genotyping by analysis of DNA extracted from plasma samples of RhD-negative pregnant women homozygous for c and/or e alleles of the RHCE gene using real-time PCR and primers and probes targeted toward the RHD (exon 7 and exon 10) and RHCE (exon 2 and exon 5) genes.
Materials and Methods
Blood samples from Caucasoid blood donors with D-negative (ccddee, Ccddee, ccddEe) and D-positive phenotypes (CCDee, ccDEE, ccDee) were used to establish and validate the efficacy of RHD (exon 7 and exon 10) and RHCE (exon 2 and exon 5) real-time PCR assays.
Forty-five consecutive RhD-negative pregnant women including those alloimmunized (1X anti-D, 3X anti-D+C, 1X anti-Kell) at risk for HDN at a gestational stage ranging from 11 to 40 weeks were recruited for the study.
The study was approved by the Ethics Committee of our local institution, and informed consent was obtained from all study patients.
DNA Extraction from Plasma Samples
Ten milliliters of maternal peripheral blood from pregnant women was collected into EDTA-containing tubes and processed within a few hours (maximally 24 hr). In detail, blood samples were centrifuged first at 1200 × g (protocol 1) (Hahn et al. 2000; Hromadnikova et al. 2003) and then at 3000 × g (protocol 2) for 10 min. (Lo et al. 1998b). Plasma samples were then recentrifuged, and the supernatants were collected and stored at −80C until further processing. DNA was extracted from 400 μl plasma using QIAamp DNA Blood Mini Kit (Qiagen; Hilden, Germany) according to the manufacturer's instructions. To minimize the risk of contamination, DNA was isolated in laminar airflow and aerosol resistant tips were used. DNA was eluted in 50 μl Buffer AE. Five μl of DNA were used as a template for the RHD PCR reaction and 2.5 μl of DNA for the β-globin (GLO) PCR reaction.
Real-time PCR Analysis
Real-time PCR analysis was performed using ABI PRISM 7700 Sequence Detection System (Applied Biosystems; Branchburg, NJ).
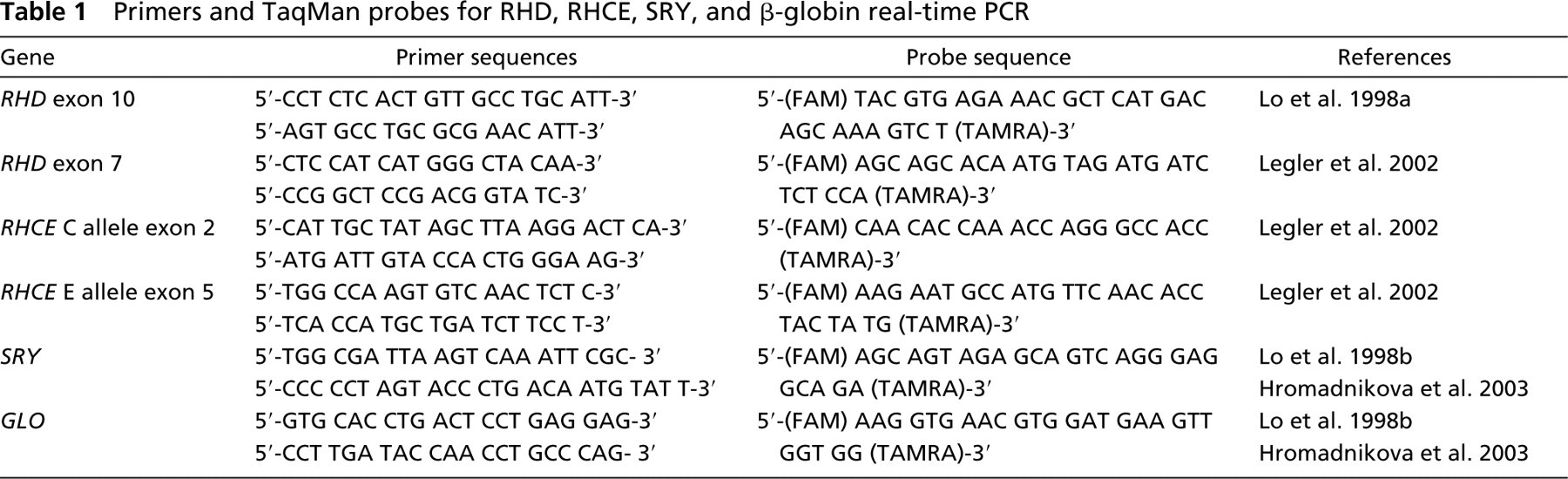
Primer and probe sequences are shown in Table 1. The GLO gene served as a control to confirm the presence and quality of DNA in each sample (Lo et al. 1998b; Hromadnikova et al. 2003). Amplicons for the GLO control gene were detected in all analyzed samples.
