Abstract
We elucidated the localization of Thy-1–positive cells in the perichondrium of fetal rat limb bones to clarify the distribution of osteogenic cells in the process of endochondral ossification. We also examined the formation of calcified bone-like matrices by isolated perichondrial cells in vitro. At embryonic day (E) 15.5, when the cartilage primodia were formed, immunoreactivity for Thy-1 was detected in cells of the perichondrium adjacent to the zone of hypertrophic chondrocytes. At E17.5, when the bone collar formation and the vascular invasion were initiated, fibroblast-like cells at the sites of vascular invasion, as well as in the perichondrium, showed Thy-1 labeling. Double immunostaining for Thy-1 and osterix revealed that Thy-1 was not expressed in the osterix-positive osteoblasts. Electron microscopic analysis revealed that Thy-1–positive cells in the zone of hypertrophic chondrocytes came in contact with blood vessels. Perichondrial cells isolated from limb bones showed alkaline phosphatase activity and formed calcified bone-like matrices after 4 weeks in osteogenic medium. RT-PCR demonstrated that Thy-1 expression decreased as calcified nodules formed. Conversely, the expression of osteogenic marker genes Runx2, osterix, and osteocalcin increased. These results indicate that Thy-1 is a good marker for characterizing osteoprogenitor cells.
Recent advances in stem cell biology have improved the feasibility and effectiveness of cell-based therapy. The mesenchymal stem cell (MSC) is a potential tool for such therapy. MSCs have been identified in various tissues, such as bone marrow and adipose tissues, and possess the ability to give rise to myoblasts, chondrocytes, osteoblasts, and adipocytes in vitro (Pittenger et al. 1999; Clausen et al. 2006; Harting et al. 2008). However, the developmental process of osteoprogenitor cells has not been well characterized. Furthermore, although MSCs are reported to express the membrane marker proteins CD44, Thy-1 (CD90), CD106, CD146, and CD166 (Pittenger et al. 1999; Arai et al. 2002; Clausen et al. 2006; Park et al. 2007; Harting et al. 2008), their distribution in vivo and morphological characteristics have not been fully elucidated. Thy-1 is a glcycosylphosphatidylinositol-anchored protein located in lipid rafts (Haeryfar and Hoskin 2004). Various cell types, including T-cells, neurons, endothelial cells, mesangial cells, and fibroblasts express Thy-1 at their surface. This molecule is reported to be involved in multiple biological processes, such as T-cell activation, neurite growth, wound healing, and fibrosis (Haeryfar and Hoskin 2004; Rege and Hagood 2006a,b). Although Thy-1 is expressed in MSCs (Pittenger et al. 1999; Arai et al. 2002; Clausen et al. 2006; Park et al. 2007; Harting et al. 2008) and osteoblast lineage cells (Chen et al. 1999), the localization of Thy-1–positive cells during endochondral ossification in vivo has not been determined.
We performed immunostaining of Thy-1 to clarify its cellular distribution during endochondral ossification. We also examined the expression of osteoblastic marker genes by RT-PCR during the development of the osteogenic phenotype in vitro to address whether perichondrial cells contribute to the formation of bone.
Materials and Methods
Animal Experiments
All procedures for animal care were approved by the Animal Management Committee of Matsumoto Dental University.
Six pregnant Lewis rats, after 15.5, 17.5, and 20.5 days (Japan SLC, Inc.; Shizuoka, Japan) of gestation [hereafter referred to as embryonic day 15.5 (E15.5), E17.5 and E20.5, respectively], were used in this experiment. After an intraperitoneal injection of sodium pentobarbital (40 mg/kg/body weight) (Dainippon Sumitomo Pharma Co., Ltd.; Osaka, Japan), pups were obtained. Upper and lower limbs were dissected and fixed with 4% paraformaldehyde in 0.05 M phosphate buffer (pH 7.4) at 4C for 24 hr for immunohistochemical analysis.
Light Microscopy
Specimens were demineralized with 10% EDTA (pH 7.4) at 4C for 1 week. After dehydration in graded ethanol, the samples were embedded in paraffin. Five-μm-thick sections were obtained by microtome.
Immunohistochemistry for Thy-1
The sections were treated with 0.3% H2O2 in a solution of 0.01 M phosphate-buffered saline (PBS, pH 7.4) for 15 min at room temperature. They were pretreated in 3% bovine serum albumin (Sigma-Aldrich; Tokyo, Japan) in PBS for 30 min at room temperature and then incubated with mouse anti–Thy-1 monoclonal antibody (1:100; Santa Cruz Biotechnology Inc., Santa Cruz, CA) for 12 hr at 4C. They were finally incubated in Histofine Simple Stain rat MAX-PO MULTI (Nichirei Co.; Tokyo, Japan) for 1 hr at room temperature. After sections were washed with PBS, immuno-reactivity was visualized by immersion in a DAB-H2O2 solution (0.05% diaminobenzidine and 0.01% H2O2 in 0.05 M Tris-HCl buffer, pH 7.6) for 5 min at room temperature. Immunostained sections were then counter-stained with methyl green.
