Abstract
Spexin is a highly conserved peptide which was recently identified through the bioinformatics approach. Immunohistochemical analysis of its expression has not yet been performed. Thus, in this study, we examined spexin location in a wide range of rat organs by both RT-PCR and IHC. RT-PCR identified spexin mRNA in all tissues examined. Spexin immunoreaction was mainly cytoplasmic. Spexin was immunohistochemically detected, although with different staining intensities, in epithelia and glands of skin and respiratory, digestive, urinary, and reproductive systems. Smooth muscle cells showed weak immunostaining, and connective tissue was negative. In the central nervous system, neuronal groups showed different intensities for reaction product. Immunoreaction was also found in ganglionic cells of both trigeminal and superior cervical ganglia and in photoreceptor, inner nuclear, and ganglionic layers of the retina. In the endocrine system, spexin immunoreaction was detected in the hypothalamic paraventricular and supraoptic nuclei; adenohypophysis, thyroid, and parathyroid glands; adrenal cortex and medulla (mainly ganglionic cells); Leydig cells; and thecal, luteal, and interstitial cells of the ovary. Because of its widespread expression, spexin is probably involved in many different physiological functions; in particular, location of spexin in neurons and endocrine cells suggests its roles as neurotransmitter/neuromodulator and endocrine factor.
S
These preprohormones contain a signal peptide sequence and putative dibasic prohormone cleavage sites. A small amino acid region between dibasic cleavage sites (called spexin or NPQ peptide) is highly conserved and is considered the most likely active neuropeptide (Mirabeau et al. 2007; Sonmez et al. 2009), although three other short potential peptides have also been suggested to have biological activity (Sonmez et al. 2009). These peptides have no homologies to other proteins. Spexin peptide, like many other peptide hormones, contains numerous aromatic amino acids and is probably amidated, as shown by the presence of a glycine residue at its end (Mirabeau et al. 2007). The rat spexin peptide differs from the human and mouse spexin peptides only by one amino acid at the C-terminal portion, ending with the sequence GHR instead of GRR, and this change has been suggested to prevent peptide amidation (Sonmez et al. 2009).
ISH identified spexin mRNA in the submucosal layer of esophagus and stomach fundus (Mirabeau et al. 2007). Its expression in this layer has been suggested to be related to the presence of the submucous plexus of the enteric nervous system (Mirabeau et al. 2007), which is known to produce neuropeptides involved in the control of smooth muscle contractility (Costa et al. 2000). Expressed sequence tags for spexin in GenBank indicate its presence in brain, ovary, kidney, and lung cancer cells. Preliminary RT-PCR investigations also showed the presence of spexin transcripts in human, mouse, and rat brains, and Northern blot analysis identified spexin expression in kidney, brain, pancreas, and, to a lower extent, placenta. The subregional distribution of spexin in the rat brain has also been studied by ISH, showing different expressions in the various nuclei (Sonmez et al. 2009). Moreover, RT-PCR and immunohistochemical analysis in the rat adrenal gland mainly identified spexin expression in the adrenal cortex (Rucinski et al. 2010).
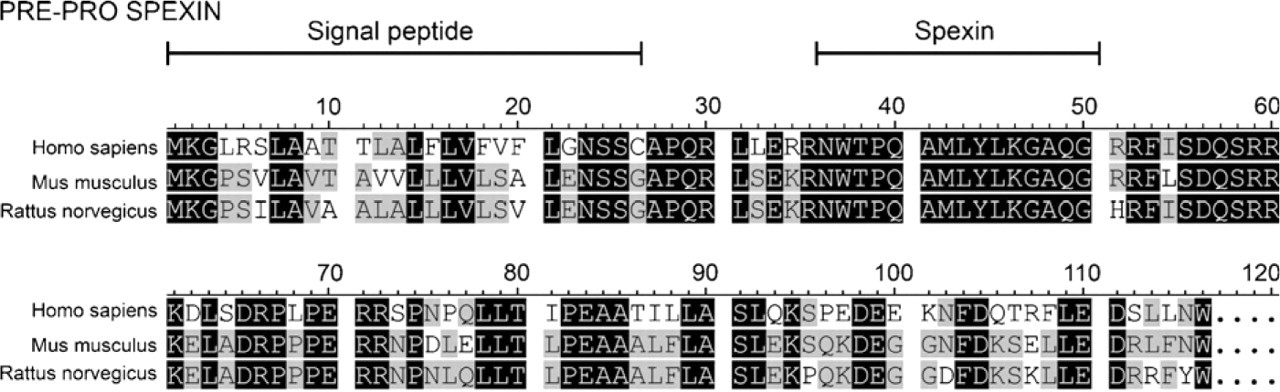
Amino acid sequences of human, mouse, and rat spexin preprohormones. The signal and mature peptides are marked. Black color indicates 100% homology. Gray color indicates homology more than 50%, but less than 100%.
Little is known about spexin's functions. The synthetic amidated spexin peptide NWTPQAMLYLKGAQ-amide has been found to dose-dependently induce muscle contraction in a stomach explant contractility assay (Mirabeau et al. 2007). We also recently found that spexin reduces the rate of adrenocortical cell proliferation and may evoke an increase in aldosterone and glucocorticoid secretion in some rat adrenocortical cell cultures (Rucinski et al. 2010). Apart from the actions mentioned, other spexin functions have not yet been demonstrated, but only supposed. Identification of the tissue or cell types that express spexin would contribute toward clarifying its physiological function. The aim of this study was to analyze spexin expression in a wide series of normal rat tissues by RT-PCR and IHC to establish its expression profiles in the rat.
Materials and Methods
Rat tissues were obtained from six 7-week-old intact rats of Wistar strain, three males and three females, kept under a 12-hr light/dark cycle (illumination onset at 8:00 am) at 23C and maintained on a standard diet and tap water ad libitum. Animals were sacrificed by decapitation. The study protocol was approved by the local Ethics Committee for Animal Studies. For real time-PCR studies, tissues were immediately placed in RNAlater (Qiagen; Hilden, Germany) and frozen at −80C. Tissues for IHC were fixed in 10% buffered formalin for 48 hr and paraffin embedded.