TaqMan amplification reactions were set up in a reaction volume of 25 μl using the TaqMan Universal PCR Master Mix (Applied Biosystems). Primers and probes were optimized to determine the minimum primer and probe concentrations that give the maximum normalized reporter. RHD exon 10 probe was used at concentrations of 100 nM and RHD exon 7, RHCE, and GLO probes at concentrations of 200 nM. PCR primers were used at a final concentration of 200 nM and 300 nM. DNA amplifications were carried out in 8-well reaction optical tubes/strips (Applied Biosystems). The TaqMan PCR conditions were used as described in TaqMan guidelines, using 50 cycles of 95C for 15 sec and 60C for 1 min, with 2-min preincubation at 50C required for optimal AmpErase UNG activity and 10-min preincubation at 95C required for activation of AmpliTaq Gold DNA polymerase. Each sample was analyzed in at least five replicate settings. A patient's specimen was considered positive if one or more individual replicates was positive [threshold cycle (Ct) <40].
Results
Fetal RHD, RHC, and RHE Genotyping from Maternal Plasma
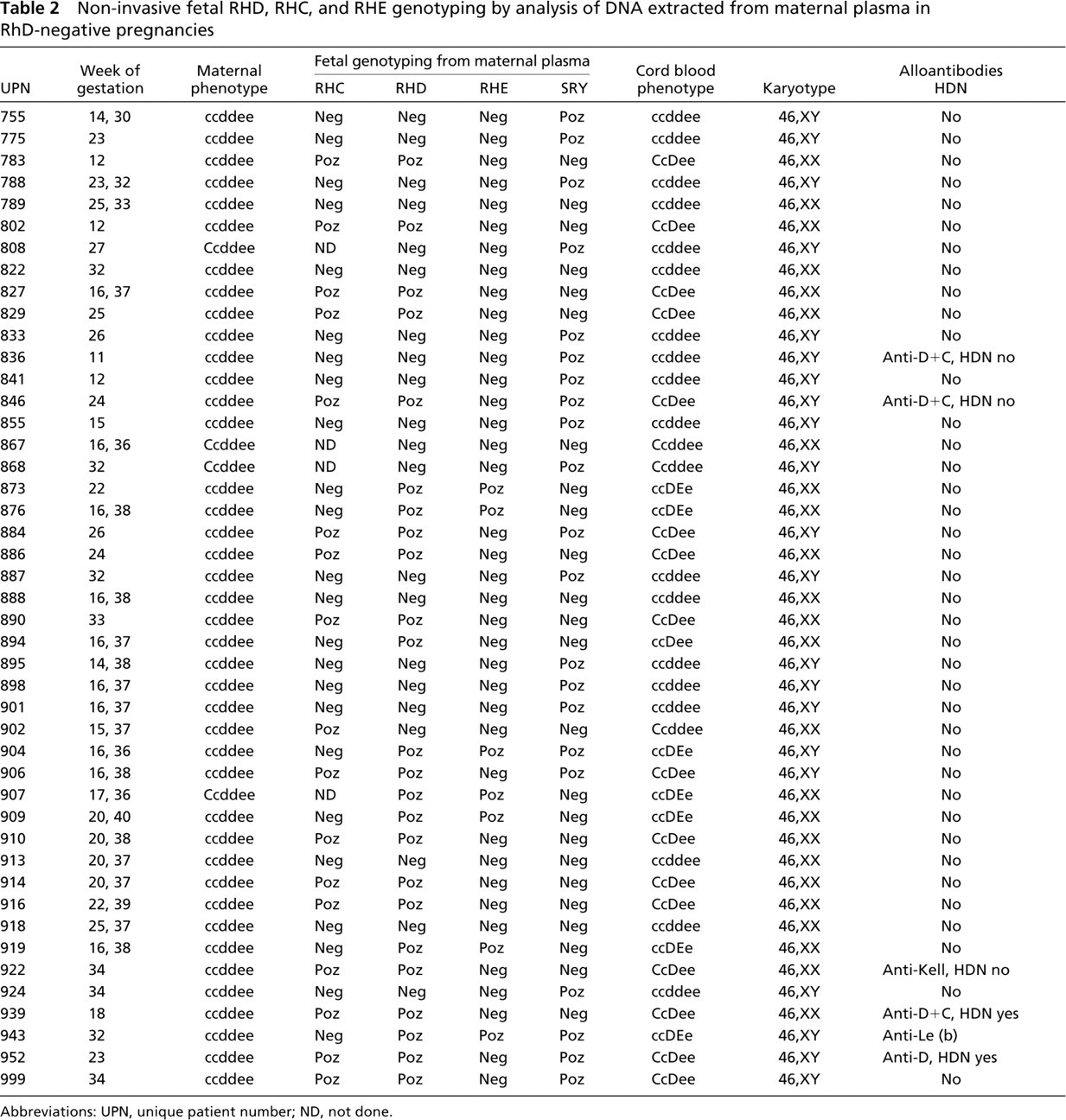
We analyzed plasma samples of 45 pregnant women with ccddee (n=41) and Ccddee (n=4) phenotypes in the 11th to 40th weeks of pregnancy and correlated results with the serological analysis of cord blood.
Non-invasive prenatal fetal RHD exon 7 and exon 10 genotyping analysis of maternal plasma samples was in complete concordance with the analysis of cord blood in 45 out of 45 RhD- negative pregnant women delivering 24 RhD-positive- and 21 RhD-negative newborns. Non-invasive prenatal fetal RHC exon 2 genotyping