Non-immune mouse sera were used in place of the primary antibody as negative controls. These sections did not show any specific immunoreactivity.
Double Fluorescent Staining
For the detection of Thy-1 and osterix, paraffin sections were incubated with mouse anti–Thy-1 antibody (1:100) and rabbit anti-osterix polyclonal antibody (1:100; Abcam., Cambridge, UK) for 12 hr at 4C. They were immersed in Alexa-Fluor-488–conjugated anti-mouse IgG (1:200; Molecular Probes, Inc., Eugene, OR) and Alexa-Fluor-594–conjugated anti-rabbit IgG (1:200; Molecular Probes). The fluorescence of specimens was observed under a fluorescence microscope (Axioplan 2; Carl Zeiss, Oberkochen, Germany) with the appropriate filter combinations: BP485/20 and BP515-565 for Alexa-Fluor-488 and BP546/12 and LP590 for Alexa-Fluor-594.
Immunoelectron Microscopy
Approximately 50-μm-thick sections were cut using a vibrating blade microtome (VT1000S; Leica Microsystems K.K., Tokyo, Japan). These sections were incubated in anti–Thy-1 antibody for 48 hr at 4C, followed by horseradish peroxidase–conjugated anti-mouse IgG for 24 hr at 4C. Immunoreactivity was visualized by immersion in DAB-H2O2 solution. After postfixation with 1% OsO4 in 0.1 M phosphate buffer (pH 7.4) for 1 hr at 4C, the tissue slices were dehydrated in graded acetone and embedded in Epon 812 (TAAB Laboratories Equipment Ltd.; Berkshire, UK). Ultrathin sections were cut using an ultramicrotome (Ultracut UCT; Leica Microsystems K.K.) and stained with lead citrate. These sections were observed under a transmission electron microscope (H-7600; Hitachi High-Technologies Co., Tokyo, Japan) at an accelerating voltage of 80 kV.
Dissection of Perichondrial Cells and Culture
For the analysis in vitro, perichondrial cells were obtained from rat fetal limbs at E17.5 according to a previous report (Colnot et al. 2004). Briefly, the perichondrium was removed using thin forceps after the dispase treatment (1 mg/ml in PBS for 5 min at room temperature) and transferred into α-modified Eagle's medium (α-MEM; Sigma-Aldrich, St. Louis, MO) supplemented with 0.1 mg/ml collagenase and 0.05% trypsin (Gibco; Grand Island, NY). After 30 min at 37C, the perichondrium was loosened by pipetting, and perichondrial cells were obtained. The perichondrial cells were cultured in α-MEM supplemented with 10% fetal bovine serum (FBS) in a humidified atmosphere of 5% CO2 in air.
RT-PCR primers for osteogenic differentiation

GAPDH, glyceraldehyde-3-phosphate dehydrogenase.
Alkaline Phosphatase (ALPase) and Alizarin Red Staining
Subcultured cells originating from the perichondrium were plated in 48-well plates (5 × 104 cells/well) and cultured in α-MEM containing 10% FBS, 5 mM β-glycerophosphate, 100 μg/ml ascorbic acid, and 10−7 M dexamethasone (osteogenic medium) for 1, 2, or 4 weeks. These cells were rinsed with PBS and stained with nitro blue tetrazolium chloride/5-bromo-4-chloro-3-indolyl phosphate toluidine salt (Roche Diagnostics; Indianapolis, IN) at 37C for 30 min to detect ALPase activity or with 0.1% Alizarin Red S solution (Wako Junyaku; Tokyo, Japan) at room temperature for 5 min to analyze calcified nodules.
RT-PCR
Total RNA was isolated using Isogen (Nippongene; Tokyo, Japan), and single-strand cDNA was synthesized by SuperScriptIII Reverse Transcriptase (Invitrogen; Rockville, MD). The primer sets used for PCR are provided in Table 1.
