Abstract
Hepatoblastoma is a pediatric liver tumor with epithelial components resembling embryonal and fetal liver cells. The existence of teratoid hepatoblastoma suggests the presence of stem cells in hepatoblastoma. The aim of this study was to analyze the expression of stem cell markers in hepatoblastomas. We studied specimens from 10 hepatoblastomas. Five of the hepatoblastomas were of epithelial and five of mixed type. Immunohistochemistry (IHC) for the stem cell markers CD34, Thy1, c-kit, and the hepatic or biliary lineage markers CK-18, OCH, CK-7, and CD56 was performed. Double IHC for stem cell and lineage markers was used to identify putative liver stem cells. The different markers showed distinct distributions on the tumor cells. Cells in atypical ducts were found to express simultaneously stem cell markers and hepatocytic or biliary lineage markers. Other cells in connective tissue showed c-kit expression, but not hepatic or biliary marker expression. The data show the presence of different cell populations bearing stem cell markers in human hepatoblastoma. Ductal cells co-expressing stem cell markers and hepatic lineage markers phenotypically resemble hepatic stem-like cells. These findings support the thesis that stem cells play a role in the histogenesis of hepatoblastoma.
H
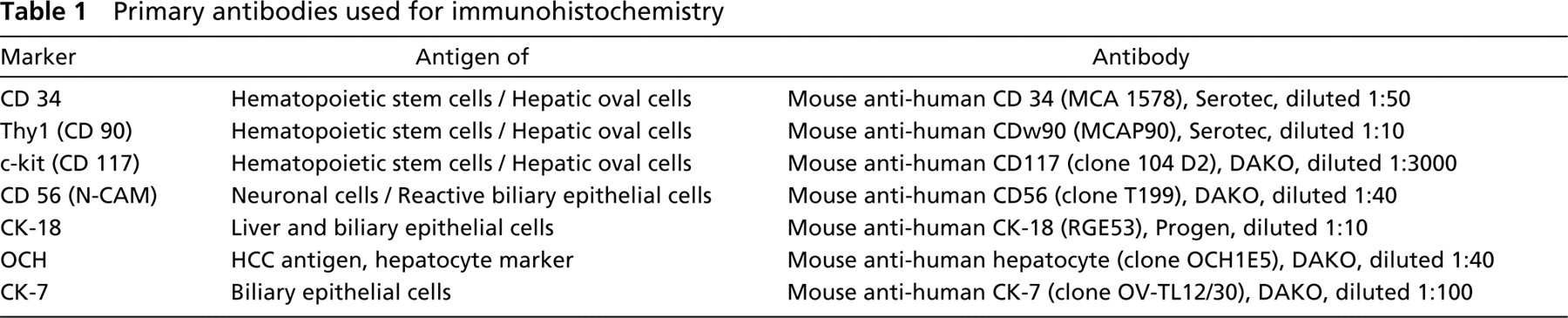
Primary antibodies used for immunohistochemistry
Materials and Methods
Harvest and Preparation of the Hepatoblastomas: H and E Staining
Ten hepatoblastomas (from patients aged 2 weeks to 12 years) resected after chemotherapy or directly as liver explantation after LTX were investigated. The tissue was snapfrozen in liquid nitrogen and stored at −80C for further processing. Cryostat sections were cut at 2 μm and were fixed with acetone at 4C for 90 sec. Slides were either stained directly or stored frozen at −20C until staining. Hematoxylin and eosin staining was performed, and the hepatoblastomas were classified histologically after the system of Ishak and Glunz (1967).
Immunohistochemistry
IHC analysis was performed using mouse monoclonal antibodies (MAbs) specific for CD34, Thy1, c-kit, CK-18, OCH, CK-7, and CD56 (specification and details in Table 1). Slides were rinsed with Tris buffer (Tris-buffered saline 0.05 M, pH 7.5) between the incubation steps.
Staining was performed using the alkaline phosphataseanti-alkaline phosphatase (APAAP) technique. Incubation with primary antibodies was 30 min. Antibodies were differently diluted (Table 1). Secondary marking was done with a rabbit anti-mouse IgG MAb diluted 1:50 for 30 min. Slides were then incubated with mouse-APAAP complex diluted 1:100 for 30 min. The alkaline phosphate substrate, New Fuchsin, was prepared as described elsewhere and the enzymatic reaction was allowed to proceed for 20 min. After rinsing in distilled water, slides were counterstained with hematoxylin.