Conventional RT-PCR and Quantitative PCR
The applied methods have been described in detail in previous studies (Tortorella et al. 2003; Rucinski et al. 2005,2007,2008,2009a,b,2010; Ziolkowska et al. 2007). Briefly, total RNA was extracted by using TRI Reagent (Sigma; St. Louis, MO) and purified on columns (RNeasy Mini Kit; Qiagen). mRNA was extracted from the total RNA by using PolyATtract mRNA Isolation System III (Promega; Madison, WI), the amount of total mRNA was determined by measuring optical density at 260 nm, and purity was estimated by 260/280 nm absorption ratio, which was consistently higher than 1.8 (measured using NanoDrop spectrophotometer; Thermo Scientific, Northumberland, UK). RT was performed using AMV Reverse Transcriptase (Promega) with Oligo dT (PE Biosystems; Warrington, UK) as primers. The reaction was performed at 42C for 60 min (thermocycler UNO II; Biometra, Goettingen, Germany). The primers used were designed by Primer 3 software (Whitehead Institute for Biomedical Research; Cambridge, MA), and the reaction amplified a fragments spanning introns (Table 1). They were purchased from the Laboratory of DNA Sequencing and Oligonucleotide Synthesis, Institute of Biochemistry and Biophysics, Polish Academy of Sciences, Warsaw, Poland. For all tested genes, 35 cycles of the following sequences were applied: denaturation, 94C for 30 sec; annealing, 58C for 60 sec; and extension, 68C for 60 sec. In control reactions, water (instead of cDNA) and omission of RT were used. Reaction products were separated on ethidium bromide containing 2% agarose gel and photographed in a transilluminator.
Quantitative PCR (qPCR) was performed by means of the LightCycler 2.0 Instrument with software version 4.05 (Roche; Milan, Italy). SYBR green detection system was used with the above mentioned primers, which were also used for conventional RT-PCR. Each of the 20 μl reaction mixture contained 4 μl of template cDNA (or standards or control), 0.5 μM of each gene-specific primer, and a previously determined optimal MgCl2 concentrate (3.5 μM for one reaction). LightCycler FastStart DNA Master SYBR Green I mix (Roche) was used. The real-time PCR program included a 10 min denaturation step to activate the Taq DNA polymerase followed by a three-step amplification program (denaturation at 95C for 10 sec, annealing at 56C for 5 sec, and extension at 72C for 10 sec). Specificity of reaction products was checked by determining the melting points (0.1C/sec transition rate). Obtained results were normalized with porphobilinogen deaminase (PBGD) reference gene.
RT-PCR and quantitative PCR primers of spexin and PBGD
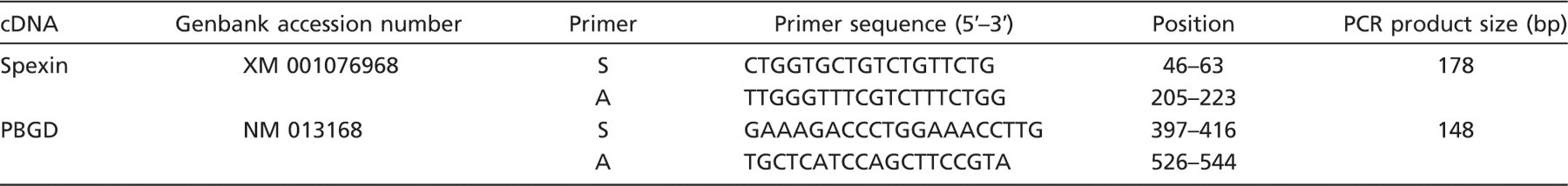
Oligonucleotide sequences for sense (S) and antisense (A) primers are shown. PBGD, porphobilinogen deaminase.
PCR efficiency was assessed by a serial dilution method. Products of traditional RT-PCR reaction were separated in 2% agarose gel, and specific bands were extracted by DNA gel extraction kit (Millipore). The amount of extracted DNA was estimated spectrophotometrically. The extracted DNA was diluted (10-fold serial dilutions) to get a standard curve for calculation of efficiency. The applied version of LightCycler 4.05 Software allows the evaluation of amplification efficiency plots.
Immunohistochemistry
Sections were hydrated gradually through decreasing concentrations of ethanol and then washed in deionized H2O. They were incubated in 0.3% hydrogen peroxide for 10 min at room temperature, to avoid endogenous peroxidase activity, and then in blocking serum (2% normal goat serum) for 30 min. Sections were incubated with primary rabbit polyclonal antibody recognizing human, rat, and mouse spexin (H-023–81; Phoenix Pharmaceuticals, Burlingame, CA) diluted 1:600 in PBS for 1 hr at room temperature. Sections were then washed three times for 5 min in PBS, incubated with anti-mouse/rabbit serum for 30 min (catalog no. MP-7500; Vector Laboratories, Burlingame, CA), and then developed in DAB (Sigma-Aldrich; Milan, Italy). Finally, sections were counterstained with hematoxylin. Negative controls were performed by omission of primary antibody. Spexin immunoreaction detected in human stomach was used as positive control, as suggested in the datasheet of the antibody. To verify the immunohistochemical specificity of the reaction, absorption tests with spexin (Phoenix Pharmaceuticals; No. 023–81) were also performed.
Immunohistochemical staining for spexin was evaluated by scanning entire tissue specimens under low power magnification (5×) and later confirmed under high power magnifications (10×, 20×, 40×). Immunohistochemical evaluation included number of positive cells, pattern of immunostaining (nuclear, cytoplasmic, and nucleocytoplasmic), and staining intensity. Overall staining intensity was stratified as follows: −, absent; +/ −, barely detectable; +, weak; ++, moderate; or +++, strong (Porzionato et al. 2006). Data of these studies are presented in Table 2.
Results
Classic RT-PCR revealed the presence of spexin mRNA in all organs and tissues studied: esophagus, stomach, small intestine, liver, pancreas, lung, skeletal muscle, heart, uterus, thymus, spleen, kidney, urinary bladder, brain, hypothalamus, adenohypophysis, thyroid, adrenal gland, testis, and ovary (Figure 2).