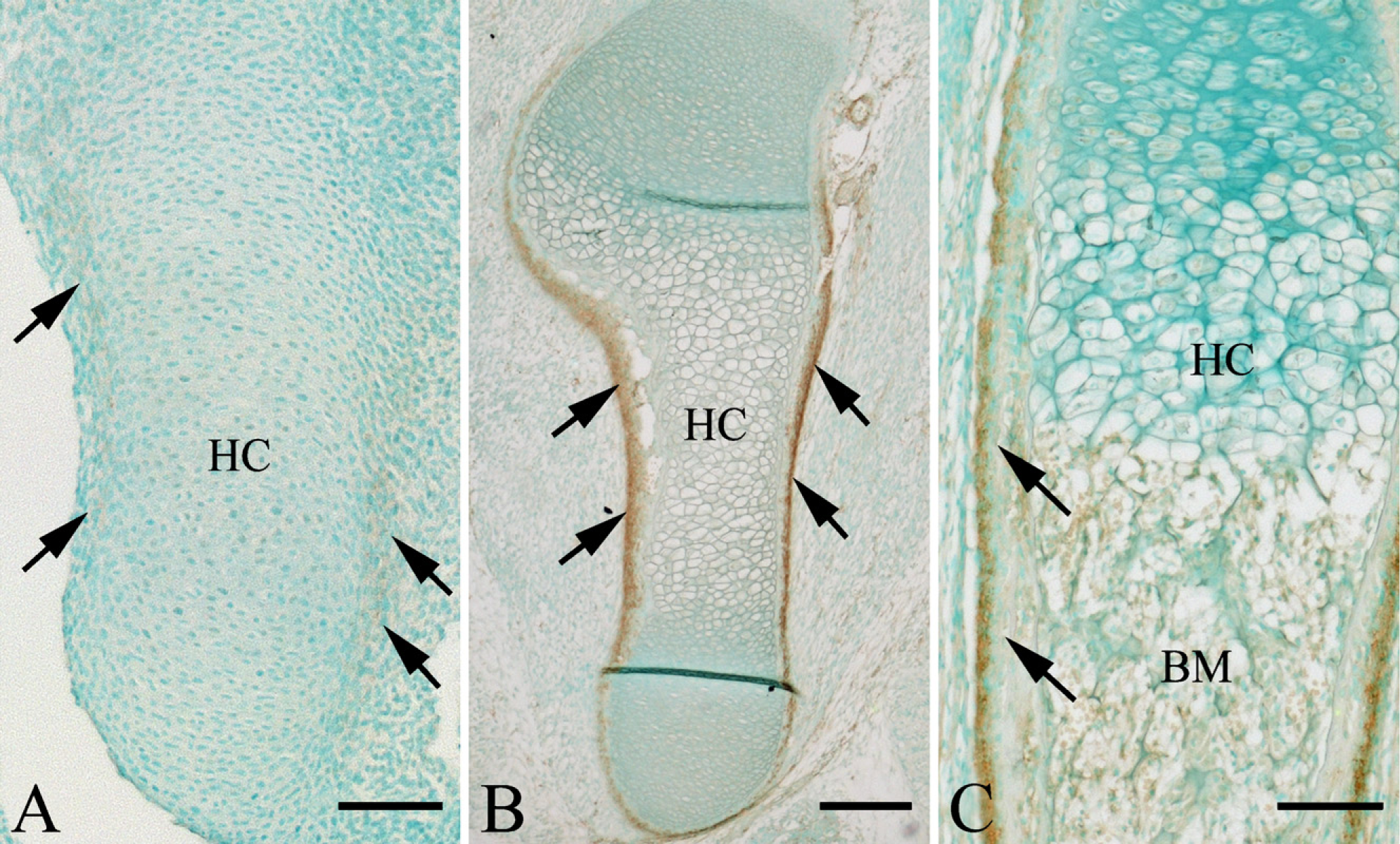
Light micrographs indicating the distribution of Thy-1–positive cells.
Results
Light and Electron Microscopic Observation
We observed immunolocalization of Thy-1 in humerus and femur because these showed similar processes of endochondral ossification during similar developmental stages. At E15.5, the cartilage primordium of limbs was surrounded by the perichondrium, consisting of flattened cells. Chondrocytes in the middle of the cartilage had a hypertrophic appearance. The perichondrium close to hypertrophic chondrocytes tends to be thicker than that in distal and proximal regions. No bone collar was observed at this stage. Immunoreactivity for Thy-1 was first detected in the perichondrium adjacent to hypertrophic chondrocytes (Figure 1A). Thy-1–positive cells had a spindle shape. In contrast, little reactivity was seen in the perichondrium of the distal and proximal regions of limbs. Immunoreactivity for Thy-1 was undetectable in chondrocytes at any stage. At E17.5, the bone collar had formed in the middle of the limbs. Round-shaped osteoblasts lined the bone matrix. Vascular invasion was seen in the zone of hypertrophic chondrocytes. Intense labeling of Thy-1 was seen in the periosteum (Figure 1B). Thy-1–positive flattened fibroblastic cells constituted the outer layer of the perichondrium. However, osteoblasts on the bone matrices scarcely showed any reactivity. At E20.5, primary trabecular bone and the marrow cavity were established. Blood vessels were also seen in the marrow space. Bone matrices formed by osteoblasts were deposited on the cartilage cores. Thy-1–positive fibroblastic cells were observed in the bone marrow as well as periosteum (Figure 1C).