Double Labeling of Thy1 and CK-18, Thy1 and CD56, or CK-18 and c-kit
For double staining cells were first stained with Thy1- or CK-18-APAAP as described above. After Thy1/CK-18 staining, slides were incubated with 20% horse serum (Vectastain; Vector Laboratories, Burlingame, CA) for 20 min. After rinsing in TRIS, the avidin and biotin blocking was performed for 15 min each. Slides were incubated with CK-18, CD56, or c-kit MAb for 30 min. A biotinylated antibody (Vectastain ABC kit) was then used as secondary antibody for 20 min. Avidin-biotin complex (Vectastain ABC kit) was prepared, and the enzymatic reaction was allowed to proceed for 20 min. The DAB staining reaction lasted for 10 min. Slides were counterstained with hematoxylin.
Results
Histological Classification of the Hepatoblastomas
Five of the 10 hepatoblastomas were classified as epithelial type and five as mixed type hepatoblastoma (Table 2). In two cases the epithelial type hepatoblastomas were pure fetal and in three cases mainly fetal. In the mixed type hepatoblastomas, the epithelial component was pure fetal in one case and mainly fetal in two cases (Table 2).
IHC for Hepatic or Biliary Epithelial Markers CK-18, OCH, CK-7, and Neural Cell Adhesion Molecule N-CAM (CD56)
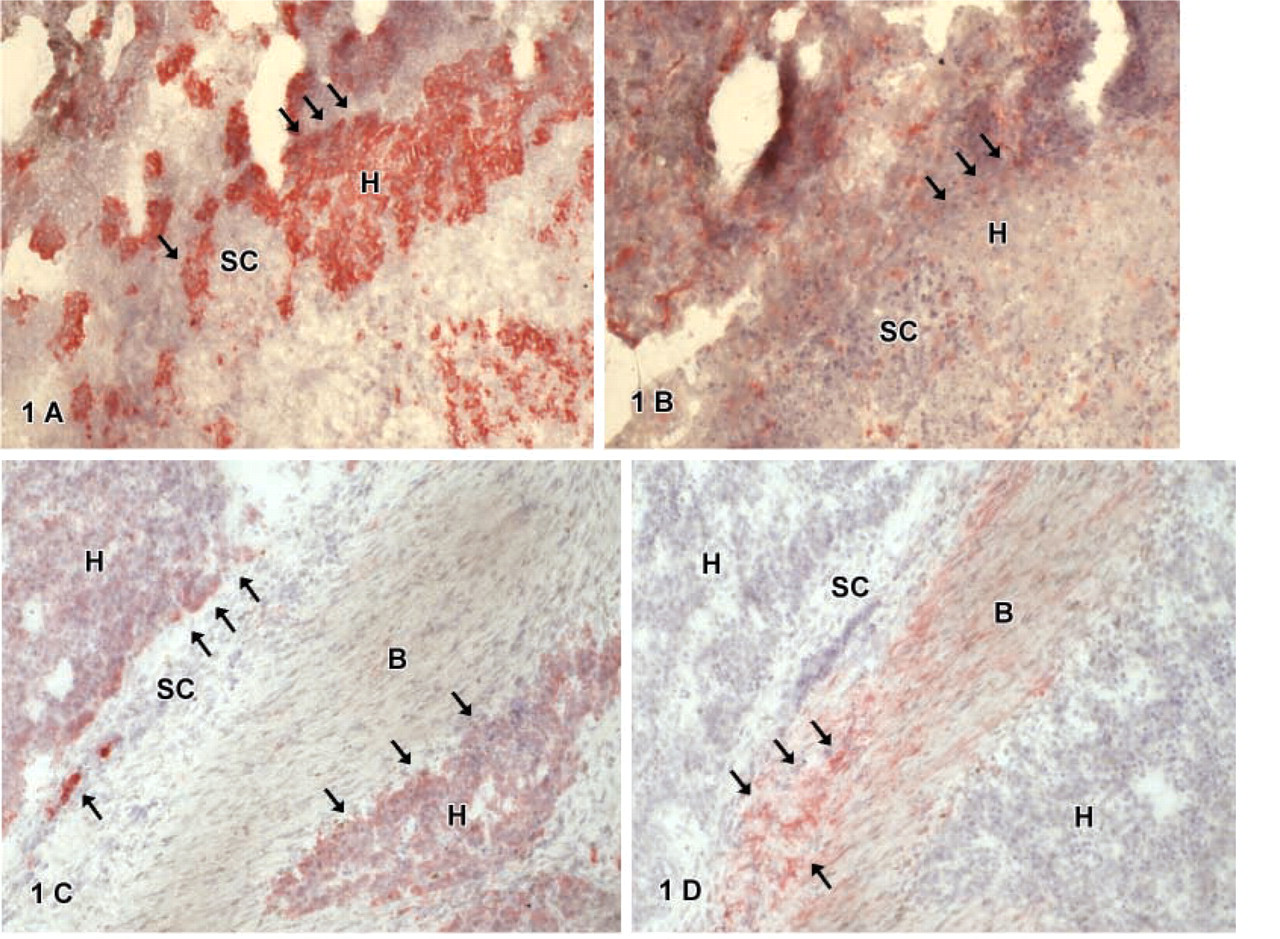
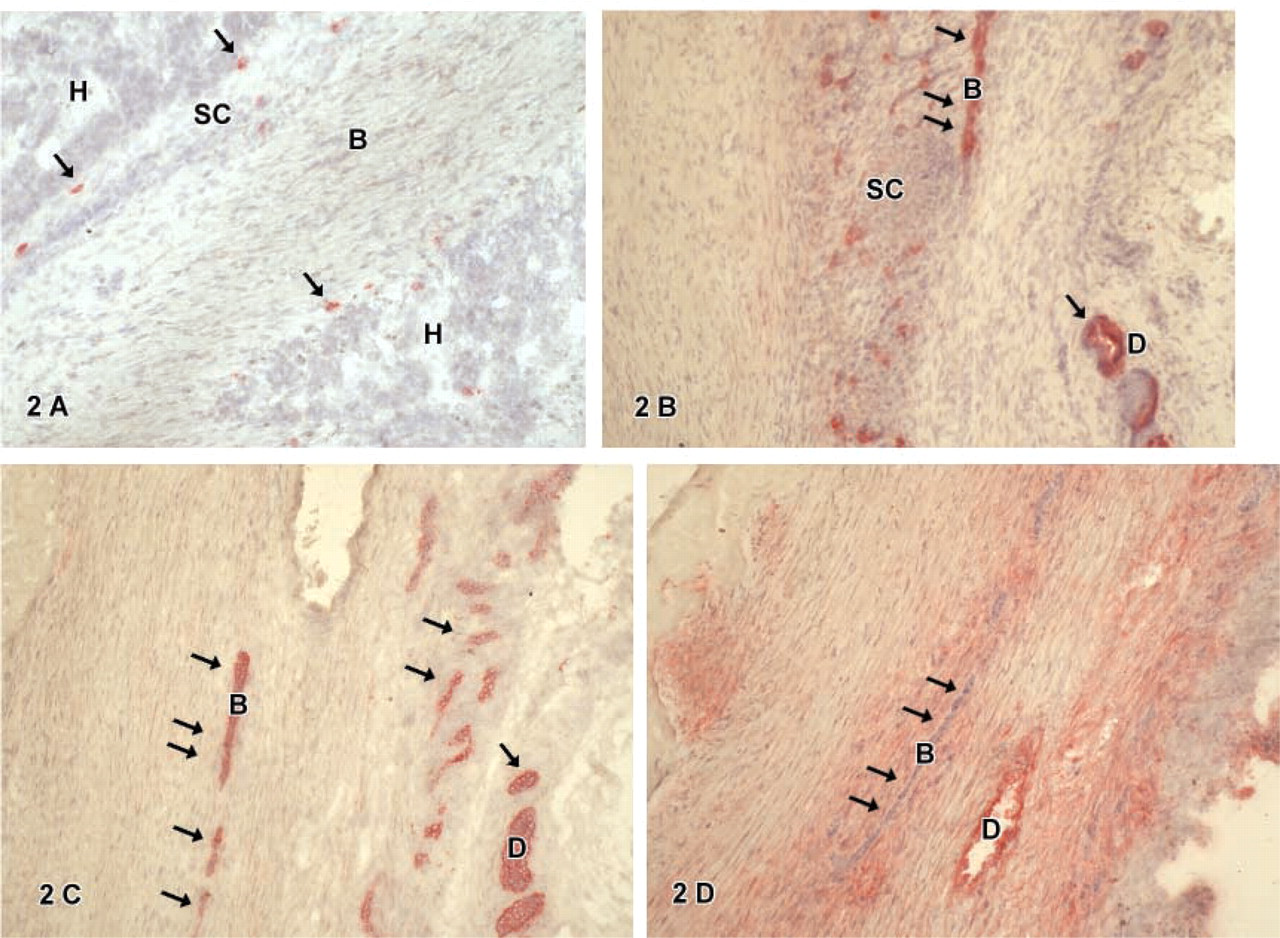
Cytokeratin-18-positive cells were found in all tumors. These cells were epithelial cells with a regular-shaped round nucleus, a small cytoplasm:nucleus ratio, and a polygonal cell shape resembling embryonal or fetal hepatocytic cells (H). Such cells were arranged either in sheets or groups surrounded by smaller cells (SC) (Figure 1A) or embedded in connective tissue (Figure 1C) not expressing CK-18. Furthermore, CK-18-positive cells were also found in atypical ducts (
Human hepatoblastomas analyzed a
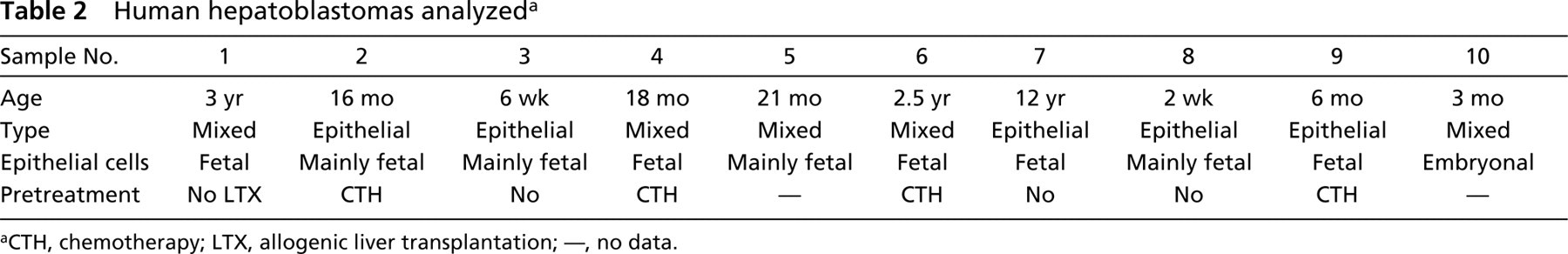
aCTH, chemotherapy; LTX, allogenic liver transplantation; —, no data.
IHC for Hematopoietic Stem Cell Markers CD34, Thy1, and c-kit
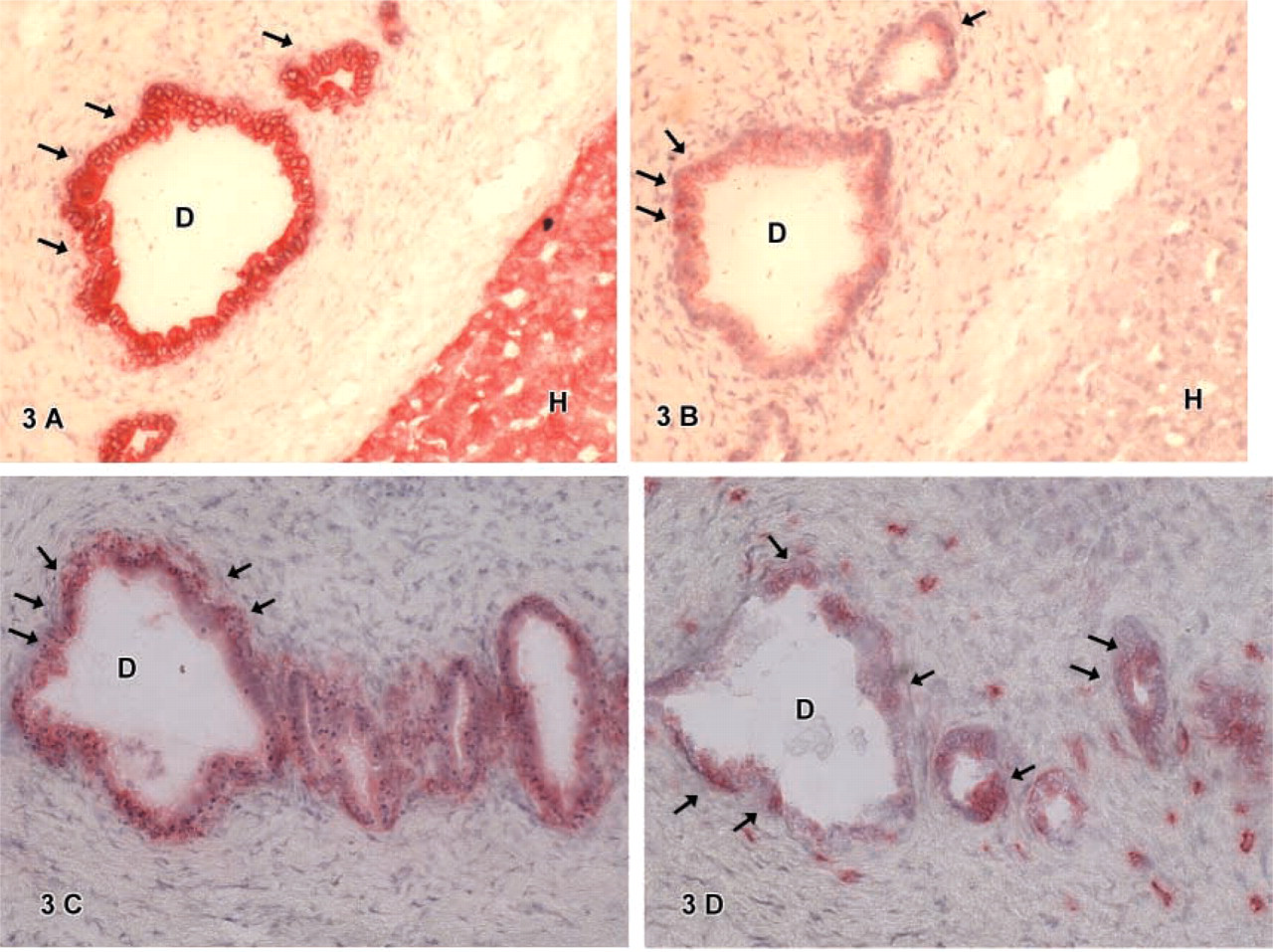