Subsequently, by means of qPCR, we studied the relative expression levels of spexin gene. As shown in Figure 3, in relation to PBGD gene, spexin gene expression was at least 10-fold higher in esophagus, liver, pancreas, kidney, brain, hypothalamus, thyroid, and ovary. In stomach, lung, skeletal muscle, heart, uterus, spleen, adrenal gland, and testis, expression of spexin gene was 5–10-fold higher than that of PBGD gene, and lower expression levels were found in small intestine, thymus, urinary bladder, and adenohypophysis.
Immunohistochemistry
We found widespread spexin immunostaining in the various tissues and organs examined, although expression levels differed among cell populations. Spexin immunoreactivity mainly involved epithelial cells and neurons. Regarding intracellular location, spexin-like immunopositive substances were mainly found in the cytoplasm.
Skin
Intense spexin-like immunopositivity was found in cytoplasm of epidermal cells. Inner and outer root sheaths of hair follicles also showed strong cytoplasmic positivity, whereas hair shafts and dermal papillae showed negative staining. Sebaceous glands showed intense cytoplasmic immunostaining (Figure 4A).
Respiratory System
The tracheal, bronchial, and bronchiolar epithelial cells exhibited strong cytoplasmic staining. No staining was seen in tracheal or bronchial cartilage. In lung parenchyma, the great majority of pneumocytes were negative, although rare alveolar lining cells with weak/moderate cytoplasmic immunoreaction were found (Figure 4B).
Expression of spexin-like immunoreactivity in normal rat tissues

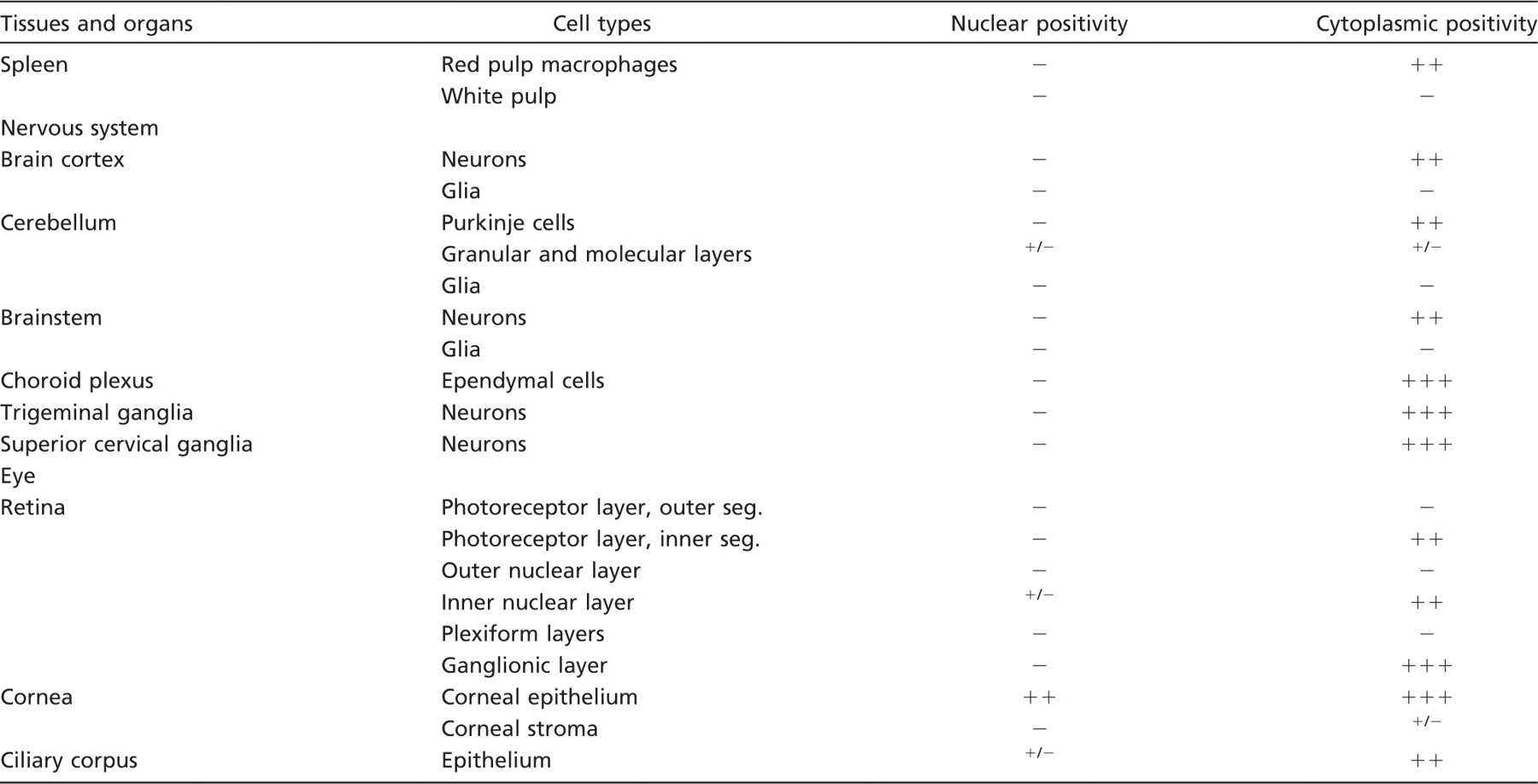
Immunostaining intensity graded as follows: -, negative; +/-, barely detectable; +, weak; ++, moderate; +++, strong.
Muscles
Skeletal and heart muscle cells showed heterogeneous staining, some fibers being negative and others showing weak or moderate immunostaining (Figure 4C-D). Smooth muscle cells of the gastrointestinal tract, urinary system, and blood vessels showed weak or moderate cytoplasmic immunostaining.
Cartilage
Epiphyseal cartilage cells were mainly negative, but sometimes cells showing moderate cytoplasmic staining were also visible (Figure 4E).
Bone
Osteoblasts, osteocytes and osteoclasts showed weak or moderate immunoreaction (Figure 4E). In bone marrow, the majority of cells were negative. Intense immunostaining was detectable in the cytoplasm of megakaryocytes (Figure 4F).