To address the relationship between Thy-1–positive cells and osteoblast lineage cells, we performed double immunostaining of Thy-1 and osterix at E20.5. Osteoblasts scarcely showed any labeling of Thy-1 (Figure 2A). Intense immunoreactivity for osterix was detected in the nucleus of osteoblasts on bone surfaces (Figure 2B). However, cytoplasmic and nuclear labeling of osterix was seen in Thy-1–positive cells adjacent to osteoblasts (Figure 2C).
We attempted immunoelectron microscopy at E17.5 because the bone collar's formation and vascular invasion had begun. Flattened cells in the periosteum showed Thy-1 labeling on their plasma membranes (Figure 3A). These cells had a fibroblastic appearance, with rough endoplasmic reticuli, the Golgi apparatus, and mitochondria (Figure 3B). They were surrounded by collagen fibrils. Thy-1–positive cells were also seen adjacent to blood vessels in the perichondrium (Figure 4A). At the site of vascular invasion in the zone of hypertrophic chondrocytes, Thy-1–positive cells were occasionally accompanied by blood vessels (Figure 4B).
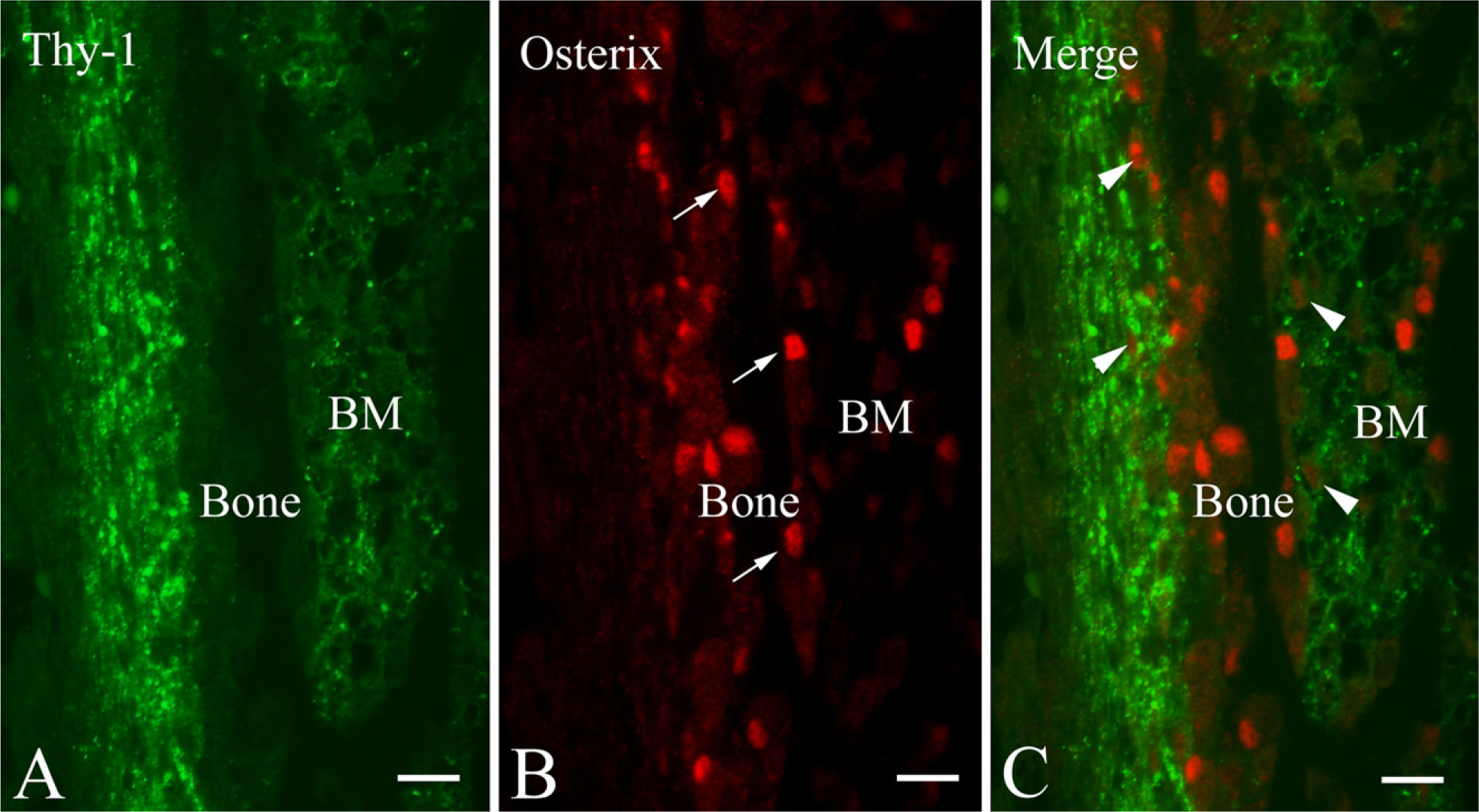
Fluorescent micrographs indicating the localization of Thy-1 and osterix at E20.5.