CD34-positive cells were found in some hepatoblastomas. Cells expressing CD34 were mainly located in atypical ducts (
C-kit-positive cells showed a different distribution. Cells in atypical ducts (
Thy1-positive cells were either in atypical ducts (
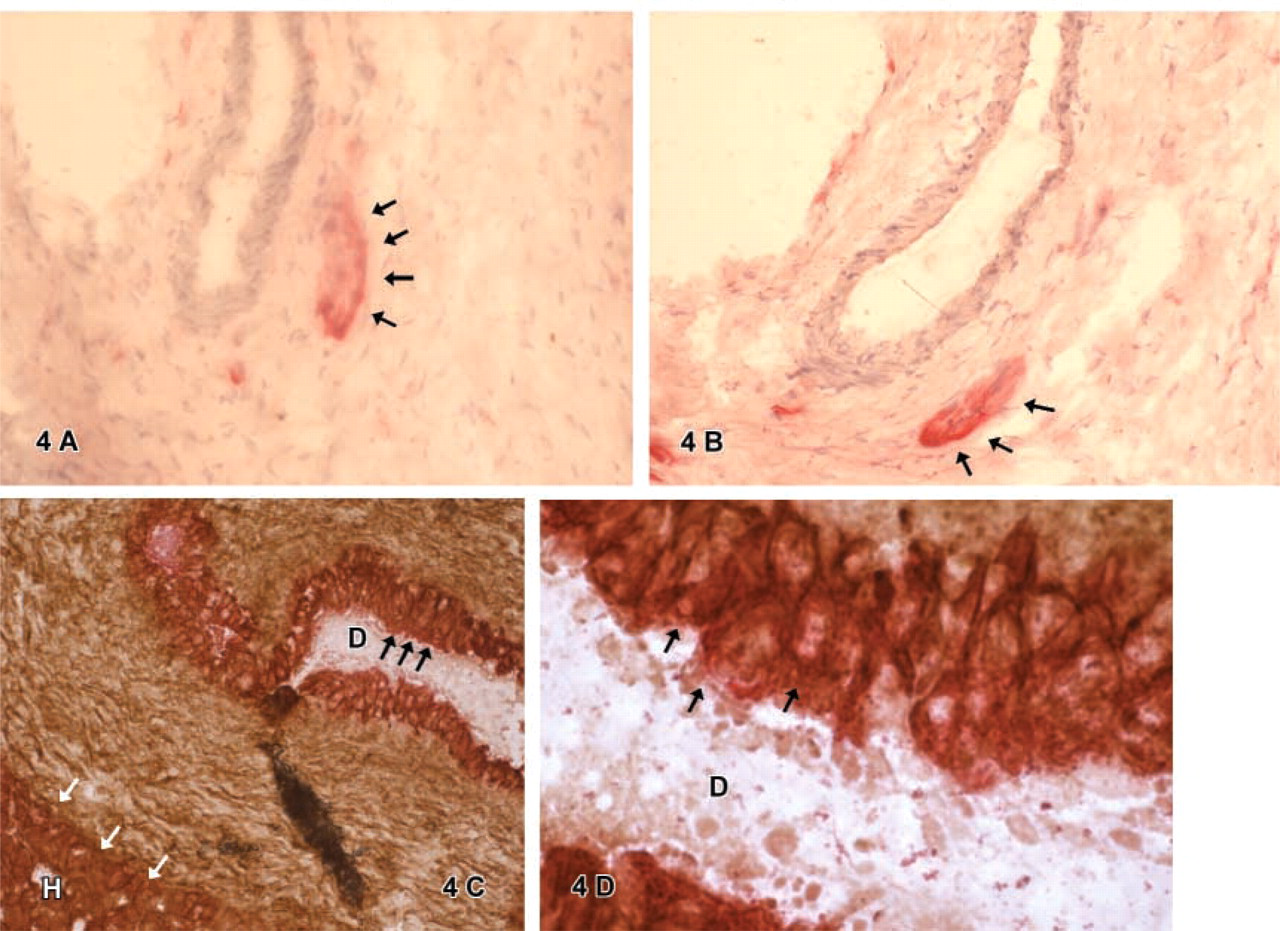
Double IHC for CK-18 and Thy1 or CK-18 and c-kit
Double IHC for CK-18 and Thy1 showed a positive reaction for CK-18 of hepatocytic cells (
Discussion
Stem Cell Markers Are Found on Different Hepatoblastoma Cells
Hematopoietic stem cell markers were used to identify possible candidates for stem cells in human hepatoblastomas. The results showed that the different stem cell markers studied were present in distinct varying cell types. CD34-positive cells were rarely observed in hepatoblastoma. We observed CD34 positive cells mainly located in atypical ducts (Figures 3 and 4). Furthermore, some small cells in one tumor were also found to be CD34-positive. Ruck et al. (1995) investigated the expression of the stem cell marker CD34 and additional markers of endothelial cells (CD31, von Willebrand factor) by IHC in hepatoblastoma. In contrast to our study, they found CD34-positive staining of liver sinusoids and capillaries in one of the studied specimens. This might be due to the different type of the tumors investigated. In the study of Ruck et al. the tumors were mainly anaplastic, whereas we investigated mainly fetal type tumors. IHC for the stem cell marker Thy1 showed a cell-specific reaction for Thy1 in atypical biliary ducts as well as in some ganglionic cells. Furthermore, a diffuse reaction of the connective tissue was observed. The positive staining reaction of the connective tissue for Thy1 was also found by others and was explained by an unspecific binding by the fibers of the connective tissue or by extracellular Thy1 accumulation (Morris and Ritter 1980; Morris and Beech 1984). C-kit-positive cells were found in atypical biliary ducts, as observed with CD34 or Thy1 positive cells. Additionally c-kit positive cells were found within the connective tissue: These were small cells located as isolated cells within the connective tissue fibers (overview Table 4). Baumann et al. (1999) identified c-kit positive cells in tissue specimens of biliary atresia in a previous study. Double staining showed that biliary CK-19 positive ductal cells occasionally expressed the stem cell marker c-kit. Some cells located around the portal tract also expressed both markers. From these observations, we concluded that cells co-expressing both markers might be progenitor cells of the human liver. Interestingly, these stem cells were found in ductal structures, but not in periportal areas or within hepatocytic cells. This was also observed in our study. Stem cell marker-positive cells for CD34, Thy1, and c-kit were located mainly in atypical ducts. On rare occasions, c-kit-positive cells were found as single cells within the connective tissue, or Thy1-positive cells were found in ganglionic groups. No such cells were observed in hepatocytic tumor cells, and only in one specimen were small cells positive for CD34 or c-kit found.