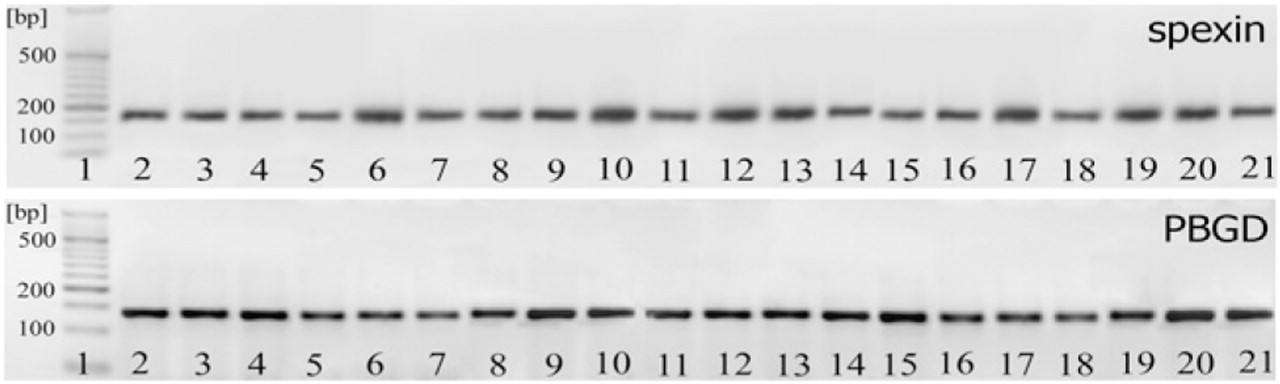
Qualitative RT-PCR analysis of spexin and housekeeping gene (porphobilinogen deaminase; PBGD) expression in different tissues. 1, DNA molecular marker size; 2, esophagus; 3, stomach; 4, small intestine; 5, liver; 6, pancreas; 7, lung; 8, skeletal muscle; 9, heart; 10, uterus; 11, thymus; 12, spleen; 13, kidney; 14, urinary bladder; 15, brain; 16, hypothalamus; 17, hypophysis; 18, thyroid; 19, adrenal gland; 20, testis; and 21, ovary.
Lymphatic Tissues
Moderate/strong immunostaining was detected in thymic epithelial cells, with particular reference to the medullary compartment. Hassal's corpuscles also showed positive immunoreaction, whereas lymphocytes showed negative immunoreaction (Figure 4G).
In the spleen, cells in the white pulp were mainly negative to immunostaining, whereas in the red pulp some cells, probably macrophages, showed moderate or strong cytoplasmic immunostaining (Figure 4H).
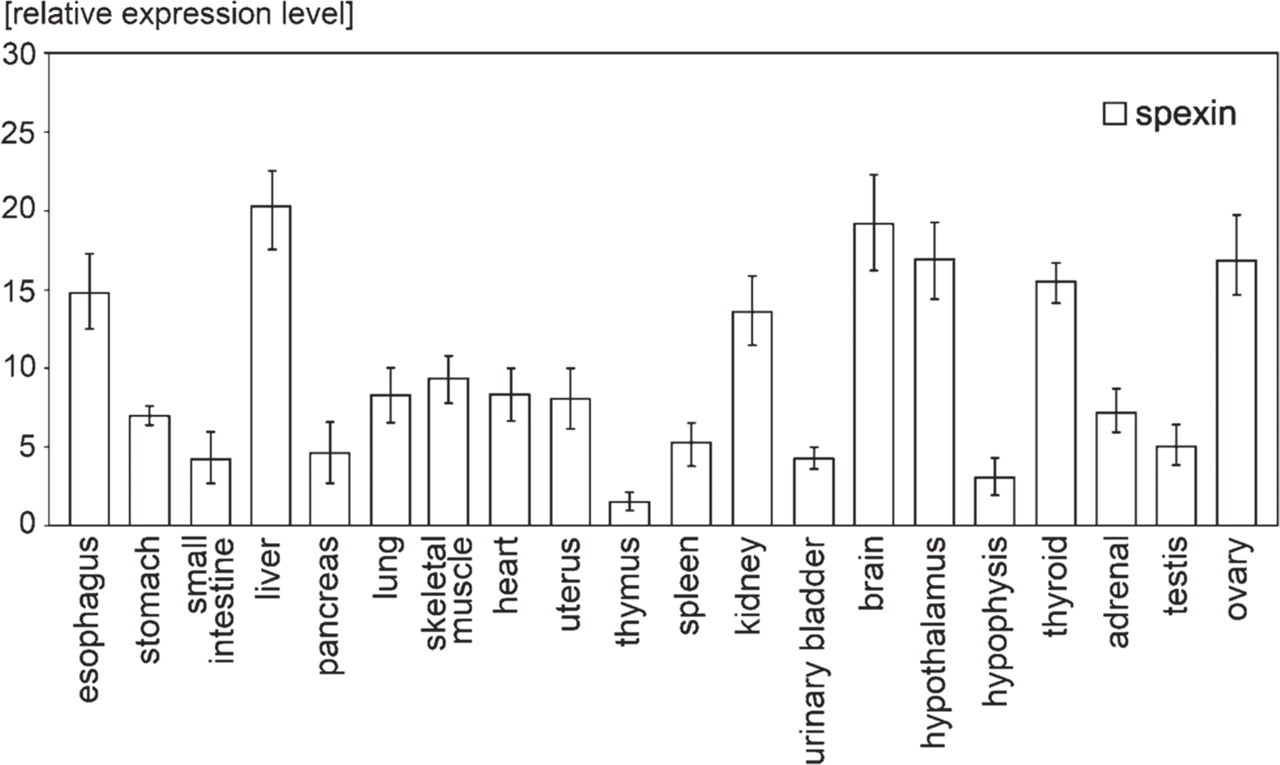
Quantitative PCR results of relative mRNA expression level in different tissues. Obtained results were normalized to PBGD reference gene.