In Vitro Analysis of Perichondrial Cells
To investigate the relationship between Thy-1 expression and osteoblastic differentiation more precisely, we cultured perichondrial cells under osteogenic conditions and analyzed the expression levels of Thy-1 and osteoblastic markers. Perichondrial cells showed ALPase activity, and this activity increased during the culture. Calcified nodules started to form at 2 weeks in the presence of β-glycerophosphate, ascorbic acid, and dexamethasone, and mineralization was apparent at 4 weeks (Figure 5A). Semi-quantitative RT-PCR revealed that the expression of Thy-1 mRNA in perichondrial cells cultured with osteogenic medium decreased over time. On the other hand, the expression of the osteoblastic markers Runx2, osterix, and osteocalcin was increased at 1 week and continued during the culture (Figure 5B).
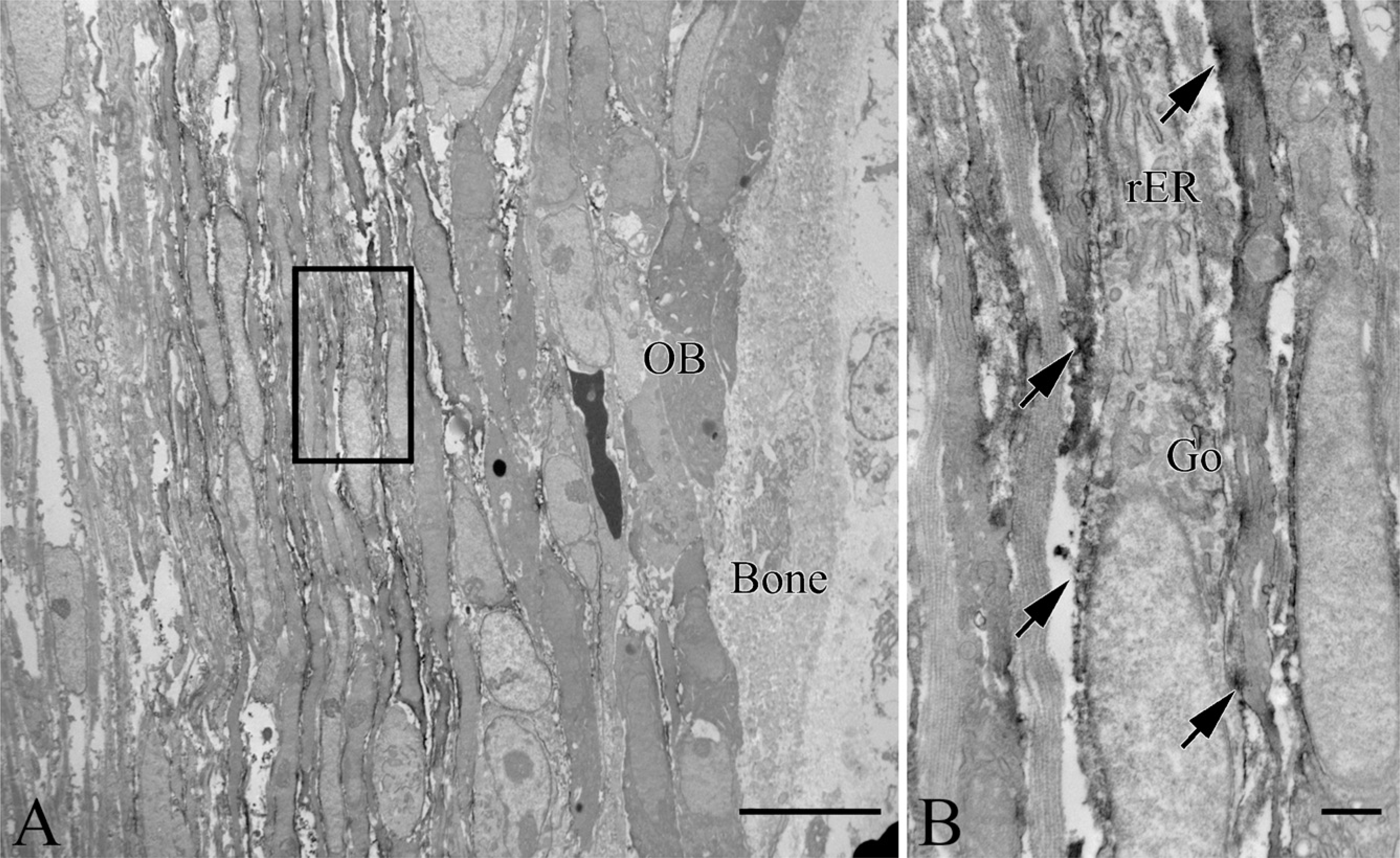
Immunoelectron micrographs indicating localization of Thy-1 in the periosteum.