(
Marker expression in human hepatoblastoma tissue a
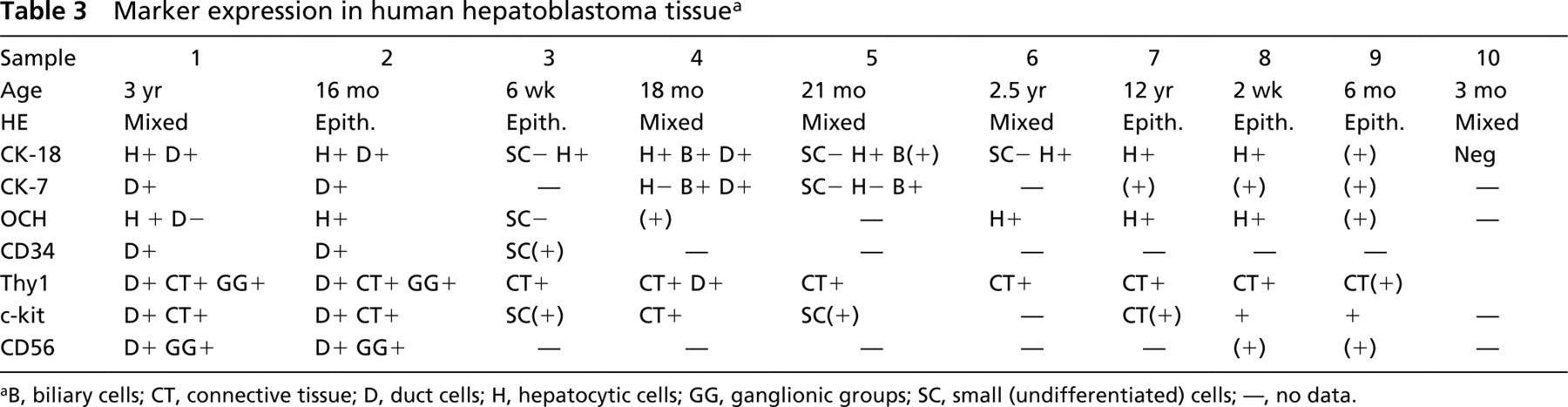
aB, biliary cells; CT, connective tissue; D, duct cells; H, hepatocytic cells; GG, ganglionic groups; SC, small (undifferentiated) cells; —, no data.
Stem-like cells Resembling Oval Cells Are Located in Atypical Ducts of Human Hepatoblastomas
Double IHC for the hepatic lineage marker CK-18 and the stem cell markers c-kit or Thy1 showed cells staining positive for both markers located in atypical bile ducts. Single staining furthermore characterized these duct cells as CD34-, CK-7-, and CD56-positive (Figures 3 and 4). The phenotypical marker expression of these cells identified in human hepatoblastoma equals the pattern of oval cells observed in adult human liver, which typically show the co-expression of stem cell markers (e.g., CD34, Thy1, c-kit) and hepatic lineage markers (Alison et al. 1996; Thorgeirsson 1996; Sell 2000; Vessey and Hall 2001). Other groups investigated the presence of the oval cell-associated antigen OV-6 to identify possible liver stem cells in hepatoblastoma by IHC studies (Crosby et al. 1998). Ruck et al. (1997) found small epithelial cells expressing CK-7, albumin, and OV-6. On this basis, it was suggested that the small epithelial cells described may be tumorous equivalents of adult oval cells in hepatoblastomas. Our study shows that small cells in hepatoblastoma do not regularly bear stem cell markers; therefore, we assume that these cells are not tumorous equivalents of oval cells. Cells with a similar pattern of marker expression, such as oval cells, were found to be distinct cells located in atypical ducts. This is consistent with findings published recently by Badve et al. (2003), who analyzed CD34 and bcl-2 expression in hepatoblastomas. They found no stem cell marker expression in small epithelial cells or in HepPar-1 (as hepatocytic marker) or CK-19 (as biliary marker)-positive small epithelial cells. From these observations it was speculated that small epithelial cells might be more primitive stem cells of hepatoblastoma than oval cell-like cells. Our data may strengthen this hypothesis. Well-characterized oval-cell-like cells co-expressing stem cell and hepatic lineage markers were located in atypical ducts. Small cells observed in our study were found in different locations. In rare instances they expressed stem cell markers (CD34 or c-kit) but never liver-like differentiation markers.
Other Stem Cell Marker-bearing Tumor Cells in Hepatoblastoma
Staining for stem cell marker Thy1 identified positive periductular clusters of cells resembling ganglionic groups (Figure 4). These cells also expressed the neuronal cell marker N-CAM (CD56). The presence of neuronal cells in close relationship to developing bile ducts was observed by Libbrecht et al. (2001) and Fabris et al (2000). These findings indicate that these groups might be neuronal progenitor cells. Furthermore, we observed distinct CD56-positve cells in atypical ducts. N-CAM (CD56) was first established as a marker for reactive biliary epithelial cells by Van den Heuvel et al. (2001). By immunocytochemical analysis of normal and diseased human liver, CD56 was found to be expressed by reactive bile ducts in most liver diseases. Normal biliary cells did not express CD56. Therefore, the CD56 expression of biliary cells might be explained by a reactive state of the tumorous bile ducts.