was performed as well in 41 out of 41 Rhc homozygote pregnant women delivering 17 RhC-positive- and 24 RhC-negative newborns. Similarly, noninvasive prenatal fetal RHE genotyping was done as well in 45 out of 45 Rhe homozygote pregnant women delivering 7 RhE-positive- and 38 RhE-negative newborns. Results are summarized in Table 2.
In 12 pregnancies (e.g., 755, 775, etc.) male fetus with cde paternally inherited haplotype was identified. In these cases RHD-, RHC-, and RHE-negative results may be considered as specific, because SRY amplification confirmed the presence of fetal DNA in maternal plasma samples (Table 2).
Fetal RHC and RHE genotyping is also important in pregnancies bearing an RhD-negative female fetus because amplification of another paternally inherited allele (RhC or RhE positivity) proves the presence of fetal DNA in maternal circulation, as we showed in the case of patient no. 902.
Primers and TaqMan probes for RHD, RHCE, SRY, and β-globin real-time PCR
Non-invasive fetal RHD, RHC, and RHE genotyping by analysis of DNA extracted from maternal plasma in RhD-negative pregnancies
Abbreviations: UPN, unique patient number; ND, not done.
SRY real-time PCR analysis of maternal plasma was in complete concordance with fetal sex in all 45 RhD-negative pregnant women (Table 2). We confirmed the sensitivity and specificity of the system previously in a cohort of pregnancies at risk for X-linked hemophilia and chromosomal aneuploidies when we determined fetal sex by analysis of DNA in maternal plasma using real-time PCR and the SRY gene as a marker for the detection of the fetal Y chromosome (Hromadnikova et al. 2003).