Discussion
The present study revealed that a specialized area in the perichondrium and the periosteum of rat limbs expresses Thy-1, a marker of MSCs. The perichondrium and periosteum serve as sources of osteoprogenitor cells (Cormack 1987). Previous research in vitro found that bone marrow–derived mesenchymal stromal cells and periosteal-derived cells also appeared to express Thy-1 (Chen et al. 1999; Pittenger et al. 1999; Clausen et al. 2006; Park et al. 2007; Harting et al. 2008). Immunoreactivity for Thy-1 was first detected in the perichondrium at E15.5 and was absent in chondrocytes, indicating that Thy-1 is a good marker of osteoblast lineage cells. Consistent with this, Chan et al. (2009) reported that CD105+ Thy-1+ cells in the perichondrium could not differentiate into chondrocytes but contributed to bone formation.
Our immunoelectron microscopic observations revealed that Thy-1–positive cells occasionally came into close contact with blood vessels. Additionally, Thy-1–positive cells appeared in the zone of hypertrophic chondrocytes when vascular invasion occurred in the process of endochondral ossification. This would suggest that the osteoblasts forming primary trabecular bone originate from the perichondrium. In fact, Colnot et al. (2004) concluded in their ex vivo experiment that the perichondrium is the source of both cortical and trabecular osteoblasts. Our in vitro experiment also indicated that Thy-1 is associated with potential precursors of osteoblasts and is involved in bone formation during endochondral ossification. Thy-1–positive cells in the perichondrium might differentiate into osteoblasts, and were required for the formation of bone during endochondral ossification. Thy-1–positive cells in the perichondrium showed fibroblastic characteristics under the electron microscope. Because it is difficult to distinguish Thy-1–positive cells from fibroblasts in connective tissues on the basis of morphological features, Thy-1 is a useful marker for osteogenic cells.
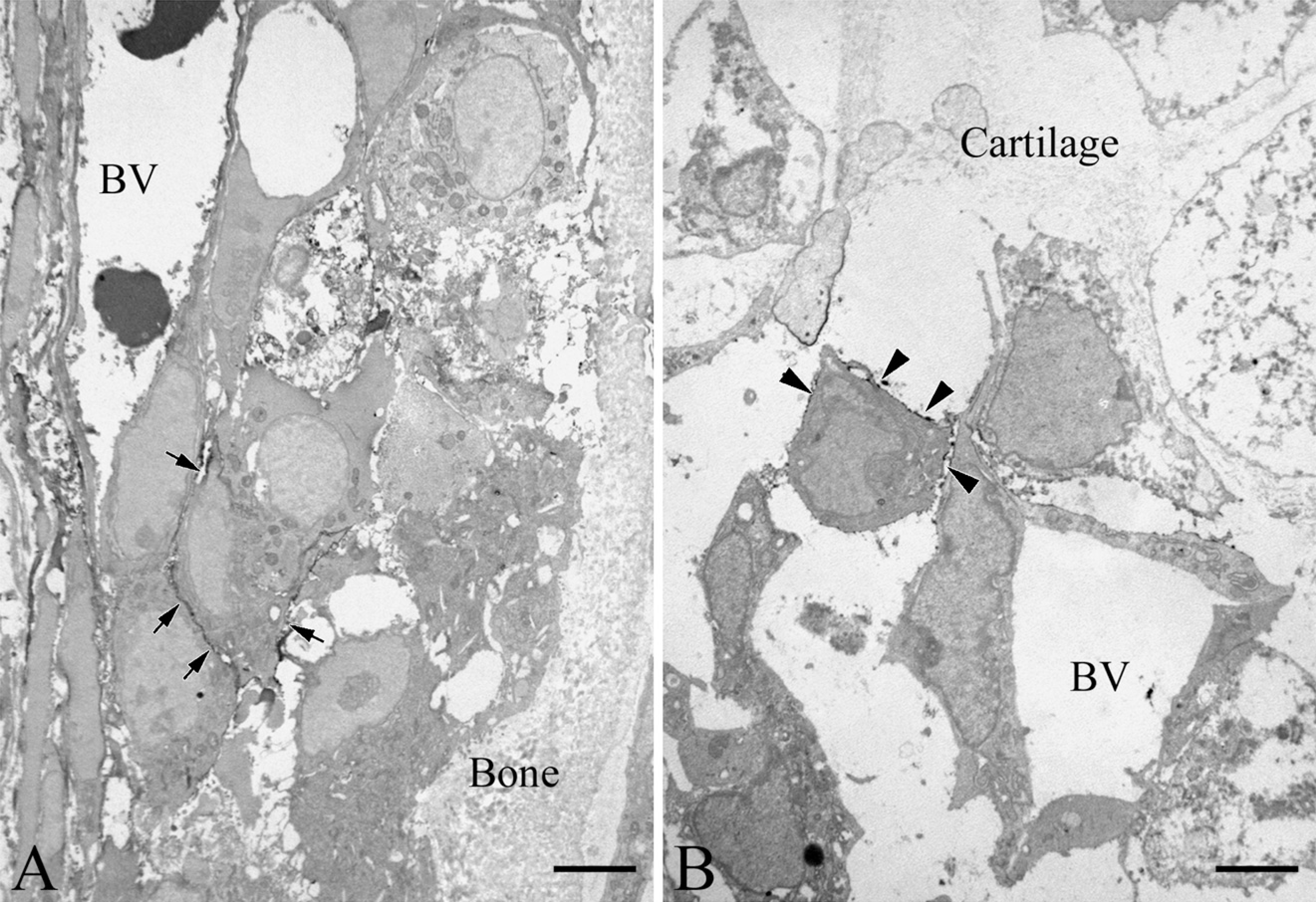
Immunoelectron micrographs indicating localization of Thy-1 at the vascular invasion site.