(
(
Stem cell marker-bearing cells represented in hepatoblastoma
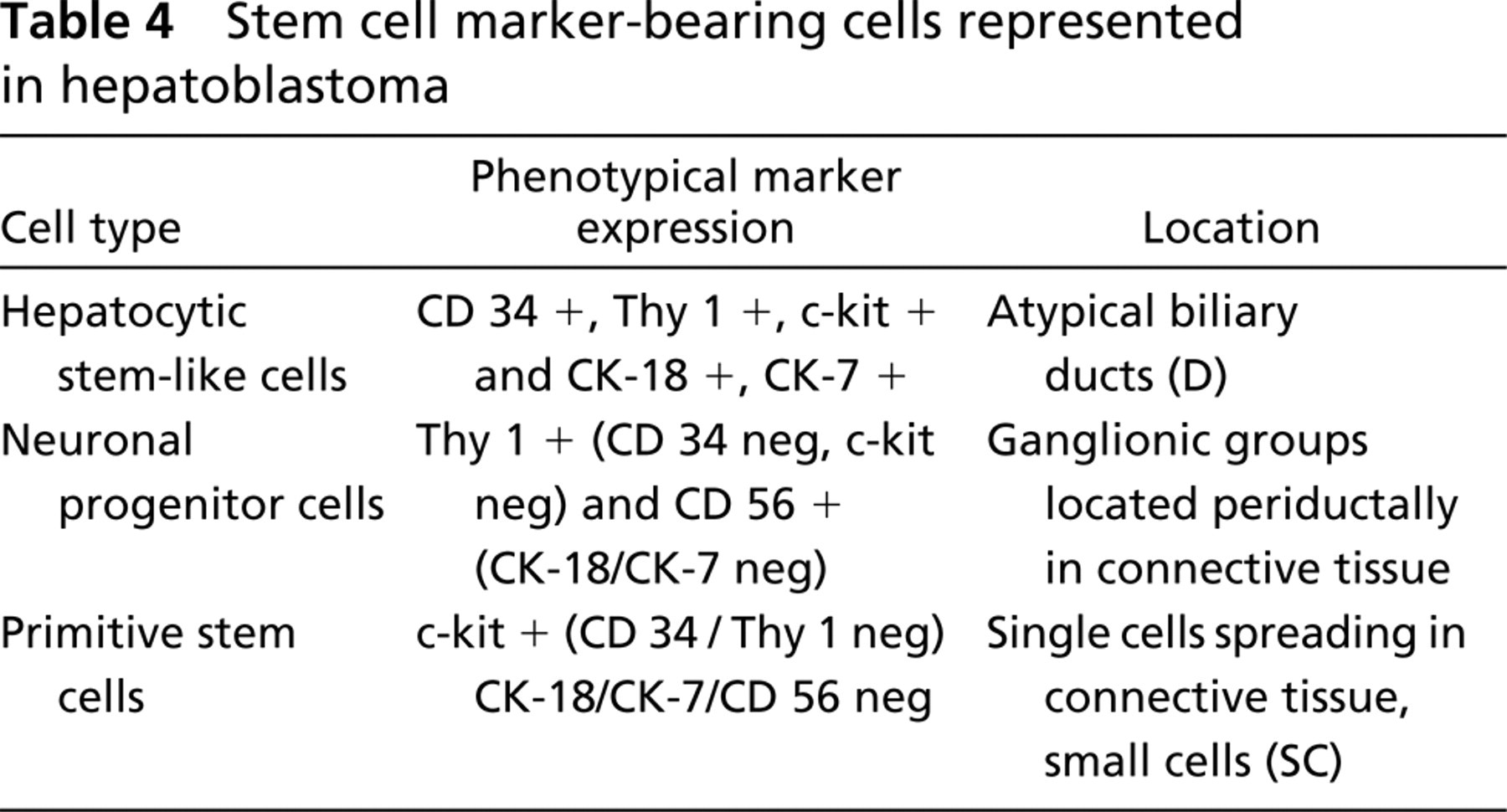
In short, the IHC analysis for the hematopoietic stem cell markers Thy1, c-kit, and CD34 and the hepatic or biliary lineage markers CK-18, CK-7, and CD56 revealed the presence of different types of stemcell marker positive cells in human hepatoblastoma (see Table 4). We observed cells positive for CK-18 and Thy1/c-kit resembling liver stem-like cells. These cells also expressed the stem cell marker CD34, the biliary marker CK-7, and CD56. Furthermore, we found cells positive for CD56 and the stem cell marker Thy1, which might be progenitors of neural cell lineages, and c-kit positive cells within the connective tissue negative for all other markers. The data indicate the presence of different types of stem cells during the histogenesis of hepatoblastoma.
Footnotes
Acknowledgements
We wish to thank Mrs B. Teichmann, Dept. of Pediatric Surgery, Medizinische Hochschule Hannover, for technical assistance.