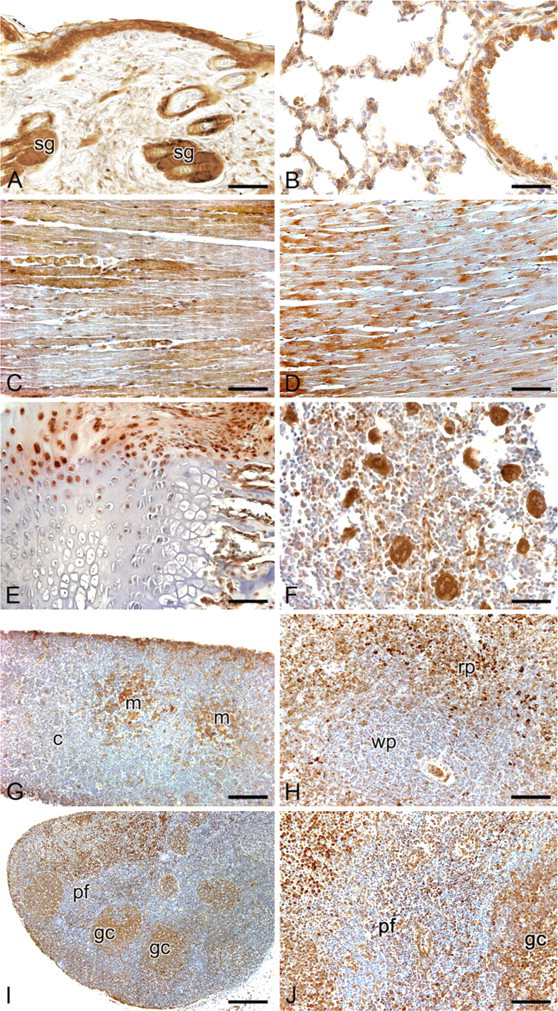
Anti-spexin immunohistochemical staining of rat normal tissues. (
In lymph nodes (Figure 4I-J), the majority of cells of the germinal centers of the lymphatic nodules showed moderate cytoplasmic immunostaining, whereas lymphocytes in the mantle zones showed mainly negative immunostaining, although some cells with moderate cytoplasmic staining were also visible. Rare cells showing nuclear positivity were also found. Aggregates of lymphocytes along the alimentary canal were mainly negative to immunostaining, with some cells showing moderate cytoplasmic staining.
Digestive System
In the salivary glands, ductal epithelia showed strong cytoplasmic immunostaining. Acinar secretory cells showed moderate/strong cytoplasmic positivity, mainly involving the basal compartments of the cells, whereas connective septa were negative to immunostaining (Figure 5A).
With the exception of superficial cells, esophageal epithelium showed intense cytoplasmic staining throughout its thickness, whereas nuclei showed negative staining. Lamina propria and submucosal layer were negative to staining. Smooth muscle cells of the muscularis mucosae and muscle layer showed weak or moderate cytoplasmic staining (Figure 5B).
In the stomach, most superficial and glandular epithelial cells showed moderate or strong cytoplasmic immunostaining. At the base of the gastric glands, positivity was more intense (Figure 5C).
In the small intestine, epithelial cells showed moderate/strong cytoplasmic immunostaining, whereas crypt cells showed less intense positivity (Figure 5D). In the large bowel and rectum, epithelial cells showed moderate cytoplasmic staining, whereas mucus droplets of goblet cells showed negative staining. Along the alimentary canal, spexin cytoplasmic immunoreaction was also identified in ganglionic cells of submucosal and muscle plexuses, whereas connective tissue structures (lamina propria and submucosal layer) showed negative staining and muscle layers showed weak/moderate staining.
In the exocrine pancreas, quite strong cytoplasmic immunoreaction was found in acinar secretory cells. In these cells, immunostaining mainly involved the basal compartment of the cells, whereas the connective tissue septa were negative to staining (Figure 5E).
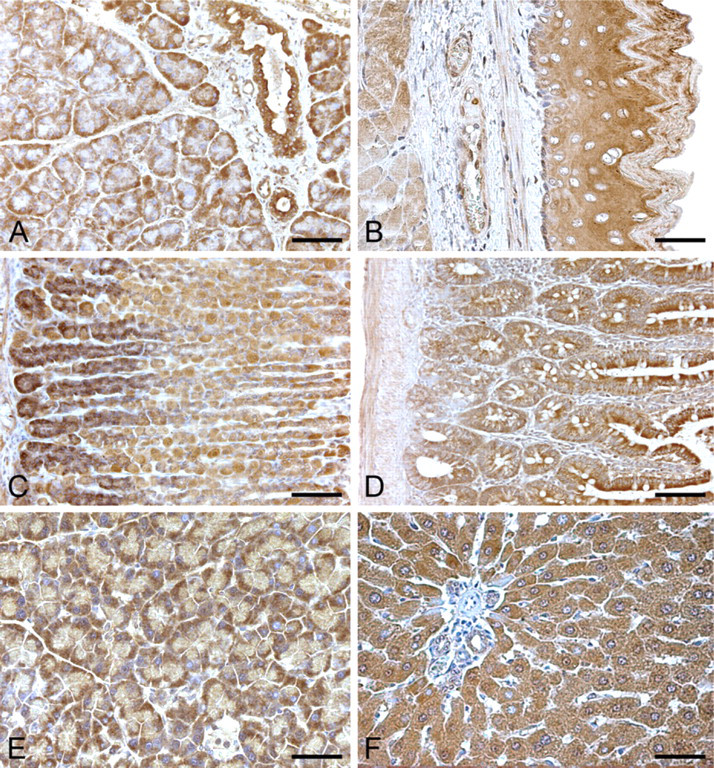
Anti-spexin immunohistochemical staining of rat digestive system. (
Hepatocytes showed intense cytoplasmic staining without nuclear immunoreaction. Connective tissue in the portal spaces showed negative staining. Sinusoidal lining cells, portal tracts, terminal hepatic venules, and hepatic arterioles were negative. In bile ducts, spexin-like immunoreactivity was seen in some epithelial cells (Figure 5F).
Urinary System
The renal corpuscles were usually negative to staining. Positive cytoplasmic immunoreaction was observed only in the epithelium of the loop of Henle and of distal tubules (Figure 6A). The surrounding stroma was negative. Urether and urinary bladder (Figure 6B) epithelium showed moderate cytoplasmic immunostaining.
Female Reproductive System
The epithelial cells of oviduct and endometrium (Figure 6C) showed moderate or strong cytoplasmic immunoreactivity, whereas stroma showed negative and muscle layers showed weak cytoplasmic staining. Vaginal epithelium showed strong cytoplasmic immunostaining throughout its layers. Lamina propria was negative to staining (Figure 6D).