The amplification efficiencies of the assays specific for SRY, RHD exon 7, RHD exon 10, and RHC were comparable. The amplification occurred on a similar Ct (the difference less than 1.5 Ct). However, a lower efficacy was observed for RHE system. The amplification of fetal DNA occurred 1.0 to 4.0 (mean 2.0) Ct later.
Discussion
We confirmed that prediction of fetal Rhesus D status from maternal plasma is highly accurate and enables implementation into clinical routine (Lo et al. 1998a; Hahn et al. 2000; Chiu et al. 2001; Zhong et al. 2001; Finning et al. 2002; Legler et al. 2002). To avoid false-negative results the assay performing fetal RHD genotyping using maternal plasma samples should involve the amplification of at least two RhD-specific products. We suggest that safe, non-invasive prenatal fetal RHD genotyping using maternal plasma should follow the recommended procedure for RHD genotyping of partial D and weak D (Flegel and Wagner 2002).
We suggest checking one of the RhD-specific nucleotides in RHD exon 7 and at least one second region: exon 4, intron 4 (Cotorruelo et al. 2002) or exon 10 of RHD gene. RHD genotyping based solely on RhD-specific sequences in the 3' untranslated region of RHD exon 10 is not considered a safe procedure (Flegel and Wagner 2002). When D-positive fetuses are identified and anti-D alloantibodies are already present, it is important to carefully monitor anti-D alloantibody titer variations during the pregnancy so that fetal-blood sampling can be performed in time to determine the level of fetal hemolysis and to start early treatment of sensitized RhD-negative pregnancies (ultraviolet phototherapy or exchange or intrauterine transfusion, if required). On the other hand, detection of a D-negative fetus excludes the risk of HDN caused by anti-D alloantibodies that may be present in the maternal circulation from previous pregnancies for a variety of reasons, including non-administration of Rh immunoglobulin, unrecognized miscarriage, leakage of fetal RBCs into the maternal circulation in late pregnancy, etc. (Avent 1998; Avent and Reid 2000). However, the identification of a D-negative fetus should be verified if another fetal marker is detected in maternal plasma samples. SRY-positive- and RHD-, RHC-, and RHE-negative genotyping results on the same maternal plasma sample identify a male fetus with cde paternally inherited haplotype. SRY positivity proves the presence of fetal DNA in a maternal plasma sample and may confirm the specificity of the RHD-, RHC-, and RHE-negative results. Similarly, in a case of an identification of an RhD-negative female fetus, amplification of another paternally inherited allele (RHC or RHE positivity) proves the presence of fetal DNA in the maternal circulation.
In other cases (e.g., a female fetus with ccddee phenotype), the specificity of RhD-negative results remains uncertain, inasmuch as we do not have any other marker to confirm the presence of fetal DNA in maternal circulation.
We recommend performing fetal RHD, RHC, and RHE genotyping together with fetal sex determination in alloimmunized pregnancies at risk of HDN due to the presence of anti-D, anti-D+C, or anti-D+C+E alloantibodies. When maternal anti-D+C are identified in sera from alloimmunized women, it is important to genotype fetuses for RhD, RhC, and RhE status to establish whether they express Dce, DCe, dCe, DcE, DCE, or dCE paternally inherited haplotypes and are at risk for HDN (Avent 1998). While larger and confirmatory studies are required, these data present a compelling argument that this type of testing may be incorporated into our clinical diagnostic algorithm for following pregnancies at risk for hemolytic disease of the newborn.
Anti-E rarely produces clinically significant alloimmunization (Avent 1998; Avent and Reid 2000).
Footnotes
Acknowledgements
This project was supported by the 2nd Medical Faculty, Charles University, Prague (No: VZ 111300003). This research was also supported by MSM 0021620806.