Osteoprogenitor cells sequentially differentiate into osteoblasts. Runx2 and osterix, specific transcription factors in osteoblast lineage cells, regulate the gene expression of bone matrix proteins, including osteocalcin, osteopontin, and bone sialoprotein (Aubin et al. 2006). We performed an experiment in vitro to address the differentiation of Thy-1–positive cells into osteoblastlike cells. The perichondrial-derived cells exhibited ALPase activity and generated mineralized nodules in osteogenic medium. We also examined the expression of Thy-1 and several osteoblastic marker genes by RT-PCR. The expression of Thy-1 decreased as calcified nodules formed, whereas that of the osteoblastic markers increased in culture with osteogenic medium. The results suggest that the expression of Thy-1 in perichondrial cells decreased with the differentiation into osteoblast-like cells. We also attempted double staining of Thy-1 and osterix to clarify the relationship between Thy-1–positive cells and osteoblasts in vivo. Osteoblasts on the bone surface were positive for osterix in the nucleus but negative for Thy-1. Although most Thy-1–positive cells in the periosteum did not exhibit staining for osterix, the periosteal cells close to osteoblasts showed osterix-positive labeling. In combination, our in vitro and in vivo data suggest that the perichondrium serves as a source of osteoprogenitor cells, and Thy-1 expression in osteoprogenitor cells is gradually decreased with their differentiation into osteoblasts. On the other hand, we cannot completely rule out that Thy-1–negative perichondrial cells might differentiate into osteoblast-like cells. To characterize the progenitors for bone formation, it is necessary to isolate Thy-1–positive cells by fluorescence-activated cell sorter and clarify their differentiation into osteoblast-like cells forming calcified matrices.
Vascular pericytes may also represent a potential source of osteoprogenitor cells in adults (Doherty et al. 1998; Kalajzic et al. 2008). Our previous study indicated that fibroblastic cells expressing α-smooth muscle actin (α-SMA), a marker of myofibroblasts and pericytes, in the periodontal ligament might differentiate into osteoblasts (Hosoya et al. 2006,2008). Although vascular pericytes are capable of differentiating into osteoblastic cells in vitro and in vivo, our immunohistochemical observation revealed that Thy-1–positive cells do not show α-SMA immunoreactivity (data not shown). Therefore, Thy-1–positive osteogenic cells in the perichondrium are distinguished from myofibroblasts and endothelial pericytes. Recent studies have demonstrated that circulating bone marrow–derived stem cells participate in ectopic bone formation and fracture healing (Matsumoto et al. 2006,2008; Eghbali-Fatourechi et al. 2007; Otsuru et al. 2007,2008). We could not completely exclude the participation of circulating stem cells as a source of osteoblasts in the process of endochondral ossification. Whether circulating stem cells express Thy-1 is unknown. The physiological relevance of Thy-1–positive cells as circulating osteoprogenitors must be studied.