Male Reproductive System
Epididymal epithelium showed moderate/strong cytoplasmic immunostaining (Figure 6E). Glandular epithelial cells of the seminal vesicles and prostate (Figure 6F) also showed moderate or strong cytoplasmic staining.
Brain
In the whole central nervous system, many neurons with moderate cytoplasmic immunostaining were visible. Glial cells were usually negative. Conversely, ependyma showed strong cytoplasmic immunoreactivity in all the regions examined. In the brain cortex, there were neurons with moderate/strong immunoreactivity and others were negative. Distribution of positive and negative neurons frequently followed the layer organization of the cortex. Neurons of the hippocampus (CA1, CA2, CA3, CA4, dorsal, and ventral blades of dentate gyrus) showed moderate cytoplasmic immunostaining (Figure 7A).
In the cerebellar cortex, Purkinje cells showed moderate cytoplasmic immunostaining. In the granular and molecular layers, the majority of cells were negative to staining, although some cells showing cytoplasmic or nucleo–cytoplasmic immunostaining were also detectable (Figure 7B). In the brainstem, different nuclei showed different percentages of immunostained neurons (Figure 7C). In the choroid plexuses, ependymal cells were strongly positive in the cytoplasm, whereas vascular structures were negative or weakly immunostained (Figure 7D). As regards the peripheral nervous system, ganglionic cells of both trigeminal (Figure 7E) and superior cervical ganglia (Figure 7F) showed strong cytoplasmic immunoreaction.
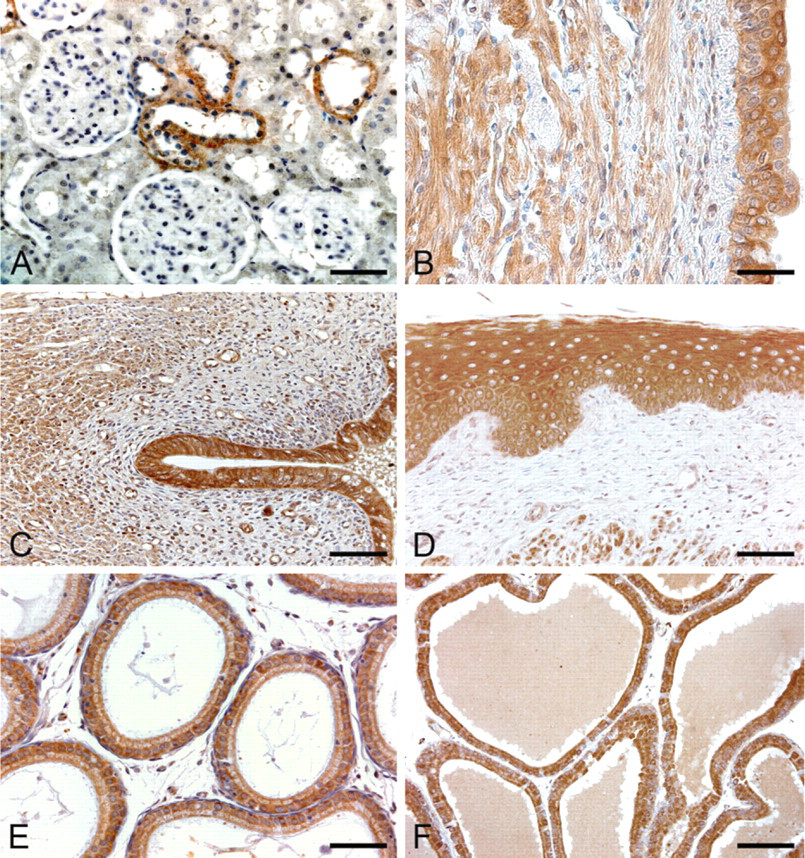
Anti-spexin immunohistochemical staining of rat genitourinary system. (
In the retina (Figure 7G), photoreceptor layer showed moderate staining in the inner segments, outer nuclear layer was negative, inner nuclear layer showed moderate/strong cytoplasmic or nucleo–cytoplasmic immunoreactivity, ganglionic cells showed intense cytoplasmic immunostaining, and plexiform layers were negative. Corneal epithelium (Figure 7H) showed very intense cytoplasmic and nuclear immunostaining, corneal stroma was mainly negative, and some sparse cells showed moderate/intense cytoplasmic immunostaining. Epithelium of the ciliary body showed moderate cytoplasmic immunoreactivity, and some cells also showed moderate/strong nuclear positivity.
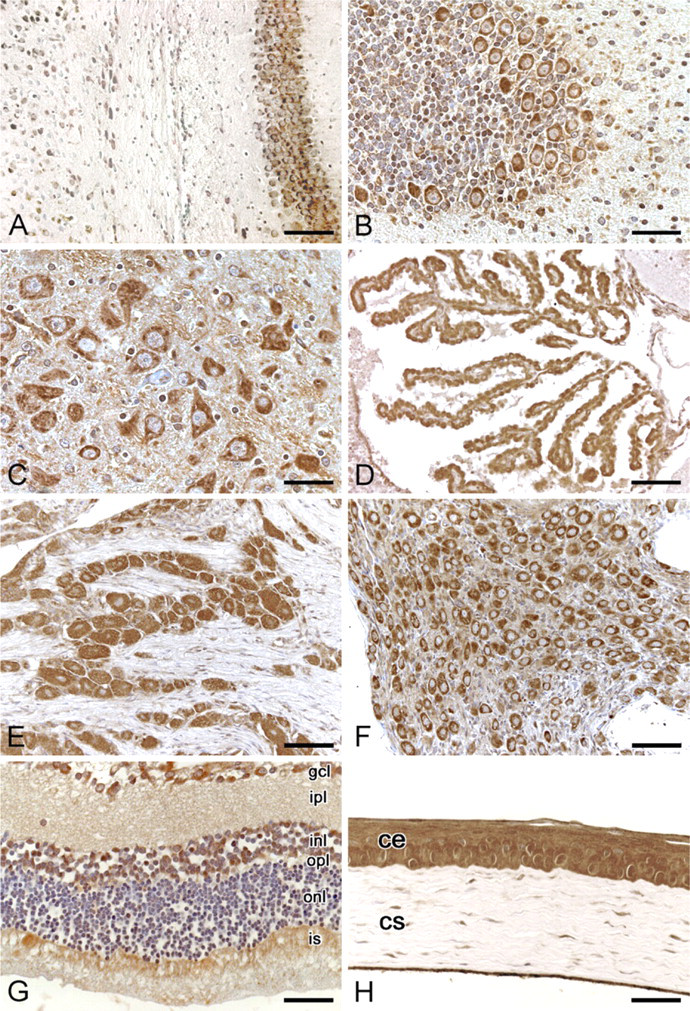
Anti-spexin immunohistochemical staining of rat nervous system and eye. (
Endocrine Glands
In the hypothalamus, both magnocellular and parvocellular neurons of the paraventricular nucleus showed moderate cytoplasmic positivity, although negative neurons were also visible (Figure 8A). Neurons of the supraoptic nucleus showed quite strong cytoplasmic immunoreactivity (Figure 8B). The other hypothalamic nuclei showed various degrees of spexin expression. Median eminence did not immunostain in either the internal or the external layer.
In the adenohypophysis, some large cells (basophilic cells) showed moderate or strong cytoplasmic immunostaining, whereas others did not show immunostaining. In the neurohypophysis, fibers showed weak/moderate immunostaining and some pituicytes showed nuclear positivity. In the pars intermedia, some cells showed cytoplasmic, nuclear, or nucleo-cytoplasmic immunoreaction (Figure 8C).
In the thyroid, follicular epithelial cells showed heterogeneous staining. In some follicles, epithelial cells showed moderate cytoplasmic staining, whereas in others, they did not show immunostaining. Sometimes, thyreocytes showing nuclear immunoreaction were also detectable. Parafollicular cells showed moderate/strong cytoplasmic immunoreactivity (Figure 8D). The chief cells of parathyroid gland presented moderate nucleocytoplasmic immunostaining.
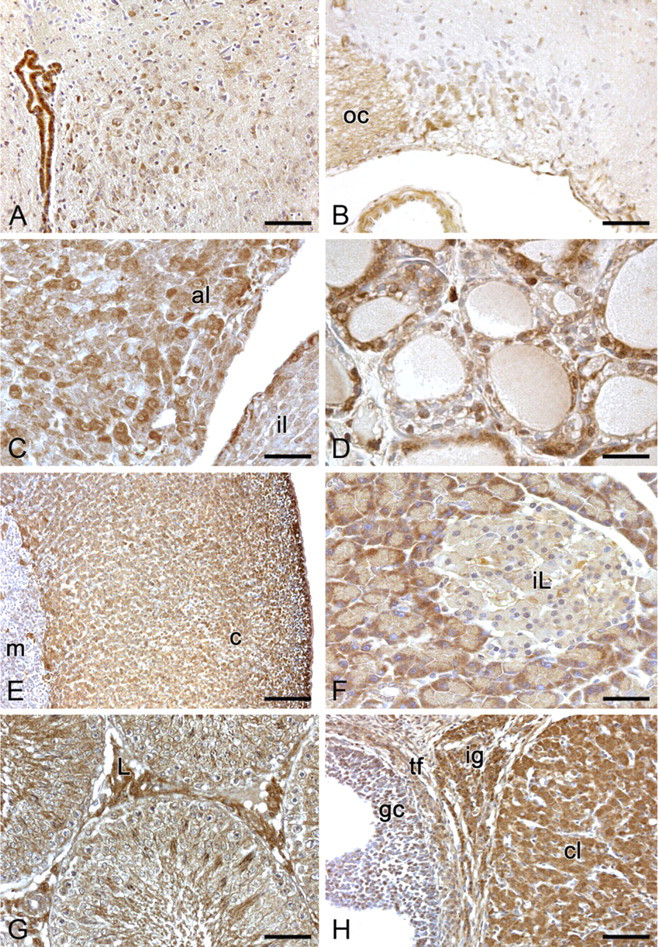
Anti-spexin immunohistochemical staining of rat endocrine tissues. (
Adrenal cortex cells showed moderate/strong cytoplasmic spexin immunostaining. Adrenal medullary cells were mainly negative or showed weak cytoplasmic positivity. In the adrenal medulla, ganglionic cells showed strong cytoplasmic positivity (Figure 8E).
The cells of the islets of Langerhans showed no spexin-like immunopositive substance (Figure 8F).
In the testis, spermatogonia and spermatocytes did not immunostain. Conversely, Sertoli cells and spermatids showed weak/moderate cytoplasmic immunostaining, and all Leydig cells showed strong cytoplasmic positivity. An intense spexin-like immunoreactivity was also present in residual bodies of the seminiferous tubules (Figure 8G).
In the ovary, oocytes were mainly negative. Granulosa cells were mainly negative, although some cells showed moderate or strong cytoplasmic or nuclear immunostaining. Thecal cells showed weak or moderate cytoplasmic staining. Luteal and interstitial gland cells showed intense cytoplasmic immunostaining, whereas ovarian stroma was mainly negative (Figure 8H).
Discussion
To our knowledge, very few reports are available on spexin expression (Mirabeau et al. 2007; Sonmez et al. 2009; Rucinski et al. 2010). Our RT-PCR and ICC studies revealed peptide expression in epithelial, muscle, endocrine, and nervous tissues and in numerous other organs. In the comparison between RT-PCR and immunohistochemical results, some small discrepancies were found in terms of levels of expression. Such different results between the two methods may be mainly due to posttranscriptional regulatory mechanisms of protein synthesis. As demonstrated in our assays, the highest expression of spexin-like immunoreactivity was present in various epithelia, somewhat lower expression in the muscular tissue, whereas only some cells were stained in the connective tissue. Characteristic structure of these connective tissue cells suggests that part of them are similar to fibroblasts, whereas the other population of spexin-positive connective tissue cells may belong to the mononuclear phagocyte system (MPS).
The highest level of spexin expression in both simple and stratified epithelia suggests its involvement in transepithelial transport, including secretion (Mirabeau et al. 2007), and may also be connected with regulation of the rate of epithelial cell renewal. All epithelia are characterized by high proliferation rate, and we cannot exclude the role of spexin in modulation of proliferation, differentiation, and death of cells in both surface and glandular epithelia. The role of spexin has recently been observed in the regulation of adrenocortical cell proliferation (Rucinski et al. 2010), suggesting a possible involvement of spexin also in the regulation of proliferation of other cell types.