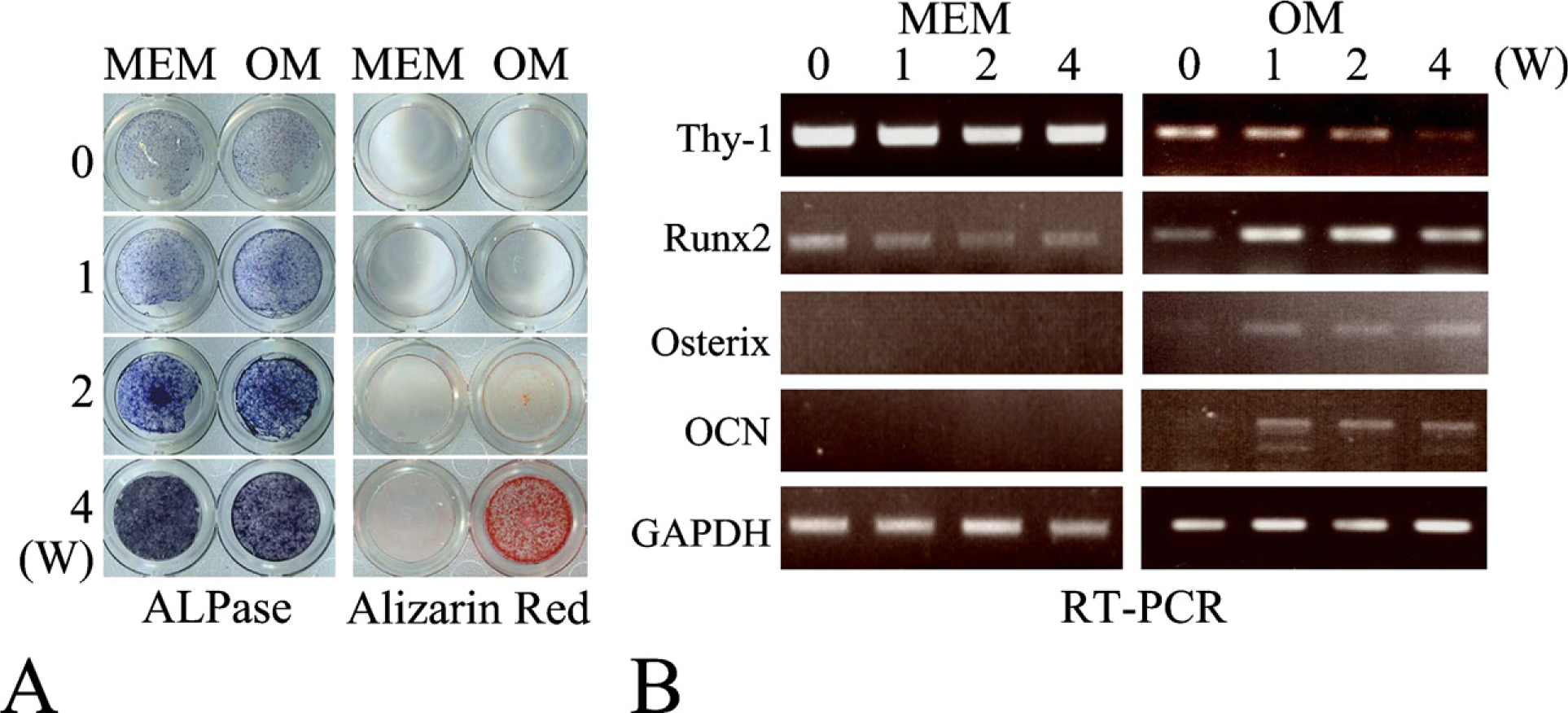
In vitro analyses of perichondrial cells. (
Several lines of evidence indicate that many molecular signals are involved in endochondral ossification. Indian hedgehog (Ihh) secreted by prehypertrophic chondrocytes appears to determine where a bone collar develops (St-Jacques et al. 1999; Colnot et al. 2005), because Ihh-deficient mice have no bone collar (Colnot and Helms 2001) and overexpression of Ihh by retroviral infection induced an ectopic bone collar (Long et al. 2004). Thus, Ihh signaling is essential for normal osteoblastic differentiation in endochondral bones. Moreover, FGF (Eswarakumar et al. 2002; Yu et al. 2003), PTH/PTHrP (Chung et al. 2001), and canonical Wnt/β-catenin also seem to play crucial roles at specific stages of the osteogenic program (Day et al. 2005; Rodda and McMahon 2006). It remains to be elucidated whether these signaling molecules participate in the differentiation of Thy-1–positive cells into osteoblast-like cells.
Recently, numerous articles have documented the importance of MSCs for bone tissue engineering (Bernardo et al. 2009; Deschaseaux et al. 2009; Menicanin et al. 2009). Osteoprogenitors, rather than pluripotent cells, are required for cell-based therapy in bone regeneration (Matsumoto et al. 2008; Luan et al. 2009). However, the transcription factors maintaining the multi-potency of MSCs and regulating the initiation of osteogenic differentiation are not fully understood. Thy-1–positive cells might be a valuable model for evaluating osteoprogenitors for bone tissue engineering.
In conclusion, we have defined a population of Thy-1–positive cells in the perichondrium. The perichondrial Thy-1–positive cells possess potential osteo-genic activity and participate in the formation of both the bone collar and primary trabecular bone during endochondral ossification. Thy-1–positive cells might be a good tool for investigating the characteristics of osteoprogenitor cells.
Footnotes
Acknowledgements
This work was supported by Promotion Research Expenses from the Graduate School of Oral Medicine, Matsumoto Dental University.