RT-PCR identified spexin expression in rat brain, and immunohistochemical analysis also confirmed expression of spexin in many neuronal populations of both the central and peripheral nervous system. As mentioned earlier, spexin expression has already been identified in rat brain through RT-PCR and Northern blotting analysis (Sonmez et al. 2009). ISH analysis also showed differential expression of spexin in various nervous centers. An initial mapping study of spexin mRNA demonstrated its expression in the mesopontine tegmentum. At the caudal extent of mesopontine tegmentum, spexin mRNA was specifically located in the Barrington's nucleus, which can be identified from the expression of corticotrophin-releasing hormone mRNA. Regional distribution of spexin mRNA overlapped closely with that of corticotrophin-releasing hormone, suggesting possible cellular colocalization of these two factors. In contrast, spexin signal was distinct from the tyrosine hydroxylase signal, which is selectively expressed in locus coeruleus and is closely related but not overlapping with choline acetyltransferase, which is expressed in the laterodorsal tegmental nucleus. Spexin transcripts have also been found to be localized at the ventrolateral quadrant of the caudal periacqueductal gray (Sonmez et al. 2009). In keeping with preceding studies, we revealed differential spexin expressions in different nervous nuclei. The widespread distribution of spexin in nervous tissues together with its capability to be secreted (Mirabeau et al. 2007) suggests a role of this peptide in neuromodulation. In the central nervous system, some nuclei showed high percentages of positivity and strong immunostaining, whereas others showed negative staining. A specific mapping of the different structures of the nervous system will be necessary to better evaluate the nervous structures in which spexin plays a more important role and in which neural processes it is involved. Also, in cerebellar and cerebral cortex, positive neurons were found to be disposed along specific anatomical patterns. For instance, in cerebral cortex, superficial and deep layers were found to show higher percentages of neuronal positivity, whereas intermediate layers were mainly negative. Also, in the cerebellar cortex, Purkinje cells showed the most intense immunoreaction.
A really suggestive pattern of anti-spexin immunostaining was also found in the retina: photoreceptor layer showed moderate staining in the inner segments, whereas the outer segments showed negative staining; inner nuclear and ganglionic cell layers showed moderate/strong immunoreactivity, whereas outer nuclear layer showed negative immunoreactivity. Our immunohistochemical analysis also identified spexin expression in a series of structures of the peripheral nervous system. In particular, neurons of submucosal and muscle layers ganglia, the alimentary canal, and the superior cervical and trigeminal ganglia showed strong immunoreactivity. Positivity of neurons of submucosal ganglia confirms the hypothesis by Mirabeau et al. (2007) that identification of spexin mRNA in the submucosal layer of mouse esophagus and stomach by ISH may be ascribed to the local presence of the submucosal plexus.
RT-PCR analyses revealed spexin expression in all endocrine glands studied, with thyroid gland, ovary, and hypothalamus showing the higher levels of expression. Of special interest is distribution of spexin-like immunoreactivity in rat testes. Reaction was observed in Sertoli cells and spermatids and in residual bodies. Thus, it seems that spexin expression in seminiferous tubules is related to the late stages of spermatogenesis.
In steroid-secreting cells, an intense cytoplasmic spexin-like immunoreactivity is expressed in adrenocortical cells; testicular Leydig cells; and luteal, thecal, and interstitial cells of the ovary. In contrast, only few granulosa cells of the ovary were stained. Significance of this unexpected finding requires further investigation.
Also, in the adenohypophysis and in the thyroid gland, spexin-like immunoreactivity is heterogenous. In the pituitary gland, the highest reaction intensity was observed in basophilic cells. Intense reaction was observed in all parafollicular cells of the thyroid gland, whereas only a small fraction of thyroid epithelial cells were stained.
Earlier studies with anti-FLAG ICC following transfection of FLAG-spexin into rat pancreatic β-TC3 cells revealed colocalization of spexin into dense core granules of the secretory pathway, and Western blotting analysis of supernatant from N-FLAG-spexin-transfected cells also revealed FLAG-immunoreactive bands demonstrating secretion (Mirabeau et al. 2007). Thus, spexin expression in various endocrine cells and its capability to be secreted suggest a role of spexin in modulation of different endocrine functions, as previously described for other neuropeptides (Malendowicz 1993; Vinson et al. 1994; Tóth and Hinson 1995; Ehrhart-Bornstein et al. 1998; Mazzocchi et al. 1998; Nussdorfer and Gottardo 1998; Nussdorfer and Malendowicz 1998a,b; Nussdorfer et al. 1999; Bornstein et al. 2000; Spinazzi et al. 2005; Heinonen et al. 2008). Such a function of spexin has already been observed in the adrenal gland, as it has been demonstrated that spexin may stimulate aldosterone secretion by freshly isolated zona glomerulosa and corticosterone by an adrenocortical cell primary culture (Rucinski et al. 2010).
Expression of spexin mRNA and protein in the lymphatic system is very heterogeneous. In this system, spexin-like immunoreactivity is present mainly in cells of the MPS of the splenic red pulp and in epithelial cells of the thymic medulla. Spexin-like immunoreactivity is also present in germinal centers of the lymphatic nodules of the lymph nodes, whereas the white pulp of the spleen and gut-associated lymphatic tissue was negative.
In general, in the entire digestive system, respiratory conducting airways, and urinary and reproductive tracts, epithelial cells' spexin-like immunoreactivity is very intense; however, in epithelia composed of different cell types, staining intensity varies depending on the cell type. For example, negligible reaction was observed in goblet cells of the alimentary tract. In the kidney, spexin-like immunoreactivity is localized primarily to epithelial cells of the loop of Henle and of distal tubules, whereas proximal tubules were not stained.
In conclusion, our studies revealed the widespread expression of spexin mRNA and protein in the rat tissues. Expression of this highly conserved protein in some tissues and organs is heterogenous. Its location in neurons and endocrine cells and its capability to be secreted suggest roles as neurotransmitter/neuromodulator and as endocrine factor. Moreover, an important role of spexin in the functioning of epithelial cells is also suggested. The physiological and/or pathophysiological role of spexin in cells remains to be elucidated.
Footnotes
Acknowledgements
The authors are grateful to Anna Rambaldo and Gloria Sarasin for their skillful technical assistance.
